Abstract
We report the exceptional occurrence of murine double minute clone 2 amplification in an atypical meningioma and its recurrent anaplastic meningioma arising in the right frontal lobe of a 75-year-old man. Murine double minute clone 2 amplification was shown by array comparative genomic hybridization and confirmed by fluorescence in situ hybridization. This is a rare finding with only one similar report in the literature. Awareness of this finding is indicated and should not lead to misdiagnosis of other entities that more commonly show this feature.
Introduction
Murine double minute clone 2 (MDM2) overexpression by amplification is typically described in a well-defined subset of sarcomas (e.g. dedifferentiated liposarcoma) and is not a typical feature of meningioma.
Yet, we report the exceptional occurrence of MDM2 amplification in an atypical meningioma and its recurrent anaplastic meningioma. This is a rare finding with only one similar report in the literature. 1
Case report
A 75-year-old man was admitted with lethargy, headache, confusion, and behavioral disinhibition. The patient was known with hypercholesterolemia, aortic valve insufficiency, lumboischialgia, and rapid eye movement (REM) sleep behavior disorder. The patient’s family history was negative. There were no arguments for neurofibromatosis type 2 in particular. Computed tomography (CT) and magnetic resonance imaging (MRI) revealed a large, extra-axial, circumscribed, contrast enhancing, subdural 5.2 cm mass located in the right frontal cerebral cortex. The tumor was broadly attached to the dura and showed a typical dural tail. The lesion showed prominent perilesional cerebral edema and extended into the adjacent brain parenchyma. The tumor mass led to compression of the anterior horn of the right lateral ventricle and slight deviation from the midline to the left. No other brain lesions were detected. The tumor was completely resected with a margin of 1 cm surrounding its dural attachment (Simpson grade 1). Adjuvant fractionated external beam radiation therapy was administered to the right frontal cortex field. A total dose of 56 Gy was delivered in 2 Gy per fraction over a period of 6 weeks. Histological examination revealed a highly cellular spindle cell lesion. The nuclei showed prominent nucleoli and an increased mitotic count (9 mitoses per 10 high power fields (HPF)). Multiple small foci of necrosis were identified. Immunohistochemistry for epithelial membrane antigen (EMA) showed both tumor regions with an intense and diffuse positivity, and regions with only minimal positivity. Somatostatin receptor subtype 2A (SSTR2A) showed diffuse and strong membranous and cytoplasmic immunoreactivity. STAT-6 was negative. A diagnosis of an atypical meningioma, World Health Organization (WHO) grade II was made (Figure 1). One year later, MRI showed subdural relapse of the tumor at the same location. The recurrent tumor was broadly attached to the dura, showed uniform contrast enhancement, and invasion of the right frontal cortical brain parenchyma on MRI. The tumor was completely resected (Simpson grade II). Upon resection, histological examination showed distinct features of malignancy including extensive necrosis, high mitotic count (44 mitoses per 10 HPF), and prominent cytological atypia. EMA showed partial positivity that was less pronounced than the primary tumor. SSTR2A showed membranous and cytoplasmic reactivity in the majority of the tumor. PR, S100 and STAT-6 were negative. The tumor showed only very focal keratin expression. The diagnosis was anaplastic meningioma, WHO grade III (Figure 2). The patient died 1 month later. In the pathology department of UZ Leuven, unusual or poorly differentiated tumors are frequently subjected to array comparative genomic hybridization (aCGH) for molecular karyotyping. aCGH was carried out on the frozen tumor specimen from the recurrent, anaplastic meningioma using the Oxford Gene Technology (OGT) CytoSure™ ISCA oligoarray set (Oxford Gene Technology, Oxford, UK) containing 180k DNA oligonucleotides with a minimum resolution of 200 kb. The detailed aCGH results are depicted in Supplementary Table 1. In short, copy number gains were found in a range of chromosomes, including 8q, 12q, and 17q, while losses occurred in chromosomes 1p, 6q, 7p, 10, 12, 14q, 17q, 18, and 22q. Chromosomes 12 and 17 were affected by chromothripsis. The former was associated with the high-level amplifications of CDK4/12q14.1 and MDM2/12q14.3-15 genes, while chromosome 17q23 region contained amplified CLTC, PTRH2, and RPS6KB1 genes. Heterozygous losses of PTEN/10q23, NDRG2/14q11, AKT1/14q32, MEG3/14q32, NF1/17q12, DAL1/18p11, SMARCB1/22q11, TIMP3/22q12, NF2/22q12, and MN1/22q12 were also detected. The CDKN2 C gene showed heterozygous loss, and the CDKN2A gene was affected by a small heterozygous intragenic deletion, while the CDKN2B and RB1/13q14 regions were not affected.

(a) Hematoxylin and eosin stain (40x magnification) from the first, atypical meningioma showed a highly cellular tumor with large nuclei and prominent nucleoli, and an increased mitotic count. (b) The tumor showed a varying expression pattern for EMA (20x magnification). (c) SSTR2A showed diffuse and strong membranous and cytoplasmic positivity (20x magnification). (d) MDM2 amplification resulted in MDM2 overexpression, since additional immunohistochemistry showed nuclear positivity for MDM2 (40x magnification).

a) Hematoxylin and eosin stain (40x magnification) from the recurrent, anaplastic meningioma showed a frankly malignant tumor with prominent cytological atypia, high mitotic count, and extensive necrosis. (b) The tumor showed only partial positivity for EMA (20x magnification). (c) SSTR2A showed membranous and cytoplasmic positivity in the majority of the tumor (40x magnification). (d) MDM2 amplification resulted in MDM2 overexpression, since additional immunohistochemistry showed nuclear positivity for MDM2 (40x magnification).
Dual-color fluorescence in situ hybridization (FISH) was subsequently performed on 4-μm paraffin sections using commercial MDM2 (SO)/SE12 (SG) [12q15/SE12,Kreatech] and LSI DDIT3 (DC BA) [12q13, Abbott] probes, by standard procedures. FISH also showed gross CDK4 amplification in 73% and gross MDM2 amplification in 76% of the tumor cells from the first, atypical meningioma (Figure 3).
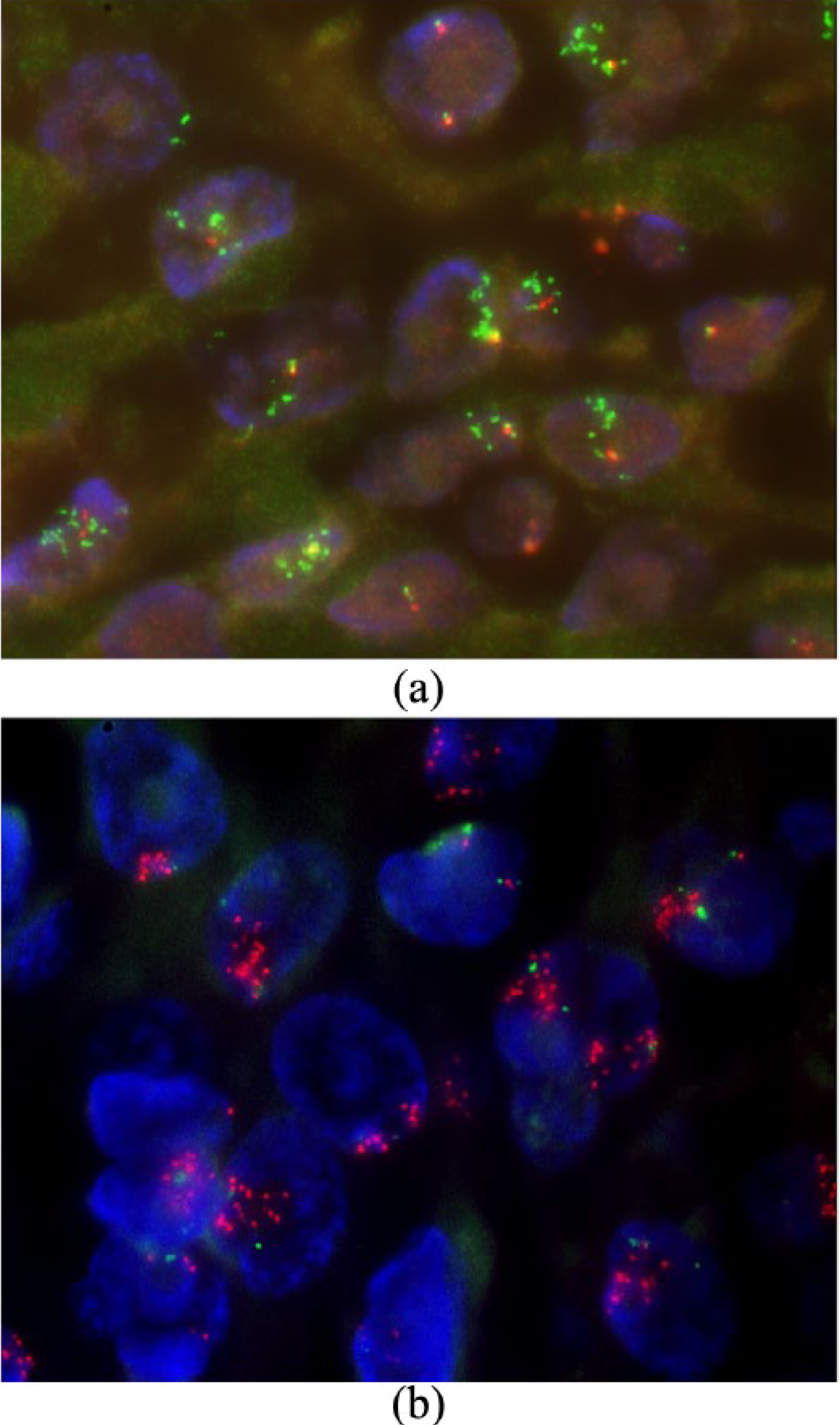
Representative FISH images from the first, atypical meningioma. (a) Amplification of CDK4 gene, as indicated by the multiple green signals from the Spectrum-Green (SG)-labeled, telomeric to DDIT3/12q13 probe (which covers CDK4 gene) in reference to the Spectrum-Orange (SO)-labeled, centromeric to DDIT3/12q13 probe. (b) Amplification of MDM2 gene, as indicated by the multiple red signals from the SO-labeled MDM2/12q14 loci-specific probe in reference to the SG-labeled chromosome 12 centromeric probe.
MDM2 amplification was shown to be accompanied by MDM2 overexpression, since additional immunohistochemistry for MDM2 showed nuclear expression in the first atypical meningioma and its recurrence, the anaplastic meningioma (Figures 1 and 2).
Discussion
Murine double minute clone 2 (MDM2) is a proto-oncogene located on chromosome 12q14.3-15, and drives cells into S-phase by direct interaction with E2 F1, and by inhibition of RB1 binding with E2 F1. 2 MDM2 is also an important negative regulator of TP53 tumor suppressor activity. 2 Levels of MDM2 and TP53 remain balanced by a negative feedback loop. 2 Since MDM2 plays an important role in controlling TP53 activity, tumors that show MDM2 amplification do not generally show TP53 mutations. 2 MDM2 overexpression by amplification is very typically described in a well-defined subset of sarcomas such as well-differentiated liposarcoma/atypical lipomatous tumor, dedifferentiated liposarcoma, intimal sarcoma, and low-grade central and parosteal osteosarcoma. MDM2 amplification is not a typical feature of meningioma, which is the most common primary brain tumor in adults. 3 While most meningiomas are benign (WHO grade I), about 20%–25% and 1%–6% of meningiomas correspond to WHO grade II and III, respectively. The latter are associated with worse clinical outcomes, and affect men more than women. 3 Atypical meningioma is associated with high recurrence rates even after complete resection, hence it is a grade II lesion.3,4 Anaplastic meningioma features frank anaplasia, and/or significantly elevated mitotic activity (≥20 mitoses per 10 HPF) and is more aggressive, corresponding to a grade III tumor. Because an anaplastic meningioma morphologically resembles carcinoma, melanoma, or high-grade sarcoma, a history of meningioma at the same site or immunohistochemical and/or genetic support is required to establish the diagnosis.3,4 Meningiomas typically express EMA, somatostatin receptor subtype 2A (SSTR2A), and progesterone receptor (PR), however, this immunoreactivity is less consistent in atypical and malignant lesions. 4 Treatment for atypical and anaplastic meningioma consists of surgical resection followed by radiation therapy. 4
Loss of heterozygosity (LOH) of NF2/22q12 region, which is the most common genetic alteration and a typical initiation event in meningioma, was detected in our case.3,4 Of note, losses of 1p, 6q, 10, 14q, and 18, and gains at 12q and 17q were described in atypical and anaplastic meningiomas as recurrent events associated with progression.3,4 Furthermore, as in most anaplastic meningiomas, the CDKN2A gene was affected in our case, showing a small heterozygous intragenic deletion.4,5 The CDKN2 C gene was also affected, showing heterozygous loss. Moreover, there was heterozygous loss of PTEN/10q23, a rarely described anomaly in anaplastic meningioma. 6 The TERT/5p15, SMO/7q32, CDKN2B/9p21, KLF4/9q31, RB1/13q14, TRAF7/16p13, and TP53/17p13 genes showed no copy number alterations.
We documented chromosome 12 and 17 chromothripsis, associated with high-level amplifications of CDK4/12q14.1 and MDM2/12q14.2. This is an unusual and exceptional finding in meningioma, with only one reported case in the literature, 1 which did not show homozygous losses of CDKN2A, p14ARF, CDKN2B, or CDKN2 C genes. Anaplastic meningiomas frequently show aberrations of CDKN2A, p14ARF, and CDKN2B genes indicating that both RB1 and TP53 pathways are affected, and that inactivation of the G1/S-phase cell cycle checkpoint is an important step for malignant progression. 5 Other members of these pathways, including CDK4 and MDM2, are rarely altered. 5 Chromosome 17 chromothripsis was in our case associated with amplification of CLTC, PTRH2, and RPS6KB1 genes on the 17q23 region. RPS6KB1 amplification has been described in anaplastic meningiomas. 7
Conclusion
In sum, we report the rare occurrence of MDM2 and CDK4 co-amplification in a recurring meningioma that evolved from a grade II to a grade III lesion. This genetic event may contribute to the pathogenesis of the malignant variant of meningioma. Awareness of this finding is indicated and should not lead to misdiagnosis of other entities that more commonly show this feature.
Technical information concerning the used antibodies for immunohistochemistry:

Supplementary Material
Supplementary Material, SupplementaryTable1WyllemanR – A rare case of atypical/anaplastic meningioma with MDM2 amplification
Supplementary Material, SupplementaryTable1WyllemanR for A rare case of atypical/anaplastic meningioma with MDM2 amplification by Robbert Wylleman, Maria Debiec-Rychter and Raf Sciot in Rare Tumors
Footnotes
Acknowledgements
R.W. and R.S. participated in conceptualizing this case report, and interpreted the hematoxylin and eosin slides and immunohistochemical results. R.W. researched the literature and wrote the first draft of the manuscript. M.D. R. analyzed, interpreted, and reviewed the cytogenetic results. All the authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval to report this case was obtained from the advisory committee of the master of specialist medicine of KU Leuven University (Approval Number/ID is mp13960).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
No written consent from the patient or a legally authorized representative was required since we retrospectively used existing, residual tissue that remained after surgical resection and diagnosis in the pathology department. Neither the patient nor his representative opposed the use of this residual tissue for scientific purposes.
Supplementary material
For a detailed aCGH result see separate document containing Supplementary Table 1.
Web references
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
