Abstract
Floor of mouth lesions in the paediatric population are uncommon. The spectrum of pathology that afflicts the floor of mouth spans inflammatory conditions, developmental anomalies, vascular malformations and benign tumours or malignancies. We report a rare case of Gardner-associated fibroma (GAF) presenting as a slow-growing floor of mouth mass in a 10-year-old boy. GAF is associated with Gardner’s syndrome (GS) and familial adenomatous polyposis (FAP), both of which are associated with multiple colonic polyps and increased risk of colorectal malignancy. To our knowledge, this case report represents the first case in the literature of a GAF presenting in the floor of mouth of a paediatric patient, and discusses the clinical implications of this rare diagnosis.
Introduction
The floor of mouth represents the inferior border of the oral cavity and comprises soft tissues between the medial border of the mandibular body superiorly and the hyoid bone inferiorly. Specialised anatomical structures and tissues found within include the sublingual and submandibular salivary glands and associated vasculature, musculature (genioglossus, geniohyoid and mylohyoid), lymphoid, nervous structures and connective tissue.
Lesions occurring in this region may affect critical function such as swallowing, speech or airway patency. Computed tomography (CT) or magnetic resonance (MR) imaging of the neck are often employed to better delineate such lesions and aid in work-up of the diagnosis.
Particular to the paediatric age group, diagnostic investigations are challenging due to the lack of amenability of lesions in this anatomical region to fine-needle aspiration cytology. Surgical access is also limited in the smaller paediatric oral cavity, making full extirpation of such masses difficult. Surgical approaches to lesions of the floor of mouth, depending on the pathology of the lesion, include marsupialisation, excision via various surgical approaches (intraoral, transcervical or combined) or sclerotherapy. There have been varying reports of recurrences and surgical complications with these approaches.
We present the first case of a floor of mouth Gardner-associated fibroma (GAF) in a paediatric patient. This rare diagnosis of GAF is a consideration as a differential when approaching lesions in this anatomic location and in the paediatric age group. Patients with GAF may have underlying Adenomatous Polyposis Coli (APC) genetic mutation associated with Gardner’s syndrome (GS) or familial adenomatous polyposis (FAP) and a resultant increased risk of colorectal malignancy. As such, genetic screening for such mutations is advised in patients with GAF.
report
A 10-year-old boy with no known past medical history of note presented to KKH Otolaryngology clinic with a 2-year history of a painless slow-growing swelling at the right floor of mouth. This was first noted by the child and then highlighted to his parents. He was otherwise well with no apparent swallowing, speech or breathing issues.
On examination, he had a smooth submucosal mass at the right floor of the mouth, measuring 2.5 cm in diameter, extending slightly beyond the tongue frenulum to the contralateral side (Figure 1). Bimanual palpation revealed it to be firm, irregular, mobile and extending to the right submandibular space. His ear, nose, throat and cranial nerve examination were otherwise unremarkable, and he had no enlarged cervical lymphadenopathy. Submucosal mass at the right floor of the mouth extending to the contralateral side.
A CT scan of his neck showed an ovoid mass at the right floor of the mouth, centred between the right genioglossus and right hyoglossus muscle, containing soft tissue, fat and coarse calcifications within it and measuring approximately 5.6 × 2.7 × 3.8 cm. It caused midline deviation to the left and displacement of the right mylohyoid muscle inferiorly, as well as right submandibular duct compressive dilation (Figure 2). Initial differential diagnosis was of a dermoid cyst based on clinical and radiological features of the lesion. Coronal view of CT scan floor of mouth showing ovoid mass centred between the right genioglossus and right hyoglossus muscle, containing soft tissue, fat and coarse calcifications within it, measuring approximately 5.6 × 2.7 × 3.8 cm. It causes midline deviation to the left and displacement of the right mylohyoid muscle inferiorly, as well as right submandibular duct compressive dilation.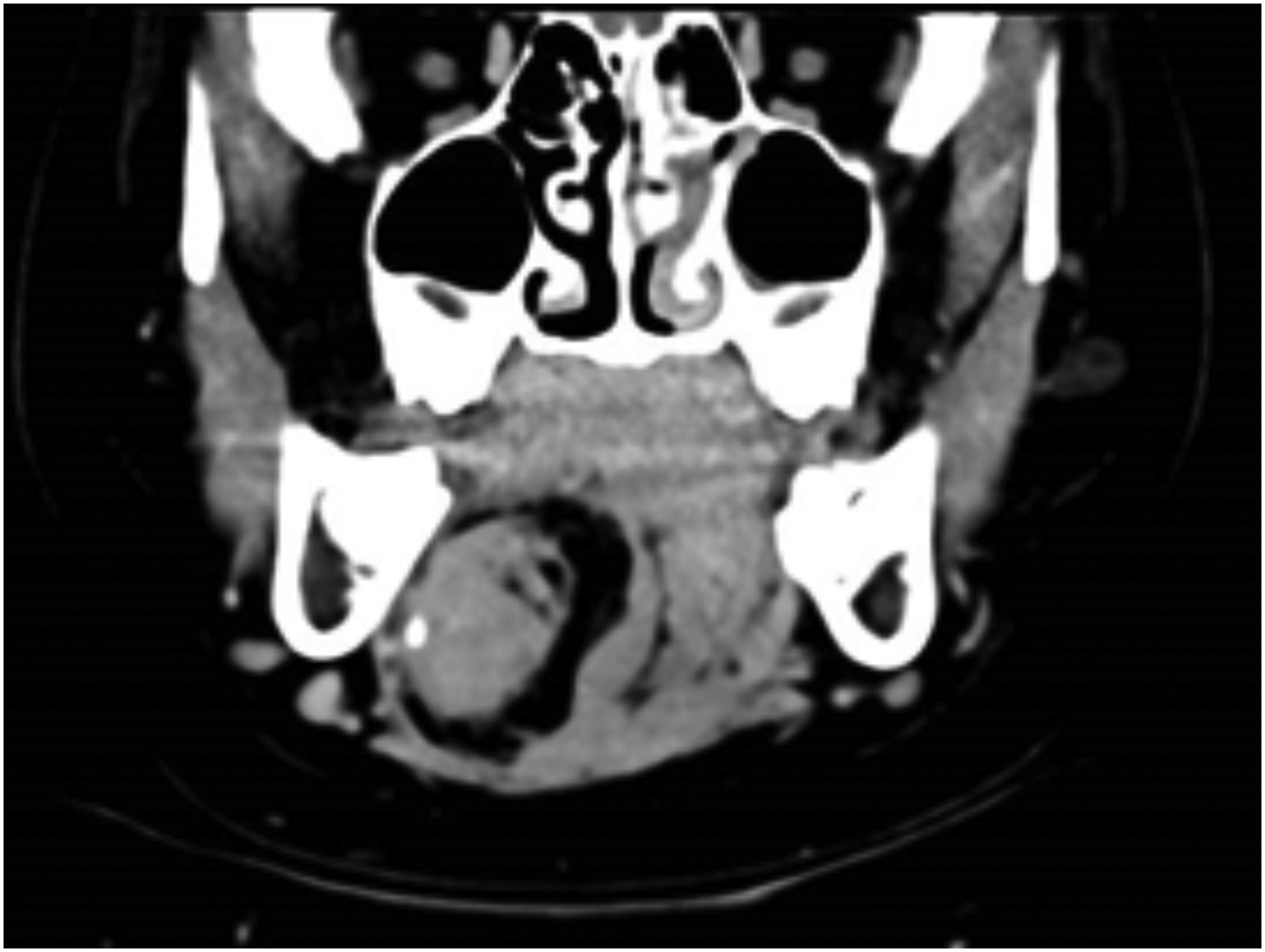
He underwent a transcervical excision of his right floor of mouth mass. This was carried out under general anaesthesia via a right skin crease incision. This approach was opted for given the large size of the lesion (> 5 cm) and concern regarding the ability for complete extirpation of the mass via an intra-oral approach. Endotracheal intubation proceeded smoothly. Access to the lesion involved raising subplatysmal flaps and retraction of the mylohyoid muscle with minimal division of mylohyoid muscle fibres. There was evidence of fibrosis of the mass and adherence to the surrounding muscles, but it was macroscopically completely excised. The marginal mandibular branch of the facial nerve, lingual and hypoglossal nerves as well as the submandibular duct were individually identified and preserved. The mass had a yellowish firm fibrotic appearance (Figure 3). The patient was discharged well on the first post-operative day following removal of the surgical drain which had less than 10 mL of hemoserous output. Intra-operative photo showing right floor of mouth mass being excised via transcervical approach.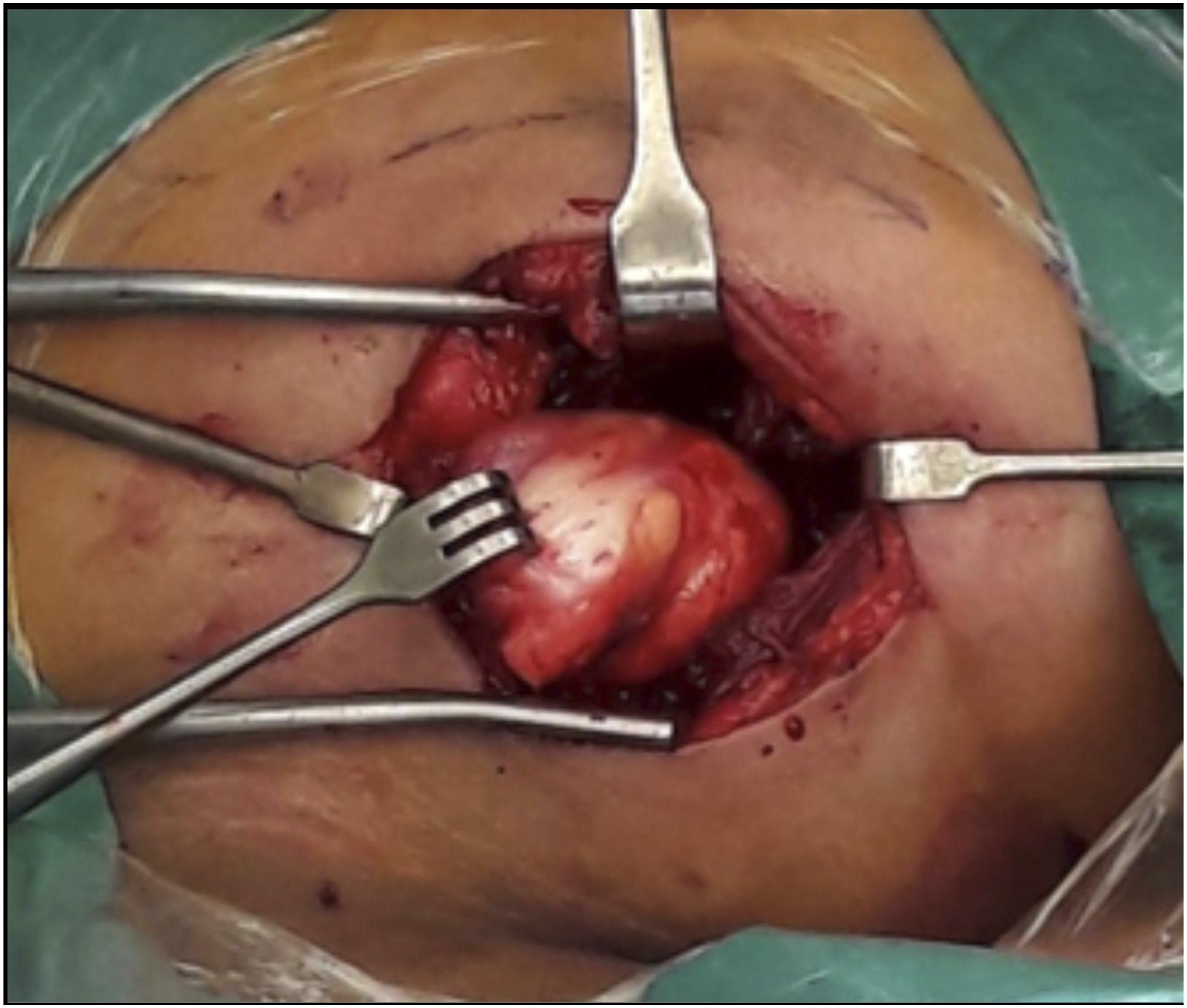
He was well at post-operative review a week and a month following surgical excision of his floor of mouth mass. Initial mild post-operative right floor of mouth oedema had resolved.
Histopathological diagnosis of the lesion returned as a 6 × 5 × 3 cm lesion with a cut section showing a tan white and whorled appearance with scattered fatty tissue and calcification. Microscopically, the lesion comprised thick, haphazardly arranged collagen bundles which infiltrates and entraps neighbouring adipose tissues and skeletal muscle bundles. The collagen bundles contained low-to-moderate number of spindle cells without significant atypia. There were areas of osseous metaplasia and calcifications. Spindle cells showed positive staining for CD34 and negative staining for SMA and Desmin. Cyclin D1 was positive. Rare cells showed positive staining for C-MYC immunostain. Occasional cells showed nuclear beta-catenin immunoreactivity. FISH assay for MDM2 amplification was negative. These findings were consistent with a diagnosis of Gardner-type fibroma.
A detailed personal and family history of desmoid tumours or colonic malignancies was enquired after and this was negative. Referral for further genetic counselling was declined by the patient and family. The patient is on continual clinical surveillance for local recurrence or development of other tumours of gut symptoms.
Discussion
Floor of mouth lesions in the paediatric population are uncommon. More commonly reported conditions affecting the floor of mouth in children include dermoid cysts, ranulas, submental or submandibular lymphadenitis or cellulitis (Ludwig’s angina) and salivary gland tumours or lymphatic malformations. 1 Rarer differentials include ectopic thyroid gland and thyroglossal duct cyst. 2
Gardner-associated fibroma (GAF) is a rare benign fibroblastic tumour and the most common fibroma seen in childhood. They affect males and females equally. GAF presents more frequently in children under 10 years of age, although they have been reported to occur in a large range of ages. 3
Clinically, it may present as a lesion in isolation or as multiple soft tissue fibromas. GAF may occur sporadically. It has also been found to be related to APC genetic mutations in up to 70–90% of cases or desmoid fibromatosis in 18–45% of cases.4,5 Gardner’s syndrome (GS) is a form of familial adenomatous polyposis (FAP) characterised by multiple colorectal polyps and various benign or malignant tumours. People with GS are at high risk of developing colorectal cancer at a young age. Additionally, GS has an autosomal-dominant inheritance pattern.
According to the World Health Organisation (WHO) classification, desmoid fibromatosis is a locally aggressive fibroblastic and myofibroblastic tumour of intermediate malignancy that arises from muscle and fascia, with high propensity for recurrence following surgical excision.3,6
The majority of GAF is located on the trunk, in particular the paraspinal region, and only 15% of GAF have been reported to occur in the head and neck. 3 Our patient is the first reported GAF presenting in the floor of mouth. Pinto et al. 7 reported a retropharyngeal GAF presenting with upper airway obstruction in a 16-month-old.
Fibroblastic and myofibroblastic tumours of the head and neck do not have specific features on imaging investigations; however, imaging provides information such as the size, anatomical extent and invasiveness of such lesions. 8
The majority of fibroblastic/myofibroblastic tumours are benign or of intermediate prognostic categories. They present a diagnostic challenge due to varying clinical presentation and histopathological characteristics of increased extracellular matrix with reduced cellular components. Fine needle aspiration cytology may be of value if taken in the clinical context of patient age at presentation as well as location of tumour and can offer differential diagnoses for such lesions. 9
Macroscopically, GAF is described as a subcutaneous or dermal, poorly defined paucicellular proliferation of stromal fibroblasts, mixed with collagen bundles with evidence of trapped adipose tissue or skeletal muscle. 10 The stromal cells of such lesions are positive for CD34 on immunohistochemistry. Nuclear β-catenin stains are positive in two-thirds of GAF. Biallelic APC inactivation is found in FAP-associated GAF. 4
There have been reports of GAF recurrence after surgical excision and desmoid fibromatosis arising at the same location of up to 50% of excised GAF. 11
There is a high association of GAF with APC germline mutations, and GAF may be a presenting lesion for FAP. In view of this, all patients diagnosed with GAF, along with their parents, are recommended to undergo genetic counselling.4,12
Conclusion
This case report highlights an unusual diagnosis of GAF in an uncommon anatomical location with strong association with APC germline mutations. Fibroblastic and myofibroblastic neoplasms of the head and neck, particularly in the paediatric age group, are rare and a diagnostic and management challenge. Awareness of their clinical behaviour is important in ensuring adequate surgery, involvement of multidisciplinary care such as the geneticist where clinically warranted, and appropriate follow-up.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Singhealth IRB does not require ethical approval for reporting of an individual case.
Informed consent
Written informed consent was obtained from a legally authorised representative(s) for anonymised patient information to be published in this article.
Trial registration
Not applicable.
Availability of data and materials
Data sharing is not applicable to this article as no datasets were generated or analysed during the current study. NOTE: Please do not leave any blank spaces. Appropriate wording can be found in the PoSH declaration policy document
