Abstract
Prolactin, a hormone secreted by the anterior pituitary, exists in three major forms in circulation. The macroprolactin form is biologically inactive but contributes to elevated serum prolactin concentrations. Precipitation of macroprolactin by polyethylene glycol (PEG) is widely used in clinical laboratories for the screening of macroprolactin. The aim of this study was two-fold. Firstly, we sought to establish locally relevant reference intervals for serum prolactin and post-PEG precipitation prolactin concentrations in both genders on the Roche Cobas Prolactin II electrochemiluminescence immunoassay. Secondly, the prolactin concentrations after precipitation by PEG dissolved in phosphate-buffered saline (post-PEG(PBS)) were compared with those after precipitation by PEG dissolved in deionised water (PEG(diH2O)). Prolactin concentrations were measured by using the Prolactin Gen II assay on a Roche Cobas e601 analyser. Recoveries of prolactin after precipitation by PEG in PBS and PEG in diH2O were 64.7%–115.8% and 60.9–100.8%, respectively. Post-PEG (PBS) prolactin concentrations were not statistically different from post-PEG (diH2O) prolactin concentrations in either gender.
Introduction
Prolactin is a hormone secreted by the anterior pituitary and exists in three major forms in circulation: monomeric prolactin, big prolactin and macroprolactin.1,2 Macroprolactin is a complex with molecular weight >150 kDa, consisting of monomeric prolactin (approximately 23 kDa) and immunoglobulins. Unlike monomeric prolactin, macroprolactin exhibits little biological activity.3,4 However, macroprolactin molecules, which have longer renal clearance due to their larger size, can accumulate in blood and contribute significantly to serum prolactin concentrations.4,5
The Endocrine Society’s clinical practice guideline on the diagnosis and treatment of hyperprolactinaemia (2011) recommends that screening for macroprolactin be performed in asymptomatic hyperprolactinaemic subjects. 6 In comparison, the Pituitary Society recommends assaying for macroprolactin in a hyperprolactinaemic patient who has moderately elevated levels (25–150 μg/L) and atypical symptoms. 3 Other investigators have recommended routine screening of all hyperprolactinaemic sera for the presence of macroprolactin.7,8 Gel filtration chromatography (GFC) is considered the gold standard method for detecting prolactin variants but the GFC method is costly, labour-intensive and not routinely available in most clinical laboratories. By contrast, precipitation of macroprolactin by polyethylene glycol (PEG) is relatively simple to perform, inexpensive and widely used as an alternative for the screening of macroprolactin in clinical laboratories.
The objective of this study was two-fold. Firstly, we aimed to establish locally relevant reference intervals for serum prolactin and post-PEG precipitation prolactin concentrations in both genders. Secondly, the prolactin concentrations after precipitation by PEG dissolved in phosphate-buffered saline (PBS) were compared with the prolactin concentrations after precipitation by PEG dissolved in deionised water (diH2O).
Methods
To determine their suitability for inclusion in this study, 271 healthy volunteers in Singapore were asked to complete a lifestyle and health questionnaire. Exclusion criteria included recent hospitalisation in the previous month or acute illness in the last 2 weeks, pregnancy or any gynaecological conditions such as irregular menses and polycystic ovary syndrome, any endocrinological conditions, for example, diabetes, thyroid disease, pituitary disorders and other acute or chronic medical conditions, and current intake of prescribed medications including oral contraceptive pills and antipsychotic drugs. A total of 223 adults (146 females and 77 males) were included in this study to establish reference intervals for prolactin. Approval from the central institutional review board (central institutional review board (CIRB) reference number 2017/2339) had been obtained for this study before recruitment of volunteers.
Venous blood was collected into serum gel tubes (Vacutainer SST II Advance; BD Diagnostics, Franklin Lakes, NJ, USA). After collection, blood specimens were centrifuged, and serum was divided into aliquots of 500 μl and stored at −70°C before analysis. Prolactin concentrations were measured by using the Roche Cobas Prolactin II electrochemiluminescence immunoassay on a Cobas e601 analyser (Roche Diagnostics, Mannheim, Germany). PEG solution was prepared by dissolving 1.25 g of PEG 6000 (Sigma-Aldrich, St Louis, Missouri, USA) in 5 mL PBS or deionised water (diH2O). Serum (250 μL) was mixed with an equal volume of PEG solution, vortexed for 1 min and incubated for 10 min at room temperature before centrifugation at 3500 rpm for 5 min. Prolactin was measured in the supernatant. Prolactin results were reported in µg/L (conversion formula: concentration in µg/L × 21.2 = concentration in mIU/L). Results of post-PEG prolactin concentrations were corrected by a factor of two to account for the dilution factor. The methods for preparation of both types of PEG solutions as well as serum treatment with PEG solution were referenced to the Roche Cobas Prolactin II package insert 2015-08 V6 and supported by review of available literature. Within-laboratory imprecision coefficients of variation of the prolactin assay were 0.8%, 1.7% and 1.1% at mean prolactin concentrations of 8.6 μg/L, 28.1 μg/L and 109 μg/L, respectively. The assay’s limit of quantitation is 1 μg/L and the analytical measuring range is 1–470 μg/L (1–4700 μg/L with 10 times dilution with universal diluent). The assay is unaffected by high-dose hook effect at prolactin concentrations up to 12,690 μg/L.
Statistical analyses were performed using the Analyse-it software version 2.03 (method validation edition, Analyse-it Software, Leeds, UK). Before calculation of reference intervals, outliers were detected by inspection of distribution histograms and using the recommendations from the Clinical and Laboratory Standards Institute (CLSI) guideline C28-A3c. 9 Reference intervals (2.5th to 97.5th percentiles) were determined using a nonparametric method based on the recommendations from the International Federation of Clinical Chemistry 10 and the CLSI guideline C28-A3c. 9 Normality of distributions was tested by the Anderson–Darling test. The Wilcoxon test was used to compare the medians of post-PEG prolactin concentrations. p<0.05 is considered statistically significant.
Results
Serum prolactin concentrations (µg/L) in healthy volunteers before and after precipitation by polyethylene glycol.
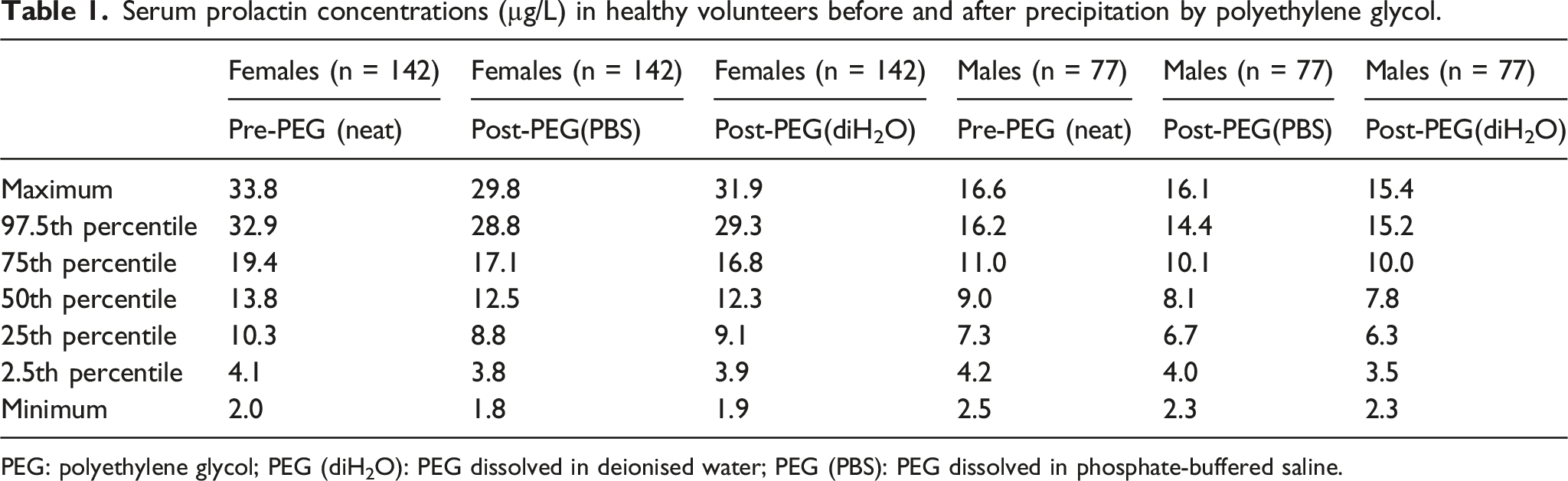
PEG: polyethylene glycol; PEG (diH2O): PEG dissolved in deionised water; PEG (PBS): PEG dissolved in phosphate-buffered saline.
Subsequently, we compared concentrations of post-PEG (PBS) prolactin with those of post-PEG (diH2O) prolactin. In both females and males, median post-PEG (PBS) prolactin concentrations were not statistically different from the corresponding median post-PEG (diH2O) prolactin concentrations (p>0.05). Mean differences between paired post-PEG (PBS) and post-PEG (diH2O) prolactin concentrations on Bland–Altman plots (not shown) were 0.12 μg/L (95% CI, p = 0.3396, −0.12 to 0.35 μg/L) in females and 0.10 μg/L (95% CI, p = 0.3414, −0.09 to 0.29 μg/L) in males. The agreement between post-PEG (PBS) and post-PEG (diH2O) prolactin concentrations was, in general, very good. Passing–Bablok regression analysis demonstrated no statistically significant differences between post-PEG (PBS) and post-PEG (diH2O) prolactin concentrations in either gender; the regression equations are as follows: post-PEG (diH2O) prolactin = 1.01 × post-PEG (PBS) prolactin − 0.06 μg/L in females and post-PEG (diH2O) prolactin = 1.04 × post-PEG (PBS) prolactin − 0.27 μg/L in males.
Discussion
In this study, we sought to establish locally relevant reference intervals for serum prolactin concentrations and post-PEG precipitation prolactin concentrations in both females and males on the Roche Cobas Prolactin II assay. As expected, serum prolactin concentrations in female adults were, in general, higher than those in male adults (Table 1). No subjects were found to have significant macroprolactinaemia, which can potentially increase the pre-PEG prolactin concentrations and affect the reference intervals.
Currently, different protocols for preparation of PEG and precipitation of macroprolactin are in use in different clinical laboratories. Pairwise comparison in this study showed no statistically significant differences between post-PEG (PBS) and post-PEG (diH2O) prolactin concentrations measured by the Roche prolactin II assay in either gender.
The reason for more than 100% prolactin recovery after PEG precipitation in some samples in this study is not entirely clear. Precipitation by PEG solution may have altered the matrix effect on the Roche prolactin II assay, leading to erroneously high post-PEG prolactin results. Imprecision due to manual dilution may also contribute to recovery slightly higher than 100%. Over-recovery of prolactin after PEG precipitation has also been reported in other studies using either a radio-immunoassay (up to 142%) 11 or other immunoassays (up to 261%). 12
Besides the diluent used for the preparation of PEG solution (PBS versus diH2O), other factors may potentially affect the precipitation of macroprolactin by PEG and thus post-PEG prolactin results. Those factors include assay design of different immunoassays,13,14 molecular weight of PEG used (6000 vs 8000), 15 storage conditions of prepared PEG solution (temperature and duration), duration of vortex mixing and speed and duration of centrifugation. Therefore, it is important that each clinical laboratory should establish its own reference intervals specific for its prolactin immunoassay and the PEG precipitation protocol used.
One of the limitations of our study is that it included only healthy adult volunteers and thus the reference intervals established in this study cannot be applied to paediatric populations. Our study also has a relatively small sample size for males (n=77).
In conclusion, gender-specific prolactin and post-PEG prolactin reference intervals were established in a local adult population for the Roche Cobas Prolactin II assay. Post-PEG (PBS) prolactin concentrations were not statistically different from post-PEG (diH2O) prolactin concentrations in either gender.
Footnotes
Acknowledgments
None.
Author contributions
C.K.M.H., C.P.Y. and P.C. researched literature and conceived the study. C.K.M.H. and P.C. was involved in protocol development and data analysis. P.C. was involved in gaining ethical approval, patient recruitment and wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Ethical approval
Ethical approval for this study was obtained from SINGHEALTH INSTITUTIONAL REVIEW BOARD (CIRB reference number 2017/2339).
Informed Consent
Written informed consent was obtained from the patient(s) for their anonymised information to be published in this article.
Availability of data and materials
The datasets generated and/or analysed during the current study are available from corresponding author.
Trial Registration
Not applicable because this study is not a clinical trial.
Conflict of interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
