Abstract
Introduction
Single-sided deafness (SSD) is associated with significant functional impairment such as listening speech in noise and localizing sound. To date, studies on the efficacy of CI and BCI for single-sided deafness are conflicting.
Objective
Thus, the objective of our study was to describe and compare the functional outcomes between these devices.
Methods
Thirteen subjects with SSD were prospectively recruited for our study. Six underwent CI and seven received BCI. Word recognition scores (WRS) and disease specific outcome measures were obtained pre-implantation, at 6, and 12 months.
Results
WRS improved both in quiet and in noise for CI and BCI recipients. On the contrary, CI recipients displayed improvement in Speech Spatial Quality (SSQ) scores. A decreasing trend of improvement in APHAB scores were observed for the BCI group.
Conclusion
BCI recipients showed a significant improvement in WRS (in noise). Conversely, CI recipients showed a great improvement in SSQ scores. These preliminary findings suggest that true binaural hearing can only be restored with CI for better SSQ performance. However, BCIs could be recommended to a sub-group of patients, if listening to speech in noise is a priority.
Introduction
Single-sided deafness (SSD) or unilateral severe to profound sensorineural hearing loss is associated with significant functional impairments. Patients with SSD often face difficulties with localizing sound,1–3 loss of head shadow and binaural summation,4,5 leading to poorer speech in noise abilities. 6 SSD is defined as severe to profound hearing loss in the affected ear, with normal or near-normal hearing in the other ear. There are many pathophysiological causes of SSD, but often it is idiopathic and occurs suddenly.7–9
Treatment options include contralateral routing of signal (CROS) hearing aids, bone conduction implants (BCI) and cochlear implants (CI). CIs are implanted in the affected ear and restore binaural hearing after a period of rehabilitation. Conversely, BCIs though implanted on the affected side, transfers sound over to the unaffected ear trans-cranially. As such, true binaural hearing is not restored with the BCI. CI, BCI or CROS devices were shown to improve the auditory ability, sound localization and quality of life (QoL) in patients with SSD in several studies.10–16 Although CROS and BCI are more established treatment options historically, CI as an option has grown in viability in recent times. 17 Past studies have suggested that binaural hearing advantages can be restored even if the hearing asymmetry is not fully corrected.18,19 Thus, the objective of our study was to evaluate the functional outcomes of the CI and BCI on patients with SSD.
CROS and BCI transmit sound signals from the poorer hearing side to the better hearing ear without restoring hearing in the affected ear. Both have been found to significantly reduce the head shadow effect when sound is presented on the poorer hearing side, 20 but their effects on sound localization is not well studied.21–23 CROS systems transmits sound from a microphone on the side of the affected ear to a receiver on the better hearing ear, with newer models having wireless capabilities. 24
In contrast, BCI systems transmit sound through inducing vibrations of skull bone. These vibrations are transmitted from an internally implanted transducer coil to an external sound processor, connected either percutaneously (piercing skin) or transcutaneous (transmitted through skin without direct physical connection).
The Bonebridge (MED-EL) is a transcutaneous BCI that consists of an implanted floating mass transducer and an external processor connected by retention magnets. It is suggested that the transcutaneous Bonebridge (BB) have fewer skin complications, as compared to percutaneous Bone-Anchored Hearing Aid (BAHA). 25 Despite this, recent literature suggests that BB and BAHA both provide audiological equivalent outcomes. 25
Cochlear implantation has been growing in acceptance in the past decade with increasing number of indications for its use in SSD. Its mechanism of action improves auditory nerve stimulation on the affected ear, which theoretically restores physiological binaural hearing with bilateral brainstem stimulation. BCI and CROS systems do not possess this theoretical advantage. A recent systematic review suggests benefit of CI in sound localization, QoL and tinnitus reduction in SSD patients, although the benefit for speech perception in noise is inconsistent. 26
To date, other single-center evaluation studies between BB and BAHA, 25 BCI and CROS,20,27 and CI and CROS/BAHA in the same patient,7,26,28 have been conducted, but there are no direct comparison studies between BB and CI. A randomized controlled trial in the Netherlands started in 2015 (CINGLE-trial) is underway to compare CI to “BCI-then-CROS” and “CROS-then-BCI” although no results have been published at the time our study. 29
Materials and methods
Recruitment
This study was approved by the local Centralized Institute Review Board D (2013/2250). Informed consent was obtained for all participants. A total number of 13 subjects with SSD were prospectively recruited into this single-center study. Six (four males, two females; median age of 49 years; range 38–66) were implanted with CI and seven with BCI (7 males; median age of 22 years; range 20–26). Duration of hearing loss was a median of 10.2 years and 16 years for CI and BCI recipients respectively. For the BCI candidates, Bonebridge with transcutaneous active transduction or percutaneous BAHA were used. In percutaneous BAHA, the skin thickness would have affected power of transduction and reduced power by about 10–15 dB. However, even the smallest BAHA would allow for contralateral bone conduction up to 45 dB. Whereas for bonebridge, although there is no skin attenuation due to direct bone conduction drive, a hearing loss of up to 35 dB is allowed in the contralateral ear. Despite the differences due to implant types, all patients in this study had a contralateral hearing of less than 25 dB pure tone average (PTA). This would make implant type irrelevant to clinical outcomes.
The recruitment period for the study was from March 2013 to December 2016 in a local Singapore hospital. Randomization was not done because that would have removed the patient’s choice in the patient-centric care approach. As CI truly restores binaural hearing while BCI does not, the exact mechanisms must be clearly explained to the patient for their due consideration, or else this may be unethical. Furthermore, regulatory policy did not allow for financial subsidy for SSD patients. This complete out-of-pocket cost meant that patient’s decision on implant type had to be duly considered. During the recruitment period of this study, no financial subsidies were given as the cost-benefit analyses for SSD then were lacking robustness in evidence. Over the years, local hearing health professionals have been lobbying and appealing for financial subsidies with emerging evidence in the literature. Hence, financial subsidies for SSD patients were only granted by our local governing body as of 1 April 2023, specifically only for BCI.
SSD inclusion criteria was defined by an audiometric severity of at least 80 dBHL in the implanted ear, with better than or equal to 30 dBHL in the contralateral (better) ear. All subjects had either magnetic resonance imaging (MRI) of the internal auditory meatus or computed tomography (CT) temporal bone done for any anatomical abnormalities. Patients who reported sudden SNHL usually undergo MRI to rule out any space occupying lesion, vascular conflict or pathology. While those with established history of progressive hearing loss, may or may not require an MRI. Hence, CT of the temporal bone to check for round window patency will suffice. It is of note that all patients had an unremarkable radiographic imaging.
All patients also underwent pre-implant counseling by the inter-professional team of surgeons, audiologists, and speech therapists before surgery to understand the surgical and aural rehabilitation expectations. Most BCI candidates had childhood onset of progressive hearing loss, while CI candidates had sudden SNHL of mostly idiopathic origins in their adults years. While the difference in onset of hearing loss may be a confounder, the BCI candidates in this study did not have any language acquisition or academic problems throughout their formative educational years. It is well supported in the literature, that most of our language mastery and acquisition is completed by the time we reach age six. Hence, we believe that there is no salient evidence to suggest a congenital hearing loss in the BCI candidates.
Selection of implant protocol
All patients who present with SSD will be seen by an audiologist for hearing aid evaluation. Three intervention options including, CROS hearing aids, BCI and CI will be presented to them. Patients will undergo a trial and demonstration of CROS hearing aids in both quiet and dynamic listening environment during the evaluation. A soft band with BCI will also be used to perform aided speech discrimination in both quiet and in noise with speech signals to the poor ear and noise (+10 dB SNR) to the better ear at 90° azimuth to the patient respectively. This will allow them to appreciate the effects of contralateral routing of sound via digital signals or transcranial conduction. As CI cannot be effectively demonstrated and trialed, patients will be briefly counselled on the mechanisms and indications for CI and be arranged to return for a pre-implant counselling session for further deliberation over CI. All patients will undergo extensive counselling on managing expectations from different devices, financial cost, maintenance, follow-up care and rehabilitation if needed by a trans-disciplinary team comprising of otologists, audiologist, and speech therapists. Some considerations of selection include financial cost as regulatory policies did not allow for cost subsidy in SSD patients then. Furthermore, younger patients who may have a hectic schedule may be more inclined to choose BCI, as it did not require an extensive period of aural rehabilitation. An aided speech discrimination score of less than 50% in the poorer ear was taken as part of the candidacy for CI implantation.
Word recognition scores
Word recognition scores (WRS) (expressed as a percentage of correctly identified words from the Arthur Boothroyd Short Word List) were acquired in both quiet at 60 dB SPL (conversational levels), and in noise at +10 dB SNR (50 dB noise). Speech stimuli was presented on the side of the poorer hearing ear at 60 dB SPL and noise was presented on the side of the normal hearing ear at 50 dB SPL, using free field speakers at 90° azimuth in an acoustic booth.
Speech spatial qualities of hearing scale and abbreviated profile of hearing aid benefit
Functional outcomes measures such as the Abbreviated Profile of Hearing Aid Benefit (APHAB) and Speech, Spatial and Qualities of Hearing Scale (SSQ) were also administered. The SSQ consists of three sections assessing speech understanding, spatial hearing and hearing quality, with a score of 0 (unable to hear anything) to 10 (heard perfectly). 21 A global score is calculated from the mean scores of all responses.
The APHAB consists of four subscales assessing ease of communication (EC), background noise (BN), reverberation (RV) and averseness (AV). Responses to degree of functional impairment are scored on a 7-point ordinal scale transformed to intervals of 99% (always), 87% (almost always), 75% (generally), 50% (half the time), 25% (occasionally), 12% (seldom) and 1% (never). A global score is calculated from the mean scores of the first three categories (EC, BN and RV) both with and without hearing aid. Hearing aid benefit is calculated from subtracting the aided average from the unaided average with the lower score suggesting better benefits achieved. 22
Tinnitus handicap inventory
Participants in the CI arm of the study also completed the Tinnitus Handicap Inventory (THI) as a few patients had significant tinnitus prior to implantation. Tinnitus measurements were only given to the CI group as none of the patients in the BCI group complained of experiencing tinnitus. Severe tinnitus on the visual analogue scale of at least six to seven upon 10 is also an indication for the selection of implant type, as CI is believed to restore true binaural hearing and may help patients with tinnitus perception. The THI is a 25-question screening tool with each question worth four points to give a score of 100 for maximum handicap and 0 for no handicap. It is used as a measure of the impact of tinnitus on activities of daily living. 23 For each variable in this study, measurements were taken at baseline before activation of the hearing aid, as well as at 6 and 12 months after activation.
Statistical analyses
Assumptions of normality and equal variance in the distribution were checked before determining the use of parametric or non-parametric tests. Wilcoxon Signed Ranked tests of Significance were used to determine if there was a significant change in pre and post scores after surgical intervention. A significance level of p < 0.05 was used to determine statistical significance. Mann-Whitney U tests were used to determine if there were significant differences observed in the baseline scores. Bi-variate correlation matrix was further used to elucidate any correlating patterns.
Results
Demographic of recruited participants.

Abbreviated profile of hearing aid benefit (6 months)
Marginal benefit was seen for the subscales Reverberation (RV) = 3.3 and Background Noise (BN) = 12.4 for CI recipients’ at 6 months (Figure 1(a)). BCI recipients showed a significant benefit in RV = 18.8 and improvement in BN at 6 months but not statistically significant (Figure 1(b)). (a) Abbreviated profile of hearing aid benefit (APHAB) subscales for cochlear implant at 6 and 12 months. (b) Abbreviated profile of hearing aid benefit (APHAB) subscales for bone conduction implant at 6 and 12 months. AV: Aversiveness of Sounds; RV: Reverberation; BN: Background Noise; EC: Ease of Communication.
Abbreviated profile of hearing aid benefit (12 months)
No improvements were observed across the four subscales for CI recipients at 12 months (Figure 1(a)). BCI recipients showed a decreasing trend for subscales RV (12.6), BN (8.2) and Ease of Communication (EC) = 6.5 at 12 months (Figure 1(b)).
Speech spatial qualities of hearing scale (6 months)
Outcomes at 6 and 12 months (SSQ).

Speech spatial qualities of hearing scale (12 months)
For BCI patients, global mean SSQ score rose from 63.6% (48.9%–75.8%) pre-implant to 69.4% (55.2%–94.7%) at 12 months. This was, however, not statistically significant. For CI patients, global mean SSQ score rose significantly from 43.6% (30.6%–49.8%) pre-implant to 64.0% (43.9%–72.4%) at 12 months (Figure 2). Speech spatial qualities of hearing (SSQ) at 6 and 12 months between cochlear and bone conduction implant recipients.
Word recognition scores (6 months)
Outcomes at 6 and 12 months (WRS in quiet and in noise).
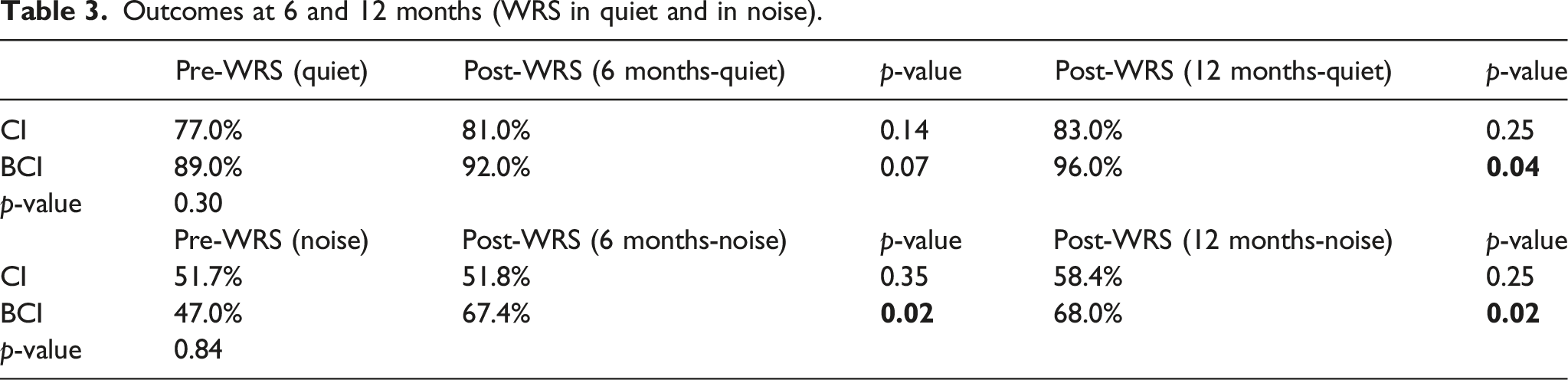
Word recognition scores (12 months)
Average WRS for BCI patients improved significantly in WRS in both quiet (Pre: 89.0% vs Post: 96.0%) and noise (Pre: 47.0% vs Post: 68%) at 12 months (Figures 3 and 4). Although CI recipients showed improvements in quiet from 77.0% to 83.0%, and in noise from 51.7% to 58.4% at 12 months, this did not reach statistical significance. Word recognition scores (WRS) in quiet at 6 and 12 months between cochlear and bone conduction implant recipients. Word recognition scores (WRS) in noise at 6 and 12 months between cochlear and bone conduction implant recipients.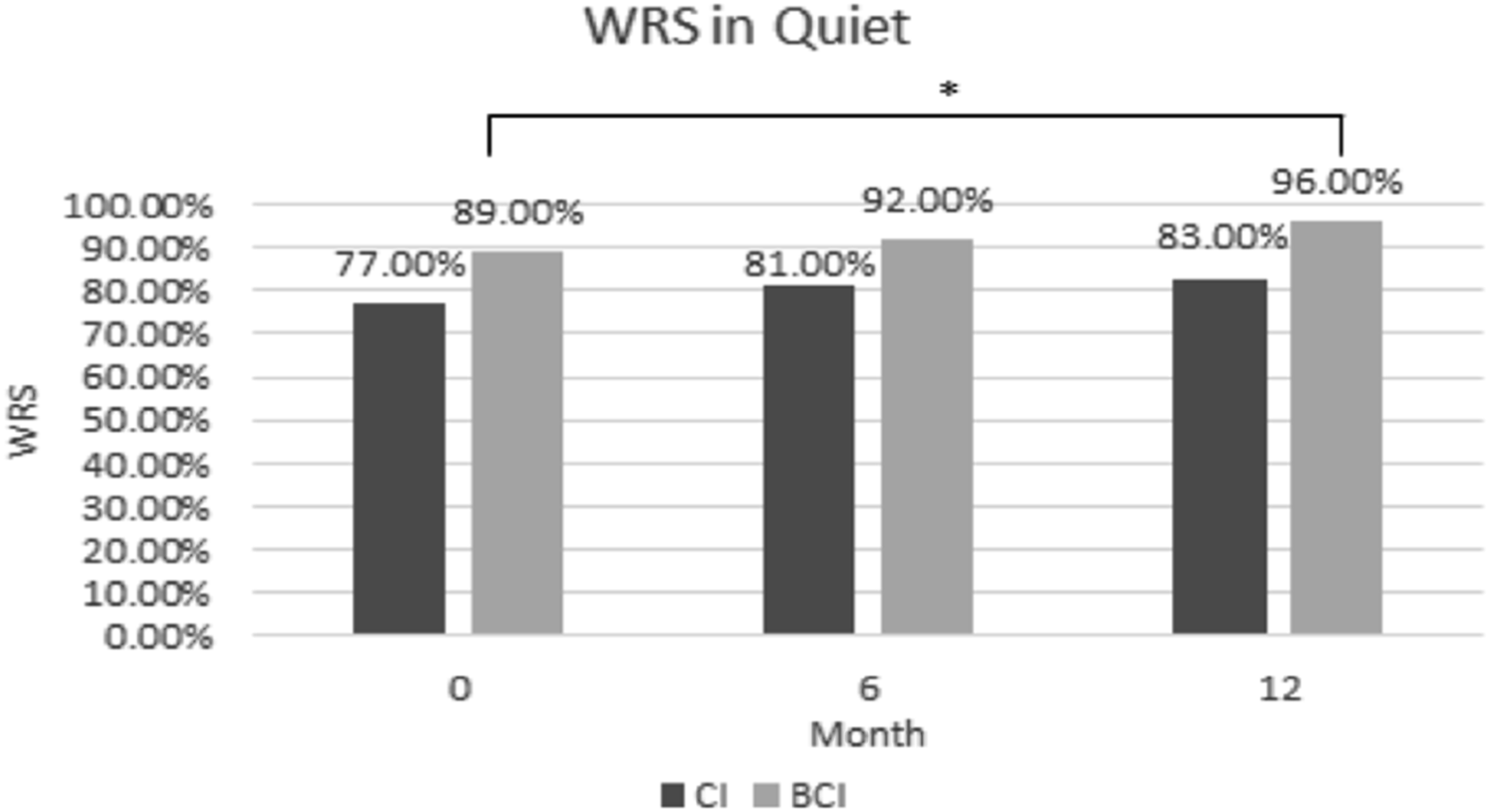
Tinnitus handicap inventory
Additional questionnaires were administered for CI recipients. A general decreasing trend of tinnitus handicap was observed, with a pre-implant mean THI score (scored out of 100) of 48 (range 12–74) falling to 32 (range 10–76) at 6 months and 32 at 12 months (range 12–42) (Figure 5). This observation was marginally significant at both 6 and 12 months (Table 4). Tinnitus handicap inventory scores at 6 and 12 months for cochlear implant recipients. Outcomes at 6 and 12 months (THI).

Discussion
In our study, BCI patients showed significant improvement in WRS in noise and quiet at 6 and 12 months. In addition, BCI patients showed benefits for the subscales RV, BN and EC at 12 months, although not statistically significant. On the contrary, BCI patients showed no significant improvement in SSQ. This is expected as BCI does not restore hearing in the implanted ear and this effect was not observed.
Contrarily, CI patients showed slight improvement in monosyllabic word recognition score (WRS) in noise and quiet at 6 and 12 months. This could be because of binaural interference in the early rehabilitative phase of CI, where the brain is unable to fully process binaural summation and squelch, which is an effect seen from brainstem processing of timing, amplitude and spectral differences in binaural hearing and a release from masking.
Even though the CI recipients were older on average and could have poorer cognitive processing, age alone contributes very little to the variance in CI outcomes,15,16 as does duration of hearing loss. 30 CI recipients also showed significant improvements in SSQ scores at 12 months. This is not surprising as sound localization can only be truly achieved with restoration of binaural hearing through CI.
A possible reason behind this disparity in benefit between the BCI and CI could be a longer rehabilitation period required for CI. However, it remains to be seen whether the difference in benefit decreases with a longer follow-up period.
CI users may require a longer time for neuro-adaptation to their device and restoration of hearing function of the affected ear. In addition to these shared benefits, CI patients also saw improvement in tinnitus handicap over 12 months with a clear reduction in mean THI score over time. Although this is a well-studied effect of CI at reducing tinnitus perception,14–16 it should be noted that participants in this study were followed only up to 12 months. Most studies on tinnitus interventions select participants with more than a year of tinnitus, as it is suggested that the majority of new-onset tinnitus self-resolve within a year. Hence, a longer follow-up period will provide better clarity on the effect of tinnitus suppression with CI in SSD. It is also not uncommon for tinnitus to be more severe post-cochlear implantation as suggested by some studies.31–33
Although WRS should also be performed with different speakers’ orientation (different azimuths), Távora-Vieira et al. 15 demonstrated that post-lingual SSD CI recipients’ speech in noise performance improved regardless of spatial arrangements. Hence, spatial arrangement may not be a limiting factor on WRS. Multi-array speakers were also not used in a laboratory setting to test localization because the SSQ questionnaire would have reflected a more accurate effect of localization.
Age of implantation, duration of deafness, family support, aural rehabilitation and personal motivation are amongst the many factors to be considered for the outcome of CI as well as BCI. Inter-professional collaboration in a trans-disciplinary team comprising of surgeons, audiologists and aural rehabilitation speech therapists play a pivotal role in the CI aural rehabilitation outcomes. 34 It remains to be seen if CI recipients can perform better across all outcome measures as CI recipients may need a longer-term aural rehabilitation. A recent local study published also concurred that SSQ improvements were seen in CI recipients, although not statistically significant. 35 Furthermore, although post-operative WRS increased in their study, it was still not within the serviceable hearing range. 35 Hence, further studies are warranted to compare different implant types available as evidence for good outcomes of CI in SSD are not robust yet.
A systematic review of CI use among SSD patients could not conclude a clear benefit for speech perception in noise, instead describing outcomes as varied, citing 9 out of 11 reports with follow-up periods of less than 12 months (two reports with follow-up ranging from 12 to 24 months). 36 Long-term follow-up studies for CI users with SSD are less common in the literature and hence make it difficult to draw significant conclusions.
Some authors have reported that the benefits from single-sided CI only become more obvious beyond 12 months. Both Mertens et al. 37 and Gartrell et al. 38 observed similar significant benefit in head shadow after 12 months. As for single-sided BCI, the learning curve for BCI use appears to be significantly shorter. Laske et al. 39 followed 9 BCI patients with SSD for 11–22 months and concluded the presence of a learning curve up to 6 months; most audiological benefit seen at 6 months of testing.
Due to the small sample size and heterogeneity in demographics, a direct comparison between the two arms could not be established. In our study, the younger patients (median age = 22 years) tend to choose BCI while the older patients (median = 49 years) tend to choose CI. The younger age of the BCI patients could have contributed to their better scores in both speech and self-report measures. Although patients effectively assigned themselves into each study arm, health professionals’ bias about information provided and portrayal of relative benefits and costs may have contributed to their final decision and cannot be fully accounted for.
A multitude of factors may affect device compliance and vary greatly across patients, including age, gender, expectations, motivation and compliance to long-term rehabilitation programs. 40 At the time of the study, automated data-logging capability was not yet available for most of the CI and BCI sound processors.
Future studies could include the number of hours of device use as a measured variable to account for possible differences in outcomes when most CI and BCI sound processors come with data-logging capability. In addition, patients’ subjective report on hours of use should also be included as not all device have data logging capabilities. In our study, we depended on subjective ratings on the SSQ as we have limited space for the set-up of multiple speakers in different azimuths; which is one of the limitations of our study. It would be ideal in a laboratory set-up to have multiple signals to noise configurations coming from different azimuths for sound localization test. However, it is of note that laboratory results may not always correlate to real-life experiences, hence subjective feedback from questionnaires may provide a more comprehensive understanding of sound localization. The recent results of the CINGLE-trial published in 2021 suggests that CI is beneficial in most domains, including WRS, sound localisation and tinnitus perception as compared to BCI with variable word recognition scores in noise. 41 This is in contrast to our study suggesting better benefits of BCI for word recognition in noise. We acknowledge our small sample size and agree that longer term follow ups are needed to truly understand the benefits of speech understanding (in noise) in both implant groups. What is in concordance is the improvement in subjective speech spatial qualities in our study and the objective sound localisation for CI patients in the CINGLE-trial at 6 and 12 months respectively.
Hearing aids may not be an intuitive option for SSD patients, given that one has to wear hearing aids in the good ear. Since implantable devices are significantly more expensive than hearing aids, stronger evidence of its benefits in SSD are needed to help justify the cost and for policymakers to support subvention. The influence of financial funding on device acquisition should not be underestimated, as revealed in a recent local study. 42
Conclusion
In conclusion, CI appears to reduce tinnitus and improve sound localization. Conversely, BCI appears in this preliminary analysis to be more beneficial for hearing speech in noise and for deriving subjectively greater hearing aid benefit in different listening situations. However, further studies with a larger cohort must be undertaken with age-matched and confounding variables adjusted to establish a direct comparison among implant types available for patients with SSD.
Footnotes
Acknowledgements
The authors sincerely thank all members from the Department of Otorhinolaryngology-Head and Neck Surgery. The authors would like to thank all aural rehabilitation speech therapists who work together with us on the rehabilitation of implanted recipients.
Author contributions
S.W.L. made a substantial contribution to the concept and design of the work acquisition, analysis and interpretation of data, drafted the article and revised it critically for important intellectual content. H.W.Y., D.L., and S.K. contributed to the analysis and interpretation of data, and intellectual input to the manuscript. KC. contributed to the analysis and interpretation of data, and revised it critically for important intellectual content. All authors reviewed and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
