Abstract
Background
Cancer care requires coordinated, multi-disciplinary teams and incurs rising costs. Time-driven activity-based costing (TDABC) calculates the costs of healthcare resources consumed along the care process. We used TDABC to evaluate the cost of selected adjuvant and palliative regimens in breast, colorectal, and lung cancer at a single institution to inform resource allocation.
Methods
Process maps of the care delivery cycle were developed using TDABC, detailing each clinic or ambulatory treatment unit visit. Resources—including personnel, equipment, and supplies—were identified at each step. Per-minute costs were generated based on time requirements, and total costs calculated by summing resource expenses per episode of care.
Results
The total cost per chemotherapy regimen varied significantly across cancer types and settings. For breast cancer, adjuvant Adriamycin-Cyclophosphamide-Paclitaxel over 24 weeks cost SGD$17,075.43, driven by manpower utilization (2.976 man-units) and chair time (4350.93 minutes). Metastatic Docetaxel, given every three weekly, cost SGD$3174.65 over 6 weeks, while weekly Paclitaxel cost SGD$6810.79 due to increased chair time (2606.57 minutes). For lung cancer, Pemetrexed-Carboplatin-Pembrolizumab cost SGD$25,711.17, versus SGD$13,992.05 for Osimertinib. Gastrointestinal regimens ranged from SGD$9548.43 for adjuvant Capecitabine-Oxaliplatin to SGD$1264.52 for palliative Capecitabine. Outpatient base costs averaged SGD$310 per visit, with additional costs driven by chemotherapy type, frequency, manpower, and adverse drug reaction management.
Conclusion
TDABC application in oncology is limited but integral to value-based care. Understanding actual costs and cost drivers in outpatient cancer care may better inform resource use during times of constraint.
Keywords
Introduction
Cancer care is a complex model, requiring multi-team coordinated care for the patients. The cost of cancer care has increased substantially over the past decade, with expenditures projected to continue rising due to novel therapies, personalized medicine, and an aging population. This necessitates a shift toward value-based care, where treatment decisions balance cost, effectiveness, and patient outcomes to optimize resource allocation in a financially sustainable manner.1–3
The COVID-19 pandemic further highlighted the need for efficient resource allocation and strategic decision-making in healthcare. Cancer patients, being particularly vulnerable due to immunosuppression from malignancy and anticancer treatments, required special consideration in healthcare prioritization. Oncology care had to be adapted by reducing clinic visits, modifying chemotherapy regimens, and optimizing care pathways to minimize exposure while ensuring treatment efficacy.4,5
In the 2020 ASCO Recommendation from the Oncology Community during the COVID-19 Pandemic, 4 it affirms that allocation decision processes should not deny patients with cancer consideration for access to scarce resources however the duty to steward limited resources is urgent to maximally benefit the greatest number of patients.5,6 This tension between equality and equity orientation of public health ethics, expressed through fair allocation of limited resources, a focus on public safety as well as patient-centred orientation of clinical ethics is repeated in The Hastings Centre publication on responding to the ethical considerations during the Coronavirus Pandemic. 7
A key gap in current cancer care models is the limited application of rigorous cost-accounting methods to quantify the real-world economic burden of treatment. Time-driven activity-based costing (TDABC) is a methodology that allows for a detailed understanding of healthcare resource utilization, enabling hospitals to make informed decisions regarding cost-effective treatment approaches. 8 This method consists of seven steps: (A) selecting a medical condition, (B) defining the clinical pathway, including the mapping of the entire care process and all key activities, (C) creating the clinical process maps included in each activity in the patient clinical pathway, incorporating all direct and indirect resources, (D) obtaining time estimates for each process for activities and resources used for each patient, (E) estimating the cost of using patient care resources, the cost of all direct and indirect resources involved in care delivery, (F) estimating the capacity of each resource and calculating the use cost rate and (G) calculating the total cost of patient care.
By implementing TDABC, healthcare institutions can transition toward value-based care by accurately measuring the cost of care and optimizing resource allocation. The ability to link cost data with clinical outcomes enables decision-makers to assess cost-benefit ratios of different treatments, ensuring that financial resources are allocated in ways that maximize patient benefit. 9 In this project, we aim to describe the use TDABC in the active engagement of health care providers in costing activities and care redesign for the ambulatory chemotherapy unit.
Methodology
National Cancer Centre Singapore (NCCS) sees close to 65% of Singapore’s public sector oncology cases. 9 Patients who require intravenous chemotherapy undergo treatment in the Ambulatory Treatment Unit (ATU) in NCCS. During the COVID-19 pandemic, a study team consisting of data-analysts, public health physicians and ambulatory chemotherapy unit staff was formed in April 2020 to review care-delivery processes between October 2019 to December 2019, over a period of 3 months prior to the COVID-19 Pandemic, providing a baseline for cost analysis before COVID-19-induced disruptions in oncology services. While this period does not capture direct pandemic-related resource constraints, it serves as a reference point for pre-pandemic cost structures, against which post-pandemic evaluations can be compared in future research. The costing analysis was conducted from the healthcare provider’s perspective, focusing on direct medical costs incurred by the institution.
Table showing the cost derived via TDABC for an individual process care map- using the care process of Adriamycin/Cyclophosphamide/Taxane (AC-T).

Although the review period was 3 months, regimen durations reported reflect the standard clinical cycles for each chemotherapy regimen. For consistency, cost calculations were based on complete regimen cycles, which range from 6 weeks for metastatic disease to 12-24 weeks for a complete adjuvant regimen, to provide an accurate assessment of total treatment costs
Diagnostic phase
Detailed process maps were created to capture the entire care pathway, encompassing initial clinic presentation, scheduling of chemotherapy, preoperative chemotherapy counselling, chemotherapy check-in, administration, and discharge. The cost inputs included staff salaries, equipment, and consumables. Additionally, the model accounted for the frequency and severity of adverse drug reactions over a 3-month period, incorporating manpower requirements to manage these complications.
Multiple techniques were employed to identify potential cost constraints, including multidisciplinary process mapping sessions to examine workflow inefficiencies, direct tracking of ambulatory treatment unit (ATU) nurses involved in patient care, and time-motion studies to assess variations in protocol execution. Additionally, audits of daily run sheets were performed to determine the incidence of adverse drug reactions and the associated costs of medical interventions and personnel allocation.
Evaluation phase
The TDABC model was applied to evaluate specific chemotherapy regimens across three major cancer types: breast, lung, and gastrointestinal cancers. For breast cancer, the methodology was used to assess the adjuvant Adriamycin-Cyclophosphamide-Paclitaxel (AC-T) regimen, as well as the metastatic treatment options of Docetaxel, Paclitaxel, and Capecitabine. In lung cancer, adjuvant treatment was evaluated for Gemcitabine and Cisplatin, while metastatic regimens included Pemetrexed-Carboplatin-Pembrolizumab, Pembrolizumab monotherapy, and Osimertinib. For gastrointestinal cancers, TDABC was applied to analyse the adjuvant regimen of Capecitabine-Oxaliplatin, along with metastatic treatments comprising Capecitabine alone, 5FU-Folinic Acid-Oxaliplatin, and Capecitabine-Oxaliplatin. All costs are reported in SGD based on 2019 price levels (Figures 1 and 2). Value added process map planning (using TDABC) for clinic visits. Value Added process map planning (Using TDABC) for Chemo Regimens – using the examples of (i) Adriamycin/Cyclophosphamide/Taxane (AC-T) (ii) Paclitaxel and (iii) Docetaxel.
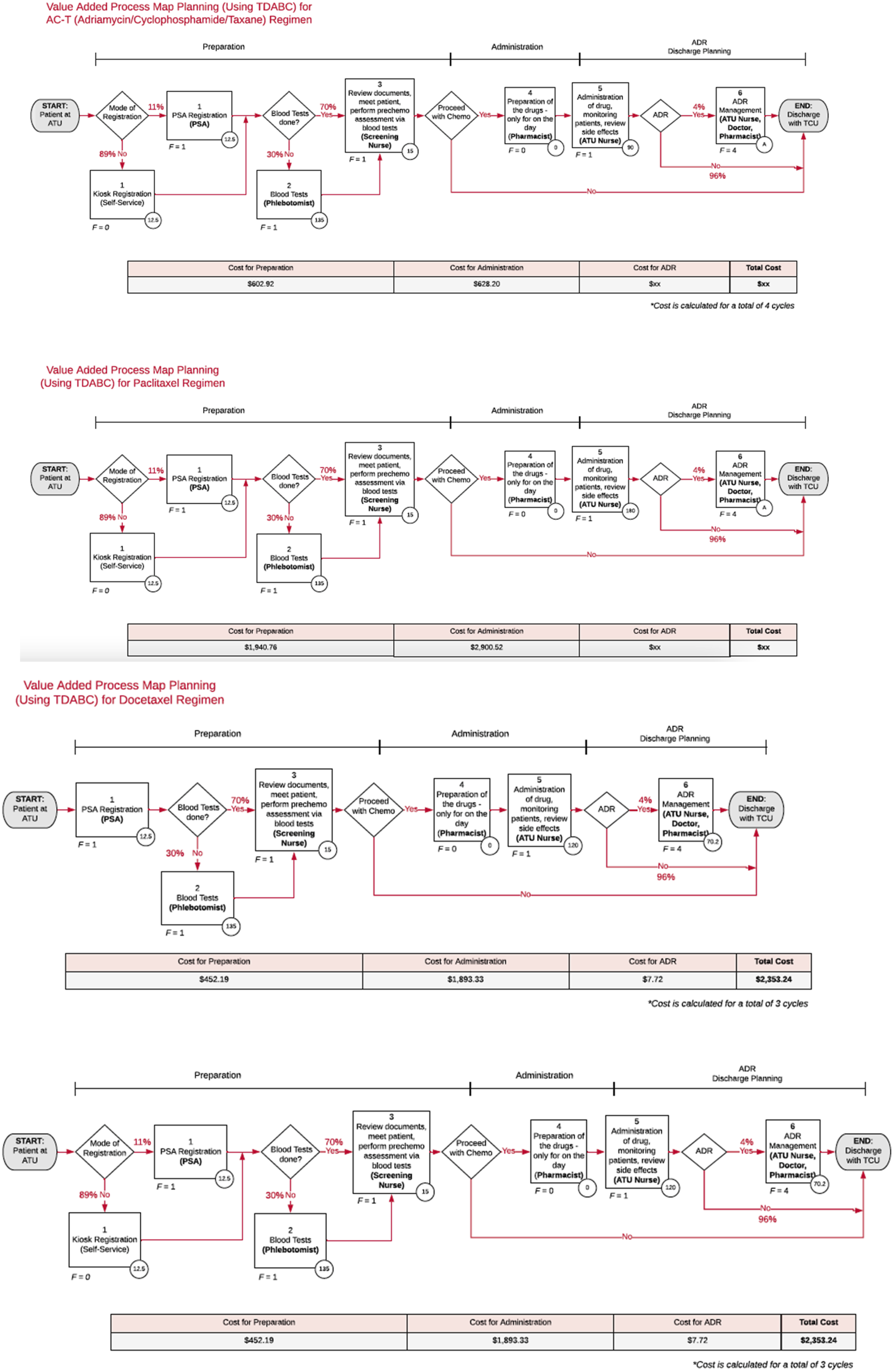
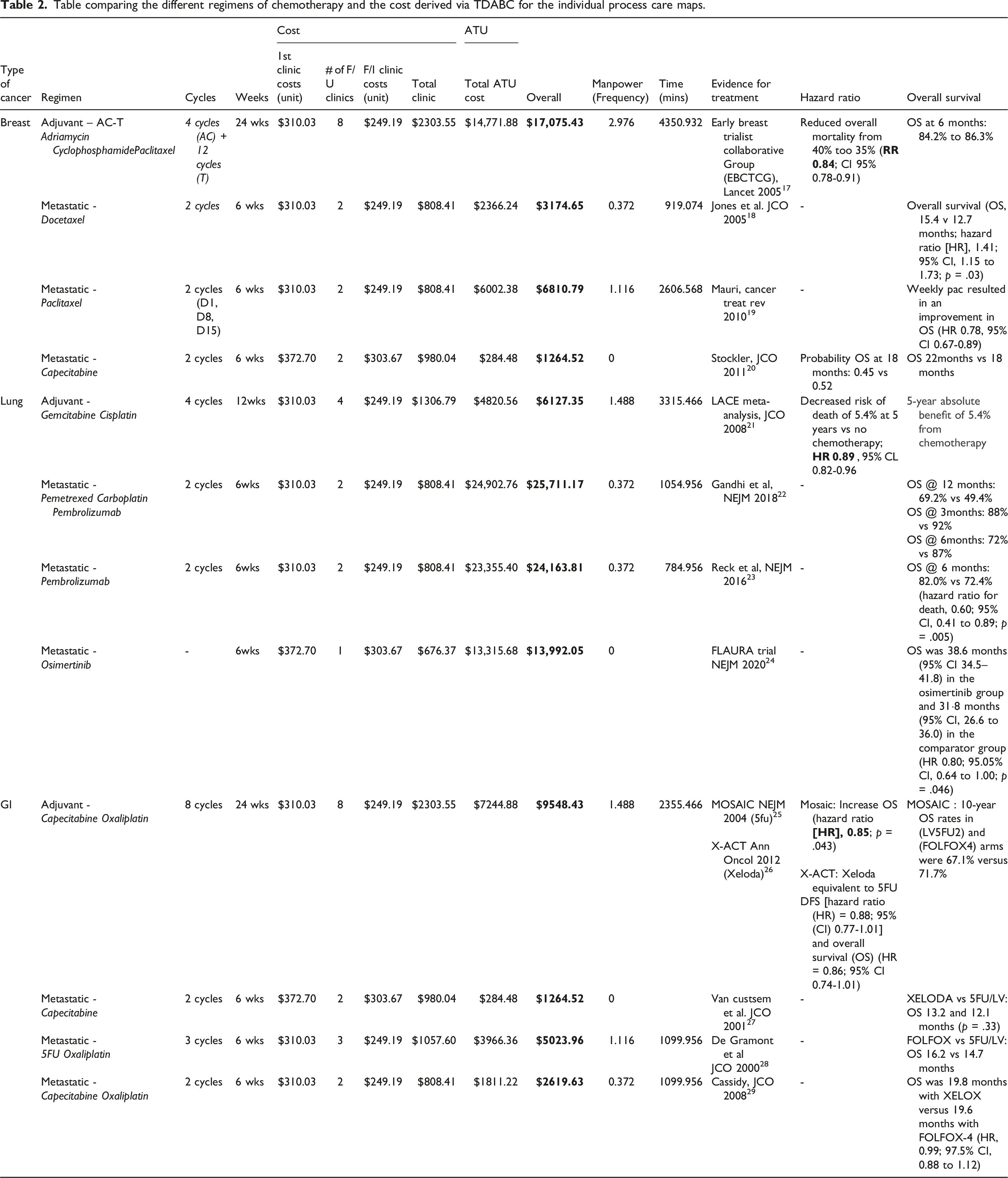
Table comparing the different regimens of chemotherapy and the cost derived via TDABC for the individual process care maps.
Results
A total of nine chemotherapy regimens across breast, lung, and gastrointestinal cancers were evaluated using TDABC, and their associated costs were compared. The total cost for each regimen was derived by summing costs related to initial consultation, follow-up clinics, ambulatory treatment unit (ATU) utilization (including manpower and chair time), and management of adverse drug reactions. Our findings highlight the variation in resource utilization and cost across different chemotherapy regimens, with oral regimens generally associated with lower manpower and infrastructure costs.
For adjuvant breast cancer treatment, the Adriamycin-Cyclophosphamide-Paclitaxel (AC-T) regimen over 24 weeks incurred a total cost of SGD $17,075.43. This cost was driven by high ATU manpower utilization (2.976 units) and a total chair time of 4350.93 minutes, reflecting the intensive nature of this regimen. In the metastatic setting, Docetaxel administered every 3 weeks was associated with a total cost of SGD $3174.65 over 6 weeks, requiring 0.372 man-units and 919.07 minutes of chair time. Comparatively, weekly Paclitaxel resulted in a higher total cost of SGD $6810.79, with greater manpower utilization (1.116 man-units) and 2606.57 minutes of chair time due to increased frequency of administration. Capecitabine, administered orally, was the most cost-efficient breast cancer regimen, with a total cost of SGD $1264.52 and minimal manpower or chair time requirements.
For lung cancer, adjuvant chemotherapy with four cycles of Gemcitabine and Cisplatin over 12 weeks resulted in a total cost of SGD $6127.35. This was attributed to moderate manpower requirements (1.488 man-units) and 3315.47 minutes of chair time. In the metastatic setting, combination therapy with Pemetrexed, Carboplatin, and Pembrolizumab over 6 weeks had the highest total cost at SGD $25,711.17, followed closely by single-agent Pembrolizumab at SGD $24,163.81. Both regimens required similar manpower utilization (0.372 man-units), though chair time was slightly lower for Pembrolizumab monotherapy (784.96 minutes) compared to the combination regimen (1054.96 minutes). Osimertinib, an oral targeted therapy, was associated with a total cost of SGD $13,992.05 and did not require additional manpower or chair time.
In gastrointestinal cancers, adjuvant treatment with Capecitabine and Oxaliplatin (XELOX) over 24 weeks incurred a total cost of SGD $9548.43, requiring 1.488 man-units and 2355.47 minutes of chair time. In the metastatic setting, oral Capecitabine was the most cost-efficient option, with a total cost of SGD $1264.52. The FOLFOX regimen (5FU, Folinic Acid, Oxaliplatin) over 6 weeks was associated with a total cost of SGD $5023.96, requiring 1.116 man-units and 1099.96 minutes of chair time. In comparison, Capecitabine and Oxaliplatin (XELOX) incurred a lower total cost of SGD $2619.63, with reduced manpower requirements (0.372 man-units) and equivalent chair time.
Discussion
This study demonstrates the feasibility and practical application of time-driven activity-based costing (TDABC) in quantifying the financial burden of various chemotherapy regimens within an ambulatory oncology setting in Singapore. Unlike top-down costing methodologies that rely on aggregate averages, TDABC offers a granular, bottom-up approach that enables precise attribution of resource utilization at each step of the care pathway.12,13 Its use in this study highlighted important variations in resource consumption and cost-efficiency across treatment regimens, supporting its utility as a tool for driving value-based oncology care.10,12
A distinguishing feature of this study is the integration of adverse drug reaction (ADR) management costs within the TDABC model, a feature less commonly observed in other oncology-focused TDABC applications. 11 By integrating ADR frequency and severity into cost estimations, we provide a more complete picture of the real-world resource burden.
Despite its strengths, TDABC remains underutilized in oncology due to several limitations. First, the dynamic evolution of cancer therapies—including antibody-drug conjugates and immunotherapy combinations—introduces new variables such as preparation complexity, toxicity management, and specialized personnel requirements.14,15 Prior studies12,16 also noted that TDABC implementation often lacks standardization and integration with clinical outcomes, limiting cross-institution comparability. Future research should embed clinical and health utility data to enhance economic evaluations.
While this study demonstrates the feasibility of applying TDABC to quantify resource utilization and costs associated with various chemotherapy regimens in an ambulatory treatment unit, several limitations should be acknowledged. Firstly, the study was conducted within a single institution, which may limit the generalizability of the findings to other healthcare settings with differing workflows, staffing models, or cost structures. Resource allocation and cost patterns observed at the National Cancer Centre Singapore may not reflect those in other institutions, particularly in healthcare systems with different reimbursement models or infrastructure capacities. Furthermore, we focused on selected regimens in breast, lung, and gastrointestinal cancers due to their prevalence, but more resource-intensive regimens—such as those used in hematologic malignancies—warrant future inclusion. These combinations require more preparation time and have increased the chances of adverse drug reactions which will also increase the cost of these care processes bother directly and indirectly, particularly with the need of more trained personnel. In addition, protocols which are time-intensive i.e. Rituximab infusion and resource-intensive protocols in the treatment of haematological malignancies were not included in this study. As with most TDABC applications, formal sensitivity analyses were not performed; future iterations should incorporate uncertainty analyses to model variation in ADR rates, drug prices, and manpower costs.
Another important limitation is the absence of direct incorporation of clinical outcomes data—such as progression-free survival, overall survival, or quality-of-life measures—into the TDABC model. Although clinical trial data were referenced to contextualize regimen efficacy, integrating real-world patient outcomes would enable more robust cost-effectiveness analyses. Future work should link cost data with patient-level clinical outcomes to support value-based decision-making and facilitate comparisons based on both economic and health outcomes.
The current care delivery model at the National Cancer Centre Singapore (NCCS), while efficient in several aspects, presents areas for further improvement. Recommendations include: (1) Standardizing time-motion tracking across regimens to ensure consistency. (2) Developing automated data capture from electronic medical records to reduce manual mapping burden. (3) Incorporating cost-utility modelling that integrates quality-adjusted life years (QALYs) or progression-free survival metrics into TDABC outputs.
Such enhancements would align Singapore’s oncology costing practices with international best practices while supporting policy shifts toward bundled payments and population-based reimbursement models.9,13
From a practical standpoint, the findings of this study offer actionable insights for hospitals and healthcare systems seeking to optimize resource allocation. By identifying cost drivers, such as high manpower utilization and chair time in specific regimens, institutions can redesign workflows, reallocate resources, or consider alternative regimens that balance clinical benefit with financial sustainability.10,11 Singapore’s healthcare system is progressively adopting bundled payment models, making cost transparency crucial for both providers and policymakers. TDABC offers a robust methodology to support this transition, as it allows for precise cost attribution across different phases of treatment.8,10,12 Future studies should incorporate computational models to predict cost variations based on adverse event frequencies, regimen modifications, and evolving treatment guidelines.
TDABC establishes a capacity cost for each resource consumed through key activities in a care process. This provides a blueprint to assist providers in making cost-conscious decisions, not just in cancer treatment but can be used across other specialities. At an institution level, it helps to analyse alternative treatments and provides transparency to help in cost reduction. From a national level, it will help policy makers in cost-efficient interventions.9,13 A multi-disciplinary, value-added framework in the cancer care incorporating TDABC will provider oncologists, insurance payers and institutional leaders the tools to provide value-added care at the institutional level and beyond.
Footnotes
Acknowledgments
The authors would like to thank National Cancer Centre Singapore for the support.
Author contributions
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
