Abstract
We present a 70-year-old gentleman with chronic cough with haemoptysis found to have left lower zone collapse on imaging. The bronchoscopy showed an endobronchial mass histologically of a non-secreting atypical carcinoid. Poor baseline lung function excluded surgical resection, and instead, he successfully underwent a bronchoscopic debulking procedure which improved his FEV1 to 84% and DLCO 83% predicted. Unfortunately, the tumour regrew, but the calculated percentage predicted postoperative for FEV1 (64%), and DLCO (65%) enabled definitive surgical resection to take place. Surgical resection remains the definite curative option for localised endobronchial atypical carcinoid tumours. Achieving this in proximal endobronchial carcinoid tumour is a challenge. We described an endoscopic tumour debulking procedure as a bridge for curative surgical resection in a patient with an inoperable proximal atypical carcinoid tumour due to poor predicted postoperative lung function. We highlighted the need to assess predicted postoperative lung function with functional and quantitative perfusion methods to aid surgical planning. Debulking the tumour by endoscopy can be used as a conduit to curative surgical resection in bronchial carcinoid tumour. The choice of calculating the percentage predicted values by either or both methods must be individualised based on tumour location and the probability of converting a lobectomy to a pneumonectomy. This precautionary approach could evaluate the postoperative lung function and morbidity and mortality risk if considering a pneumonectomy. Endoscopic debulking can be a successful bridge to a curative surgical resection aided by comprehensive preoperative lung function tests to predict postoperative lung values
Keywords
Introduction
Surgical resection is the definite curative option for localised endobronchial atypical carcinoid tumours. The resection aims to remove the tumour altogether and preserve as much normal lung tissue as possible. However, resecting a proximal endobronchial carcinoid tumour would be a challenge. Patients may instead need functionally debilitating pneumonectomy as intraoperative findings pose technical difficulty performing lesser lung preserving surgery. Therefore, preoperative lung function testing should include both surgical options of pneumonectomy and lobectomy to stratify surgical risk and predict postoperative dyspnoea and morbidity. The operation should only proceed if it is functionally acceptable to the patient’s well-being postoperatively.
We described an endoscopic tumour debulking procedure as a bridge for curative surgical resection in a patient with a functionally inoperable proximal atypical carcinoid tumour. We would also highlight both the functional and quantitative perfusion methods for calculating percentage predicted postoperative values when assessing the patient for surgery, including the debulking procedure and the later decision to completely resect the tumour.
Case report
A 70-year-old gentleman presented with cough for 5 months and was associated with intermittent haemoptysis and breathlessness. He has childhood bronchial asthma with well-controlled symptoms before presentations. Family history of malignancy or previous exposure to tuberculosis not reported. He was an ex-smoker and stopped for more than 30 years with a history of 10 pack-years. He is a retiree and previously had no occupational or environmental exposure to chemical fumes or radiation.
On clinical examination, vitals were stable. Oxygen saturation was 96% on room air. The patient was not cachexia, and finger clubbing was not present. Cervical or axillary lymph nodes were not palpable. Chest examination revealed reduced chest expansion, air entry and bronchial breathing over the left lower zone. Chest radiograph showed a classical sail sign indicating left lower collapse (Figure 1). Sputum for acid-fast bacilli was negative. Initial chest radiograph of our patient showing the typical sail sign of left lower collapse.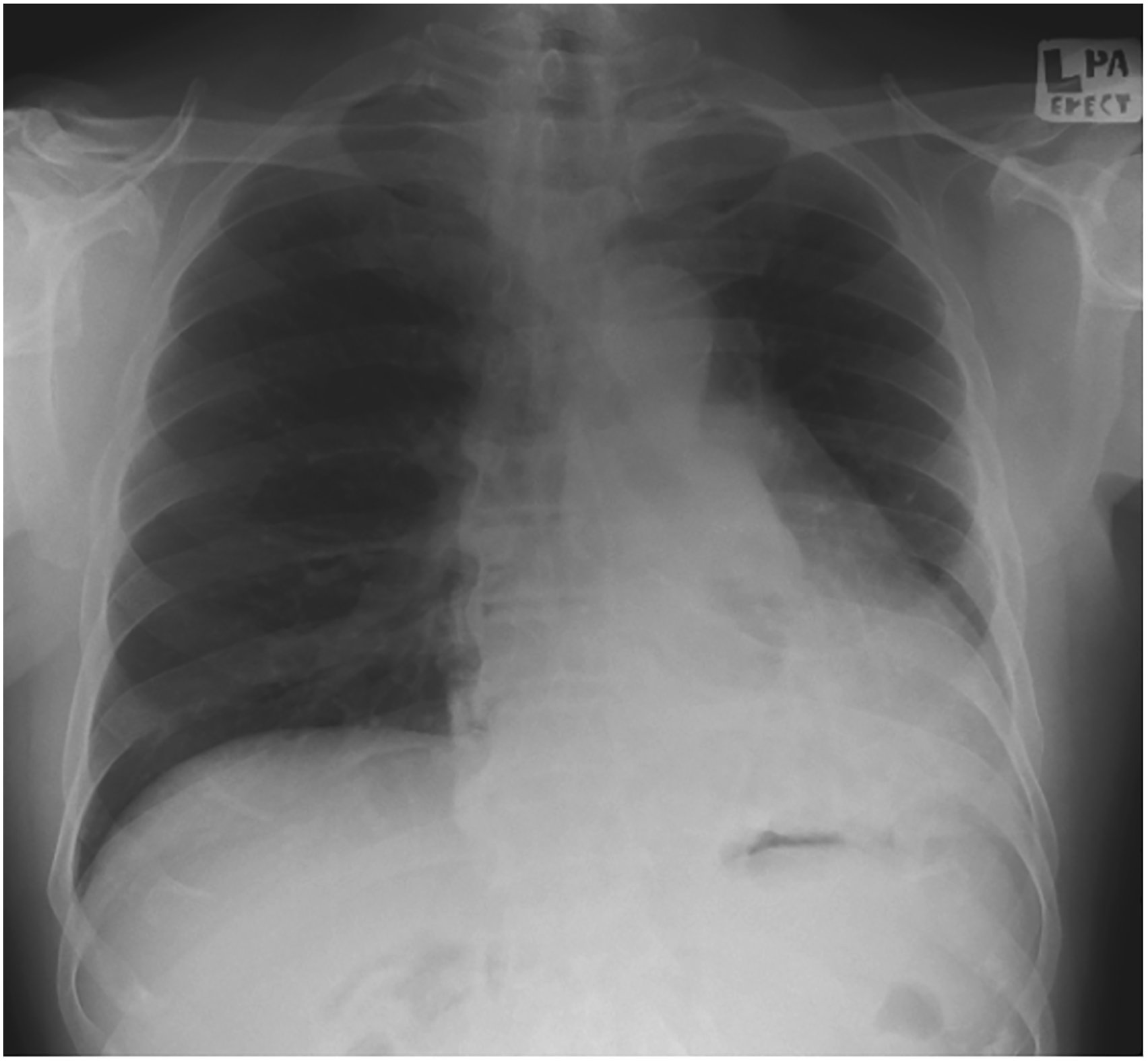
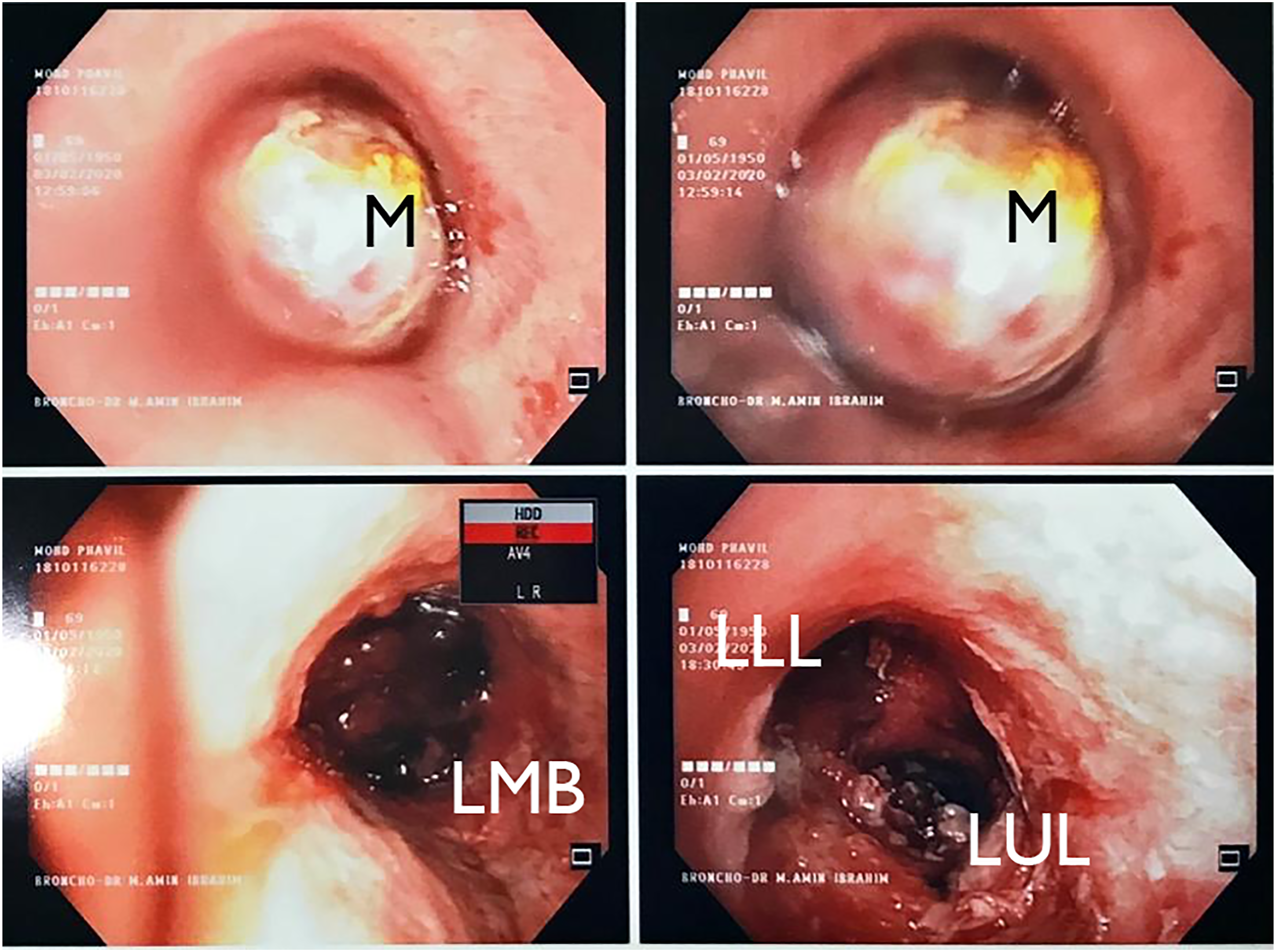
Subsequent CT Thorax confirmed a left endobronchial lesion causing left lower lobe collapse (Figure 2). Subsequent bronchoscopy showed a pedunculated mass at the left main bronchus about 5 cm from the carina and taking multiple biopsies from it (Figure 3). The right lung was unremarkable. Histopathology revealed atypical carcinoid cell and immunohistochemistry testing positive for neuroendocrine markers chromogranin and synaptophysin. Functional scan using Gallium-68 DOTATATE Positron Emission Topography (PET) CT scan showed increased uptake at the left endobronchial mass (SUV max 29.7) measuring 5.6 cm X 1.9 cm parallel to T7 vertebrae extending posteroinferior into the left lower lobe. The scan indicated evidence of a localised somatostatin receptor avid primary disease of the mass. At this point, the diagnosis was an endobronchial atypical carcinoid tumour with the stage of T2N0M0. The urine 5-hydroxy-indole-acetic acid excretion was normal, indicating that this was a non-secreting carcinoid tumour that would not be treatable with medical therapy. Computed tomography lung window view at carina level showing left bronchus mass with patent airways beyond the mass. Lower images showing a patent left main bronchus revealing left upper and lower lobes after tumour debulking procedure. LMB: left main bronchus; LUL: left upper lobe opening; LLL: left lower lobe opening; M: endobronchial mass.
The baseline lung function was prohibitive for any degree of surgical resection with FEV1 of 47% predicted and FVC of 47% predicted. We did not perform a perfusion study in this patient due to limited access to the facility, hence the inevitable waiting time, as it would invariably delay the resection. An echocardiogram showed good cardiac reserve. Instead, he first underwent a bronchoscopic tumour debulking procedure, and the left main bronchus became patent. The procedure was performed via flexible bronchoscopy and snare electrocautery and securing the bleeding of the tumour base by argon plasma coagulation. Post-procedure, his symptoms and exercise tolerance improved. However, at follow-up assessment, his symptoms recurred, and a repeat CT scan, including a bronchoscopy, showed the tumour had regrown and again causing a partial obstruction. The repeat lung function test showed improved FEV1 at 84% and DLCO 83% predicted. The calculated percentage predicted postoperative (%PPO) FEV1 was 64%, and %PPO for DLCO was 65% for left lower lobectomy using functional segment technique. Lung perfusion scan showed an analysis of quantitative perfusion of the left lung was about 23.5%, and the right lung contributed 76.5% of the total pulmonary perfusion. In comparison, quantitative ventilation analysis showed the left lung contributing 21.6%, and the right lung accounted for 78.4% of the entire pulmonary perfusion. Therefore, the calculated percentage predicted postoperative FEV1 was 64.3% and DLCO of 63.5% for left pneumonectomy based on the perfusion values.
Based on the respectable predicted postoperative lung function, the patient underwent curative lung resection via left thoracotomy approach with the initial plan of lobectomy. Intraoperatively, however, the tumour was found to be at the distal left main bronchus close to the branching junction. The inferior medial lower lobe adhered to the aorta, and there was a generalised dense adhesion surrounding the mass. Subsequently, converting the operation to a left pneumonectomy due to the technical difficulty of performing a sleeve lobectomy. Gross histology showed polypoidal intrabronchial nodule 55x30x35 mm with clear surgical margin and no metastasis in 5 hilar lymph nodes. These findings indicated pathological staging 2B (T3N0M0) and achieving curative surgery. Postoperatively, the patient denied undue exertional shortness of breath nor limitation in doing regular daily activity. Postoperative FEV1 was 43.6%, and DLCO manoeuvre was not acceptable due to short breath holding time. We follow up the patient after 4 months of surgery with serum Chromogranin A.
Discussion
Bronchial carcinoid tumour represents a rare type of neuroendocrine epithelial lung cancer. It accounts for 0.5–2.5% of all primary lung tumour and is considered low to intermediate grade malignant neoplasm arising from the surface of the bronchial glandular epithelium. 1 Using The American Joint Committee on Cancer TNM system for staging typical and atypical carcinoid tumours. 2 However, functional syndromes might occur in the setting of bronchial carcinoid tumours. Therefore, biochemical testing including 24-h urine 5-hydroxy-indole-acetic acid and serum Chromogranin A should be assessed,3,4
Surgical resection offers cures for localised atypical carcinoid tumours to preserve normal lung tissue in the process. 5 General follow-up recommendations include a reassessment once between 3–6 months after curative resection, then every 6–12 months for at least 7 years after that with serial serum Chromogranin A and DOTATATE scan to detect recurrence. 6 However, the immediate curative resection was impossible for our patient due to poor predicted postoperative lung function. Poor lung function has shown poor postoperative morbidity, postoperative dyspnoea and long-term survival. 7 In our patient, this could be explained by the CT and bronchoscopy findings of a fungating tumour to the left main bronchus causing total occlusion. At this point, we performed endoscopic tumour debulking as a palliative measure to alleviate the symptoms, and only later that we realised the procedure bridged the possibility of curative resection.8,9 Thus, endobronchial debulking should be reserved for patients who are considered unacceptably high risk for surgical resection or as a possible brdige to surgery.
However, the tumour had regrown and caused partial obstruction found on repeat CT scan and bronchoscopy. Thus, enacting a curative surgery option; in the case of localised disease, the surgical techniques of choice were planned to be lobectomy or sleeve resection based on established literature10,11 where lung parenchymal-sparing surgery should be preferred over pneumonectomy.12,13
The tumour location in our patient was at the distal left main bronchus, which was too close at the branching junction, which posed technical difficulties for lung preserving surgery, and possibly that the patient would end up with pneumonectomy. Alternative to a curative surgical intervention was repeated endoscopic tumour debulking, but this was only palliative and likely needing multiple debulking procedures.
Any patient planned for lung resection surgery should undergo complete lung function testing and quantitative calculation of predictive postoperative lung function to evaluate the mortality and morbidity risks and stratify patients who will benefit from surgery. 14 The formula for calculating %PPO FEV1 and %PPO DLCO is either with the anatomic or perfusion methods. Segment counting is used for lobectomy, while calculating pneumonectomy values is done by the perfusion method. 15 With %PPO values >60%, patients can undergo lung resection. In comparison, patients with %PPO values <60% were tested further by a six-minute walk test, stair climbing test and cardiopulmonary exercise test. 15
The lung function had improved due to tumour debulking, which opened the left main bronchus. Thus, this case presents an exciting finding that an endobronchial resection of an airway blocking tumour could serve as a bridge to definitive resection in patients with an initially poor lung function secondary to blocked airways. However, a postoperative lung function test was performed 1 month after the operation. Therefore, his functional status and respiratory mechanics could not have been fully optimised at the time, and this could have explained his lower than predicted postoperative FEV1.
Conclusion
Utilising endoscopic debulking is a bridge to curative surgical resection in bronchial carcinoid tumour. Patients planned for lobectomy or pneumonectomy should have a quantitative calculation of the percentage predictive postoperative lung function by either the anatomical or perfusion method or both. They must be individualised based on tumour location and the probability of converting a lobectomy to a pneumonectomy. This precautionary approach could evaluate the postoperative dyspnoea and mortality risk if considering a pneumonectomy.
Footnotes
Acknowledgements
The authors would like to acknowledge Univeriti Teknolohi MARA Sungai Buloh for supporting the submission of this case report.
Author’s contributions
M.S.M.R was responsible for analysis and drafting the manuscript. M.A.I was responsible for the images and editing the manuscript. A.A.M.R and M.F.A.R for revision of the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval to report this case was obtained from Universiti Teknologi MARA (UiTM) Sungai Buloh Ethics Committee
Informed consent
Written informed consent was obtained from the patient for his anonymised information to be published in this article.
Availability of data and materials
The images that support the findings of this study are available from UiTM Sungai Buloh, but restrictions apply to the availability of these images, which were used under licence for the current study and so are not publicly available. Images are, however, available from the authors upon reasonable request and with permission of UiTM Sungai Buloh.
