Abstract
Sarcomas are cancers that arise in soft tissues or bone and make up a small percentage of malignancies. In an effort to identify potential genetic targets for therapy, this study explores the genomic landscape of a metastatic undifferentiated pleomorphic sarcoma (UPS) with spindle cell morphology. Thick sections (50 µm) of formalin-fixed, paraffin-embedded tissue from a primary, recurrent, and metastatic tumor were collected and processed from a single patient for DNA content-based flow-sorting and analyses. Nuclei of diploid and aneuploid populations were sorted from the malignant tissues and their genomes interrogated with array comparative genomic hybridization. The third sample was highly degraded and did not contain any intact ploidy peaks in our flow assays. A 2.5N aneuploid population was identified in the primary and recurrent sample. We detected a series of shared and unique genomic aberrations in the sorted aneuploid populations. The patterns of aberrations suggest that two similar but independent clonal populations arose during the clinical history of this rare tumor. None of these aberrations were detected in the matching sorted diploid samples. The targeted regions of interest might play a role in UPS and may lead to clinical significance with further investigation.
Keywords
Introduction
Sarcomas are rare, malignant tumors that mostly occur in Caucasians older than age 50 and make up an estimated 1% of malignancies in the United States. 1 Clinically, sarcomas pose unique challenges due to poorly understood oncopathologies, over 50 described subtypes, and a diverse range of origin. 2 Sarcomas develop in bone or soft tissues that surround, connect, or support other structures and organs.3,4 Furthermore, it is possible for affected cells to become less differentiated and revert to a state that contain a population of varying morphological cell types (such as spindle, histiocyte, or giant). 5
When this occurs, the resultant diagnosis is undifferentiated pleomorphic sarcoma (UPS) as there is a lack of defined characteristics for a specific sarcoma subtype. 2 UPS has not been well characterized and little is known about both the molecular evolution and genomics of this rare cancer.
Historically referred to as malignant fibrous histiocytoma, in 2002 the World Health Organization (WHO) re-defined this as UPS. UPS now accounts for 5% of all malignant sarcoma incidences (30 cases per million) since the new classification was defined.3,6 UPS most commonly originates in the trunk or extremities (46%) and rarely in the head and neck region (3%).6–8 UPS of the head and neck have a poorer five-year survival (48%) compared to those with UPS of the extremities or trunk (77%). 8 One-third of patients diagnosed with UPS develop metastasis, and these occur more often in the lung, bone, and liver, but rarely in the regions identified in this study. 9
This study investigates a case of UPS in the left maxillary sinus that metastasized to the left jugulodigastric lymph nodes, gluteus muscle, and sacral bone. The aim was to examine the genomic landscape of sarcoma in a patient developing metastatic UPS and to identify genomic aberrations that may play a role in the development of this rare cancer to potentially be considered as candidate for therapeutic targets.
Materials and Methods
A healthy 33-year-old man presented with left-sided facial pain and was ultimately diagnosed with UPS with spindle cell morphology of the left maxillary sinus. A board-certified pathologist confirmed the diagnosis. The patient denied tobacco use and reported occasional alcohol use. Family history was significant for a grandfather with gastrointestinal cancer and mother with an unspecified bone cancer.
An initial debulking procedure was performed to remove the 5.1×4.2 cm tumor that was identified by computed tomography (CT) scan (UPS1) and post-operative intensity-modulated radiation therapy (IMRT) at a total dose of 6000 cGy followed (Figure 1).
Computed tomography images of undifferentiated pleomorphic sarcoma. The left panel depicts a coronal slice image of the left maxillary sinus primary mass. The right panel depicts the left gluteal metastasis. Red arrows point to the tumor.
Three months later, an MRI showed disease progression with tumor extending throughout the maxillary sinus and into the skull base. Another debulking procedure of the left maxillary sinus was performed (UPS2) followed by IMRT to a total dose of 4140 cGy.
Fifteen months later, metastatic disease was identified in the left jugulodigastric lymph nodes, gluteus muscle, and sacral bone. A biopsy of the left gluteal muscle confirmed metastatic disease (UPS3). The patient succumbed to the disease eight months after metastatic disease was confirmed.
Tissues were obtained with approval of the local institutional review board [Scottsdale Healthcare, Scottsdale, AZ (SHC)] before any analysis was conducted. Three samples were collected from the patient at varying time points during treatment. Treatment and follow-up care was delivered at SHC. The first sample (UPS1) was collected in May 2004 and was taken from the primary tumor located in the left maxillary sinus. The tumor was debulked and was formalin-fixed and paraffin-embedded (FFPE). Three months later in August 2004, the tumor recurred locally and was excised and FFPE archived (UPS2). A third sample (UPS3) was collected as a needle biopsy from metastatic tumor in the left gluteus muscle fifteen months later in November 2005, but was omitted from further the study due to extensive tissue degradation.
Fifty µm sections were de-waxed prior to isolation of nuclei from each sample using established protocols. Briefly, each section was washed three times for 5 min with 1 mL Xylene then rehydrated in sequential ethanol washes (100% 5 min ×2, then 95%, 70%, 50% and 30% ethanol). Samples were suspended in 1 mM EDTA pH 8.0 then incubated at 95°C to remove cross links. Samples were then digested overnight (6–17 hours) in 1 mL of a freshly prepared enzymatic cocktail containing 50 U/mL of collagenase type 3, 80 U/mL of purified collagenase, and 100 U/mL of hyaluronidase in PBS ph 7.4/0.5 mM CaCl2 buffer. Nuclei were disaggregated in NST buffer then filtered through 40 um mesh, stained with DAPI prior to flow sorting with an Influx cytometer (Becton-Dickinson, CA) with UV excitation and DAPI emission collected at >450 nm. DNA content and cell cycle analysis was performed using the software program MultiCycle (Phoenix Flow Systems, CA).
DNAs from diploid and aneuploid populations sorted from biopsies UPS1 and UPS2 were extracted using Qiagen micro kit (CA). For hybridization, DNA from 50,000 sorted nuclei and genomic DNA of pooled commercial 46, XX reference (Promega, Wisconsin) was digested with DNaseI and labeled with Cy-5 dUTP and Cy-3 dUTP, respectively, using a BioPrime labeling kit (Invitrogen). All labeling reactions were assessed using a Nanodrop assay before mixing and hybridization to 400,000 feature CGH arrays (Agilent Technologies, California). The aCGH data was assessed with a series of QC metrics then analyzed using an aberration detection algorithm (ADM2).
Ethical considerations
The study was approved by the Scottsdale Healthcare institutional review board (IRB) under Exemption 4 of Title 45 Code of Federal Regulations (CFR) concerning retrospective study of existing data. Patient consent is not required under this exemption and was not obtained for this study, as Title 45 CFR Part 46 does not apply. All lung tumor tissues and images were analyzed in this study were retrospectively obtained after prior approval of the IRB.
Results
We identified a 2.5N aneuploid population in biopsies from UPS1 and UPS2 (Figure 2). The histograms of the UPS3 biopsy showed high levels of subcellular debris consistent with excessive tissue degradation. Thus, we did not detect or sort any intact diploid or aneuploid populations in the UPS3 biopsy.
UPS1 (first sample) and UPS2 (second sample) clone comparison. This figure illustrates the presence of unique and shared genetic aberrations between the two populations. A) and B) provide a comparison between the histograms that were generated from flow-sort of each sample. C) and D) indicate which focal aberrations are present in UPS1 and UPS2 respectively and if they are unique or shared between both sample populations.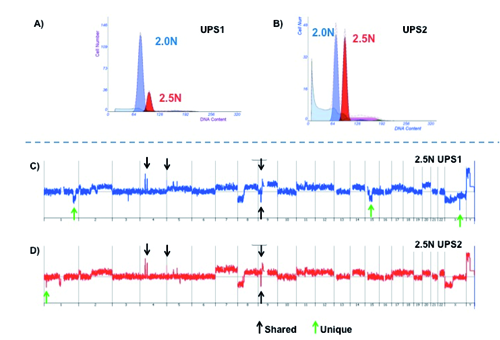
A series of regions targeted by focal copy number aberrations were identified in the 2.5N populations [Amplified: 4q12 (LNX1), 4p14 (UBE2K), 5p15.33 (hTERT)] [Deleted: 9p21.3 (CDKN2A), Xq23 (TRPC5*), 1p36.13 (PAX7)]. In addition, we detected an interstitial 1q32.2-q42.13 deletion with a breakpoints located at the TAF5L locus (q42.13) and HHAT* (q32.2). This latter deletion, the one targeting TRPC5, and a second interstitial deletion at 15q14-q21.3 were unique to UPS1. Two of the deletions (CDKN2A, TRPC5) were homozygous based on a rigorous cut off of log2ratio <−3.0 in our analysis. The presence of these shared and unique focal deletions seen in UPS1 and UPS2 suggests that the 2.5N populations represent two distinct clonal populations that arose during the clinical history of this tumor.
Discussion
Synopsis of key findings
We report our evaluation of the genomic landscape in a case of metastatic UPS of the head and neck. Gene targets that have been previously associated with cancer were identified and could influence UPS carcinogenesis and progression. Previous reports have shown that genetic amplifications and deletions have a direct effect on protein expression, which can result in cancerous phenotypes when cancer-promoting regions are affected by these copy number changes.10–13
Through analysis of aCGH results for targeted regions, it appears that the recurrent tumor was a related but independent clone with similar characteristics to the primary tumor. Evidence of this claim is fortified by the loss of three loci (interstitial 1q32.2-q42.13 and 15q14-q21.3, and the homozygous deletion of TRPC5) in UPS1 that were not detected in UPS2. It is unlikely that genomic regions that have been deleted to reinstate themselves back into the genome once lost, thus a reasonable explanation is that an independent but similar clone developed when the tumor locally recurred. Furthermore, the aberrations that did persist between UPS1 and UPS2 seem to have particularly important roles in the development and progression of this UPS.
Target loci functions and relevance to undifferentiated pleomorphic sarcoma/cancer
The ligand of Numb protein X or E3 ubiquitin-protein ligase LNX (LNX1) is suggested to encode a scaffold for a multi-protein complex that interacts with the protein numb homolog (NUMB), which is required for embryonic neurogenesis and maintenance of neural progenitor cells. This LNX1 complex mediates ubiquitination and targets specific isoforms of NUMB for degradation. Studies pertaining to LNX1 show that alterations of this gene are often found in glial tumors, but not much work has been done to show alterations/expression in other tissue types. 14 Therefore, future investigation may show that NUMB does not only regulate neural progenitor cells. Amplification of LNX1 may lead to an increase of NUMB degradation and higher susceptibility to unstable regulation of progenitor cells in other tissues such as mesenchyme that lead to soft tissue sarcomas like UPS.
Further supporting favorable conditions for tumor formation, the ubiquitin-conjugating enzyme E2K (UBE2K aka Hip2) gene encodes the UBE2K protein, which is involved in the cell cycle and significantly suppresses apoptosis by promoting proteasomal degradation of the proapoptotic molecule second mitochondria-derived activator of caspase (Smac/DIABLO). Normally Smac inhibits caspase activity by interacting with members of the inhibition of apoptosis (IAP) family, but when UBE2K is present, Smac is destabilized, suggesting that UBE2K can regulate Smac-mediated apoptosis. 15
Hedgehog acyltransferase (HHAT) has been mostly studied in melanoma and is being evaluated as a target for tumor vaccine therapy. 16 HHAT catalyzes the palmitoylation of sonic hedgehog protein (SHH), which is required for effective SHH pathway signaling. SHH signaling is essential for many processes such as cellular differentiation, growth, and embryogenesis. Furthermore, SHH has been seen to have a role in the formation and progression of many cancers (liver, pancreatic, prostate, etc.). 17
Transient receptor potential cation channel, subfamily C, member 5 (TRPC5) belongs to the transient receptor family that is thought to form a calcium permeant cation channel. This protein can function alone and also has the ability to form a heteromultimeric assembly with other related proteins. 18 Additionally, TRPC5 and other members of this family have been observed to be involved with abnormal proliferation, differentiation, and growth in cancer when deleted. 19
Paired box (PAX) genes comprise a gene family that encodes transcription factors essential for development. These genes are most active during embryogenesis, regulating cellular differentiation, tissue formation, and proliferation, but re-expression can occur later in life. The PAX gene family is classified into four subgroups, and groups II and III expression has been seen in a variety of cancer types. The PAX gene targeted in this study (PAX7), has been associated with melanoma, lung cancer, as well as, with multiple sarcomas. Furthermore, PAX7 (PAX3 can as well) can form chimeric genes with FKHR and is strongly associated with another sarcoma called alveolar rhabdomyosarcoma. It should also be noted that PAX7-mutant mice develop abnormalities involving the maxilla and nose, but no additional studies have been done to relate these findings to human UPS in the maxillary sinus. 20
Cyclin-dependent kinase inhibitor 2A (CDKN2A, p16) is a well-known tumor-suppressor that is often mutated or deleted in many cancer types. Deletion of CDKN2A has also been identified in other cases of UPS, a broad range of sarcomas, as well as, in many other cancers. 21 A deletion of CDKN2A leads to a loss of cell cycle regulation and can result in tumorigenesis.
Telomerase reverse transcriptase (TERT) is a highly essential ribonucleoprotein polymerase that maintains telomere length by adding a repeated TTAGGG sequence known as the telomerase RNA component (TERC). TERT is usually not active in adult somatic cells, which leads to telomere shortening that occurs progressively after each round of DNA replication. Furthermore, when telomeres become to short, DNA damage response signals are released to trigger senescence and apoptosis. As discussed previously, amplification of a genomic region also suggests increased expression, and the overexpression of TERT has thought to be involved in oncogenesis and cellular immortalization due to the prevention of telomere shortening. 22
Amplification of TERT is thought to occur through the homozygous deletion of CDKN2A (p16) seen in both UPS1 and UPS, which is an upstream suppressor of TERT and provides an explanation for a proposed pathway leading to cellular immortalization and progression to sarcomagenesis for this instance of UPS. Normally, the famed tumor suppressor p53 induces the transcription of cyclin-dependent kinase inhibitor 1A (CDKN1A aka p21), which binds to CDKN2A and sets off a cascade that suppresses TERT transcription (Figures 3–5).23,24 Due to the homozygous deletion of CDKN2A, the CDKN1A/CDKN2A complex, as well as, an alternative p53/p73/CDKN2A transcriptional inhibition complex of TERT cannot form and effectively suppress TERT expression. 25 There are other transcriptional and post-translational mechanisms for TERT inhibition but it is believed by our group that the loss of these two complexes in combination with TERT amplification is enough for TERT to play a role in cellular immortalization and work in conjunction with other cancer-promoting factors leading to sarcomagenesis.
Telomerase complex regulation of cellular senescence and apoptosis. This figure illustrates how telomerase complex regulation is involved in cellular senescence and apoptosis. Figure derived using the Functional Ontology Enrichment Tool in MetaCore from GeneGo, Inc. (v6.8; Thomson Reuters Business, Philadelphia, PA).
Telomerase complex regulation of telomere maintenance and cellular immortalization. This figure illustrates how telomerase complex regulation is involved in telomere maintenance and cellular immortalization. Figure derived using the Functional Ontology Enrichment Tool in MetaCore from GeneGo, Inc. (v6.8; Thomson Reuters Business, Philadelphia, PA).

Conclusions
This case of UPS has cancer-promoting genomic aberrations that have been observed in other cancers. Further investigation may provide additional information on the frequency of these gene targets. This study provides the first instance of identifying genomic alterations that potentially play an active role in a sarcoma that is both rare and relatively unexplored in basic research.
Consent
All tissues analyzed in this study were retrospectively obtained after prior approval of the Scottsdale Healthcare IRB (#2010-025) under Exemption 4 of Title 45 Code of Federal Regulations (CFR) concerning retrospective study of existing data. Patient consent is not required under this exemption and was not obtained for this study, as Title 45 CFR Part 46 does not apply. The clinical information associated with these specimens is not individually identifiable, and was collected in such a manner that the subject cannot be identified either directly or indirectly through identifiers linked to the subject.
Footnotes
Acknowledgements:
we would like to thank Kelly Sheff, Irene Cherni, Shilpi Arora, Brock Armstrong, and Aaron Fowler for technical assistance. IBIS Foundation of Arizona for funding support. Ethical approval: Scottsdale Healthcare IRB (#2010-025).
