Abstract
Osimertinib is a third-generation thyroxine kinase inhibitor (TKI) used in treating advanced non-small cell lung cancer (NSCLC) patients with epidermal growth factor receptor (EGFR) mutation. Osimertinib is used as a second line treatment for patients who develop EGFR T790 M resistance mutation rather than first line due to financial constraints. We present a case of a 60-year-old Chinese gentleman who diagnosed as stage 4 adenocarcinoma with EGFR Exon 19 deletion. His disease progressed after a year of treatment with afatinib. His plasma T790 M was negative and due to delay in tissue biopsy, he was empirically started on osimertinib. However, in view of financial concerns, he opted a dose reduction of 80 mg every alternate day rather than the conventional dose of 80 mg daily. Serial CT Thorax showed partial response based on RECIST 1.1 criteria and he has 9 months of progression free survival. This case report shows that Osimertinib in reduced dose may achieve good partial response and progression free survival in patients.
Introduction
Non-small-cell lung cancer (NSCLC) is one of the main cancer causing deaths worldwide. Prior to the introduction of genomic EGFR inhibitor therapy, despite platinum-based combination chemotherapy, the overall survival of most patients with advanced non-small-cell lung cancer is less than a year. 1 After the discovery of activating mutations in EGFR, TKIs have been introduced into the first-line treatment of NSCLC with 19 deletion and L858 R mutation. TKI has improved survival of patients with advanced NSCLC with EGFR gene mutations substantially in the last decade. 2
Gefitinib and Afatinib are examples of tyrosine kinase inhibitors approved for the first-line treatment of NSCLC patients with EGFR mutation, while osimertinib is the second line treatment for patients who developed T790 M mutation after failed first generation EGFR TKI. 3
Case report
We present a case of a 60-year-old Chinese gentleman who is a nonsmoker, with a background history of diabetes and hypertension presenting with shortness of breath for a 3-month duration associated with loss of weight and loss of appetite.
Chest radiograph revealed right pleural effusion and CT thorax and upper abdomen showed massive right pleural effusion with a right upper lobe lung mass measuring 3.6 × 5.3 × 6.0 cm in size, multiple right pleural nodules and mediastinal lymphadenopathy (Figure1(a)). Pleural biopsy from pleuroscope showed metastatic adenocarcinoma, TTF1 and CK 7 positive and EGFR mutation testing confirmed Exon 19 deletion. After discussion with patient, he opted oral chemotherapy rather than conventional platinum-based chemotherapy due to logistic issue as platinum-based chemotherapy needs to be administered in hospital-based setting and patient is also worried of its side effects. He was then started on Afatinib since May 2020 with repeated CT thorax in August 2020 showed good partial response based on RECIST 1.1 criteria. Surveillance CT in January 2021 (Figure 1(b)) showed resolved right pleural effusion with reduction of right upper lobe mass. However, a follow up scan in June 2021 showed disease progression with marked enlargement of multiple right lung nodules and presence of new lung nodules (Figure 1(c)). Liquid biopsy for both 19 deletion and T790 M mutation were not detected. (a) CT Thorax upon diagnosis: massive right pleural effusion with right upper lobe lung mass measuring 3.6 × 5.3 × 6.0 cm, multiple right pleural nodule and mediastinal lymphadenopathy. (b) After 8 months of Afatinib: Resolved pleural effusion with reduction of right upper lobe mass more than 30%. (c) After 1 year of Afatinib: marked enlargement of the metastatic right lung nodules, largest 1.8 × 2.2 cm with several new lung nodules, suggestive of disease progression.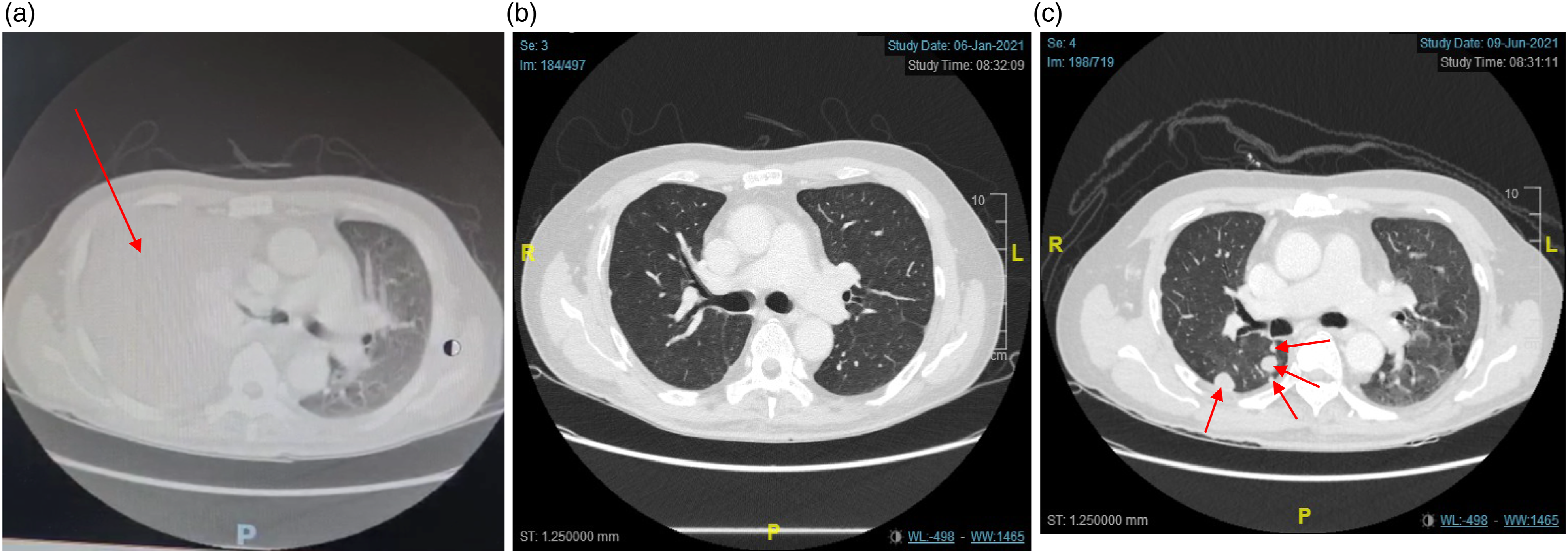
After discussion with the patient, he opted to switch to Osimertinib while waiting for CT guided biopsy. He was taking Osimertinib starting from July 2021, with a reduced dose of 80 mg every alternate day (EOD) rather than the usual recommended dose of 80 mg daily in view of financial constraints.
He was subsequently planned for CT guided biopsy of the new lung nodules for T790 M mutation. However, the procedure was abandoned as bilateral lung nodules have reduced significantly in number and size. Bronchoscopy showed multiple small nodules on both bronchi and repeated HPE showed no malignant cells seen. Repeated CT scan after 4 months of osimertinib in Oct 2021 showed partial response with marked reduction in the size of lung nodules based on RECIST 1.1 criteria (Figure 2(a)). Whereas a follow up CT thorax in Feb 2022 (Figure 2(b)) showed partial response and as of now he has more than 9 months of progression free survival. (a) 4 months post Osimertinib in reduced dose: Marked reduction in right lung nodule. (b) 9 months post Osimertinib in reduced dose: Enlarged right subpleural nodules but rest of the lung nodules has almost resolved suggestive of partial response to treatment.
Discussion
Osimertinib is an oral, third-generation epidermal growth factor receptor tyrosine kinase inhibitor that is used for the treatment of advanced non-small cell lung cancer (NSCLC). Based on AURA trial that includes two treatment naive cohorts with 60 patients treated with osimertinib in first line settings, the results indicate that osimertinib had a deep and durable response to EGFR-mutated NSCLC regardless of the presence of T790 M. 4 The overall response rate (ORR) and median progression free survival (PFS) with osimertinib, across doses were 77% and 20.5 months respectively. 4 However, at current costs, by World Health Organization cost-effectiveness threshold criteria, osimertinib is not cost effective for first-line therapy of EGFR mutated NSCLC. 2 One month of osimertinib will cost around RM 8000 (USD 2000) in Malaysia even with patient assisted programme. If osimertinib is given as second line, the cost is RM 14,000 a month.
Further studies of Phase II extension of AURA trial that involve screening of total of 401 patients with EGFR-mutated NSCLC progressing after first- or second-generation EGFR-TKIs, from 327 tissue samples, 207 were positive for the T790 M mutation and finally 201 patients received osimertinib at the recommended dose of 80 mg. Efficacy results were closed to the phase I data with an ORR of 62%, with no difference between second and third-line or more (62% and 61%), and a disease controlled rate of 90%. The median PFS was 12.3 months.
Consistent with the results of the AURA trial, the FLAURA trial demonstrated that osimertinib was effective regardless of acquired T790 M resistance with an ORR of 80% (95% CI, 75–85%) and PFS of 18.9 months (95% CI, 15.2–21.4 months). Thus, the Food and Drug Administration (FDA) in the United States approved osimertinib for use in first-line settings for EGFR-mutant NSCLC, which now enables the use of osimertinib without testing for T790 M mutation. 4 Based on this available evidence, we can empirically use osimertinib to treat EGFR positive NSCLC patients with disease progression while awaiting for T790 M biopsy results.
In this case report, the pre-existing EFGR mutation and T790 M mutation were not detected in the liquid biopsy which likely due to non bulky disease and tumour heterogenicity. The patient was switched to osimertinib after clinically as well as radiologically proven disease progression after being treated with Afatinib. However, in view of financial constraints, he received osimertinib at a dose reduction of 80 mg every alternate day rather than recommended dose of 80 mg daily. He was started on osimertinib at a dose of 80 mg every other day instead of 40 mg daily as tablet preparation of 40 mg is not available in our facilities and dividing the 80 mg tablet might results in inaccurate dosing. The follow up scans show a good response with his current treatment. This also correlates with AURA 1 which showed good ORR with osimertinib in 20 mg and 40 mg. This indicates that a lower starting dose can be cost saving in developing countries.
From the results of AURA phase I trial, the recommended dose of osimertinib in further trials for first or second line settings was 80 mg once daily. However, there is no significant difference in PFS between two arms: 22.1 in 80 mg vs 19.3 in 160 mg, resulting in 80 mg being the recommended dose for the further clinical trial. In clinical practice, osimertinib also permitted to be decreased to 40 mg once daily. 4 Based on AURA 1 data, ORR by dose for overall population is 43% in 40 mg and 52% in 80 mg. Whereby in T790 M positive patients, ORR is 50% in 20 mg, 59% in 40 mg, 70% in 80 mg and 61% in 160 mg. However, there is no PFS from the study.
This case report also correlates with previous study done by S. Sonobe, Y. Taniguchi and team that the efficacy of osimertinib was maintained after dose reduction in treating patients with T790 M positive advanced non-small-cell lung cancer. 5 In this study, which involves twenty-nine consecutive patients, dose reduction was done due to adverse events of regular dose of osimertinib.
Conclusion
Osimertinib in dose reduction at 80 mg EOD may be effective in treating patients with EGFR mutation at Exon 19 based on the case scenario above. It is also can be used as an empirical therapy after failure of first or second generation TKI if biopsy is delayed.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
