Abstract
Background
Sucroferric oxyhydroxide has been used in the treatment of hyperphosphataemia in adult patients with chronic kidney disease receiving haemodialysis or peritoneal dialysis since 2013. However, there is a paucity of data on sucroferric oxyhydroxide use in Asian populations.
Objective
We aim to provide real-world experience of sucroferric oxyhydroxide use in our local cohort on maintenance haemodialysis.
Methods
A retrospective clinical audit was performed to review laboratory parameters of haemodialysis patients who received sucroferric oxyhydroxide over a 14-month period from December 2020 to January 2022. Sucroferric oxyhydroxide reduced serum phosphate levels effectively, with a more than 3-fold increase in the proportion of patients who were able to achieve phosphate levels of ≤5.5 mg/dL. Ferritin levels and transferrin saturation were increased, but there were no significant differences in haemoglobin levels and erythropoietin used in the long term. We showed that sucroferric oxyhydroxide was highly effective in reducing serum phosphate to recommended levels in patients receiving haemodialysis. Further prospective studies in Asian populations are warranted to confirm our findings.
Introduction
The incidence of chronic kidney disease (CKD) is rising in Singapore, increasing from 1586 per annum in 2011 to 2079 per annum in 2019. 1 Hyperphosphataemia is a common complication faced by patients with CKD and its association with all-cause mortality has been demonstrated in various studies.2–4 Treatment of hyperphosphatemia with phosphate binders has been shown to improve survival in large cohort studies,5–7 though a recent Cochrane review found that while sevelamer may lower death (all causes) compared to calcium-based binders and incur less treatment-related hypercalcaemia, there was no clinically important benefit of any phosphate binder on cardiovascular death, myocardial infarction, stroke, fracture or coronary artery calcification. 8
It has been suggested that use of calcium-based phosphate binders results in higher mortality than either sevelamer or non-calcium-based phosphate binders in general. 9 Sucroferric oxyhydroxide (SO) was introduced in the therapeutic armamentarium for adult patients on dialysis in 2013. It is a calcium-free polynuclear iron (III)-oxyhydroxide phosphate binder shown to be non-inferior to sevelamer in a phase III study, with benefits of reduced pill burden and better adherence. 10 In addition, long term use of SO reduced serum fibroblast growth factor-23 significantly, while having minimal systemic iron absorption and therefore low risk of iron accumulation. 11 The VELREAL multicentre study provided real-life data that SO results in better serum phosphate control, reduction in the number of tablets and an improvement in therapeutic adherence, and may be beneficial with regards to secondary hyperparathyroidism and nutritional status. 12 Similar results were demonstrated in another larger European multicentre study with a longer follow up of 36 months 13 SO has been shown to have a favourable safety and tolerability profile in various clinical studies. 14 It has also been shown that patients maintained on uninterrupted SO use had fewer hospital admissions compared to patients who switched to treatment with other phosphate binders. 15
SO was introduced to The National Kidney Foundation Singapore (NKF) formulary in December 2020. Herein we report on its use in treatment of hyperphosphataemia in our local cohort, to provide real-world experience of SO use in a community-based haemodialysis program, especially given the paucity of data in southeast Asia currently.
Methods
We conducted a retrospective clinical audit of patients on haemodialysis at NKF dialysis centres, who received SO treatment over a 14-month period from December 2020 to January 2022. We included patients with at least 2 months of continuous SO usage during this period. For patients whose treatment was interrupted for more than 2 months, we only considered the period of first continuous usage. We excluded patients without repeat laboratory monitoring after treatment initiation. The dose and duration of SO was at the discretion of the treating physician.
Laboratory parameters are performed at NKF routinely on a 2-monthly basis and data collated. We compared calcium, phosphate, transferrin saturation, ferritin, haemoglobin, albumin levels and normalised protein catabolic rate (nPCR), and erythropoietin dosage at baseline, at 2 months post SO initiation and over the 14-month period of audit. Drug discontinuation rates and the reasons were reviewed.
IBM® SPSS® Statistics 26 was used for statistical analysis. Continuous variables were analyzed with the paired t test for normally distributed data, and the Wilcoxon signed-rank test for non-parametric data. All statistical tests were performed using 2-sided tests at the 5% significance level. As this was a clinical audit, institutional review board for ethics approval and informed consent were not required.
Results
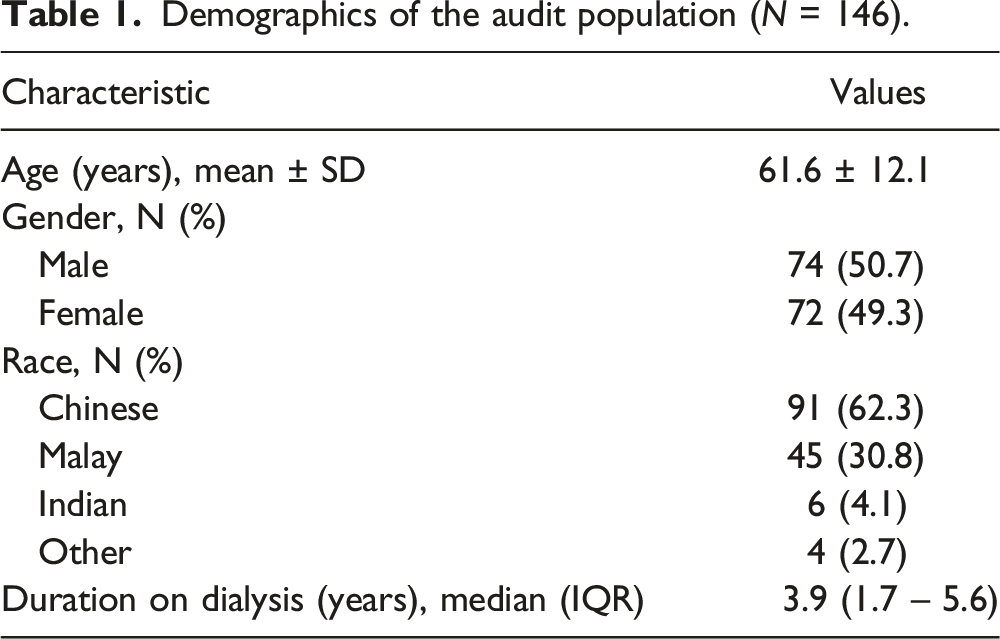
Demographics of the audit population (N = 146).
Biochemical and haematological parameters at baseline, at 2 months after sucroferric oxyhydroxide initiation, and over the 14-month period audited.
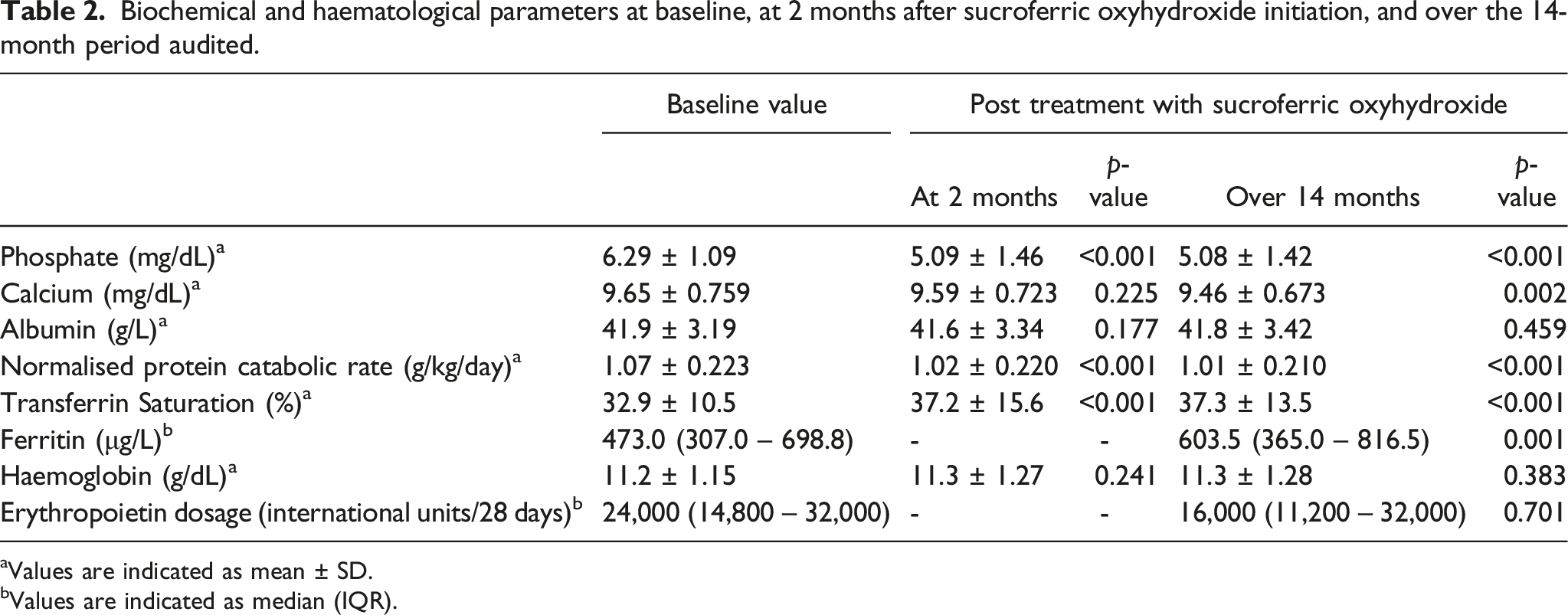
aValues are indicated as mean ± SD.
bValues are indicated as median (IQR).
Prior to the initiation of SO, 19.2% of patients achieved target phosphate levels of ≤5.5 mg/dL with existing phosphate lowering therapy. Serum phosphate improved after SO initiation, with 65.1% of patients achieving target phosphate levels ≤5.5 mg/dL over the 14-month audited period (Figure 1(a)). Proportion of patients achieving target phosphate levels ≤5.5 mg/dL after completing 2, 4 and 6 months of cumulative SO treatment also showed marked improvement at 65.1%, 60.0% and 79.3% respectively (Figure 1(b)). Percentage of patients achieving serum phosphate levels ≤5.5 mg/dl before sucroferric oxyhydroxide (SO) treatment and over the audited treatment period. (a). Percentage of patients who achieved serum phosphate levels ≤5.5 mg/dl prior to treatment initiation and over the audited 14-month period of sucroferric oxyhydroxide treatment. (b). Percentage of patients who achieved serum phosphate levels ≤5.5 mg/dl before treatment initiation after 2, 4 and 6 months of cumulative sucroferric oxyhydroxide treatment.
There was no significant difference in the serum calcium levels 2 months after initiation of SO (p = .225). A slight drop from 9.65 mg/dL to 9.46 mg/dL (p = .002) was observed over the 14-month period. Our audit cohort included patients taking calcium-based phosphate binders concurrently with SO. A sizeable number of patients (n=112) switched from calcium-based phosphate binders to SO or took a combination of calcium-based phosphate binders with SO. A small number of patients (n = 34) were not on calcium-based phosphate binders before SO initiation.
Mean transferrin saturation increased from 32.9% to 37.2% at 2 months and 37.3% over the audited 14-month period after SO administration (p < .001). There was significant increase of ferritin levels from 473 μg/L to 603.5 μg/L (p = .001) over the 14-month period. More than half of the audit cohort (57.5%) were on concomitant intravenous iron supplementation. However, there was no significant difference in the haemoglobin levels (p = .383) or the dose of erythropoietin used (p = .701) (Table 2). 14 patients were not on erythropoietin before SO initiation, while four patients discontinued erythropoietin after SO initiation. Of the remaining patients, 42 patients (28.8%) had a decrease, 25 patients (17.1%) had an increase and majority (61 patients, 41.8%) had no change in erythropoietin dosage.
The nPCR dropped for both time frames evaluated (p <.001), although the values remained above 1.0. There was no statistically significant difference in albumin levels pre- and post-SO administration.
Drug discontinuation rate was 34.9% over the 14-month period audited. After censoring for patient death, drug discontinuation was 26% with majority (60.5%) due to documented adverse drug reactions (ADR). Gastrointestinal (GI) side effects were the most common reported ADR.
Discussion
Hyperphosphataemia is common in patients with CKD, resulting in secondary hyperparathyroidism, vascular calcification and increased cardiovascular mortality. KDOQI (2003) guidelines recommended that serum phosphate levels be maintained between 3.5 and 5.5 mg/dL in patients with CKD on dialysis, 16 while KDIGO (2017) guidelines suggested lowering high phosphate levels towards the normal range in patients with CKD G3a-G5D. 2 In our audit, only 19.2% of patients were able to achieve a target phosphate level of ≤5.5 mg/dL pre-initiation of SO treatment. In patients who received and continued treatment with SO, there was a more than 3-fold increase in the percentage of patients (65.1%) who were able to achieve the target phosphate levels of ≤5.5 mg/dL over the 14-month period. The marked improvement was consistent across various cumulative periods of SO treatment over the 14-month audit period.
The use of SO in our audit reflects real-world experience, where SO may be used in various combinations in clinical practice, for example, adding to existing calcium-based binders, switching from existing calcium-based binders, or starting SO in a patient not on phosphate binders. Despite various combinations of use with SO, we demonstrated its effectiveness in treating hyperphosphataemia. The efficacy of SO in different clinical treatment settings was demonstrated in a recent review of real-world evidence of its use. 17 We found no significant difference in serum calcium levels in short term use of SO, but a slight reduction was observed in longer term use. This could be attributed to the sizeable proportion of patients (76.7%) in our audit cohort who switched from calcium-based phosphate binders to SO or took a combination of calcium-based phosphate binders with SO (with reduced dosage of calcium-based binders, presumably).
Our cohort had a significant increase in transferrin saturation in both short- and long-term use of SO, as well as raised ferritin levels in the long term. Nearly 60% of our cohort were receiving intravenous iron supplementation concomitantly. However, there was no significant difference in haemoglobin in the long term. This is consistent with results from other studies. A phase III study showed that median ferritin levels increased in both treatment groups of sevelamer and SO, while an increase in transferrin saturation was only seen in the SO treatment arm. The increases in iron parameters occurred early during treatment with SO and plateaued thereafter, indicating no iron accumulation. Similarly, there was no significant difference in the haemoglobin levels in both treatment groups. 10 Post hoc analysis of this study including its 28-week extension showed ferritin concentrations increased to a greater extent in the overall study population, more than 70% of whom received concomitant intravenous iron, compared with the subset of patients who did not receive concomitant intravenous iron, indicating the increase was primarily driven by intravenous iron use. 18 This was also reported in real-world data from the VERIFIE study. 13 Consistently, studies have found that long-term use of SO is not complicated by significant risk of iron overload.13,18–20
We did not find a change in dose of erythropoietin use in the long term. While there was a trend towards lower use of anti-anaemic products (erythropoiesis-stimulating agents and intravenous iron) among patients treated with SO compared with those treated with sevelamer carbonate in the phase III study and its extension, 18 other studies have shown that SO use did not change haematological parameters, use of iron supplementation or erythropoietin dose.19,20
Although nPCR in our cohort decreased significantly after SO use, the median nPCR values remained above 1.0. Moreover, there was no significant difference in the albumin levels. This differs from that reported in studies which have suggested that SO use may improve nutritional status.12,21 It may be that reinforcement of dietary restrictions impacted inadvertently on dietary protein intake in our patients. The lack of improvement in nutritional markers likely reflects the non-hypoalbuminaemic state of our cohort at baseline. Indeed, it has been shown that in hypoalbuminaemic patients, initiation of SO therapy was associated with increase in serum albumin, suggesting SO use may have allowed patients to increase their dietary protein intake. 22
Drug discontinuation rates have varied across studies to date, reflecting real-world experience in SO usage. Consistent with studies to date, GI adverse effects were the most frequently reported reason for treatment dropout, making up 40% of all reported ADRs in our cohort. In our clinical experience, GI side effects appear to be dose-related, as such initiating SO at a lower dose may ameliorate ADRs and improve adherence.
Limitations of the findings from our audit reflect real-world clinical practice; usage of different combinations of phosphate binders based on clinicians’ practice, unmeasured effect of dietary changes and the influence on phosphate levels, and variable adherence to prescribed therapy.
Conclusion
We demonstrated that SO is highly effective for hyperphosphataemia treatment in patients on maintenance haemodialysis. Iron indices were increased but haemoglobin levels and erythropoietin dosage were unchanged from long-term therapy with SO. Prospective studies in Asian populations are warranted to confirm our findings.
Footnotes
Author contributions
AN and BK conceived the study. AN and RT researched literature. VS and YH did the data analysis. AN, BK, TS and RT did the data interpretation. AN wrote the first draft of the manuscript. RT reviewed, edited and produced the final manuscript. All authors reviewed and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was not sought for the present study because the report was performed as part of a clinical audit. This study was completed in accordance with the Helsinki Declaration.
Informed Consent
Informed consent was not sought for the present study because the work was done as part of routine clinical auditing.
