Abstract
Background
CI in SSD strives to restore binaural hearing. With normal acoustic hearing on one ear, the benefits of rehabilitating the deaf ear with an implant are not well established.
Objectives
We investigate audiologic and quality-of-life measures, and long-term usage patterns of cochlear implantation (CI) in patients with single-sided deafness (SSD)
Methods
Eight patients with idiopathic sudden sensorineural hearing loss of less than 5-year duration were recruited. Pure tone average (PTA), speech discrimination score (SDS), hearing-in-noise test (HINT), tinnitus handicap inventory (THI), quality-of-life speech spatial qualities (SSQ) scale tests were performed before, and one-year after CI. Long-term usage of CI four-years post-operatively was determined.
Results
The median PTA of the deaf ear was 96 dBHL (IQR = 90–120) before, and 30 dBHL (IQR = 27–33) after CI (p = 0.0156). SDS improved from median of 0% (IQR = 0–3) to 33% (IQR = 24–58) (p = 0.0360). Median signal-to-noise ratio (SNR), particularly of the S0Nbetter setting of HINT showed improvement from 6.4 dB (IQR = 5.7–7) to 0.9 dB (IQR = −2.25–6.2) (p = 0.1094). Despite median THI improving from 24 (IQR = 6–47) to 4 (IQR = 2–7) (p = 0.1563), two patients experienced worsening of tinnitus. SSQ scores in all subscales showed modest improvement not approaching significance. 5 of 8 (62%) patients stopped using their implant four years after surgery.
Conclusion
Despite improved audiologic and tinnitus outcome measures, our patients’ SDS remained in non-serviceable range, while quality-of-life measures showed only modest improvement. Majority of our patients stopped using their implant four years post-surgery. Our study suggests that objective measurable benefits of CI in SSD may not translate to actual patient perceived benefits.
Introduction
Single-sided deafness (SSD) is a condition where a person has hearing loss of greater than 70 dB in one ear and normal hearing in the other. In comparison to monoaural hearing experienced in SSD, binaural hearing confers multiple benefits. Firstly, interaural time and level differences between the ears enable better sound localization. 1 Secondly, hearing in noise is improved due to the binaural summation and squelch effects. Thirdly, the reduction of the head shadow effect, although not based on binaural sound processing, permits the listener two different signal-to-noise ratios, resulting in improved speech comprehension. 2
Hearing rehabilitation options for SSD include contralateral-routing-of-signal (CROS) hearing aids, bone conduction hearing aids, and cochlear implants (CI). The former two options provide only pseudobinaural hearing, as the brain only receives and processes auditory input from one side. Cochlear implantation, on the other hand, strives to restore true binaural hearing, by centrally integrating electrical and acoustic stimulation. Additionally, purported benefits of CI in SSD include reduced tinnitus distress and improve hearing-related quality of life.3–5
Despite its theoretical benefits, CI in SSD is yet to be considered standard treatment, due to the lack of high-quality evidence, heterogeneity of studies, methodological issues inherent to retrospective studies, small sample sizes, limited follow-up, and varying results.6–9
The primary aim of our study was to evaluate audiometric and tinnitus outcomes, together with quality-of-life measures, in patients with SSD before and after cochlear implantation in our institution. The secondary aim of our study was to characterize their long-term usage patterns over a period of four years.
Methods
A prospective single-arm study was conducted at a single institution with a target sample of 10 patients. The study was designed and conducted in accordance with the Declaration of Helsinki. The study protocol was approved by our healthcare cluster’s Centralized Institutional Review Board (CIRB reference no: 2013/063/D).
Patients with stable SSD were recruited. All patients received a trial of softband BAHA and CROS hearing aid in the clinic as part of pre-operative counseling, before deciding on CI as their rehabilitation option. Inclusion criteria included age more than 18 years old, idiopathic sudden sensorineural hearing loss (SSNHL) as the cause of SSD, with normal hearing in the contralateral ear (four-frequency pure tone average of 500 Hz, 1 kHz, 2 kHz, and 4 kHz not exceeding 30 dB), and duration of deafness not exceeding 5-years duration. These were determined to be idiopathic based on a normal MRI of the internal acoustic meatus, and no other attributable cause of deafness. Only patients with less than 5-years of hearing loss were selected as prolonged auditory deprivation can affect CI outcomes.10,11 Exclusion criteria included patients with inner ear abnormalities, and poor fitness for general anesthesia. All patients provided their written informed consent prior to participating in the study.
All implants were performed by a single experienced surgeon. Cochlear implants were switched-on 3 weeks after implantation. Patients had 7 mapping sessions (at switch-on, followed by at 2 weeks, 4 weeks, 1 month, 3 months, 6 months, and 1 year after switch-on) and 3 auditory rehabilitation sessions (before CI, 1 week after CI activation, and 3 months after switch-on). Patients were given the option of more auditory rehabilitation sessions at their own expense.
Audiologic tests
Pure Tone Audiometry—unaided (pre-operatively) and aided (post-operatively) thresholds—were performed with 50 dB of noise masking the good ear presented through insert earphones. Responses from a free field speaker on the deaf side were measured. Pure tone average (PTA) was calculated from the average of four frequencies (500 Hz, 1 kHz, 2 kHz, and 4 kHz).
Speech Discrimination Score (SDS) was performed on the deaf ear using the Arthur Boorthroyd (AB) word test presented through a speaker at 65 dB. Masking of the non-deaf ear was performed with 50 dB of noise through insert earphones.
Hearing-in-Noise Test (HINT) was performed in three different configurations: (1) S0N0 with speech and noise located directly in front of the subject, (2) S0NSSD with speech in front and noise to the deaf ear 90° to the patient, and (3) S0Nbetter with speech in front and noise to the normal ear 90° to the patient. All speech sound sources were presented 1 metre away from the subject’s head. Scores were reflected as signal-to-noise ratio (SNR), where the higher the SNR, the more difficulty the patient had hearing in noise.
Subjective tests
Tinnitus Handicap Inventory (THI) assesses a patient’s physical, emotional, and functional effects of tinnitus. The score ranges from 0 to 100, with a higher score indicating greater perceived handicap. The score can be categorically reported in five categories of handicap with scores of 0–16 indicating “no or slight handicap”, 18–36 indicating “mild”, 38–56 indicating “moderate”, 58–76 indicating “severe”, and 78–100 indicating “catastrophic handicap”.
Speech Spatial and Qualities of hearing scale (SSQ) is a patient-reported 49-item questionnaire representing quality-of-life related to hearing, commonly used in SSD. It measures perceived hearing ability in different situations using a Likert scale scored from 0 to 10. Responses are averaged, and an overall score, together with three subscales—(1) speech hearing, (2) spatial hearing, and (3) other qualities of hearing (such as segregating sound, recognizing sound, clarity, naturalness and listening effort)—are derived (min 0, max 10). A higher score indicates better hearing perception.
All tests were performed before and 12 months after implantation. Median, together with interquartile range (IQR), was used to report above test scores before and after CI. The changes of these scores before and after CI were compared by means of Wilcoxon rank test. All analysis were done using R 3.6.2 (https://www.r-project.org).
Results
Patient demographics.
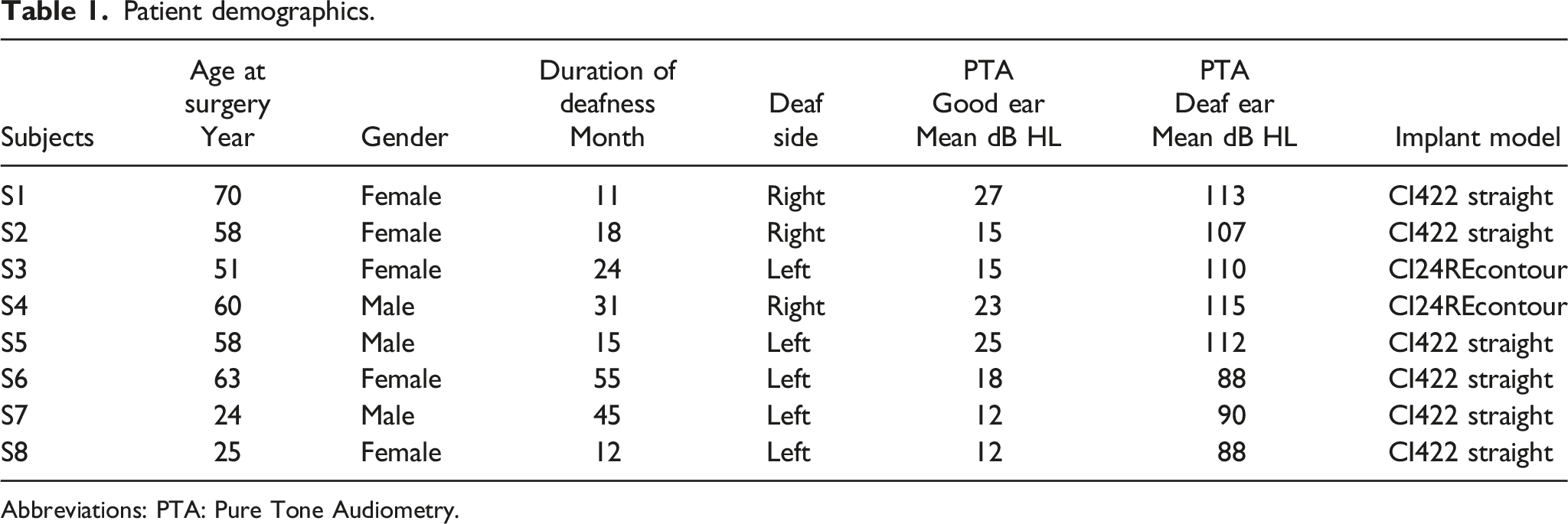
Abbreviations: PTA: Pure Tone Audiometry.
Subject 5’s data was excluded from analysis of audiologic tests and SSQ scores as he stopped using his CI 10 months after surgery due to worsening tinnitus. His THI scores remain included in analysis.
The median PTA of the deaf ear improved from 96 dBHL (IQR = 90–120) before CI, to 30 dBHL (IQR = 27–33) after CI (p = 0.0156). SDS improved from median of 0% (IQR = 0–3) before CI to 33% (IQR = 24–58) after CI (p = 0.0360).
Figure 1 shows the SNR in the 3 HINT scenarios before CI and 12 months after CI. Notably, median SNR in S0Nbetter scenario was 6.4 dB (IQR = 5.7–7) before CI and improved to 0.9 dB (IQR = −2.25–6.2) after CI (p = 0.1094). Hearing-in-Noise Test (HINT). SNR: signal-to-noise ratio. CI: Cochlear Implantation.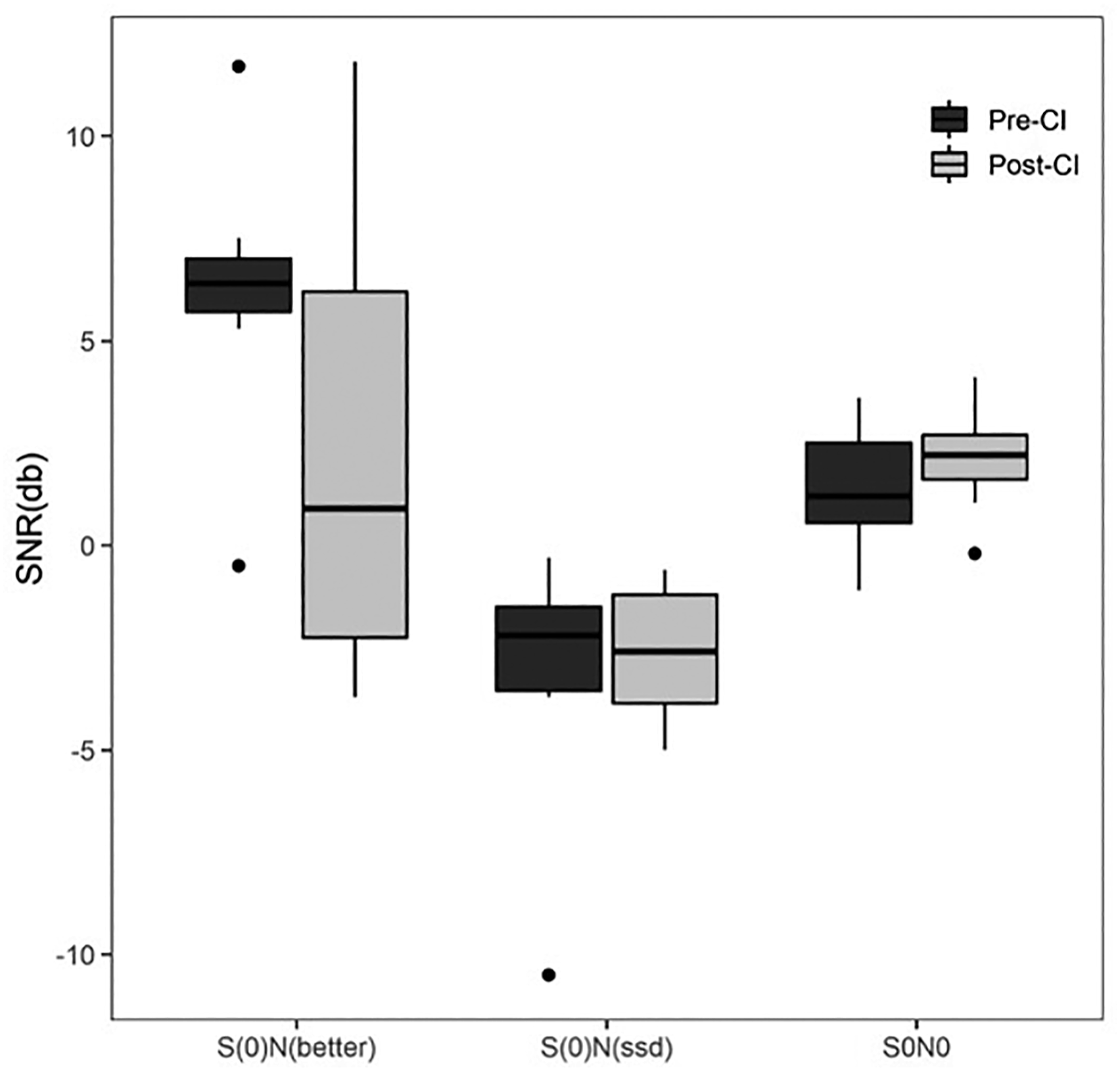
The median THI score was 24 (IQR = 6–47) before CI and 4 (IQR = 2–7) after CI (p = 0.1563). Figure 2 shows individual changes in THI score before and after CI. Tinnitus Handicap Inventory (THI). THI: Tinnitus Handicap Inventory. CI: Cochlear Implantation.
Figure 3 shows the improvement of median SSQ scores following surgery. SSQ overall score improved from a median score of 5.58 (IQR = 4.39–6.31) before CI to 6.21 (IQR = 5.67–7.09) (p = 0.2188). Of the three subscales, the spatial subscale showed the most change in median score from 3.06 (IQR = 2.86–4.87) before CI to 5 (IQR = 4.19–6.4) after CI (p = 0.1563). Speech Spatial and Qualities of Hearing Scale (SSQ). CI: Cochlear Implantation.
Audiological tests, tinnitus handicap inventory and speech spatial qualities scores before and 12 months after cochlear implantation.

Abbreviations: PTA: Pure Tone Audiometry; SDS: Speech Discrimination Score; THI: Tinnitus Handicap Inventory: HINT: Hearing-in-Noise Test; SNR: signal-to-noise ratio; SSQ: Speech Spatial and Qualities of hearing scale; CI: Cochlear Implantation.
More than 4 years following surgery, only 3 out of 8 patients (37.5%) continued using their implants. Five patients (62.5%) became non-users. The first patient stopped using the implant 10 months after the implant was switched-on as his tinnitus became louder. The second patient stopped using his implant as the tinnitus improved post-surgery even without device use and he found no additional benefit with using the implant. The third patient found no perceptible benefit too and stopped using the implant due to follow-up costs concerns. The fourth patient found the cochlear implant sound to be more like noise than speech. The fifth patient was not contactable despite repeated tries and is likely to have moved overseas.
Discussion
Systematic reviews have not shown high level evidence of benefit for CI in SSD due to small sample size and large clinical diversity between studies.7,8
Our results show significant improvement of PTA scores in the implanted ear, with the post-operative median achieving near-normal 30 dB levels. SDS scores showed statistically significant improvement; however, the median score of 33% remains in the level of non-serviceable hearing.
In the largest cohort by Sullivan et al. in 2019, 60 patients with SSD received CI, with a mean follow-up time of 37.9 months. Speech perception was noted to improve to an average of 33% at 6 months, comparable to our study, and remained below 40% at 36 months. Although they described only 4 subjects not using their implants throughout their study, the actual non-usage rate was possibly higher given a lost to follow-up rate of 42% at 24 months. The study nonetheless concluded that binaural benefit and sound localization improved gradually with time after a few years of hearing rehab. 12 Another Asian study performed in Japan with 5 cases of CI in SSD similarly demonstrated fluctuations in hearing results, possibly due to time needed for binaural integration. 13 In comparison, our study provided audiology and subjective tests up to 1-year. We speculate if our test results could have continued to improve over time beyond the duration of this study.
In the HINT results, the S0Nbetter scenario represents the configuration that typically presents the greatest challenge in SSD. In this setting before CI, noise is presented to the better hearing ear, resulting in a high median SNR of 6.4. After CI, the improvement of SNR to 0.9 suggests great functional improvement from the restoration of hearing in the deaf ear. In the S0NSSD scenario, where noise is presented directly to the implanted side, the median score of −2.2 improving to −2.6 suggest that the CI does not cause any worsening of speech intelligibility.
Tinnitus accompanying SSD can have profound effects on one’s quality of life. The use of CI in SSD began with Heyning in 2008 where he found that tinnitus from SSD improved with CI. 14 This benefit in reducing tinnitus may be seen as early as 1 month after CI surgery and can improve with time. 4 In a systematic review of CI in SSD, 34.2% of patients had no more tinnitus, while 53.7% experienced improved, 7.3% had stable, and 4.9% had increased tinnitus. No patients experienced new onset tinnitus. 15
In our study (Figure 2), 3 patients experienced marked improvement (from “moderate” or “catastrophic handicap” to “no or slight handicap”), 3 patients remained stable, while 2 experienced worsening of their tinnitus. Of note, one of the two patients who experienced worsening of tinnitus had his THI score increase from 22 to 80 (from “mild” to “catastrophic handicap”), even while not using the implant, resulting in him discontinuing use of his CI 10 months post-surgery. This patient had pre-existing depression, which together with the worsened tinnitus, caused him to stop using the implant.
Quality-of-life measures better reflect a patient’s perceived health status, encompassing many aspects of their daily life. Meta-analysis found that clinical measures of speech recognition poorly predict patient-reported quality-of-life with CI. 16 Studies have found that CI in SSD can have positive effects on one’s quality-of-life. Häußler et al. found a significant decrease in stress levels, and that depression and anxiety improved after CI.3,5 Common cochlear implant related quality-of-life measures include the Nijmegen Cochlear Implant Questionnaire (NCIQ), and the Speech, Spatial, and Qualities of hearing scale (SSQ). NCIQ looks at three main domains—hearing, social, and psychological, whereas the SSQ looks at hearing in different situations. In studying the long-term outcomes of CI users with SSD, Távora et al. found that the mean SSQ score of patients increased from 4.09 ± 1.58 to 5.68 ± 2.44 post-operatively. 17 Their patients had an average duration of CI use of 5 years (range 4–10 years). We choose to use SSQ as our instrument to measure quality-of-life.
Our median SSQ scores (Figure 3) in overall and all subscales showed global improvement although they did not approach significance in our small sample size. The speech hearing subscale showed marginal improvement, while spatial hearing and other qualities showed more obvious improvements. This suggests modest improvements in quality-of-life after CI.
A study looking at non-users in SSD, found a low non-user incidence of 5 out of 114 (4.4%). 18 In our study, 5 out of 8 (62.5%) became non-users. We postulate that our high non-user rate may be due to cultural reasons. A study in Singapore found that only 7.5% of adults with disabling hearing loss wore hearing aids. 19 This reflects the society’s resistant attitude to improving hearing even with non-invasive means like hearing aids. We also found it challenging to recruit patients due to reluctance to have surgery when they could still hear well on one side. We question if our Asian culture of inter-dependence within the large household would reduce the need for self-reliance, and hence lessen the demand for restoration of hearing.
Aural rehabilitation has been found to improve hearing outcomes for children with cochlear implants, 20 and improve speech outcomes in both children and adults.20–22 The auditory rehabilitation program has also been found to optimize outcomes in adult CI users with SSD. 23 We postulated that more auditory skills training, with goals carried over to the home setting, will improve the hearing outcomes. Two Listening and Spoken Language Specialists Certified Auditory Verbal Therapists provided the necessary rehabilitation sessions for this study.
Our patients received three auditory rehabilitation sessions funded under the study. Further sessions were offered but patients declined, citing a lack of motivation and cost issues. During informed consent to participate in this study, patients were informed there were two therapists who would help them achieve various goals with their cochlear implants over three sessions, and they could have more sessions at their own expense. A local study found that inter-professional collaboration between audiologists, otologists, and speech therapists improved post CI hearing rehabilitation attendance. 24 We informed patients that they could have their auditory rehabilitation sessions together with their CI mapping session in order to reduce the number of visits. However, uptake for extra auditory rehabilitation sessions was still low. Prior to surgery, we also explained that hearing benefits of the implant is dependent on the usage, that is the more the implant is used, the more hearing benefit there would be.
Conclusions
Despite improved audiologic and tinnitus outcome measures, SDS remained in the non-serviceable range, while quality-of-life measure in the form of SSQ showed only modest improvement. Furthermore, majority of our patients stopped using their implant four years after surgery. Our Asian study suggests that improved audiologic measures of CI in SSD may not translate to actual patient perceived benefits. Patients should be appropriately counseled to ensure their expectations of going from monoaural to binaural hearing are not overstated. Cochlear implants in the treatment of single-sided deafness has yet to be established as standard treatment.
Footnotes
Acknowledgements
Mandy Phua and Olivia Wee, our Listening and Spoken Language Specialists who supported the patients in their auditory rehabilitation for this study, and provided valuable comments in the manuscript.
Author contributions
VYJ Tan was involved in literature review, data analysis, writing manuscript, journal submission. BYB Tan was the principal investigator who designed the study, consented patients, and edited the manuscript. PS Leem, D D’Souza, Gopal Krishna S and SW Teng were involved in IRB submission, maintaining data integrity, and editing the manuscript. EZY Zhang was involved in stastistical analysis, literature review and editing manuscript. HH Li was involved in statistical analysis and editing manuscript. B Ong was involved in literature review, maintaining data integrity and editing manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of data and materials
Data is available on request from the corresponding author.
Ethical approval
Ethical approval for this study was obtained from Centralised Institutional Review Board (CIRB reference no: 2013/063/D).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Cochlear Limited and SGH Integrated Fund (GZSHF9004701)
Informed consent
Written informed consent was obtained from all subjects before the study.
