Abstract
Eosinophilic bronchiolitis (EB) is a rare disease that may mimic or coexist with asthma, but EB typically fails to improve with guideline-based asthma treatments. A 52-year-old man presented with wheezing and shortness of breath for 5 months. He was found to have elevated peripheral blood eosinophils and moderately severe airflow obstruction but did not improve with high-dose inhaled corticosteroids in combination with long-acting beta2-agonist and long-acting muscarinic antagonist. Computed tomography revealed diffuse and widespread “tree-in-bud” changes. Transbronchial lung biopsy demonstrated eosinophilic bronchiolitis. The patient improved with a prolonged course of systemic corticosteroids. EB is distinguished from eosinophilic asthma by the presence of florid bronchiolitis on radiologic imaging or histopathology. The mainstay of treatment is systemic corticosteroids, with a possible role for biologics.
Keywords
Introduction
Eosinophilic lung diseases comprise a diverse group of pulmonary disorders that can be idiopathic or secondary to other aetiologies, such as autoimmune, allergens, drugs, or parasitic infections. 1 Eosinophilic bronchiolitis (EB) was first reported in Japan in 2001 as an atypical eosinophilic lung disease. 2 The patient presented with dyspnoea, peripheral blood eosinophilia, bronchiolitis, and airflow obstruction. Initially diagnosed as having diffuse panbronchiolitis, the patient experienced clinical worsening despite macrolide therapy. A video-assisted thoracoscopic lung biopsy demonstrated diffuse eosinophil infiltration of bronchiolar walls. The patient was then started on systemic corticosteroids which resulted in rapid symptomatic, radiographic, and spirometry improvement.
In 2013, Cordier et al. reported a case series of six patients and proposed a set of diagnostic criteria for the diagnosis of EB 3 : (1) blood eosinophil count >1 G/L and/or bronchoalveolar fluid eosinophil count >25%; (2) airflow obstruction that is not improved by a prolonged course of inhaled bronchodilators and corticosteroids; and (3) characteristic direct signs of eosinophilic bronchiolitis and/or bronchiolitis on lung biopsy and/or high-resolution Computed tomography (CT), including centrilobular nodules, branching opacities, and a tree-in-bud branching pattern. 4
Here, we describe a case of EB and review the existing literature on this rare but emerging condition.
Case Report
A 52-year-old man presented to clinic with a 5-month history of productive cough associated with wheezing and shortness of breath. His symptoms woke him up at night and he reported streaks of haemoptysis twice a day over the previous 2 weeks. He did not have fever, rash, sinonasal symptoms, diarrhoea, abdominal pain, or unintentional weight loss. His past medical history included diabetes mellitus and hyperlipidemia. He denied a past history of asthma, family history of lung cancer, tuberculosis contacts, or recent travel. The patient was working as a bus driver and was an active smoker of 30 pack years. Previously, he was seen by his company doctor who had prescribed high-dose combined inhaled corticosteroids (ICS)/long-acting β2-agonist (LABA), but despite being adherent to the medication his symptoms did not improve. He also did not improve with doses of co-amoxiclav and clarithromycin, nor with the addition of long-acting muscarinic antagonist (LAMA) to his inhaler regimen.
On examination, the patient’s vital signs were within normal limits, including an oxygen saturation of 97% on room air. He was alert and comfortable with no signs of respiratory distress. There was no lymphadenopathy and the oropharynx was clear. Chest auscultation was clear with good air entry, and cardiac and abdominal examinations were unremarkable. No rash, oedema, or clubbing were detected.
The patient had a raised absolute blood eosinophil count of 2.6 × 103/μL, 19.4% of the total white blood cells. Serum creatinine was normal. Serum total IgE was slightly elevated at 268 IU/ml. Anti-myeloperoxidase and anti-proteinase antibodies were negative. Cold agglutinin titer was 1:4. Skin prick testing for house dust mites, Alternaria, cat fur, dog hair, Aspergillus, grass and cockroach were negative. Chest radiography was normal, but chest CT showed widespread centrilobular nodules, many with “tree-in-bud” branching morphology and distal airway wall thickening suggestive of widespread bronchiolitis (Figure 1). Sputum smears for acid-fast bacilli and mycobacterial cultures were negative. Spirometry performed while the patient was on LAMA/LABA/ICS demonstrated moderately severe airflow obstruction with concave expiratory flow-volume loop, FEV1/FVC of 0.55 (lower limit of normal, LLN, of 0.72), FEV1 of 1.56 L or 53% predicted (LLN 2.25 L), and FVC of 2.84 L or 79% predicted (LLN 2.75 L). Chest computed tomography at presentation (left) depicting diffuse centrilobular nodules, some with tree-in-bud configuration, and airway wall thickening. These changes improved after a course of systemic corticosteroids (right).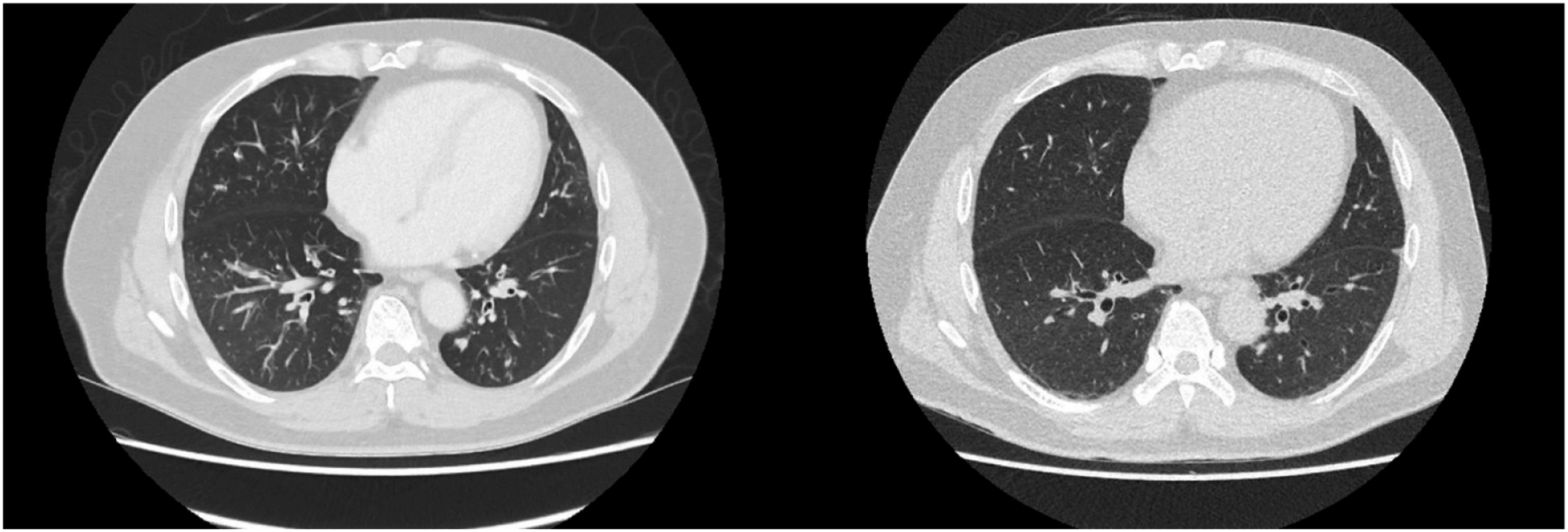
Bronchoscopy was performed and airways were unremarkable. Bacterial culture, acid-fast bacilli, mycobacterial culture, and tuberculosis DNA amplification from bronchoalveolar lavage fluid samples were negative. Transbronchial lung biopsy revealed features of eosinophilic bronchiolitis, with an increased predominance of eosinophils within the bronchiolar mucosa and Charcot-Leyden crystals (Figure 2). No established alveolar fibrous organization, tissue necrosis, diffuse alveolar damage pattern, granuloma or vasculitis was seen. There was negative staining for fungi and acid-fast bacilli. Haematology consult was sought and the opinion was that of isolated bronchiolar involvement with eosinophil-driven organ damage, not consistent with hypereosinophilic syndrome or a clonal blood disorder. Transbronchial lung biopsy specimen demonstrating an increase of eosinophils in the bronchiolar mucosa and the presence of Charcot-Leyden crystals.
The patient was subsequently started on prednisolone 40 mg/day, tapered over six months to 10 mg/day. On follow-up, the patient’s cough and breathlessness improved with systemic steroids, with no further haemoptysis reported. FEV1/FVC improved from 0.55 to 0.81, and FEV1 from 1.56 L (53% predicted) to 3.15 L (108% predicted). Blood eosinophil count decreased from 2.6 × 103/μL to 0.4 × 103/μL. Interval CT showed marked improvement in the centrilobular nodularity. LAMA was discontinued when airflow limitation had reversed, but high-dose ICS/LABA was continued. Further attempts to taper the dose of prednisolone to below 10 mg/day resulted in the recurrence of symptoms. The patient experienced weight gain due to steroids and was offered a trial of biologics for potential steroid-sparing effect, but he declined due to cost concerns.
Discussion
Eosinophilic bronchiolitis was first reported in 2001 in Japan. Since then, reported cases have an apparent confluence in East Asia. However, cases of EB have also emerged in Europe and USA, which suggests that this entity may have a wider geographical distribution and may be under-diagnosed.
Clinical features of patients with reported eosinophilic bronchiolitis, including our present case.
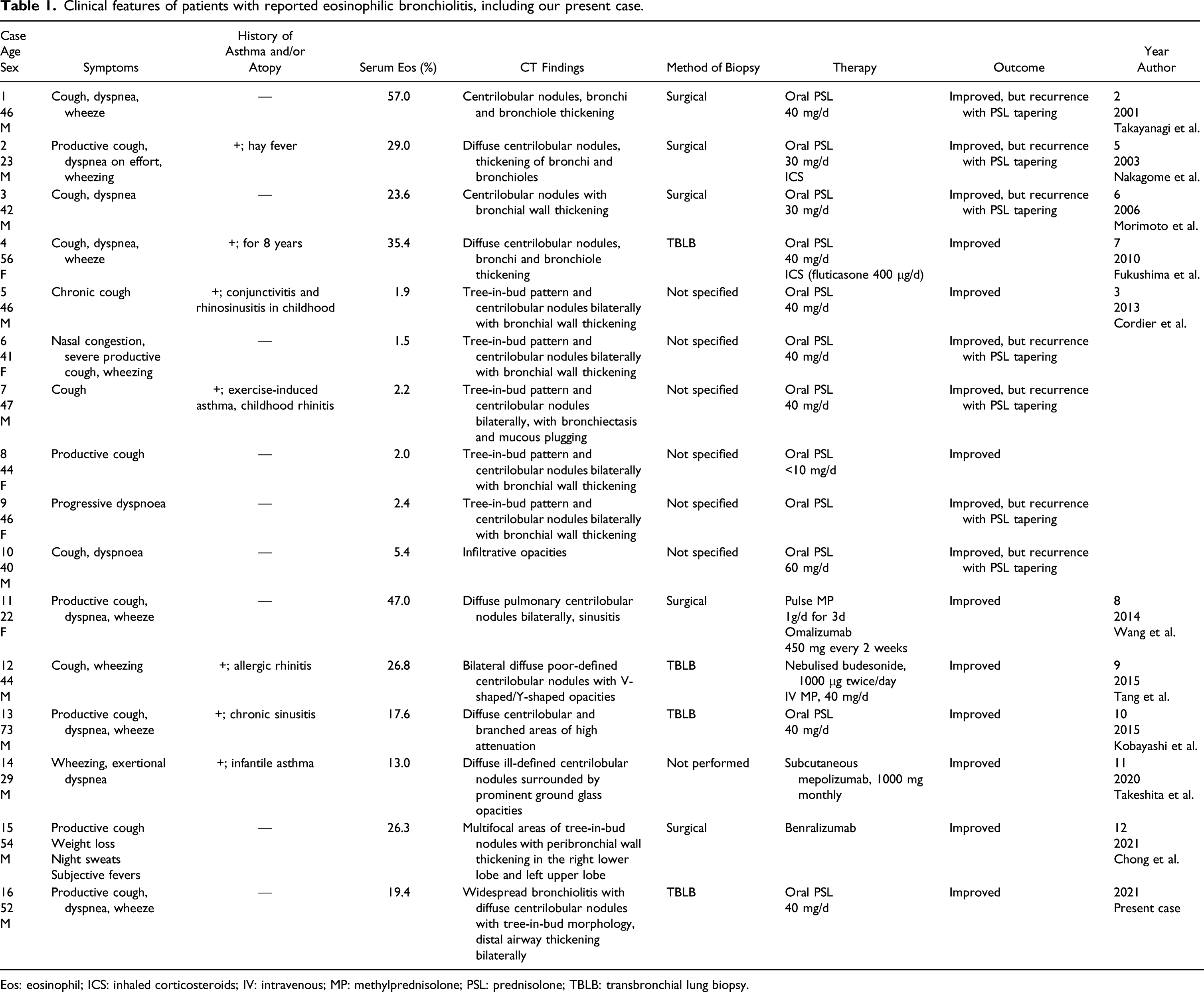
Eos: eosinophil; ICS: inhaled corticosteroids; IV: intravenous; MP: methylprednisolone; PSL: prednisolone; TBLB: transbronchial lung biopsy.
Patients typically present with nonspecific respiratory symptoms, including cough, dyspnoea and wheezing, but extrapulmonary symptoms are notably absent in all cases. There may be a history of atopy or asthma. Airflow limitation may be present on spirometry, but unlike most cases of asthma, does not normalize with high-dose inhaled corticosteroids. EB is also distinguished from asthma by the presence of florid bronchiolitis on chest CT beyond what is usually seen in severe asthma. These radiologic features of bronchiolitis include diffuse centrilobular nodules, tree-in-bud opacities, and thickening of bronchiole walls. Peripheral blood eosinophilia is invariably present. Immunological testing for allergic bronchopulmonary aspergillosis or ANCA-associated vasculitis is characteristically negative. Bronchoalveolar lavage fluid also demonstrates increased eosinophils, and lung biopsy, either bronchoscopic or via video-assisted thoracoscopy, demonstrates the major feature of prominent massive eosinophil infiltration of the bronchioles with accumulation of inflammatory cells in the bronchial lamina.
Differentials include drugs and toxins, eosinophilic granulomatosis with polyangiitis, allergic bronchopulmonary aspergillosis, hypereosinophilic syndrome, and acute or chronic eosinophilic pneumonia. An important differential to exclude is that of a parasitic infection, which can also present with eosinophilia and respiratory symptoms, for example, tropical pulmonary eosinophilia and strongyloidiasis. In particular, administration of systemic corticosteroids in Strongyloides infection could lead to fatal disseminated infection. In the present case, the was a lack of gastrointestinal and dermatologic symptoms, only a modestly elevated IgE level of 268 IU/ml (usually >1000 IU/ml is seen in tropical pulmonary eosinophilia), and an absence of helminthic structures from bronchoscopic specimens. Taken together, these findings indicated that parasitic infection was less likely.
In most of the cases reported in literature, the cause of EB is unidentified. Isolated cases have been reported in association with drug (minocycline), the lymphocytic variant of hypereosinophilic syndrome, and a lung-limited form of eosinophilic granulomatosis and polyangiitis, suggesting that EB is a syndrome that could arise from diverse aetiologies. The reason why the pathology centres on bronchioles/small airways is also unknown. As seen in the patient reported above, EB shares similar clinical features with severe eosinophilic asthma, except for the presence of florid bronchiolocentric disease which is unusual in asthma. This case serves to highlight the challenge of identifying the exact clinicopathobiologic boundaries differentiating EB and severe eosinophilic asthma, that is, whether the two diseases lie on a spectrum or if they are, alternatively, underpinned by fundamentally different biological processes. Pathologic and biological data derived from larger cohorts are needed to elucidate the mechanisms driving small airway-centric eosinophilic inflammation and any intersections with immunologic dysregulation, hypersensitivity, and autoimmunity.
The optimal treatment for EB is currently not well-defined. Systemic corticosteroids have been reported to result in marked improvement in clinical symptoms, lung function, and radiographic findings, with a corresponding decrease in blood eosinophil levels. Symptoms may recur when steroid doses are tapered, and prolonged courses may be required. Biologics targeting eosinophilic or type 2 inflammation have a potential role in the treatment of this condition, as evidenced by anecdotal reports of therapeutic response to omalizumab, mepolizumab, and benralizumab, as seen in cases 11, 14, and 15 respectively.
Conclusion
In conclusion, EB is an emerging condition characterized by peripheral blood eosinophilia, airflow obstruction, CT imaging pattern consistent with bronchiolitis and/or biopsy confirming cellular bronchiolitis which is predominantly eosinophilic. The mainstay of treatment is systemic corticosteroids.
Footnotes
Acknowledgments
None
Author Contributions
All authors have reviewed, edited, and approved the final version of the manuscript.
Availability of Data
Data sharing is not applicable to this article as no datasets were generated or analysed during the current study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not required under HBRA regulations for a case report.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Written informed consent was obtained from the patient for their anonymised information to be published in this article.
