Abstract
Background
Poor medication adherence (MA) is prevalent globally. MA assessment, often based on scales or pill count, lacks actionability. A mobile application (app), MediEasy, targeting patients on multiple daily medications, has been developed to engage their caregivers to enhance their MA.
Objectives
This pilot single-arm study aimed to determine the feasibility, acceptability, utility and glycaemic control of the MediEasy-app by patients with type-2 diabetes mellitus and their caregivers.
Methods
20 Multi-ethnic Asian patients aged 51 to 70 years with suboptimal glycaemic control (HbA1c>7.5%) and their caregivers were recruited at a Primary care outpatient clinic in Singapore. The Medieasy app that contains the following major functionalities such as the alerts to take medications, embedded medication details, personalized dosing options (such as frequency and scheduling dosage settings) is twinned to their designated caregivers. The Caregivers can remind the patients who missed the scheduled medication intake when the paired device was alerted.
Results
85% (17/20) of the participants used the app for the entire 4 weeks, reflecting feasibility; 3 dropped out due to technical glitches or failure to adhere to protocol. The app was activated with an average of 2.8 times per day showing adequate utility. The 17 participants and their caregivers agreed that the app supported medication adherence. Their mean HbA1c decreased by 0.2% after 12 weeks of app usage.
Conclusions
The results showed feasibility, utility and acceptability of the MediEasy-app, with improved glycaemic control after its usage.
Introduction
The World Health Organisation has identified poor adherence to medical treatment as a key global healthcare problem. 1 Confusion or simple forgetfulness are frequent involuntary causes of poor medication adherence (MA). 2 A local study has shown that overall poor adherence to oral anti-diabetic medication was 57.1%, with forgetfulness being the main cause. 3 Monitoring MA becomes a crucial part of patient care to optimise their health status and reduce complications. 4
MA is often measured by validated scales and prescription refill rate in research. 5 However, these methods have yet to be systematized in routine clinical practice; they can be laborious and lack actionability to promptly address the cause of poor MA. 5 Mobile health or mHealth has been increasingly used to promote MA. A systematic review showed that the use of mobile applications (app) increased MA beyond clinical setting to the residences of the patients. 6 Now there are over 700 Apple and Android apps available to users to enhance MA. 7
The simplest mHealth method is to send a message to notify a patient to take the medication based on the scheduled timing. 8 The wireless pill container is an existing simple Internet of Things (IoT) solution to monitor consumption of medication. However, most of these interventions have yet to sustain MA in the lifelong management of non-communicable diseases. 9 A review of users’ feedbacks by Jamie Park et al alluded to variable utility among these MA related apps. 7 Karla Santo et al also reported that most apps did not have actionable functions and almost half of them were classified as basic medication reminder apps. 10
MA requires behavioural adaption, which cannot be simply solved by a reminder system. 11 Bosworth et al highlighted the need to incorporate behavioural skills enhancement in innovations to facilitate the adoption of medication-taking into everyday life.11,12 They cautioned the limited effectiveness in stand-alone intervention, but advocated engaging patients, their caregivers and healthcare providers to interact and communicate on their medication intake as pivotal to enhance the effectiveness of the innovation. 11 Hence, garnering additional support from patients’ caregivers in the design and operation of the mobile app is a potential strategy to improve their MA.
A MA reminder app named MediEasy has been developed for use in primary care, targeting at older patients treated with multiple medications in the management of their type-2 diabetes mellitus (T2DM). Aside from app functions appreciated by users, such as the alerts to take medications, embedded medication details, personalized dosing options (such as frequency and scheduling dosage settings) and optimizing medication information display and input,7,10 it is twinned to their designated caregivers. The caregiver is notified by the app when the patient consumes the medication. They can remind the patient if they spot any lapse by patient in adhering to their scheduled medication intake. The MediEasy app is also synced to remote frequency tagged medication containers so that the patients do not need to manually document their medication intake.
With such a modus-operandi, the MediEasy app was postulated to enhance the MA among patients due to extra supervision and support from their caregivers. The app can be adapted to measure adherence to a single or multiple medications to manage patients on diverse range of oral medications.
This pilot study aimed to evaluate the uptake, utility, operation and functionalities of the MediEasy app in enhancing the MA of patients treated with oral medications for their T2DM and other co-morbidities. The study also aimed to assess their medication-taking behaviour (timing and frequency of medication intake), their perceptions of the mHealth in monitoring their MA and health outcome.
Methods
Study site
The study was conducted in a typical polyclinic (primary care clinic) located in Pasir Ris estate with a population of 123,700 multi-ethnic Asian residents in north-eastern region of Singapore. 13 The polyclinic attended to outpatients, 29% of whom are aged 65 years and older. 14 Dyslipidaemia, hypertension and T2DM constitute the top three medical conditions managed in this polyclinic. 15 Patients with any of such diagnosis are routinely directed to a health monitoring station (HMS), where trained staff measure their clinical parameters such as weight, height and blood pressure. These clinical data are automatically ported over to their respective electronic medical records.
Study population
A total of 20 multi-ethnic Asian patients with T2DM and 20 of their respective caregivers provided written informed consent to participate in this feasibility study over a period of 4 to 6 weeks. The rule of thumb for sample size of pilot study according to Julious is recommended to be a minimum of 12. 16 To buffer for possible 50% dropout at follow up, the sample size was further increased to 20.
Inclusion criteria
1. Aged 51 – 70 years of any gender and Asian ethnicity. 2. Minimally 1-year history after diagnosis of T2DM, with at least one previous glycaemic control index (glycated haemoglobin or HbA1c) record in the EMR in the past 6 months. 3. Treated with 2 or more medications to be consumed daily (patients treated with other medications were also included) 4. Ability to use smart phone to download the MediEasy app, and are willing to use it to check notification, input text, and connect to Internet 5. Willingness of family members or caregivers who are users of smartphones to download the MediEasy app and use it to assist the patients in their medication adherence 6. Suboptimal glycaemic control of HbA1c of >7.5% 7. Poor MA based on score of less than 25 using the Medication Adherence Report Scale for at least one of their medications (MARS-5 comprises 5 questions, each question with a maximum score of 5 for full adherence to medication) 8. No new or change in the frequency and doses of their medication in the past 3 months
Exclusion criteria
1. Failure of patients and their caregivers to download the app to their mobile phones or were unwilling to use the MediEasy app 2. Current cognitive impairment [Abbreviated Mental Test (AMT) score should be at least 8 or more] which rendered patient or caregiver incapable of using the MediEasy app nor providing informed consent to participate in the study.
17
3. Inability to adhere or complete the study protocol, such as pre-scheduled extended travel. 4. Current optimal adherence to all their medications based on score of 25 using the MARS-5 Scale for each of their medications 5. Satisfactory glycaemic control (HbA1c<7.5%) 6. Patients on injectables were excluded
Subject enrolment
A trained clinical research coordinator (CRC) screened potential subjects at the HMS. Written informed consent were obtained from both the patients and their caregivers after their queries and doubts were addressed and clarified.
The enrolled patients were shown photos of all the available diabetes-related medications in the polyclinic drug inventory to the recruited patients to identify their specific therapeutic regimens. Next, they administered the questionnaire which collected the following data from the patients: 1. Demographic characteristics: age, gender, ethnicity, housing type, occupation (if applicable), eligibility of health care financing privileges for those in the lower socioeconomic status. 2. Family structure and home care status (number of family members living together or stay alone; profile of caregivers and availability of domestic helper) 3. Type of smart phone (brand and model) and service plan 4. Abbreviated Mental Test score (AMT) 5. Medication Adherence Report Scale (MARS-5) for each of their diabetes related medications 6. Perception and attitude towards the adoption of new MediEasy App using a 5-point scale (self-developed) 7. Information of medical conditions, medications and number of tablets to be collated backend from their electronic medical records.
The CRC also collected information from the caregiver using a separate questionnaire: 1. Their demographic characteristics: age, gender, ethnicity, housing type, occupation 2. Relationship to patient 3. Type of smartphone and plan 4. Perception and attitude towards mHealth based on scale (maximum 5 points)
Development and functionalities of the MediEasy app
The investigators comprised a family physician, pharmacists and IT software engineers from a local polytechnic who designed the app using design-thinking approaches. The prototype underwent three rounds of iterations based on user requirements. The major functionalities of the MediEasy app include: 1. Enlisting the medications to be consumed daily, including the name (generic and brand names), doses, frequencies and timings of each medication; the photos of each tablet, in their blister pack or in the original containers are shown together with their names on the app. 2. The container of each medication is tagged to Near Field Communication (NFC) to sync with the app. It allows the tracking of the medication when it is taken out of the container, and data is captured in the app. 3. The data was conveyed by the app to a cloud located in a local server protected by the necessary cyber-security measures; the data could be shared with the caregiver app and the Web site for healthcare providers. 4. Notification and alerts at scheduled timings of each medication can be configured in vibratory or conventional ring tones. The MediEasy app allows these notifications and alerts to be configured in personalised voice-annotated messages in their preferred Asian language (Malay, Tamil) or in Chinese dialects. This function enables patients who are not literate in English to use the app. 5. A caregiver app is also developed to pair up with the patient app. Caregivers are able to view the date, timing and types of medication consumed when patients activate their app respectively.
Figure 1: Modus operandi of the app for the patient and the caregiver. Modus operandi of the app for the patient and caregiver.
At the end of 4 to 6 weeks, the patients completed the questionnaire to assess their MARS-5 scale score, perceptions and attitude towards the app. The CRC counted the leftover pills, which the patient returned. Each patient was reimbursed S$20 (approximately USD15) supermarket voucher for the travel expense as approved by the institution review board. The app was removed from their smart phone at this second visit. The cloud-based data base was downloaded for data audit and analysis before it was removed 3 months after the last visit by the last recruited patient.
Outcomes
The total number of tablets for each medication to be consumed within 4 weeks was computed as the denominator in the baseline data. The patients were instructed to discard any balance of medications at home at the point of study enrolment and to consume only the latest dispensed medications according to prescription regimen. 1. Feasibility of the MediEasy app was assessed by the number of patients who used the app for the entire study period as a proportion or percentage of total number of recruited patients. 80% of patients who used the app over the stipulated study period was regarded as an arbitrary indicator of feasibility. 2. The acceptability of the app was gauged by the patients’ responses to specific questions in the questionnaire, such as their perception and attitude towards the adoption of new technology scale before and after the study. 3. The app utility for the patients was assessed by computing the number of app activation per day (mean, median, minimum, maximum, timing of app activation) retrieved from the cloud-based data. 4. The effect of the app on MA was computed by the pill count, which was a direct measure of MA during the study period and change in self-reported outcomes based on MARS-5 scale. 5. The medication intake behaviour was computed by the a) Frequency of medications which were not taken over the total number of planned medications b) Frequency of medications which were not taken within 2 h of planned time over the total number of planned medications 6. The primary health outcome was defined as the change in HbA1c levels between study enrolment and 12 weeks later.
In the MARS-5 questionnaire, patients who indicated the frequency (‘always’, ‘often’, ‘sometimes’, ‘rarely’ or ‘never’) for each question, were scored from ‘always’ (1 point) to ‘never’ (5 points). Scores for each of the five questions are aggregated to give the final score which ranges from 5 to 25 points. A total score of less than 25 points is defined as low adherence to the medication. 3
Statistical analysis
Descriptive statistics were performed to determine the demographic characteristics of the patients and caregivers. Their medication adherence from the MediEasy app was calculated based on the definitions above. Spearman’s correlation was used in assessing the change in HbA1c and app activation. Mann-whitney U test was used to compare the change in HbA1c if patient missed their medication, and if patient exceeded 2 h of planned time. A p-value of less than 0.05 is considered statistically significant. All analyses were analyzed using IBM SPSS version 27.0.
Results
Figure 2: The consort flow diagram shows the number of patients who were screened, and those who were excluded based on the eligibility criteria, or declined participation. A total of 629 patients were screened, of which 66.8% failed to qualify as they had satisfactory glycaemic control (HbA1c of 7.5% or below); 10% could not download the app due to phone incompatibility; 4.6% did not have any caregivers to look after them, 3% had good medication adherence and 2.7% newly diagnosed T2DM. Out of 81 (12.8% or about one in eight subjects) who were eligible for study enrolment, 61 declined to participate and 20 patients were eventually recruited, constituting a response rate of 25% (20/81). Consort diagram showing the patient recruitment data. *Patients could have more than one exclusion criteria.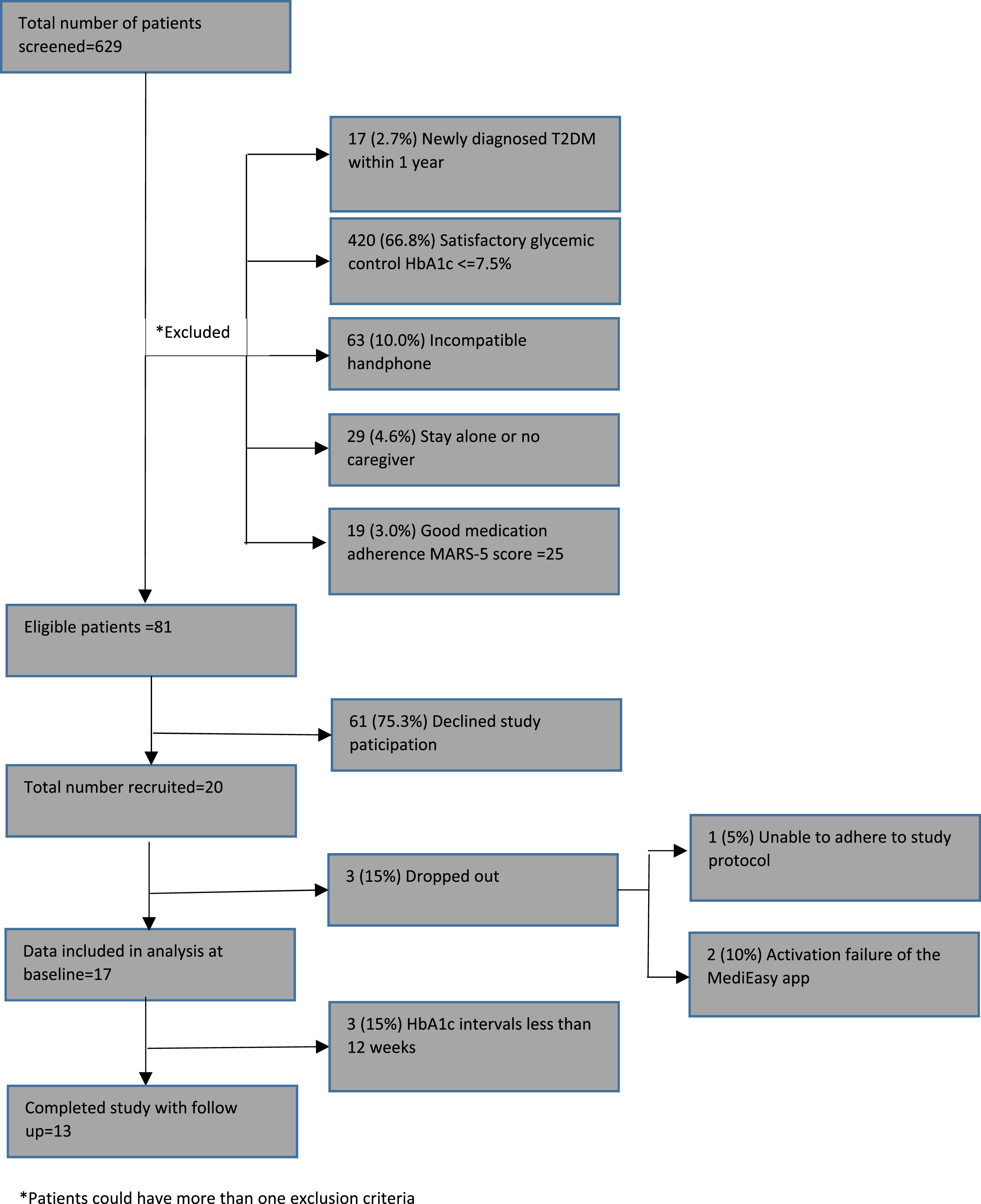
Of the 20 recruited patients, 17 of them completed the study. 3 dropped out during the study period: 1 cited inability to adhere to the study protocol due to work commitment and the remaining 2 claimed that they could not activate the app due to incompatibility of their smart phone model.
Subsequently, 13 patients had HbA1c repeated 12 weeks after study enrolment. One patient had missing baseline HbA1c and the remaining 3 had HbA1c that were less than 12 weeks of interval, and were excluded from the analysis.
Demographics of patient and caregiver.

aChange of HbA1c 12 weeks after study enrolment Subjects with less than 3 months interval of HbA1c readings were excluded.
All the patients reported poor adherence to anti-diabetic medications on enrolment but the majority (81.3%) of them reported good adherence to the same medications at the end of the study. The mean HbA1c dropped by 0.2% in the 13 patients.
Adherence of medications and patient’s app activation associating with HbA1c outcome.
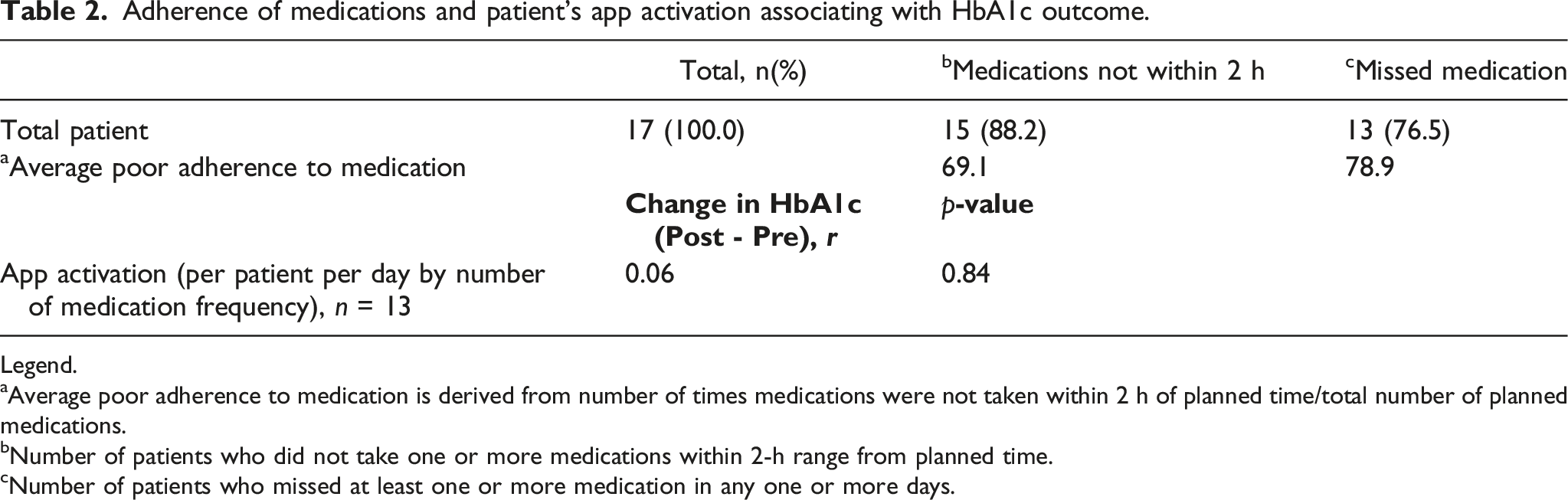
Legend.
aAverage poor adherence to medication is derived from number of times medications were not taken within 2 h of planned time/total number of planned medications.
bNumber of patients who did not take one or more medications within 2-h range from planned time.
Number of patients who missed at least one or more medication in any one or more days.
In terms of pill count at 12 weeks, all patients reported no balance of any of the prescribed medications at the last visit.
Impact of MediEasy app on short term health outcome (HbA1c).

Legend: #Change of glycaemic control (HbA1c) of patients 12 weeks after study enrolment Note: Subjects with less than 3-months interval of HbA1c readings were excluded (n = 4).
Figure 3 shows the medications were taken at the diverse timing; morning and evening were the commonest timing for the medication consumption. Outliers are noted in few patients who took their medications in the early morning. MediEasy app activation pattern.
Using a cut off of 28 days from recruitment of the 17 patients, the average number of MediEasy app activation per patient was 2.8 times per day, with a median of 2.8 times, minimum of 1.2 times and maximum of 4.8 times daily. Comparing utility of first 2 weeks and last 2 weeks, the average app utility decreased from 2.9 to 2.6 times per day.
Discussion
The results showed that 81.3% of participants reported better MA based on the MARS-5 score after using MediEasy app for 4 weeks (Table 1). The MediEasy app aligns to the recommendations by Bosworth et al to address the complex problem of MA. 11 The app alerts and engages the caregivers to pay attention to their family members with suspected poor MA. Aside from the typical buzz or ubiquitous ring tone in smart phone, the app allows personalization of the medication reminder notifications. The voice-annotated messages can be delivered to the patient in their preferred language or from their caregiver, whom the patient is familiar with. This is a boon to patients who may not be highly educated and can only speak and understand local Asian languages or dialects.
Most patients (85%) used the app for the entire 4 weeks. They provided favorable feedbacks of its operational modus. The overall system appears to be a viable platform where goals of MA between patient, caregiver and healthcare providers are shared, discussed and mutually agreed upon. Concurrently, the autonomy of the patients to select their preferred timing of medication-taking with personalized reminders can be actuated via the MediEasy app system.
Patients who missed their medication according to the MediEasy App did not return their balance medication; this shows that data could be unreliable if we depend on self-reporting and pill count. The MediEasy app not only generates data of any missed medication but also time-stamp on their intake. Figure 3 showed that patients were taking their medications in the early hours of the morning. These outlier timings could be related to their sleeping pattern, or shift work hours among the employees. Understanding the pattern of medication intake allows the healthcare provider to focus on specific MA issues faced by patients, such as home or work environmental barriers, which can interfere with the dosing frequency and timing of medication intake.
Approximately one in four patients (25%) were recruited in the study. Most of them were ineligible for the study enrolment as their MA were screened to be satisfactory. The app will not value add to patients with good MA. Nonetheless, a small subset of the target users was excluded as they did not own nor use a smartphone. They were often associated with lower level of literacy or social economic status. Understanding the profile of users who demonstrated fidelity to the MediEasy app allows the healthcare providers to identify those who are more likely to adopt the app for the intended effect. The latter should recognize that personal characteristics such as digital health literacy, mental health status and psychosocial background can also influence the use of mHealth app. Time and efforts to coach the older patient to use the app functionalities are crucial for its successful adoption and utility. The research coordinators and the developers were onsite to induct the enrolled patients and were ready to respond to their technical difficulties.
Patients in the intervention group showed improvement in their glycemic control when their mean HbA1c declined by 0.2% up to an observation period of 12 weeks. The result reflects potential benefits of MediEasy in improving health outcomes in older adults. It is non-invasive, inexpensive and easier to implement compared to the gold standard of performing blood assays of the medications or their metabolic products which are costly, requires blood sampling, inaccurate or unavailable in most primary care establishment. Nonetheless, the RFID tagging may not be practical for all patients and their acceptance of such an approach has yet to be widely assessed.
The app largely addresses the involuntary cause of MA. It is incapable of identifying a patient who activates the app by opening the NFC tagged container but without taking the medication. Other confounders such as dietary indiscretion could still interfere with the glycemic control of the patients. MediEasy should ideally be embedded in a comprehensive multi-functional app which can support a patient to manage the many facets of diabetes management.
The MediEasy app system has other limitations. mHealth apps operate on either iOS or android platforms or both. They are susceptible to malfunction after upgrading of these platforms. The failure of two patients to use the app after study enrolment highlights the importance of selecting users with compatible smartphones to download and activate the MediEasy app. The manual tagging of the medication containers with NFC is laborious. The next MediEasy prototype will be linked to a smart pillbox to reduce repetition of this manual task, especially for patients with multiple medications.
New brands of insulin were being introduced to the inventory at the time of the study, which might potentially confuse the patients: the investigators decided to exclude these parenteral medications, although they could be included in the MediEasy app.
The MediEasy system is also limited by the absence of the caregiver app utility data. This functionality will be included in the development of the next prototype, which will involve the targeted users or patients and their caregivers in the design planning stage. The scope of the app will be expanded to embed information on medication storage, potential interactions with other medication or natural products and adjustment of medication during overseas travel over different time zone.
The study population is deliberately small to test feasibility in a rapid cycle, such that the recruitment was completed in less than 2 months, despite screening over 600 patients. Nevertheless, the data from this small population provides perspectives such as effect size, profiling of suitable users, implementation issues, which are invaluable information to design an adequately powered randomized controlled trial to prove its value or effectiveness.
The MediEasy app can be used in future to provide insight into the adoption of any new medication regimen by patients over the initial 1 month. Any emerging issues on the medications can be resolved by both caregiver and healthcare provider early, so that patients regain control of their medical condition expeditiously to avoid associated morbidity or even mortality. Longer period of app utility beyond 4 to 6 weeks may lead to alert fatigue. The app can be re-introduced to patients when poor MA is suspected at any time point of their health review.
Conclusion
The results showed feasibility and utility of the MediEasy app. The improvement in glycemic control has proven its potential to improve MA and health outcomes among older patients, with the support of their caregivers and their healthcare providers. The patients accepted its use with favorable feedbacks. A randomized controlled trial using the next enhanced prototype is planned to assess its values in improving longer term health outcome.
Footnotes
Acknowledgments
The authors are also grateful to the students from Nanyang Polytechnic (Hak MN, Muhammad Zulkifli MR, Chau PY, Chin FL. Krishna Morthy SVRR, Lim E, Pang P, Soh T, Yeo A) who trained the patients and their caregivers to use MediEasy; Ng CE and Goh WTC for recruiting the patients; Sankari U for designing the questionnaire and electronic clinical research files; Caris and Patricia for assisting in the budget planning and ethics board application.
Contributorship
TNC, LZQ and DP conceptualised the design of the MediEasy app and system. GCC, IT, LZQ supervised the implementation of the feasibility study. TNC and EK analyzed the data and interpreted the results. TNC, GCC and EK drafted the manuscript. All authors reviewed, critiqued and agreed on the final version of the manuscript before journal submission.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the TOTE Board Social Innovation Research (SIR) Fund, which was awarded to the team members (LZQ and DP) from Nanyang Polytechnic.
Ethical statement
Data Availability Statement
We have included the data produced in the present work in the manuscript.
