Abstract
Transbronchial cryobiopsy (TBCB) is performed to aid diagnosis of interstitial lung disease, of which bleeding is a potentially life-threatening complication. Post-TBCB management involves temporary balloon blockade, bronchial artery embolisation (BAE) or surgery. Bronchial occlusion by endobronchial Watanabe spigot (EWS) as the definitive method of bleeding control post TBCB has not been described. A 56-year-old male underwent TBCB to aid diagnosis of interstitial lung disease. TBCB had been performed at RB4 (lateral segment of right middle lobe) with a prophylactic balloon blocker. However, prolonged bleeding was observed upon deflation of the balloon blocker. Haemostasis was secured with successful deployment of EWS into RB4, with no evidence of rebleeding in surveillance bronchoscopy and chest radiographs. EWS was kept in situ for four days and subsequently removed. The patient was discharged with good functional status. This case demonstrates that EWS placement may be considered for definitive management of low-volume post-TBCB bleeding, especially when BAE and surgical intervention are not possible.
Introduction
Transbronchial cryobiopsy (TBCB) is increasingly being performed to establish histopathological diagnosis in interstitial lung disease. 1 TBCB is not without risk, with bleeding being the most common complication. 2 The prophylactic use of an endobronchial blocker or Fogarty balloon in TBCB is thus recommended,2,3 serving as a temporary measure to prevent blood flooding from the biopsied airway before definitive treatment such as bronchial artery embolisation (BAE) or surgery. 4 The endobronchial Watanabe spigot (EWS®; Novatech, La Ciotat, France) is a silicon spigot introduced in 1991 primarily for the management of fistulous lung disease. 5 Its use as definitive therapy in haemoptysis has since been described for bronchiectasis-related and tumour bleed4,6,7 but has yet to be reported in post-TBCB bleeding.
Case report
A 56-year-old chronic smoker of 30 pack years presented to us with six months of progressive exertional dyspnoea and dry cough. Clinical examination revealed fine inspiratory crepitations at the bases of both lungs. There was no associated clubbing or features of connective tissue disease. Initial chest radiograph showed bilateral lower zone interstitial changes. Echocardiogram showed no evidence of pulmonary hypertension or reduced left ventricular ejection fraction. Spirometry was restrictive, with a forced vital capacity (FVC) of 2.76 L (66% predicted) and forced expiratory volume (FEV1) of 2.38 L (77% predicted), while the diffusing capacity of the lung for carbon monoxide was reduced at 16.14 mL/min/mmHg (63% predicted). Anti-nuclear antibody was negative, and creatine phosphokinase level was normal at 205 IU/L. A high-resolution computed tomography (CT) scan of the thorax showed predominantly sub-pleural and basal ground-glass changes and reticulations, with traction bronchiectasis and bronchioloectasis consistent with non-specific interstitial pneumonia (NSIP; Figure 1(a)). Bronchoalveolar lavage from the anterior segment of the right lower lobe (RB8) revealed a predominantly lymphocytic (50%) differential cell count. Infective screen was negative for bacterial, fungal and tuberculous organisms. The patient was counselled for TBCB.

(a) Computed tomography (CT) scan of the thorax, showing predominantly sub-pleural and basal ground-glass changes and reticulations with traction bronchiectasis and bronchioloectasis consistent with non-specific interstitial pneumonia. (b) Chest radiograph taken immediately post TBCB, showing consolidated right middle and lower lobes with the endobronchial Watanabe spigot (EWS) in place (red arrow). (c) Transbronchial cryobiopsy (TBCB) specimen, demonstrating uniform alveolar septal thickening with mild to moderate plasma cell and lymphocyte infiltration. Distribution of fibrous tissue is uniform and monotonous, with minimal collagen deposition and absence of fibroblastic foci. There is no architectural distortion. Findings are consistent with non-specific interstitial pneumonia (hematoxylin and eosin stain, 40× magnification). (d) Chest radiograph one month post procedure showing complete resolution of consolidation.
The target segment of interest was pre-identified to be the lateral segment of right middle lobe (RB4). Pre-procedurally, the patient had normal vital signs and haematological parameters. A flexible therapeutic bronchoscope (1TH190; Olympus Medical, Tokyo, Japan) was guided through the endotracheal tube (ETT) under intravenous anaesthesia. Radial endobronchial ultrasound (EBUS) examination at RB4 under fluoroscopic guidance did not reveal any significant blood vessel. The prophylactic 4Fr balloon blocker (B5-2C; Olympus Medical) was inserted into the ETT independently via a three-way adapter at the lumen of the ETT, and the tip of the balloon was guided into the ostium of RB4 and locked in place (Figure 2(a)). Subsequently, a 1.9 mm cryoprobe (ERBE; Medizintechnik, Tübingen, Germany) was passed through the working channel of the bronchoscope into the target site. Once reaching the pleura, the tip of cryoprobe was pulled back 1 cm under fluoroscopic guidance. The cryoprobe was then activated for five seconds – carbon dioxide is the cryogen in our centre. Thereafter, the bronchoscope and cryoprobe were removed together, with simultaneous inflation of the preplaced balloon for five minutes.
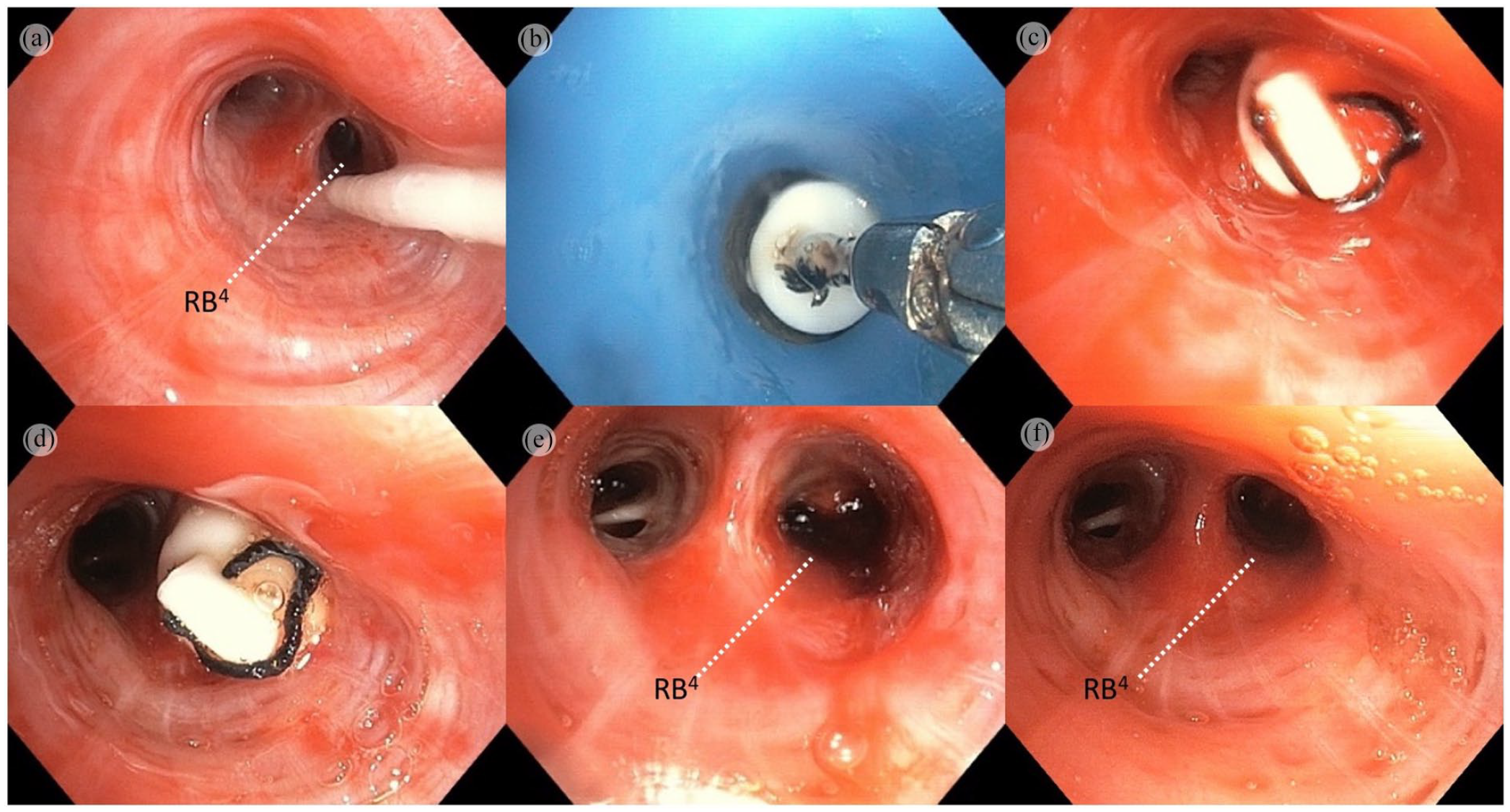
(a) Prophylactic balloon blocker placed at RB4 ostium. (b) EWS with attached nylon thread grasped by flexible forceps, being inserted into the airway via endotracheal tube. (c) EWS in the culprit sub-segment (RB4) immediately post insertion. (d) EWS in situ 72 hours post insertion. (e) Old blood clots in distal airway of RB4 observed immediately upon removal of EWS. (f) No evidence of rebleeding from RB4 after blood clot removal.
Unfortunately, upon deflation of the balloon blocker, re-examination of the airways revealed continuous ooze of fresh blood from RB4. Immediate re-inflation of the balloon was followed by 20 mL cold saline, 50 mL 1:20,000 adrenaline solution and 500 mg tranexamic acid applied sequentially through the inflated balloon catheter into the distal airway. Total balloon occlusion time was approximately 30 minutes, with subsequent significant reduction of bleed. However, three to four minutes following the balloon deflation, a persistent slow ooze was still noted. The patient became hypotensive, requiring a dose of intravenous phenylephrine. A decision was thus made to embolise the culprit segmental bronchi (RB4) using a 7 mm EWS.
A 3/0 nylon thread was pre-sutured at the distal end of the EWS for ease of retrieval prior to insertion. The EWS was grasped at the distal end of the bronchoscope with flexible forceps (Figure 2(b)). The balloon blocker was then deflated and removed to allow greater leeway for EWS insertion through the ETT. The EWS and bronchoscope were then introduced as a single unit through the ETT and into RB4 rapidly under direct vision. The EWS was inserted snugly into RB4, and no bleeding was noted from the surroundings after 5–10 minutes of observation (Figure 2(c)). All clots were aspirated from dependent airways, and no bleeding was observed upon completion of the procedure.
Post procedure, the patient was mechanically ventilated in the intensive care unit. A chest radiograph after the procedure showed right mid- and lower-zone consolidations with the EWS in situ and no pneumothorax (Figure 1(b)). BAE was not carried out in view of acute kidney injury. As there were no clinical features to suggest active rebleeding, confirmed via cautious surveillance bronchoscopy performed at six hours and on days 2 and 4 post bleed, conservative management was maintained (Figure 2(d)). The bronchoscopies showed the EWS in situ with no signs of rebleeding, no purulent discharge and no unhealthy mucosa. The decision for EWS removal was made on day 5 post procedure. Removal of the EWS was a two-step method. With the forceps grasping the pre-sutured nylon thread, the EWS was first dis-impacted from the ostium of RB4 using gentle traction. This was followed by a brief pause to observe for any bleeding – only minimal stale blood was seen. Next, the EWS was completely removed, followed by aspiration of remaining clots from the lumen until clear. There were no features to suggest active bleeding (Figure 2(e) and (f)). The patient was extubated post procedure. His blood parameters showed resolution of acute kidney injury, and he was discharged with good functional status.
Histopathological examination of the TBCB specimen showed features consistent with NSIP (Figure 1(c)). Follow-on investigations revealed myositis-associated auto-antibodies positive for anti-SRP, anti-PL7 and anti-Ku. A diagnosis of idiopathic inflammatory myositis (IIM)-related interstitial lung disease was made, and patient was started on oral prednisolone and azathioprine, with a repeated lung function a month later showing marked improvement of 530 mL in FVC and 340 mL in FEV1. A repeated chest radiograph also showed resolution of the right middle and lower zone consolidation (Figure 1(d)), while a repeated CT scan revealed no vascular abnormality. The patient was well, never manifested extrapulmonary manifestations of IIM and had no recurrence of haemoptysis.
Discussion
Based on two large studies, the incidence of post-TBCB bleeding ranges from 8.3% (58/699 patients) 2 to 16% (58/359 patients) 8 and remains the most important and potentially life-threatening complication of TBCB. Bleeding diasthesis, anticoagulant, clopidogrel use and thrombocytopaenia are absolute contraindications to performing TBCB 9 – none of which were present in our patient. Expert consensus on performing TBCB advocates advanced airway via endotracheal intubation or rigid bronchoscopy to allow easier manoeuvrability and fluoroscopic guidance to confirm sub-pleural placement of cryoprobe to avoid injury to larger-sized vasculature in too-proximal biopsies. 3 In addition, we performed radial EBUS examination to ensure the absence of significant vasculature in our pre-planned biopsy site – a technique described previously. 10 Despite all safety measures, complications are not completely avoidable. The emergent decision to attempt EWS insertion in our patient was made on the basis that post-TBCB bleeding almost always originates from the biopsied sub-segment. Thus, an optimally placed EWS should contain such localised bleed. While spontaneous resolution of bleeding may have been possible in our case, the presence of fresh oozing blood signalled an alarming risk of continuing bleeding.
There are a few limitations to EWS as definitive management for post-TBCB bleeding. First, occult rebleeding occurring distal to the EWS may not be detected. In our case, the resolving infiltrates at the site of biopsy as seen on chest radiograph and a stable haemoglobin level were reassuring indicators of resolving bleed. Second, there is a risk of EWS displacement. Hence, appropriate EWS size selection is crucial. Bronchoscopic surveillance was performed to ensure optimal, tight EWS placement. Third, EWS placement confers a risk of obstructive pneumonia at the occluded segment. Severe infections are rare in fistulous lung disease, even with permanently placed EWS, 11 but removal is advocated to avoid infections in at-risk patients. 12 Fourth, spigot placement may not be feasible in profuse bleeds, as one would anticipate difficulty in spigot placement once the balloon is deflated. Fifth, EWS placement would be challenging in bleeding from the apical segments of upper lobes. 6 Lastly, EWS as a definitive therapy without an angiographic study may miss iatrogenic vascular pseudoaneurysm.
Conclusion
EWS placement is a novel technique in the definitive management of severe post-TBCB bleeding, complementing the role of prophylactic balloon blocker by preventing airway flooding. It can be considered when BAE or surgical intervention is not feasible, provided close monitoring for persistent bleeding is available. Routine EWS usage may be a relatively cheap and accessible option in the future but requires further studies to validate this observation.
Footnotes
Acknowledgements
None.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Availability of data and materials
Data sharing is not applicable to this article, as no data sets were generated or analysed during the current study.
Authors’ contributions
S.S.K. and S.T.T. initiated the idea for case reporting, and L.E.N. prepared the main manuscript. L.E.N., S.S.K., S.K.C. and S.T.T. prepared the final copy of the manuscript. S.T.T. was the main operator of the TBCB procedure. S.S.K. was the first assistant of the procedure. S.T.T., S.S.K., C.S.C., S.K.C. and L.E.N. were involved in the overall management of the patient. All authors have read and approved the final manuscript.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Informed consent
Written consent was obtained from the patient for their anonymised information to be published in this article.
Ethical approval
Sarawak General Hospital does not require ethical approval for reporting individual cases or case series.
