Abstract
Background:
There is a lack of data regarding prevalence estimates of diabetic peripheral neuropathy (DPN) in Singapore. This study aimed to investigate the prevalence of DPN and risk factors in patients who present to a diabetic foot screening clinic and are considered low to medium risk in developing foot complications due to their type 2 diabetes mellitus.
Methods:
A one-year retrospective analysis was conducted at Ng Teng Fong General Hospital. Patients who underwent diabetic foot screening and endocrinologist review in the Diabetes and Endocrinology Specialist Outpatient Clinic during the period January 2019–December 2019 were included in this study. DPN was defined by the patient’s inability to detect ⩾1 out of 10 sites using the 10 g monofilament. Significantly associated risk factors with DPN were analysed using a multivariable logistic regression model.
Results:
Data from 479 patients were analysed. Prevalence of DPN was 28% (95% confidence interval (CI) 24.0–32.2). DPN was significantly associated with age >65 years (odds ratio (OR)=5.44, 95% CI 2.87–10.32), Indian ethnicity (OR=1.99, 95% CI 1.04–3.80), insulin use (OR=1.65, 95% CI 1.03– 2.64), diabetic retinopathy (OR=2.36, 95% CI 1.47–3.78) and stroke (OR=2.44, 95% CI 1.03–5.77).
Conclusion:
Prevalence of DPN in this study’s population sample was 28%, and the significant risk factors are age, Indian ethnicity, insulin use, diabetic retinopathy and stroke.
Introduction
Diabetic peripheral neuropathy (DPN) is the most common complication of diabetes, and is defined as a ‘symmetrical, length-dependent sensorimotor polyneuropathy attributable to metabolic and microvessel alterations as a result of chronic hyperglycemia exposure (diabetes) and cardiovascular risk covariates’. 1 It has been forecast that the prevalence of type 2 diabetes mellitus (T2DM) among adults in Singapore will increase from 7.3% in 1990 to 15% in 2050. 2 With evolving lifestyles attributed to rising affluence, the lifetime risk of Singaporeans with T2DM is expected to be one in two in the year 2020. 2 Additionally, the lifetime risk of an individual with diabetes developing a diabetic foot ulcer (DFU) may be as high as 25%.3,4 DPN, along with deformity and trauma, has been identified as one of a triad of common predisposing factors in the foot. 5 Patients with DPN may experience loss of protective sensation in their feet, numbness and impaired proprioception and gait progression. 6 Thus, they are at higher risk of developing DFUs due to altered sensation and micro-trauma over pressure sites on their feet. 6 Furthermore, DFUs are the most common cause of non-traumatic foot amputation worldwide. They lead to a significant long-term impact on the morbidity and quality of patients’ lives. 7 Hence, a better understanding of the prevalence of DPN and its associated risk factors will allow the development of preventable strategies for DPN and DFUs in Singaporean patients with T2DM. However, there are limited published data regarding prevalence estimates and the relevant risk factors in Singapore. Therefore, the aim of this study was to investigate the prevalence and risk factors of DPN in patients with T2DM who underwent diabetic foot screening in a government restructured hospital in Singapore.
Methods
We conducted a one-year retrospective analysis of medical records of patients with T2DM who presented to their annual diabetic foot screening in the Diabetes and Endocrinology Specialist Outpatient Clinic in Ng Teng Fong General Hospital (NTFGH) between January 2019 and December 2019. The population studied mainly comprised patients with a low to medium risk of developing foot complications due to their T2DM. This is because patients who were classified as high risk due to a history of lower-extremity amputation and/or active foot ulcers typically presented to the NTFGH outpatient podiatry clinics for podiatric treatment and routine surveillance, instead of the diabetic foot screening clinic. Records of patients were excluded when there were missing data and/or a diagnosis other types of diabetes, including type 1, latent autoimmune diabetes of adulthood and gestational diabetes. Demographic characteristics such as age, sex, ethnicity and body mass index (BMI) were recorded for each study subject. Blood pressure, duration of T2DM since initial diagnosis, use of insulin and/or anti-diabetic oral medication and history of smoking were also collected from their medical records when they consulted their endocrinologists within the same period between January 2019 and December 2019. The presence of co-morbidities, such as hypertension, hyperlipidaemia, diabetic retinopathy, ischaemic heart disease, chronic kidney disease and stroke, was collected for each patient. Such conditions were defined by their respective diagnosis codes and cross-checked with the patients’ medical records.
As part of routine diabetic foot screening in NTFGH, DPN was screened with two clinical tools: the 10 g monofilament and the neurothesiometer. The 10 g monofilament was used on 10 sites on each foot to screen for a loss of protective sensation. Additionally, a neurothesiometer was used on the first metatarsophalangeal joint, with the inability to feel vibration from the equipment probe at ⩽25 V indicating a loss of vibration perception. 8 As there was a considerable amount of incomplete data regarding the vibration perception threshold, DPN was defined in this study with the patient’s inability to detect ⩾1 out of 10 sites using the 10 g monofilament. 9
No personally identifiable information was collected during the study process. Only anonymised data were extracted. The Domain Specific Review Board approved this retrospective analysis study (NHG DSRB Ref: 2020/00708), and informed consent was not required.
Statistical analysis
IBM SPSS Statistics for Windows v27 (IBM Corp., Armonk, NY) was used to analyse all the data collected in this study. The mean and standard deviation of continuous variables were calculated and compared using an independent t-test. Percentages for categorical data were assessed using the chi-square test. Univariate logistic regression was used to examine association of potential risk factors with DPN. Potential risk factors that were related to DPN by a predetermined value of p<0.10 were selected and used in a multivariable logistic regression model. A final value of p<0.05 was considered to be statistically significant.
Results
The data of 479 patients with T2DM and who underwent diabetic foot screening between January 2019 and December 2019 were collected and analysed. The clinical characteristics of both patients with and without DPN are presented in Table 1.
Demographics and characteristics of study subjects.
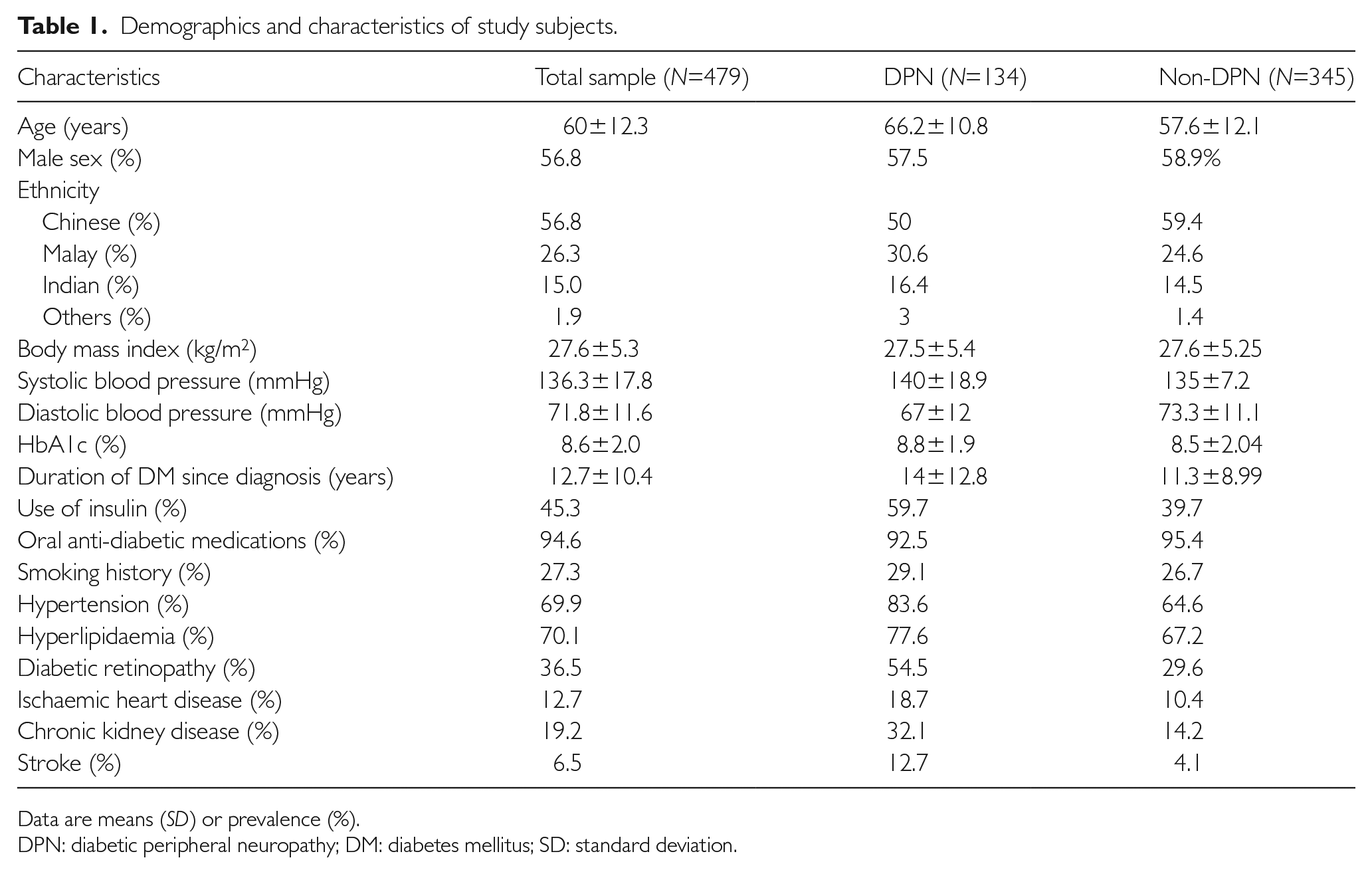
Data are means (SD) or prevalence (%).
DPN: diabetic peripheral neuropathy; DM: diabetes mellitus; SD: standard deviation.
There were 272 (56.8%) males and 207 (43.2%) females. The mean age of these 479 patients was 60±12.3 years. Males (59.0±11.9 years) were significantly younger than females (61.3±12.8 years; p<0.05). The mean duration of diabetes was 12.7±10.4 years, and this was similar in both sexes. Our study population consisted of 56.8% Chinese, 26.3% Malays, 15% Indians and 1.9% other ethnic groups.
The overall prevalence of DPN (n=134) was 28% (95% confidence interval (CI): 24.0–32.2%). Just over half (57.5%; n=77) of the patients with DPN were males, and 42.5% (n=57) were females. Sex differences had either no or a negligible effect on patients with or without DPN. Figure 1 presents the prevalence of DPN stratified by age. The prevalence of DPN in subjects aged >65 years was higher at 49.7% compared to those aged <55 years.

Prevalence of diabetic peripheral neuropathy stratified by age groups.
The results of the univariate logistic regression analyses are listed in Table 2. Age, systolic and diastolic blood pressure, the duration of DM and use of insulin were significantly associated with DPN in our study population. Diastolic blood pressure showed an inverse relationship with DPN in the patients. Risk factors such as hypertension, hyperlipidaemia, diabetic retinopathy, ischaemic heart disease, chronic kidney disease and stroke were also significantly associated with DPN. Sex, BMI, usage of oral anti-diabetic medications and smoking history were not significantly associated with DPN.
Univariate associations (Odds ratios, 95% CI) of clinical characteristics with DPN in T2DM patients.

p<0.05.
CI: confidence interval.
Overall, patients with DPN were older, had higher blood pressure, lower diastolic blood pressure and a longer duration of diabetes compared to those without DPN (p<0.05). They also exhibited a higher proportion of co-morbidities such as hypertension, hyperlipidaemia, diabetic retinopathy, ischaemic heart disease, chronic kidney disease and stroke (p<0.05).
Ethnicity and the various risk factors with significant univariate associations were entered into a logistic regression model, with DPN as a dependent variable. DPN was found to be significantly associated with age >65 years (odds ratio (OR)=5.44, 95% CI 2.87–10.32), Indian ethnicity (OR=1.99, 95% CI 1.04–3.80), insulin use (OR=1.65, 95% CI 1.03–2.64), diabetic retinopathy (OR=2.36, 95% CI 1.47=3.78) and stroke (OR=2.44, 95% CI 1.03–5.77; Table 3). Other conditions such as hypertension, hyperlipidaemia, ischaemic heart disease and chronic kidney disease were found not to be significantly associated with prevalent DPN.
Multivariate associations (Odds ratios, 95% CI) of clinical characteristics with DPN in T2DM patients.

Discussion
The overall prevalence of DPN in this study was 28%. DPN was significantly associated with older age, Indian ethnicity, use of insulin, diabetic retinopathy and stroke. The prevalence estimates are close to the findings from a study published by Nather et al. in which they found the prevalence to be 33.3% in both types of DM, with 96.58% of the study population having T2DM. 10 In another study, Nang et al. found the prevalence to be 16.6% in Singaporean patients with types 1 and 2 DM. 11
Prevalence estimates vary worldwide, ranging from 2.4% in the UK to 61.8% in Shanghai, China.8,12–14 A meta-analysis found that the pooled prevalence of DPN among patients with T2DM to be 31.5%. 15 There is high variability in the prevalence estimates, as it is highly dependent on the studies’ adopted diagnostic criteria and population. 16 Nerve conduction studies (NCS) are regarded as the gold standard to diagnose DPN. 1 However, they is not widely adopted in routine practice, as the test is time-consuming and requires trained personnel and special equipment. 1 Therefore, most research studies diagnose DPN with other clinical tools and tests. There are also studies that assessed for DPN in their participants using scoring systems such as the neuropathy disability score and the neuropathy symptom score.13,17 In the Singaporean context, Nang et al. defined DPN in their subjects using either a neurothesiometer reading of >25 V at the hallux and medial malleolus or a monofilament sensory test of fewer than four of five sites in either foot. 11 On the other hand, Nather et al. utilised the monofilament sensory test as their sole diagnostic test. 10 Both the monofilament and neurothesiometer are used in the diabetic foot screenings in NTFGH. However, during data collection, we discovered that there was a considerable amount of missing data with regards to the vibration perception test. Therefore, we decided to define DPN in our subjects as the inability to detect ⩾1 site during the 10-site monofilament test which has been found to have high sensitivity and specificity when compared to the gold standard NCS. 9
This study highlighted that age, insulin use, diabetic retinopathy and stroke are significantly associated with DPN. With regards to age, this finding is strongly supported by several studies that found age as a significant risk factor for DPN.17–20 A meta-analysis supported the significant association between DPN and diabetic retinopathy by showing a similar increase in risk of developing DPN in the patients who have both complications (OR=2.34, 95% CI 1.74–3.16). 21 Hyperglycaemia from DM is a contributing factor for the pathogenesis of both DPN and retinopathy, in which there is an association between the accumulation of advanced glycation end products (AGEs) and the activation of polyol pathway, protein kinase C and free radicals, which result in the biomolecular damages in the affected tissues. 22 Insulin use was also a significant risk factor for DPN in a few studies that involved patients with T2DM.13,23,24 It was proposed that this could be due to the fact that insulin use suggests beta cell failure in the pancreas and may reflect a later stage in the natural history of diabetes or a greater severity. 24 With regards to stroke, it was not found to be significantly associated with DPN in a large retrospective cohort study done in both Germany and the UK. 12 However, as one of the major cardiovascular diseases (CVD), it was shown that patients with both CVD and T2DM were at increased risk of developing DPN at 10 years of follow-up. 25
Interestingly, our study showed that Indians with T2DM in Singapore are at higher risk of developing DPN, but we were unable to find any other study that can support our claim. However, the Singapore Epidemiology of Eyes Diseases Study discovered that Indian Singaporeans have a higher prevalence of diabetic retinopathy compared to Chinese and Malays. 26 Disease risk can be altered by ethnicity due to complex differences in socio-economic, cultural, lifestyle or dietary or genetic factors. 27 Future studies may further investigate on the validity of this finding and how ethnicity contributes to the differences in risk of development of DPN.
This study did not show that the duration of diabetes was a significant risk factor of DPN. However, the duration of diabetes was shown to be associated with significantly increased risks of DPN among patients with diabetes by several other studies.21,24,28,29 The study’s finding may be affected by the limited accuracy of this independent variable during data extraction and collection. The duration of diabetes was calculated by subtracting the year of diagnosis from the year 2019. We encountered some instances where the year of diagnosis was not clear, as an approximate year was documented based on the patient’s memory.
As this study is a retrospective analysis of medical records, another limitation is that it can only highlight the associations between risk factors, and causality cannot be determined. Additionally, the findings are limited by the validity and completeness of data. The definition of DPN may also lead to an inaccurate estimation of the true prevalence of DPN, as other clinical tests such as the neurothesiometer or a combination of positive findings from various tests may contribute to a higher diagnostic accuracy. The sensitivity and specificity of the 10 g monofilament test is varied and dependent on the number of sites being tested on the foot. 30 This study involved the testing of 10 sites on each foot with the monofilament. The Ministry of Health Singapore Appropriate Care Guide regarding foot assessment in patients with diabetes was only published in June 2019, and it recommends that the monofilament should be used on four sites on each foot (the plantar aspect of the hallux and the heads of the first, third and fifth metatarsals). 31 However, it was demonstrated that there were no significant differences in the effectiveness between both 4- and 10-site monofilament testing for DPN screening. 32
Furthermore, the estimated prevalence of DPN in this study is to be interpreted with caution, as the study sample mainly represented patients with a low to medium risk of developing foot complications or lower-extremity amputation due to their T2DM. This is because patients who were classified as high risk due to history of lower-extremity amputation and/or active foot ulcers usually presented to NTFGH outpatient podiatry clinics for podiatric treatment and routine surveillance instead of the diabetic foot screening clinic.
As a single-site study conducted in a government restructured hospital, another limitation is that the subjects may not be representative of the overall population of T2DM patients in Singapore due to convenience sampling. This study may not represent the patients who undergo their diabetic foot screenings in government-aided polyclinics and private general practices which are also significant sectors of the primary health-care population. However, the proportion of clinical characteristics and demographics of our study subjects are comparable to a large cohort study of 14,033 Singaporean patients. 33 The large cohort of T2DM patients recorded an ethnic distribution of 59.3% Chinese, 22.7% Malays and 17.3% Indians, which is similar to what was found in this study.
Despite its limitations, this study is still useful, as it adds to the limited body of research in Singapore regarding the prevalence of DPN and its risk factors in T2DM patients. Increased age, Indian ethnicity, insulin use, diabetic retinopathy and stroke have been shown to be significantly associated with increased odds of developing DPN. Early identification and increased understanding of risk factors and co-morbidities allow clinicians to develop targeted screening of DPN and implement strategies to help to prevent DFUs and their complications. This study also supports the well-known significant association between DPN and diabetic retinopathy. Therefore, the study reinforces the importance of diabetic foot surveillance and intervention which includes increased frequency of diabetic foot screenings and foot care education for patients with T2DM.
Footnotes
Acknowledgements
We would like to thank Ms Neo Jiawei and Dr Shen Liang for their assistance and guidance in the data analysis process.
Authors’ contributions
A.H. and M.R.T. researched literature and conceived the study. A.H. and M.R.T. were involved in protocol development and gaining ethical approval. A.H. was involved in the data analysis. A.H. and M.R.T. wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Availability of data and materials
The data sets generated and/or analysed during the current study are available from the corresponding author.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from NHG Domain Specific Review Board (2020/00708).
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed consent
Informed consent was not required to be obtained due to study design.
