Abstract
Paclitaxel (P) and bevacizumab (B) are agents that provide clinical benefit in advanced angiosarcoma (AS). The objective of this study was to assess the efficacy and safety of P-B in two different scheduled regimens. Patients were to receive P 200mg/m2 IV with B 15mg/kg IV every 21 days (Regimen A) or P 90mg/m2 IV weekly D1, 8, 15 with B 15mg/kg IV D1 of a 28 day cycle (Regimen B) x6 cycles. Maintenance B followed at a dose of 15 mg/kg intravenously once every 21 days. The primary end point was 4 month non-progression rate (NPR). A total of 16 patients were enrolled. 4 month NPR was 62.5% with median overall survival 16 months and median progression free survival 5.06 months. 11 patients made it to cycle 3 and were evaluable for response with 1 CR (9%), 4 PR (36%), 2 SD (18%), and 6 PD (36%). There were ten grade 3 toxicities and four grade 4 toxicities. The breakdown between the two regimens revealed comparable efficacy and safety. Paclitaxel and Bevacizumab is an active regimen in angiosarcoma. Q3 week and weekly paclitaxel appear similar in efficacy and safety.
Background
Angiosarcoma (AS) is a rare tumor type arising from blood or lymphatic vessels, comprising approximately 1%–2% of all soft tissue sarcomas. 1 The most common sites of origin are in the head and neck, accounting for around 62% of cutaneous ASs, 2 while other locations include the breast, trunk, and visceral tissues. They can arise sporadically but also can be associated with previous radiation therapy, with breast cancer–related treatment being the most common.3,4ASs possess an aggressive behavior and outcomes tend to be poor with 5-year survival rates ranging between 30% and 50%.2,5,6
Phase II trials have shown that paclitaxel7–9 and bevacizumab 10 individually has efficacy in ASs. Paclitaxel is an anti-microtubule agent that hyperstabilizes assembled microtubules and prevents their depolymerization 11 which subsequently leads to inhibition of the dynamic reorganization of the microtubule network and halts mitosis. 12 Bevacizumab is a humanized monoclonal antibody against vascular endothelial growth factor (VEGF) which has been shown to be highly expressed in ASs. 13 We conducted a multi-center phase II trial (NCT01055028) to assess the efficacy and toxicity of combination paclitaxel and bevacizumab therapy. We tested two different regimens of paclitaxel, a q3 week versus weekly administration.
Patients and methods
Patient eligibility
Patients aged ≥18 years with histologically confirmed, unresectable, locally advanced or metastatic AS were eligible for this study. Patients must have had ≤2 prior chemotherapeutic regimens for AS which did not include paclitaxel, docetaxel, or bevacizumab. The other eligibility criteria included an ECOG (The Eastern Cooperative Oncology Group) performance status 0–2, adequate organ function including a left ventricular ejection fraction >50%, serum creatinine ≤2.0, total bilirubin <2.0 × upper limits of normal (ULN), serum glutamic-oxaloacetic transaminase (SGOT) (aspartate transaminase (AST)) <2 × ULN (if documented hepatic involvement, total bilirubin can be <3 × ULN and AST <5 × ULN), and absolute neutrophil count (ANC) ≥ 1500/mm3. Exclusion criteria included an active infection, inadequately controlled hypertension (defined as systolic blood pressure >150 mmHg and/or diastolic blood pressure >100 mmHg), prior history of hypertensive crisis or hypertensive encephalopathy, New York Heart Association (NYHA) Grade II or greater congestive heart failure, history of myocardial infarction or unstable angina, stroke or transient ischemic attack within 6 months prior to day 1, and known central nervous system (CNS) disease, except for treated brain metastasis. The protocol was approved by the Institutional Review Boards (IRB) at Stanford Cancer Institute (SCI), MD Anderson Cancer Center (MDACC), and Sarcoma Oncology Center (SOC) IRBs. All patients signed an informed consent.
Study design
Two regimens were administered. (Regimen A): paclitaxel 200 mg/m2 intravenously over 3 h followed by bevacizumab 15 mg/kg intravenously (cycle 1: 90 min; cycle 2: 60 min; cycles 3–6: 30 min) every 21 days ×6 cycles or (Regimen B): paclitaxel 90 mg/m2 D1, 8, 15 with bevacizumab 15 mg/kg intravenously D1 of a 28-day ×6 cycles. Maintenance bevacizumab (MB) started after the completion of combination of paclitaxel and bevacizumab and it was given at a dose of 15 mg/kg intravenously once every 21 days for a maximum of eight cycles. Patients were allowed to receive growth factors. Dose reductions were done based on hematologic and non-hematologic toxicities based on NCI Common Terminology Criteria for Adverse Events (CTCAE) version 3.0.
Imaging was performed with computed tomography (CT) or magnetic resonance imaging (MRI) at baseline, after cycles 3, 6, MB 4, and MB 8. Patients were followed up until progression with scans every 3 months the first year, every 4 months the second year, and every 6 months years 3, 4, and 5. Measurements were based on RECIST 1.1 (response criteria in solid tumors).
Statistical considerations
The primary objective of this study was to evaluate the 4-month non-progression rate (NPR). The secondary objectives included best response after the third and sixth cycles, median progression-free survival (mPFS), median overall survival (mOS), 6-month and 12-month OS rate, and toxicity (according to the National Cancer Institute Common Toxicity Criteria). Time to event data were summarized using Kaplan–Meier methods. Best response was assessed among patients who received at least three cycles of therapy; OS was defined as time of treatment start to date of death or date of last follow-up, PFS was defined as time of treatment start to date of disease progression or death, or censored for toxicity or last follow-up.
Results
Patient characteristics
A total of 16 patients were enrolled between June 2010 and September 2013 (SCI

Flow diagram of patient enrollment, intervention, follow-up, and data analysis.
Patient baseline characteristics.

Efficacy
The response rates and other efficacy criteria are summarized in Table 2. As an aggregate of both regimens, the 4-month NPR was 62.5% (95% confidence interval (CI): 42.8%–91.4%, regimen A: 50%, regimen B: 67%). For assessment of best response, 11 patients completed at least three cycles of therapy and were thus assessable by imaging. There was one complete response (CR) (9%), four partial response (PR) (36%), two stable disease (SD) (18%), and four progressive disease (PD) (36%) for a best response rate of 5/11 (45%). Of the five who did not make it to cycle 3, three patients died of disease progression prior to imaging (early deaths), one patient switched to taxol monotherapy until surgical resection, and one patient due to decline in functional status. The mOS was 16 months (range: 2.7–30.4 months) and the mPFS was 5.06 months (range: 0.7–12.9 months). The 6- and 12-month OS rate was 87.5% and 74.5%, respectively. A Kaplan–Meier curve of both OS and PFS is displayed in Figure 2. The mean number of chemotherapy cycles was 3.6 and four patients eventually underwent MB. In terms of differences between regimens A and B, the efficacies were comparable with relatively similar 4-month NPR (50% vs 67%), best response rate (50% vs 40%), mean number of chemotherapy cycles (4 vs 3.25), mOS (11.1 vs >13.6 months), and mPFS (3.35 vs 5.39 months).
Treatment Response by Study Group.

CI: confidence interval; PB: Paclitaxel/Bevacizumab; OS: overall survival; PFS: progression-free survival.
Of the five patients who did not make it to cycle 3, three of them died of disease prior to imaging, one of them discontinued drug due to drug fevers, and the other dropped out of the study due to decline in functional status.

Kaplan–Meier curve of overall survival and progression-free survival of the cohort.
Toxicity
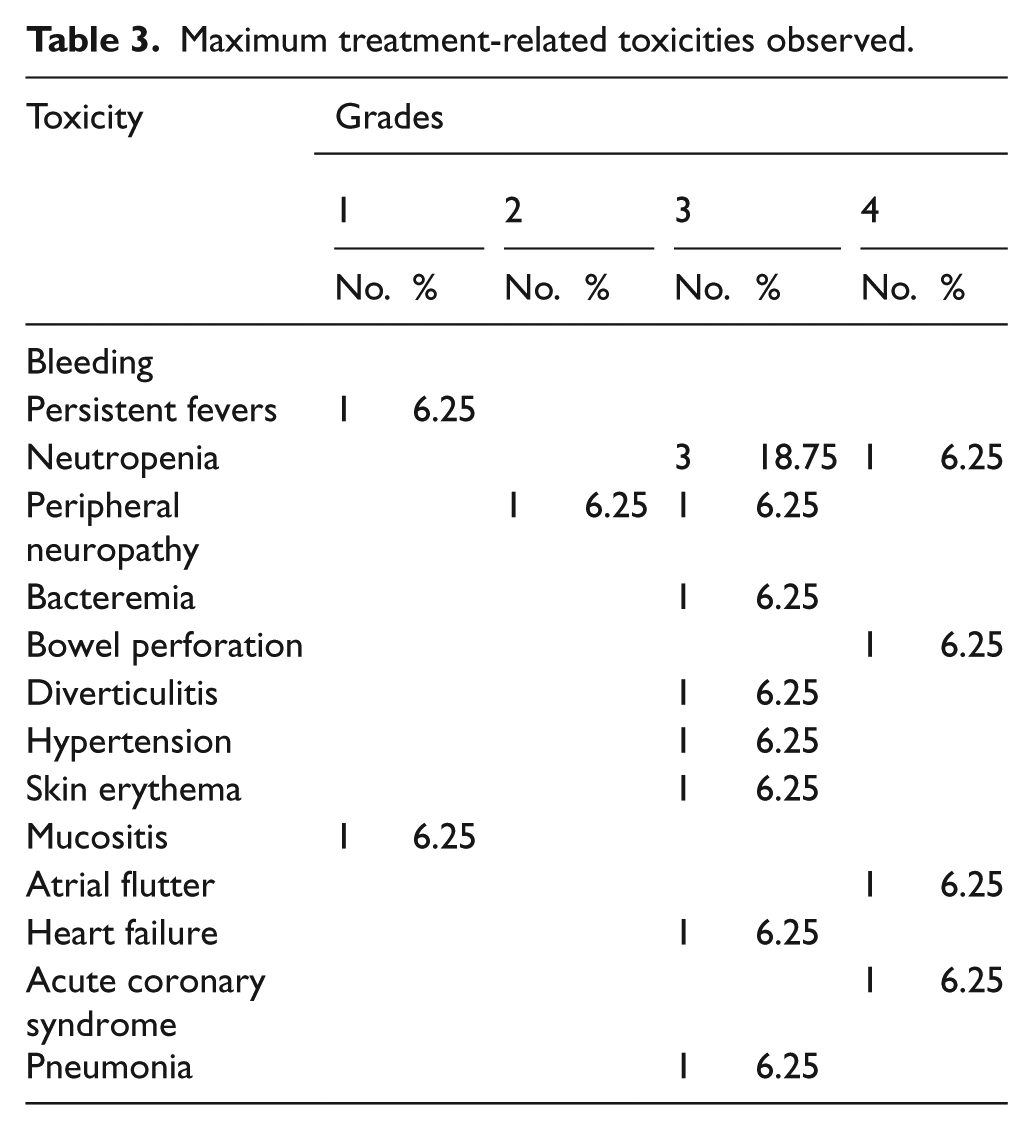
All toxicities are noted in Table 3. There were seven patients who had one or more grade 3 toxicities and two patients with one or more grade 4 toxicities. There were no grade 5 toxicities. The most common grade 3 or 4 toxicity was neutropenia (4/10) with three occurring in regimen A and one occurring in regimen B. Other grade 4 toxicities were neutropenia, bowel perforation, atrial flutter, and acute coronary syndrome. Three patients discontinued treatment due to drug toxicity. One patient quit after grade 1 drug fevers and went on to receive radiation and gemcitabine/docetaxel. Another had grade 3 neuropathy prior to cycle 6 so the final cycle was omitted and the patient moved on to MB. The last patient had grade 3 skin erythema, skin mucositis, and decrease in performance status and transitioned to gemcitabine/docetaxel. Between regimens A and B, there were an equal amount of total grade 3/4 toxicities.
Maximum treatment-related toxicities observed.
Discussion
We report the results of a multicenter phase II clinical trial using a combination of paclitaxel and bevacizumab for the treatment of advanced or metastatic AS. In the world of sarcoma research, assessing histology-specific treatment effectiveness can be difficult as the rarity of sarcoma lends itself to heterogeneous study populations. One of the strengths of this trial is that we evaluated a combination of two drugs with individual AS-specific effectiveness, in a single histology cohort. We also were able to evaluate two different dosing regimens of paclitaxel, which are both widely used in the current environment. This study shows that the combination of paclitaxel and bevacizumab is effective. The 4-month NPR was 62.5% (95% CI: 42.8%–91.4%) with an mOS of 16 months and an mPFS of 5.06 months. Toxicity was manageable although serious adverse events (SAEs) did include heart failure, acute coronary syndrome and atrial flutter, and bowel perforation. The two dosing schedules of paclitaxel, q3 week and weekly, were found to be comparable in terms of efficacy and toxicity, albeit with small sample size.
Previous studies have indicated that paclitaxel7–9 and bevacizumab 10 are individually effective in AS. In the ANGIOTAX study, Penel et al. 7 treated 30 advanced/metastatic AS patients with weekly paclitaxel and found a 4-month PFS of 45% with mOS of 8 months. Agulnik et al. treated 30 patients with advanced AS and epithelioid hemangioendothelioma with single agent bevacizumab found PRs in 13%, SD in 50%, and an mPFS of 12.4 weeks. In 2015, Ray-Coquard et al. 14 published a randomized phase II trial of combination bevacizumab and paclitaxel versus single agent paclitaxel alone in 50 patients. Although both arms were active, there was no additional benefit of adding bevacizumab, with similar 6-month PFS (54% vs 57%), mPFS (6.6 vs 6.6 months), and mOS (15.9 vs 19.5 months) for combination versus single agent, respectively. In addition, there were 10 drug-related SAEs in the bevacizumab + paclitaxel arm that were reported in eight patients, including one fatal grade 5 intestinal occlusion. The results from our trial showed that combination therapy was similarly active with mPFS of 5.06 months and mOS of 16 months. However, there were also SAEs in three patients including heart failure, acute coronary syndrome and atrial flutter, and colon perforation. We did not include a comparator arm so it is difficult to qualify or quantify the benefit of adding bevacizumab. This was an important distinction in the Ray-Coquard trial, as the single agent paclitaxel arm showed substantial improvement over the previous single agent paclitaxel study (Penel et al.) with an mPFS of 6.6 versus 3.8 months and an mOS of 19.5 vs 8.3 months despite using the same drug with the same schedule. This was discussed to be likely due to the heterogeneous population of ASs as well as the fact that the Ray-Coquard trial had more strict inclusion criteria and thus likely recruited a baseline healthier study population.
This study has several limitations. First, the sample size was small (
Soft tissue sarcomas are a heterogeneous group; however to date, most of the large therapeutic trials pool together different histologies due to the rarity of disease. Although this has proven effective for certain chemotherapy regimens such as doxorubicin15–18 or gemcitabine/docetaxel based,19–22 there is clear evidence that there is a benefit for histology-specific treatment. This has been evidenced by the identification and targeting of cKIT in gastrointestinal stromal tumor (GIST),23,24 CSF1R (colony stimulating factor 1 receptor) in tenosynovial giant cell tumor,25,26 mTORC1 (mammalian target of rapamycin complex 1) in perivascular epithelioid cell tumors (PEComas),27,28 as well as many other examples.29–31 Recent genetic characterizations of ASs have revealed novel gene fusions (NUP160-SLC43A3) 32 as well as recurrent activating mutations (PTPRB, PLCG1, and KDR)33,34 that may be potential targets for future treatment. As primary tumor and circulating tumor DNA sequencing become more prominent in the workup of patients, we imagine greater discovery of promising therapeutic options.
In conclusion, combination paclitaxel (either q3 week or weekly) with bevacizumab is an active regimen. We did not include a comparator arm, and a previous study has suggested that there is no benefit versus single agent paclitaxel alone. 14
Footnotes
Acknowledgements
This work was presented in part at the Annual Meeting of the American Society of Clinical Oncology, Chicago, IL, 2–6 June 2017. Abstract located at ![]() . K.N.G. had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis; V.R., S.C., and K.N.G. contributed to study concept and design; N.B., N.K., V.R., M.L., and K.N.G. acquired, analyzed, or interpreted the data; N.B., N.K., V.R., S.C., and K.N.G. contributed to drafting of the manuscript; N.B., V.R., S.C., and K.N.G. made critical revision of the manuscript for important intellectual content; N.B., K.N.G. performed statistical analysis; M.L. provided administrative, technical, or material support; and V.R., S.C., and K.N.G. supervised the study.
. K.N.G. had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis; V.R., S.C., and K.N.G. contributed to study concept and design; N.B., N.K., V.R., M.L., and K.N.G. acquired, analyzed, or interpreted the data; N.B., N.K., V.R., S.C., and K.N.G. contributed to drafting of the manuscript; N.B., V.R., S.C., and K.N.G. made critical revision of the manuscript for important intellectual content; N.B., K.N.G. performed statistical analysis; M.L. provided administrative, technical, or material support; and V.R., S.C., and K.N.G. supervised the study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: N.B., N.K., and M.L. report no financial disclosures; V.R. received consulting fees from Eli Lilly, royalties from UpToDate, and research funding from Novartis; and K.G. received research funding from Genentech.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Genentech.
