Abstract
Unicentric Castleman disease (UCD) is a rare benign lymphoid proliferative disease having solitary or multiple lesions in adjacent organs, limited to the same lymphoid region. Among all the affected stations, mesenteric localization of UCD is very rare and often misdiagnosed due to the absence of typical clinical and imaging features, making preoperative diagnosis difficult. Complete surgical resection remains the standard of care, which shows curative intent in 90% of patients. We report a rare case of an adolescent male with abdominal UCD who presented with intermittent abdominal pain, vomiting, and a firm, mobile lump in the right lower abdomen. Hematological parameters were within normal limits. Ultrasound raised suspicion of mesenteric mass in the right paraumbilical region which on contrast-enhanced CT scan was suggestive of benign mesenteric mass/desmoid tumor. Intraoperatively, a 10 × 9 cm mesenteric mass was noted in the right paraumbilical region attached to root of mesentery along with an inflamed appendix. Excision of the mesenteric mass with appendectomy was done. Histopathology was suggestive of Angio Follicular Lymphoid Hyperplasia (Castleman disease) and inflamed appendix. Castleman disease is an unusual medical case that poses a diagnostic challenge and it must be included in the differential diagnoses of solid abdominal mass. Though multiple treatment options are available, surgical resection is still considered the standard treatment of choice due to its diagnostic and therapeutic advantages.
Introduction
Castleman disease (CD) is a rare, benign lymphoid proliferative lesion, clinically classified into unicentric CD (UCD) and multicentric CD (MCD).1,2 UCD refers to solitary or multiple lesions in adjacent organs, limited to the same lymphoid region. MCD refers to multiple lesions involving more than two non-adjacent lymphoid regions. UCD involves usually the chest (30%), neck (23%), abdomen (20%), and retroperitoneal (17%). 3 Most patients have no specific clinical symptoms or typical imaging features. The diagnosis of UCD is generally achieved with histological and immunohistochemical findings after surgical resection. 4 Complete surgical resection is the standard treatment of UCD. 5 We herein report a rare presentation of abdominal UCD posing a diagnostic challenge.
Case report
An 18 year male presented to the surgical outpatient department with the complaint of a lump in the right lower abdomen for 3 years. It was insidious onset, gradually progressive, associated with on and off vomiting and mild pain. There was no history of alteration of the bladder, bowel habits, bleeding per rectum, or obstructive symptoms. There were no known comorbidities or significant past surgical history. There was no cervical, axillary, or inguinal lymphadenopathy. Physical examination revealed a soft abdomen, no local rise of temperature, a single 6 × 8 cm intrabdominal lump palpable in the right para umbilical and lumbar region, mildly tender with well-defined margins, firm in consistency, mobile, and no evidence of free fluid clinically, bowel sounds were heard. No features of obstruction or peritonitis. Clinical differential diagnoses of an ileal gastrointestinal stromal tumor, mesenteric cyst, desmoid tumor, or lymphoma were made.
Routine blood parameters were normal. Ultrasound imaging showed a well-defined heterogeneous mass of 59.9 mm × 98.7 mm in the right lumbar and paraumbilical region with internal vascularity suggestive of a mesenteric mass. Contrast-enhanced CT Abdomen revealed a well-marginated soft-tissue attenuation solid enhancing mass of 10 x 7.3 x 7.0 cm in the right para umbilical and lumbar region showing internal calcification and inhomogeneous enhancement displacing adjacent bowel loops suggestive of a benign mesenteric mass or a desmoid tumor (Figure 1). There was no evidence of any similar lump elsewhere clinically and radiologically. After informed consent, an exploratory laparotomy was planned, and intraoperatively, a 10 × 9 cm mass was present in the right paraumbilical region at the root of the mesentery. No retroperitoneal extension or bowel involvement was noted. Multiple dilated vessels were seen passing from the mesentery over the mass. The appendix was found inflamed and the rest of the bowel was normal. Excision of mesenteric mass with ligation of the feeding vessels and appendectomy were done. (Figure 2). The postoperative period was uneventful. CECT Abdomen showing a well-defined soft-tissue attenuating solid enhancing mass of 10 x 7.3 x 7.0 cm in the right para-umbilical and lumbar region showing internal calcification and inhomogeneous enhancement displacing adjacent bowel loops. Features suggestive of Benign mesenteric mass/Desmoid. A 10 × 9 cm of resected specimen of mesenteric mass weighing about 800gm.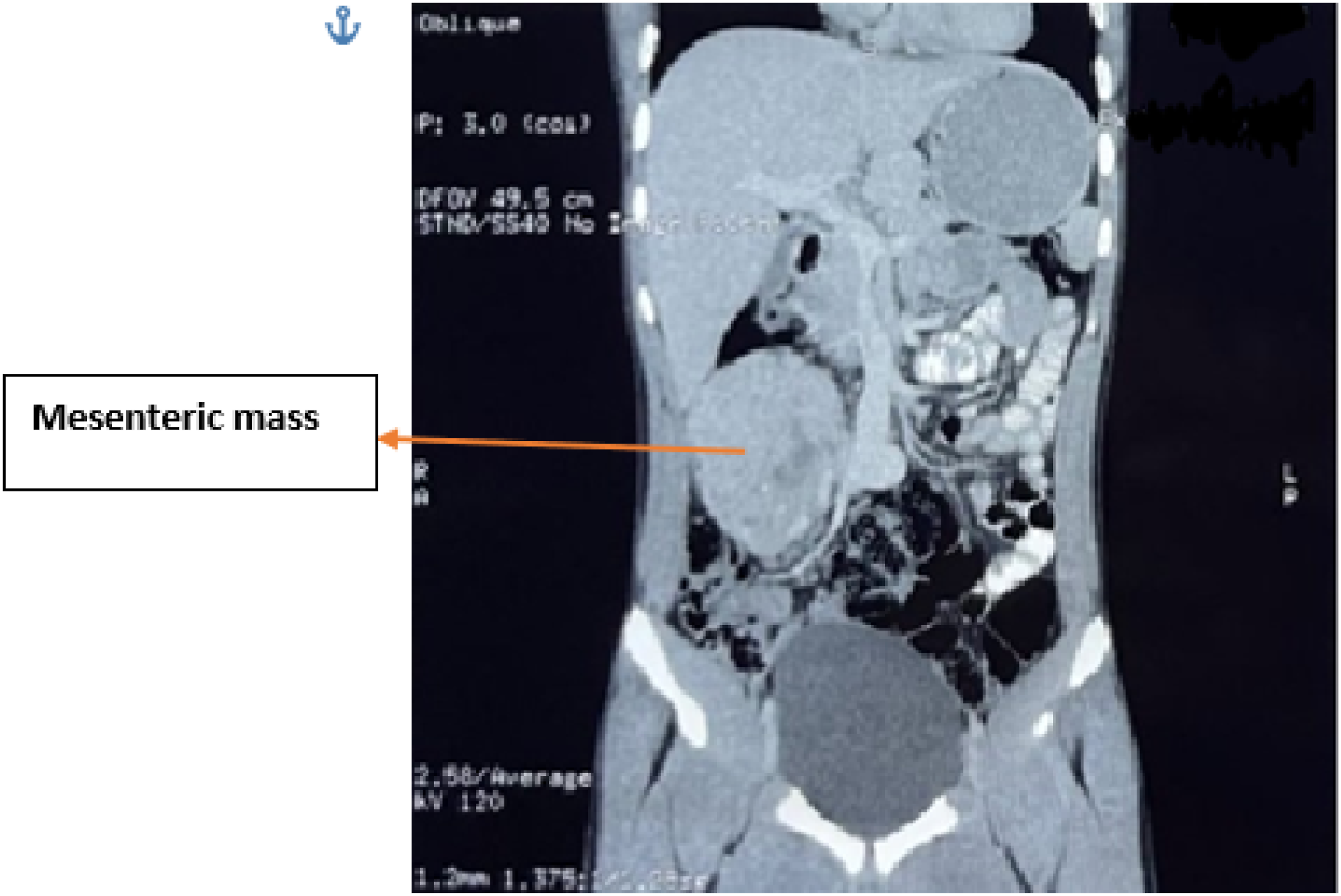

Histopathological examination revealed a lymph nodal mass with multiple variable sized lymphoid follicles of varying sizes with attenuated germinal centers traversed by hyalinized blood vessels. Mantle zones are thickened with lymphocytes arranged in concentric layers giving a characteristic onion ring appearance. Features were suggestive of Angio Follicular Lymphoid Hyperplasia (Castleman disease- Hyaline vascular type) (Figure 3). Sections from the appendix revealed chronic appendicitis. Histopathological examination (400x magnification) of the resected specimen showing Hematoxylin & Eosin staining of lymph nodal mass with multiple hyperplastic lymphoid follicles of varying sizes with atretic germinal centers traversed by hyalinized blood vessels. Foci of hyalinization were seen within the lymphoid substance. Interfollicular area hyper vascular with numerous post-capillary venules with reactive endothelium. Mantle zones are thickened with lymphocytes arranged in concentric layers giving an onion ring appearance. Lymphoid cells were polymorphous with no granuloma. Sinuses were patent.
The patient is in regular follow-up and is doing well for the past 1 year with normal annual CECT abdomen and laboratory parameters.
Discussion
Castleman disease was first described in 1954 by Benjamin Castleman, a pathologist at Massachusetts General Hospital as an uncommon lymphoproliferative disorder, which is non-malignant in nature. 6 Clinically CD is classified as UCD or MCD depending on the involvement of lymph nodes in the same or non-adjacent lymphoid regions. The MCD may present as Human Herpes Virus eight associated disease; idiopathic MCD (iMCD), when associated with thrombocytopenia, anasarca, fever, reticulum myelofibrosis, organomegaly (iMCD- TAFRO), or iMCD-NOS (not otherwise specified) or POEMS associated-peripheral neuropathy, organomegaly, endocrinopathy, monoclonal gamma patchy, skin changes. 3
Patients with UCD can be asymptomatic or have non-specific symptoms due to mass compression. Our patient presented with a lump in the lower abdomen with non-specific symptomatology.
Plain radiographs in thoracic UCD may show a solitary, rounded mediastinal, hilar, or lung mass. On ultrasonography, well-defined hypoechoic mass with prominent surrounding peripheral vessels may be demonstrated. Meadow and Mc Larney 7 reported that abdominal tumors greater than 5 cm in diameter show heterogeneous enhancement with low attenuation areas consistent with necrosis or intralesional fibrosis. Current hypotheses regarding the etiology of CD implicate either low-grade inflammation, immune deficiency, autoimmunity, hamartomatous process, or dysregulation of IL-6 overproduction.8,9 PET scan may provide additional information regarding the metabolic activity of the affected lymph nodes.
UCD in the abdomen and pelvis must be differentiated from gastrointestinal stromal tumors, lymphoma, sarcoma, tuberculosis, and autoimmune disorders.
The treatment of choice for UCD is surgical resection, which is associated with 90% relapse-free survival. Talet et al reported an overall survival of >90% in a retrospective literature study of 278 patients treated with surgery with 10 years of follow-up.
10
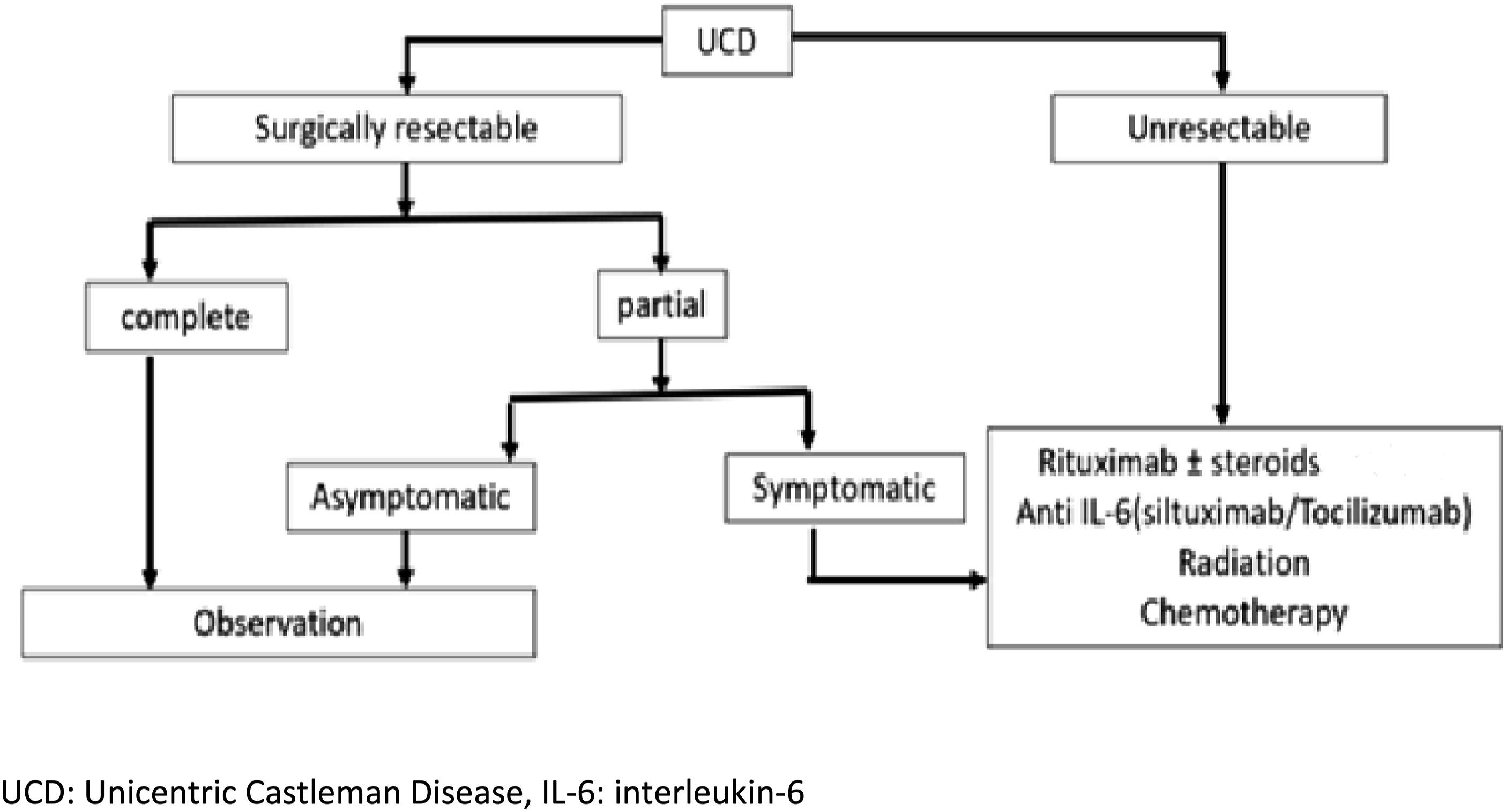
The unresectable disease can be treated with local radiation, embolization, rituximab, anti-IL-6 monoclonal antibodies, and/or steroids to downstage the disease (Figure 4). Flowchart showing various treatment options for Unicentric Castleman disease (UCD).
11
The pathological diagnosis of CD is established by Keller’s criteria: (i)The proliferation of follicle-like structures. (ii)Regressed germinal center with radially penetrating capillaries. (iii)Extensive capillary proliferation with obliteration of lymphatic sinuses. (iv)Perivascular hyalinization in both the germinal center and interfollicular area.1,11
Regular CT scan monitoring at 12 monthly intervals with annual physical and laboratory investigations is recommended. Recommended laboratory tests are hemoglobin, ESR, CRP, serum albumin, and gamma globulins. All these are markers of cytokine-induced inflammatory response. In our case, these parameters in annual follow-up were within normal limits. If the patient remains disease-free, annual imaging can be discontinued after 5 years. 12
Conclusion
Castleman disease is an unusual medical case that poses a diagnostic challenge. Hence, abdominal UCD must be included in the differential diagnosis of solid abdominal mass. Although multiple treatment options are available, surgical resection is still considered the standard treatment of choice due to its diagnostic and therapeutic advantages.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
