Abstract
Background
Bronchial asthma is characterised by chronic inflammation of both large and small airways. Extra-fine formulation of inhaled corticosteroid (ICS) can achieve a higher peripheral lung deposition leading to improved asthma control. We investigated the effect of extra-fine and fine particle size inhaled ICS with airway resistance and severity of symptoms in asthmatic patients.
Methods
Prospective interventional study of outpatient asthmatics in Universiti Kebangsaan Malaysia Medical Centre (UKMMC). Subjects were grouped based on pre-existing inhalers; extra-fine and fine inhaled corticosteroid (ICS)/long acting beta agonist (LABA). Crossover was at week 7 and treatment regime continued for 7 weeks. Subjects were assessed at week 0, 6 and 13 using asthma control test (ACT) score, forced expiratory volume in 1 s (FEV1) and impulse oscillometry (IOS) Inhaler technique was assessed periodically.
Results
Thirty-four subjects were recruited. Median asthma duration was 20 years. Twenty subjects (58%) in fine ICS/LABA and 14 (42%) in extra-fine ICS/LABA group. Thirty-one subjects were analysed before crossver. There was a significant improvement of FEV1 in the extra-fine ICS/LABA group (1.7 ± 0.47 to 1.88 ± 0.5; p = 0.01). ACT score reduced significantly in extra-fine ICS/LABA group (23 ± 5 to 21 ± 4; p = 0.04) at week 0–6. Week 7–13 showed a significant improvement of ACT score (16.5 ± 3 to 20 ± 3; p = 0.01) and FEV1 (1.71 ± 0.96 to 1.81 ± 1.02: p = 0.01) in extra-fine ICS/LABA. There was no significant difference in (IOS) parameters between the groups.
Conclusion
There was an improvement of FEV1 and ACT score with extra-fine ICS/LABA with no benefit in airway resistance.
Introduction
Asthma is a heterogeneous disease characterised by chronic airway inflammation with variable expiratory airflow limitation. The National Health and Morbidity Survey (NHMS) III in 2006 reported that the prevalence of asthma in Malaysia was 7.1%. 1
The variable expiratory airflow limitation leads to heterogeneity in respiratory symptoms. 2 A thorough history and physical examination are important. The gold standard in asthma diagnosis is spirometry with bronchodilator reversibility test. Performing a spirometry test is not easy as it requires cooperation to perform forceful expiration and inspiratory manoeuvres. 3
IOS is a method which measures airway function using tidal breathing requiring minimal cooperation without special manoeuvre. The principle of IOS is to measure respiratory resistance and reactance by producing multiple-frequency sound waves transmitted into the lung as pressure waves. 4 It is more sensitive than spirometry for detecting small airway disease. 5 The presence of small airway dysfunction evaluated by IOS is associated with poor asthma control.5,6
ICS plays an important role in asthma management. Studies have shown that early initiation of ICS in asthma leads to greater improvement in lung function.7,8 ICS treatment is recommended as early as step 1 since GINA 2019. 7 Different types of ICS have different particle sizes. The mass median aerodynamic diameter (MMAD) is a value used to measure the ICS particles' size. Fine ICS particle is defined by MMAD between 2 μm and 5 μm in diameter. Extra-fine ICS is defined by size less than 2 μm. 9 Particle size of less than 5 μm is more likely to be deposited in bronchial as well as bronchioles with internal diameters less or equal to 2 mm. 9 Different ICS particle sizes lead to a different degree of lung deposition, e.g. fluticasone dry powder inhaler (DPI) with the particle size of 5.4 μm has 15% deposition and ciclesonide hydrofluoroalkane (HFA) with the particle size of 1–2 μm has 52% deposition.6,9
Lung deposition from inhaled medication also depends on the inhalation technique, the type of the inhalers, e.g. pressurised metered dose inhaler (pMDI), DPI, and soft mist inhaler (SMI), as well as types of spacers used.9,10
In a meta-analysis involving 33,453 asthmatics, extra-fine ICS achieved better asthma control with lower exacerbation rates and prescribed doses compared to fine-particle ICS. 11 A prospective study of 30 subjects with mild asthma showed extra-fine ICS to be superior to fine particle ICS in improving airway function and inflammation. 12
IOS is relatively easier to perform than spirometry with good correlation to clinical symptoms and asthma control. We investigate the effect of extra-fine and fine-particle size ICS on airway resistance using IOS and the effect on asthma control.
Materials and Method
Study design
This was a prospective interventional, comparative, crossover study involving asthma patients under follow-up in the respiratory clinic UKMMC between April 2019 and September 2019 involving 34 patients. The crossover study model was used to reduce the number for subject recruitment. Every subject acted as their own control.
Patients aged more than 12 years with a physician diagnosis of asthma who attended the outpatient clinic were recruited if they met the following inclusion criteria: ACT score <20 for subjects on fine particle ICS/LABA and ACT score ≥20 for subjects on extra-fine ICS/LABA.
We excluded the following subjects: presence of comorbidities such as congestive cardiac failure, end-stage renal failure, pulmonary fibrosis, asthmatic with high risk of intubation, inability to perform spirometry despite counselling, poor inhaler technique and asthma/COPD overlap. The study was approved by the Research Ethics Committee, Universiti Kebangsaan Malaysia, FF-2019-232.
Procedure
The demographic data including age, gender, BMI, education level and race were recorded during a clinical interview. The inhaler technique and ACT score were recorded at baseline. Spirometry and IOS were performed.
IOS could measure the respiratory mechanical properties during quiet tidal breathing. IOS also have higher sensitivity in detecting small airway disease and is better correlated with small airway structure. In small airway obstruction, the resistance at lower frequencies increases but is unchanged at higher frequencies that do not reach the small airways. Pressure oscillations generated by a loudspeaker were superimposed onto normal tidal breathing through a mouthpiece for 30–45 s, ranging from 5 to 35 Hz. Resistance values are considered normal within 150% of predicted values.
The asthma control test score was performed in either English or Malay depending on the subject’s preference. Subjects were divided into two groups based on their pre-existing treatment regime: fine particle ICS/LABA Budesonide/Formoterol (160mcg/4.5mcg) Turbuhaler or Fluticasone/Salmeterol (250mcg/50mcg) Accuhaler DPI and extra-fine particle ICS/LABA Beclomethasone/Formoterol (100mcg/6mcg) pMDI of similar steroid potency. They were continued on these medications for a total of 6 weeks.
Subjects were reassessed at the end of week 6 with ACT, spirometry and IOS. Spirometer and IOS were performed by a trained technician using SpiroUSB (CareFusion) and JaegerTM (CareFusion). In this study, IOS was performed followed by spirometry. A crossover was performed between the two groups with no washout period. After crossover, the study was extended by another 7 weeks to reduce the effect of ICS given prior to crossover. At the end of week 13, ACT score, spirometry and IOS were re-evaluated. The inhaler technique was assessed and corrected during each review. Figure 1 summarises the study flow. Consort flow chart.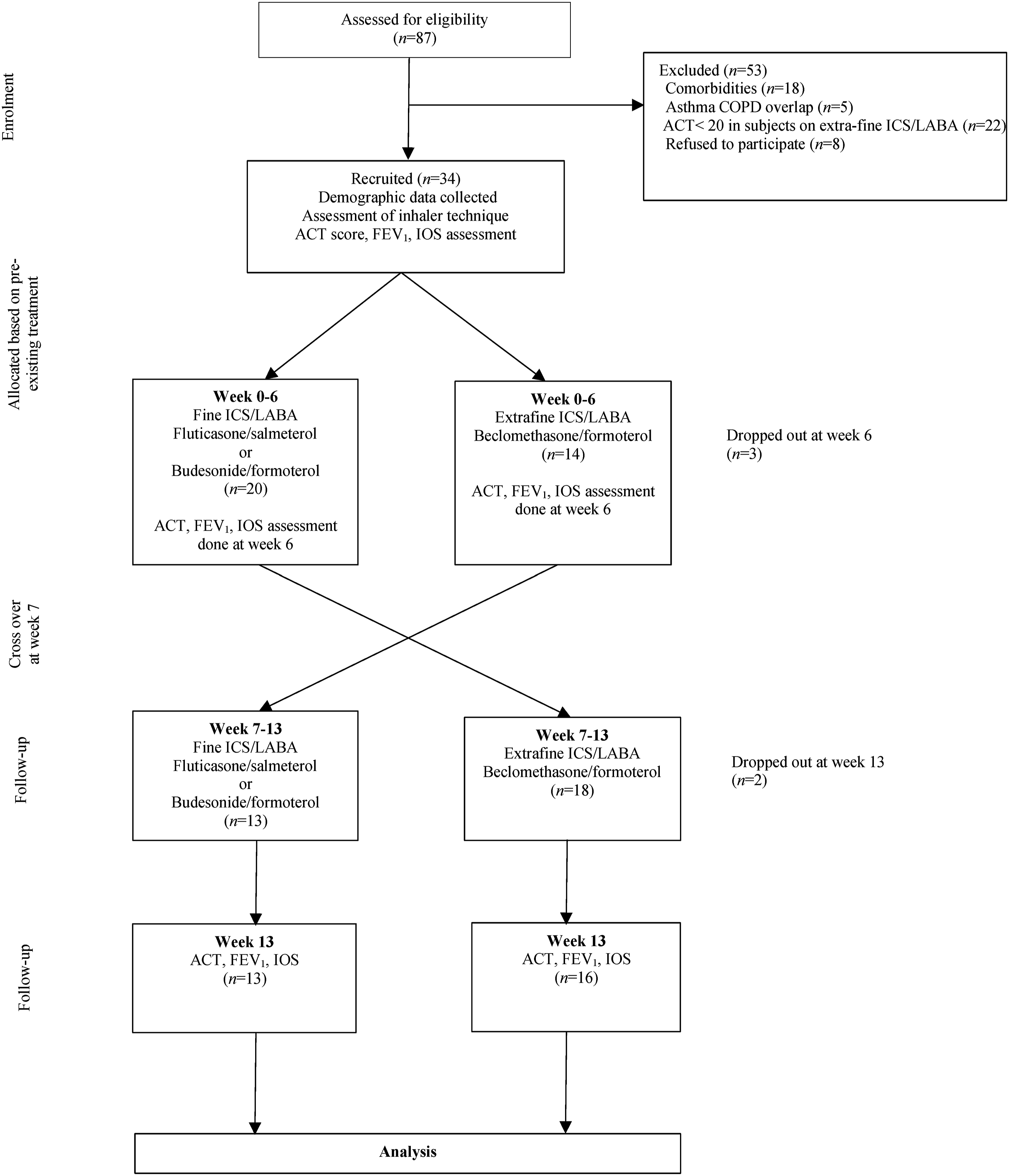
Statistical analysis
Data analysis was performed using Statistical Package for Social Sciences (SPSS) software version 20. The continuous variables were tested with a t-test for normal distribution and a Mann-Whitney U test for non-normal distribution to compare the two groups: extra-fine ICS/LABA and fine ICS/LABA. The categorical data were tested with the Chi-square test and Fisher exact test. The results of the data between the two groups were analysed using independent-sample t-test and its equivalent non-parametric Mann-Whitney U test for parameters with normal and non-normal distribution, respectively. Paired t-test and Wilcoxon signed rank test for non-parametric data were used to analyse data in each group. Correlation between variables was tested using Pearson’s correlation test. Statistical significance was declared when p < 0.05.
Results
A total of 87 subjects were screened. Fifty-three subjects were excluded (Figure 1). Thirty-four subjects were recruited and 29 subjects completed the study. None of the subjects developed asthma exacerbation or any medical adverse effect throughout the study.
The mean age for the extra-fine group ICS/LABA was 52 ± 13 years and 53 ± 13 years for the fine ICS/LABA group. Student t-test showed no significant difference between both groups. The majority of the subjects were females (70%).
Majority were Malays 29 (85%) followed by Chinese 2 (6%), Indian 2 (6%) and Sikh 1 (3%). The highest education level was tertiary 18 (53%) followed by secondary school 14 (41%) and primary school 2 (7%). Using the Fisher exact test, we found no significant difference between these two groups' ethnicity and highest education background.
Baseline demographic data.

Note: aPresented as median (interquartile range).
Data at week 0 and week 6 for extrafine and Fine ICS/LABA group.
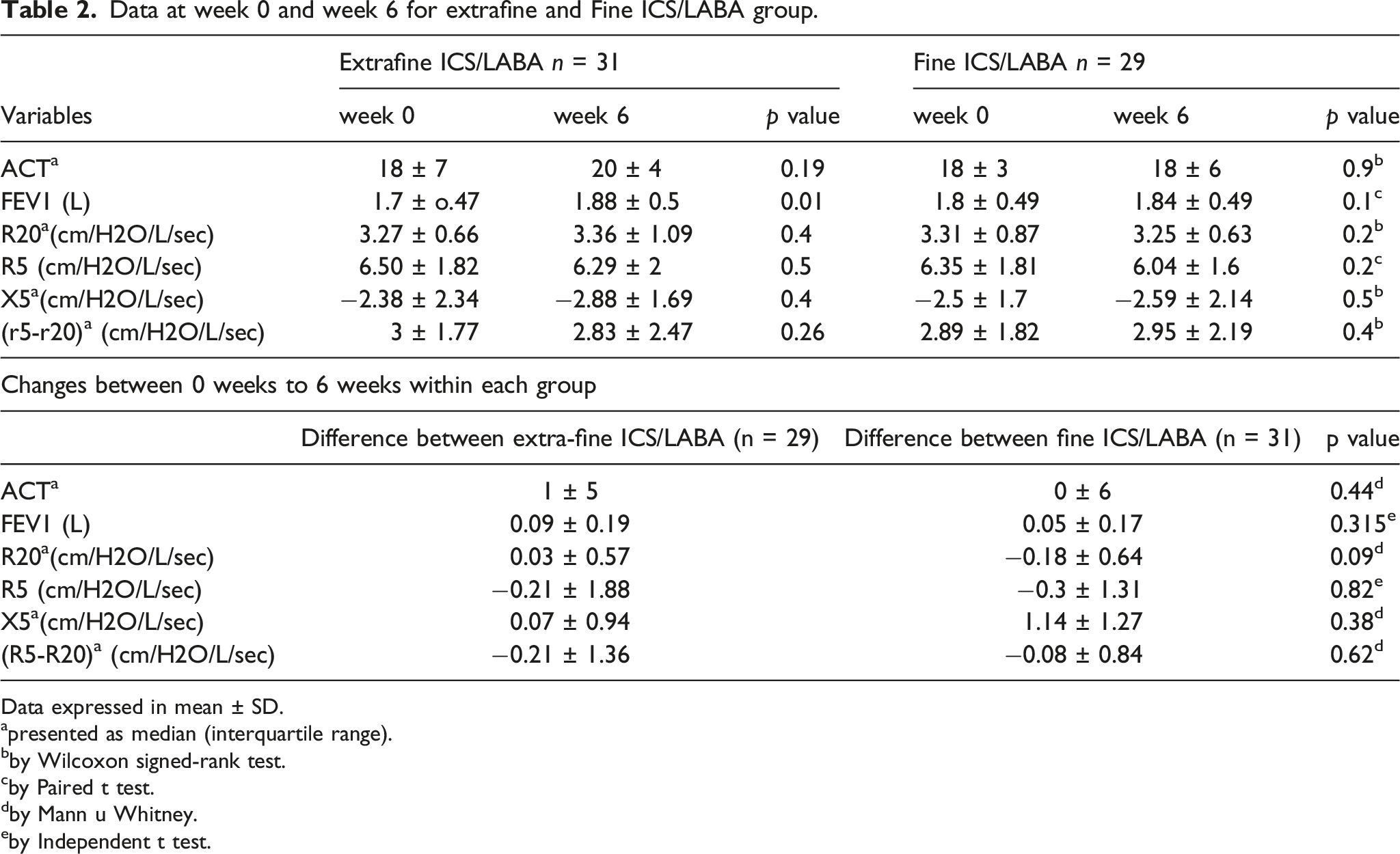
Data expressed in mean ± SD.
apresented as median (interquartile range).
bby Wilcoxon signed-rank test.
cby Paired t test.
dby Mann u Whitney.
eby Independent t test.
Data at week 7 and week 13 for extrafine and fine ICS/LABA (after crossover).
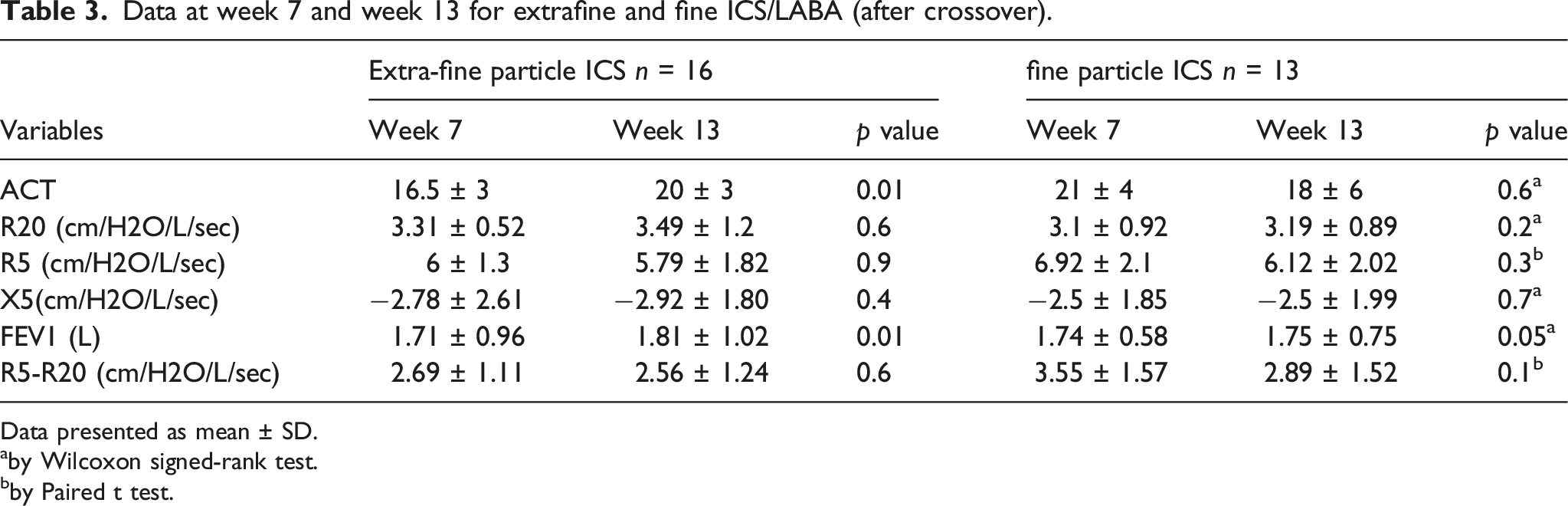
Data presented as mean ± SD.
aby Wilcoxon signed-rank test.
bby Paired t test.
ACT, FEV1, IOS pre (week 7) and post (week 13).

Data presented as mean ± SD.
apresented as median (interquartile range).
bby Mann Whitney U test.
cby Independent t test.
Correlation between ACT changes with variables (week 0 -week sixth).
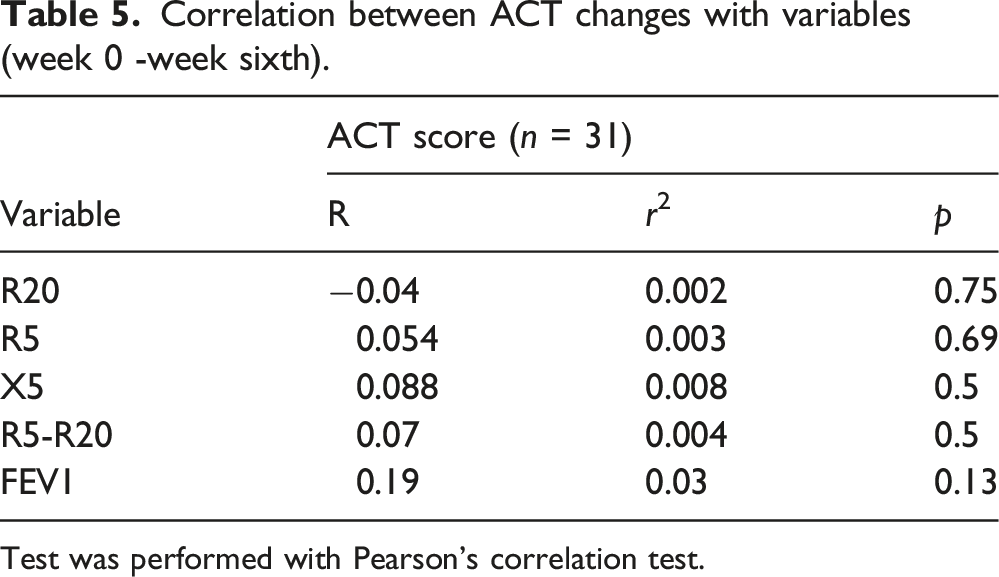
Test was performed with Pearson’s correlation test.
Correlation between ACT changes and variables (week 0 - week 6).

Test was performed with Pearson’s correlation test.
Correlation between ACT changes with variables (week 7 – week 13).

Test was performed with Pearson’s correlation test.
Discussion
To our knowledge, this is the first study done in Malaysia assessing the correlation between extra-fine and fine particle ICS/LABA in asthma control using ACT score, spirometry and IOS. Small airway disease has been related to the control of asthma, severity, and risk of exacerbation. Uncontrolled asthma has been shown to be related to small airway disease and extra-fine inhaled corticosteroid has improved capacity for distal lung deposition. 13
In this study, we assessed asthma control by looking at the effects of extra-fine ICS/LABA and fine ICS/LABA group and assessing the differences between baseline and post treatment between both groups of ACT score, spirometry and IOS. We used an extra-fine ICS/LABA (MDI beclomethasone/formoterol 100 mcg/60 mcg) and fine ICS/LABA (DPI fluticasone/salmeterol accuhaler 50 mcg/250 mcg and turbohaler budesonide/formoterol 160 mcg/4.5 mcg). We recruited asthma patients at step 3 to depict a real-life clinical situation. The ACT scores at recruitment were non-homogenous between the two groups as we wanted to observe the effect after crossover in each group. Weeks 0 to week 6 is a period given to improve the adherence and inhaler technique to reduce the confounding effect.
Analysis of each arm of the crossover sample showed a significant improvement of FEV1 in the extra-fine ICS/LABA group. The percentage of improvement of FEV1 is 10.5% from the baseline. The finding is clinically relevant based on validated MCID which is 230 mL or 10.38%. 14 A 1-year prospective observational cohort study showed similar findings with a significant positive increment of FEV1 with the extra-fine ICS/LABA. 15 Our findings are inconsistent with the systemic review of 23 trials involving adults and children, demonstrating no significant difference in FEV1 efficacy between standard and small particle size. The studies did not include IOS in their parameters. 16 In our study, We found no significant changes in other parameters between extra-fine ICS/LABA and fine ICS/LABA.
We found no significant difference in IOS throughout the study period, between the extra-fine and fine groups. This finding was consistent with a study done by Gafar et al. which showed a similar response between extra-fine and fine ICS. 17 We believe the non-significant changes from the analysis crossover sample for the other parameters were contributed by samples between weeks 0–6 as subjects were on pre-existing ICS/LABA. Thus, any improvement in the first 6 weeks would not be remarkable and will affect the data analysis.
As the crossover samples showed non-significant results in most parameters except for the FEV1, we performed a sub-analysis looking at the changes in each group and comparisons between groups were performed at every phase i.e.; week 0 to week 6 and week 7 to week 13. We observed a significant reduction in the median ACT score in the extra-fine ICS/LABA group from week 0 to week 6 (p = 0.04). The changes in the difference of ACT scores also showed a significant difference with p = 0.02 where a positive difference (increment) was observed in group fine ICS LABA (0.5 ± 2.4) and a negative (reduction) difference was observed in the extra-fine ICS/LABA group (−1.85 ± 2.91). During weeks 0 to week 6, subjects were made to continue with their pre-recruitment ICS/LABA, we believe the significant difference from the analysis was contributed by increased adherence and improved inhaler technique with counselling. Despite a reduction of ACT score with a median reduction of −1.85 ± 2.01 in the extra-fine group, the ACT score at week 6 was 21 ± 4, which was well within the level of good asthma control. The variability of asthma symptoms which gave a variable ACT score in between the 2-time frames may be explained by a reduction in ACT value in weeks 0–6 but still within good control asthma.
Data from week 7 to week 13 (after crossover) showed significant improvement in FEV1 and ACT scores in the extra-fine ICS/LABA group and comparative changes in the difference between the two groups of the ACT scores were significant (p = 0.03). This finding corresponds with the outcome of the study by Hoshino et al. 12 Another study also showed that extra-fine ICS had an impact on improving FEV1.15,18 The significant finding of the FEV1 value does not fulfil the validated MCID for FEV1. 14
Even though there is a significant finding of ACT score in a subanalysis of weeks 0-weeks 6 and week 7-week 13, based on validated MCID ACT score for asthma which is 3, the significant finding of ACT score in extra-fine ICS/LABA in week 7–13 make it more relevant and add-on value in clinical practice. 19
FEF 25-75 showed a significant improvement in both groups and significant changes in comparing both groups in this study. So far, no valid MCID value has been established to make it more significant in clinician practice. 20
This study also gives us a better understanding of the changes seen using different types of ICS preparation. A positive increment of ACT scores and FEV1 in the extra-fine arm with statically significant value was seen when the subjects were changed from dry powder inhaler to metered dose inhaler. This shows that different preparations should be a factor in the selection for patients. A multicentre study involving more than 7300 patients showed that physicians placed more importance on ease of use and only considered device suitability in older patients. 21
We assessed the correlation between ACT scores and quantitative lung function assessment using spirometry and IOS. There was a significant weak positive correlation between the ACT score difference and FEV1 difference in week 6 to week 13 (R = 0.4, r2 = 0,16 p-value = 0.03). In the conducted by Shirai et al., they recorded a similar finding that ACT score had a weakly positive correlation with FEV1 (R = 0.214, p-value = 0.03). 22
This study has several drawbacks. The sample size obtained was relatively small compared to the calculated sample size needed since this study was done in a single centre. Hence the crossover model study was built to overcome this issue as this model requires a smaller sample size than a parallel group trial. This model’s pitfalls include the lack of randomisation and small sample size giving rise to possible selection bias and confounding variables.23,24 A proper crossover model usually has a washout period. Every ICS has a different terminal half-life; for example, the terminal half-life for fluticasone is 7.8 h, and budesonide is 2.8 h 25 In view of that, it would take five terminal half-lives to reach a steady state. 26 Once a steady state is reached, it takes around 2 weeks for the effect of ICS to diminish. This made us choose to prolong the second phase with the aim of reducing the carryover effect due to the non-availability of other ICS/LABA groups as a replacement during the washout period and since it is unethical to stop all medication before initiating another inhaler, no washout period was included in this study thus it might give a carryover effect.
In conclusion, extra-fine particle ICS/LABA can improve ACT scores in patients previously on fine particle ICS/LABA. Extra-fine ICS/LABA leads to improvement of FEV1. There was no extra benefit on airway resistance from the different particle sizes of ICS.
Footnotes
Acknowledgements
We acknowledge the patients and their families who participated in this study and the respiratory lab technicians who performed the spirometry and impulse oscillometry procedures. This study was supported by University Kebangsaan Malaysia with project code FF-2019-232.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the this research received fundamental grant from Universiti Kebangsaan Malaysia (FF-2019-232.)
Authors’ contributions
AA: study protocol, study visits, and first article drafting; SAS, SA: statistical analyses; ABYL,: study protocol, concept; MFH. concept, study protocol, and principal investigator. MSMJ, NN,NBH, AAA: Formal analysis and first draft. All authors revised the article draft.
Ethical approval
Ethical approval was obtained from Research Ethics Committee of the Universiti Kebangsaan Malaysia (research code FF-2019-232)
Informed consent
Written informed consent was obtained from patients involved.
Data availability
The datasheets generated and analysed during the study are available from Dr Asyraf Ahmad
