Abstract
Purpose
Central vein stenosis is commonly encountered in end stage renal disease patients on hemodialysis. Most are manageable with repeated procedures and venoplasty, however in some cases, venoplasty may not be successful.
Method
We discuss a rare case of persistent upper limb swelling due to severe central vein stenosis from compression of an aortic arch stent.
Result
Despite attempt at prolonged balloon inflation during venoplasty of the central vein, the stenosis still persisted. The arteriovenous fistula was then ligated and other forms of vascular access for hemodialysis were sought after.
Conclusion
Central venograms may be useful in end stage renal failure patients as part of pre-operative arteriovenous fistula creation so as to anticipate the possibility of central vein stenosis and its associated complications.
Introduction
The need for longer term vascular access in end stage renal failure (ESRF) is increasing and techniques to achieve longevity of these access are continuously established. Central vein (superior vena cava, brachiocephalic, subclavian) stenoses are common obstacles to the long-term patency rate and lifespan of arteriovenous access in the upper limbs. They limit blood flow through venous hypertension and cause upper limb swelling, resulting in the need for repeated venoplasty to overcome the stenosis. However, recalcitrant upper limb edema may eventually lead to abandonment of the arteriovenous access and the creation of a new vascular access elsewhere. In the long run, a patient may eventually run out of options for the arteriovenous creation of vascular access.
We hereby discuss a rare case of a non-salvageable central vein stenosis (CVS) with immediate on-table angioplasty failure.
Case report
Our patient is an 82 year-old Chinese man with ischemic heart disease, atrial fibrillation as well as ESRF secondary to chronic glomerulonephritis. He is on hemodialysis via a left radiocephalic arteriovenous fistula (RC AVF) created in 2016 that was complicated by recurrent AVF inflow stenosis requiring repeat angioplasty every 4–6 months.
His other past medical history includes pulmonary tuberculosis which led to an incidental finding of a saccular aneurysm at the aortic arch. Endovascular repair of the thoracic aorta was performed in November 2018. Thereafter, he presented with new onset left upper limb swelling complicated by overlying cellulitis. During angioplasty, the brachiocephalic junction was noted to be stenosed but was initially left alone as the culprit vessel was attributed to a severe cephalic vein stenosis at the AVF. Figure 1 shows a cut off at the thoracic aortic graft stent. Cut off seen at the thoracic aortic graft stent.
After balloon angioplasty to the cephalic vessel, he still experienced persistent upper limb swelling. Therefore, central vein venoplasty was attempted to address the upstream CVS. Despite prolonged inflation of the venoplasty balloon, the stenosis could not be overcome. Due to extrinsic compression from the aortic stent graft in the aneurysmal aorta, it was also deemed stenting of the central vein would not be feasible. Figure 2 shows the wasting of balloon across the central vein due to compression of the left brachiocephalic vein against the manubrium by the aortic arch graft stent. Wasting of balloon across the central vein due to compression of the left brachiocephalic vein against the manubrium by the aortic arch graft stent.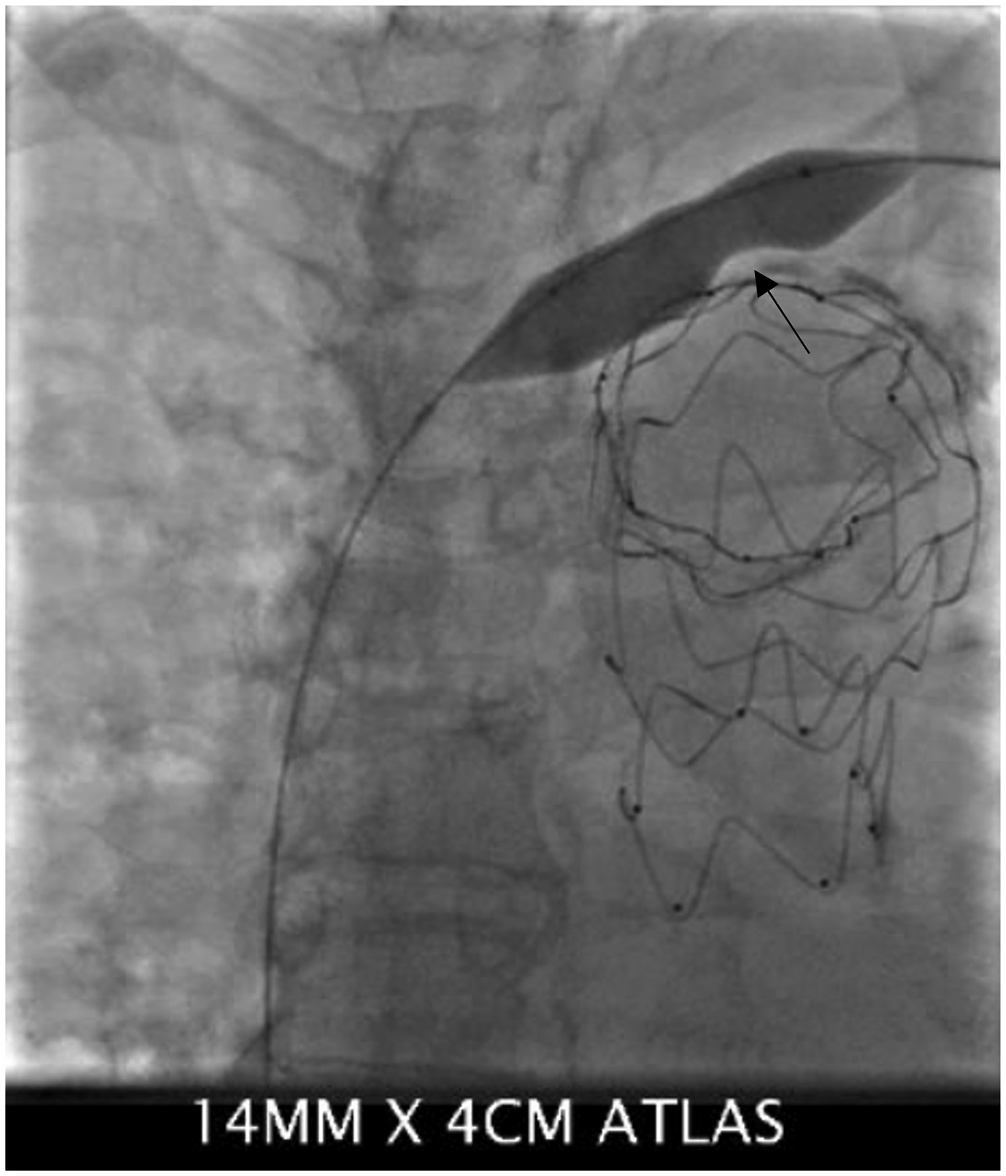
After extensive discussion with the patient, he opted for permanent dialysis through a tunneled dialysis catheter instead of ligation of his RC AVF and re-siting a new vascular access elsewhere.
Discussion
The left brachiocephalic vein joins the right brachiocephalic vein to form the superior vena cava on the right side of the upper chest. The normal course of the left brachiocephalic vein is obliquely downwards and to the right of the upper chest, passing anterior to the aortic arch and its major branches. 1
The incidence of CVS ranges from 9% to 51%, depending on patient selection, with the use of tunneled dialysis catheter identified as an independent risk factor. 2 It is theorized that trauma and inflammation from a catheter placement lead to microthrombi formation, intimal hyperplasia and fibrosis, with the subsequent development of CVS. 3 Computed tomography (CT) venogram remains the gold standard for delineating the anatomy of central veins and to localize the likely lesion. However, peripheral vascular access can be a problem for ESRF patients and renders the injection of contrast difficult. As such, magnetic resonance (MR) venography without contrast may be a better alternative and has yielded equally good results in pre-angioplasty planning. 3
Repeated angioplasty and stenting are common procedures performed in patients undergoing hemodialysis. Frequency of procedures can range from once every month up to every three to six months. 3 Treatment options for CVS include (1) angioplasty and/or stent placement (2) combined endovascular-surgical approach involving hybrid graft-catheter (3) obliteration of the vascular access in the presence of debilitating upper limb oedema. 4
In the current era, endovascular stenting or hybrid procedures combining open supra aortic vessels debranching with endovascular stenting, form the mainstay of treatment in aortic arch aneurysms. 5 Besides endoleak and access site related complications, other major complications include stroke, temporary spinal cord ischaemia and renal injury.6,7 To our knowledge, central vein stenosis from an aortic arch stent graft obstruction has never been reported in the English medical literature.
Left brachiocephalic vein compression by a mediastinal mass, leading to upper limb swelling after arteriovenous graft creation, had previously been reported by Yamaguchi et al. 8 However, our patient differed in that surgical removal of the mediastinal obstruction (aortic arch aneurysm with stent) was not a viable option. The increase in venous return after arteriovenous fistula creation is well recognized. Not uncommonly, the surge in cardiac pre-load can lead to high-output cardiac failure. 9 In our patient, once the cephalic vein stenosis at his RC AVF had been relieved, the increased venous return became the proverbial ‘straw that broke the camel’s back’ as it unmasked the venous obstruction secondary to the aortic arch stent.
Conclusion
Central vein stenosis can usually be ameliorated with repeated venoplasty. However, in our patient, venoplasty to treat the stenosis is not possible due to compression by the aortic graft in a previously repaired aneurysmal aorta. In the subgroup of patients who have had endovascular repairs of aortic aneurysms, it is important to be mindful of adjacent vascular hemodynamics and the possibility of vasculature distortion by the stent grafts. Central venograms may be needed in these patients as part of pre-operative planning before AVF creation and to avoid multiple repeat angioplasty procedures for their vascular access grafts.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Consent
No patient identifiers were used, informed consent was obtained from participating individual included in the report.
