Abstract
Background/objective:
Prevention of cytomegalovirus (CMV) infection is an important component of post kidney transplant care. We aimed to evaluate the impact of two different CMV prophylaxis protocols on the epidemiology and outcomes of CMV infections at our centre.
Methods:
This is a single-centre retrospective before/after observational study. Kidney transplant recipients who received Protocol 1, a valacyclovir- or valganciclovir-based regimen prescribed for one to three months based on the CMV risk status between 2004 and 2008, were compared to those who received Protocol 2, a valganciclovir-based regimen prescribed for three months and six months for those at moderate and high risk, respectively, between 2010 and 2014. The impact of different prophylaxis regimens on the incidence of CMV infections, disease, recurrent infections and onset of CMV infection at 24 months were reviewed.
Results:
There were 192 patients included; 106 patients received Protocol 1, 86 received Protocol 2. At 24 months, the incidence of CMV infection was 53.8% and 55.8% in Protocols 1 and 2, respectively (
Conclusion:
The incidence of CMV infection was similar in both protocols. Where valganciclovir is not available, valacyclovir may be considered over no prophylaxis. Post-prophylaxis CMV infections are not uncommon, and vigilance for it should be advocated.
Introduction
Cytomegalovirus (CMV) is a medically important pathogen in kidney transplantation, and it is associated with graft rejection, graft dysfunction, graft loss and mortality.1–4 Therefore, CMV prevention is an important component of post-transplant care.
CMV prevention strategies vary within and between institutions. Either universal prophylaxis or pre-emptive therapy is practiced. 5 For universal prophylaxis, antiviral agents such as oral ganciclovir, valganciclovir and valacyclovir have been used. 6 Oral ganciclovir is not recommended due to its lower bioavailability which may predispose to emergence of resistance. 7 Valganciclovir, a prodrug of ganciclovir, has good bioavailability and is the most potent and preferred prophylaxis agent in most transplant centres. However, it is expensive, and prophylaxis duration may sometimes be truncated due to valganciclovir-induced leucopaenia. High-dose valacyclovir is a cost-effective alternative for CMV prophylaxis but can be associated with neurotoxicity.8,9 Selection of CMV prophylaxis strategy and agents is also influenced by local prescribing patterns and available resources.
At Singapore General Hospital (SGH), universal prophylaxis is practised. There was a change in CMV prophylaxis recommended for kidney transplant, and this was more widely implemented after 2009 (see Table 1). Before 2009, patients at moderate risk for CMV (R+ recipients) received one month of CMV prophylaxis with valacyclovir, while those at high risk (R+ recipients who received thymoglobulin, D+/R– recipients) received three to six months of valacyclovir- or valganciclovir-based prophylaxis. After 2009, the protocol was intensified. Valganciclovir was exclusively used for prophylaxis, and prophylaxis duration was extended to three months for those at moderate risk and to six months for those at high risk. The change in prophylaxis regimen was driven by data which showed that the rates of CMV disease and viraemia were reduced when prophylaxis duration was prolonged in high-risk transplant recipients. 10 This study aimed to evaluate the impact of intensifying CMV prophylaxis on our local renal transplant population.
CMV prophylaxis strategies practiced at SGH during the different study periods.
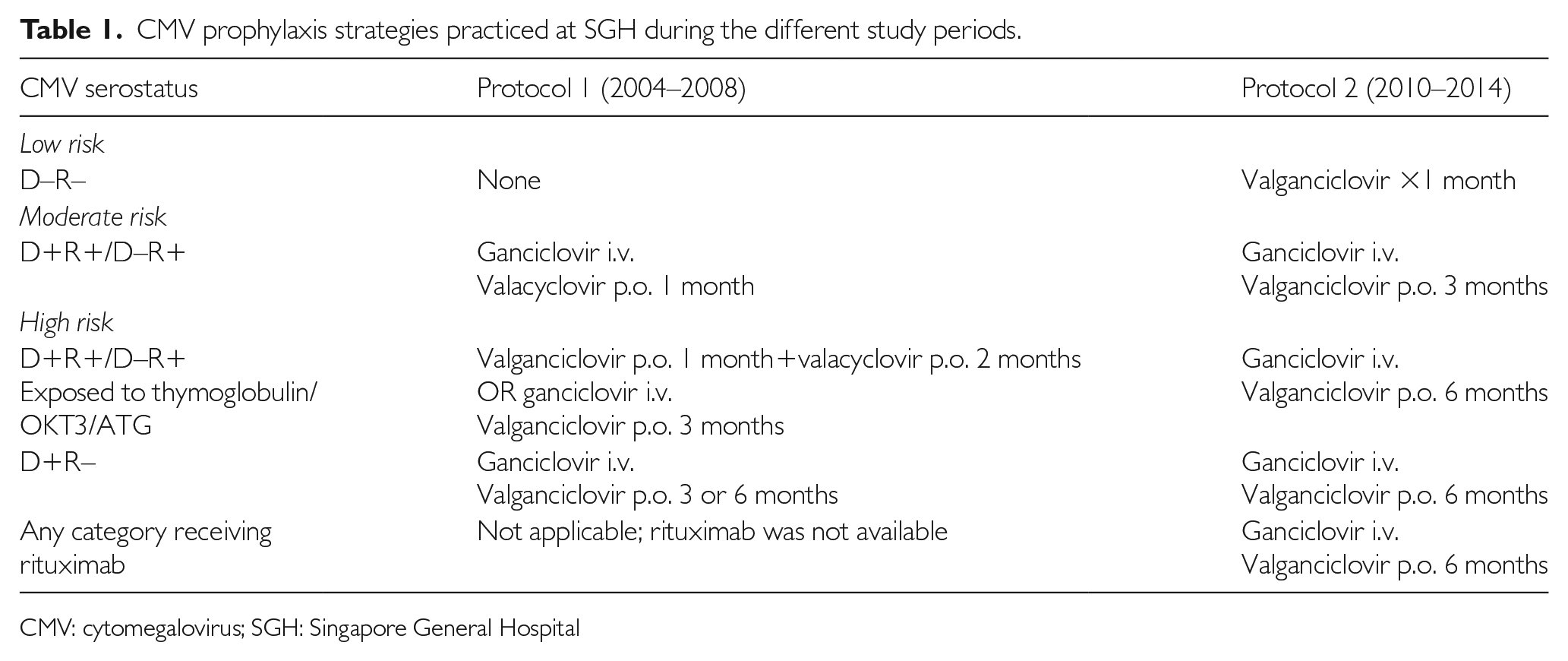
CMV: cytomegalovirus; SGH: Singapore General Hospital
Methods
This is a single-centre before/after retrospective cohort study to evaluate the impact of different CMV prophylaxis protocols on the epidemiology and outcomes of CMV infection in the kidney transplant recipients treated at SGH. The Singhealth Institutional Review Board approved this study (CIRB Ref No. 2018/2367).
Kidney transplant recipients treated at SGH from 2004 to 2008 and from 2010 to 2014 were screened for inclusion and exclusion criteria. A one-year transition period in 2009 was factored into the study design to ensure that the new protocol was more widely implemented. Patients were included if (a) they were aged ⩾18 years at the time of kidney transplant, (b) both donor and recipient CMV serology were available, (c) they complied with the prophylaxis protocol for the designated period and (d) they were followed up at our hospital for at least 24 months. Patients were excluded if they (a) had their transplant performed outside of SGH or Singapore, (b) received a re-transplant during the study period, (c) did not comply with unit prophylaxis protocols and (d) were followed up for <24 months at SGH.
Medical records, laboratory investigations and pharmacy records were reviewed. Patient demographics, transplant data, CMV prophylaxis and clinical outcomes at 24 months were collected and managed using the Research Electronic Data Capture (REDCap) tool hosted at our institution.
The risk of CMV infection was stratified based on CMV serostatus and if lymphocyte-depleting thymoglobulin was given. A recipient is considered high risk if CMV serostatus is donor positive and recipient negative (D+/R–), or if he or she has received thymoglobulin. Risk is considered moderate if CMV serostatus is recipient positive (D+/R+ or D–/R+). This risk stratification applies to both time periods/protocols. The recommended prophylaxis for each risk stratum in the two protocols is detailed in Table 1. Valacyclovir was dosed at 2000 mg q.d.s. in patients with an estimated glomerular filtration rate (eGFR) >75 mL/min, and renally adjusted for those with a lower eGFR. Valganciclovir was dosed at 900 mg o.m. in patients with an eGFR >60 mL/min, and was also renally adjusted for those with a lower eGFR.
CMV infection is defined by the presence of detectable CMV in body fluid or a tissue specimen (with viral culture, antigen tests or nuclear acid amplification tests).1,11,12 Although recent evidence suggests detection of CMV virus, antigen or DNA in the blood may not necessarily mean active CMV replication, 12 this distinction was not applied for the purpose of this study. The diagnosis of definite and probable CMV end-organ disease has to fulfil preset criteria as defined by Humar et al.11,13 ‘Delayed-onset’ CMV infections refer to infections occurring one to six months after the cessation of prophylaxis. ‘Late-onset’ CMV infections refer to those occurring more than six months after the cessation of prophylaxis. 14
During the study period, CMV was detected using either the Light Diagnostics CMV pp65 Antigenemia IFA Kit (EMD Millipore, Temecula, CA), or an in-house quantitative real-time CMV polymerase chain reaction. Primary outcomes reviewed include (a) the incidence of any CMV infection occurring over a 24-month follow-up period, (b) incidence of CMV disease (CMV syndrome as well as tissue-invasive disease), (c) recurrence of CMV infection and (d) time to first CMV infection. Tolerability of different CMV prophylaxis was reviewed as a secondary outcome.
Results are expressed as the median and interquartile range (IQR; 25th, 75th percentile) for continuous data and as frequency and percentage for categorical data. The Mann–Whitney
Results
There were 192 subjects included in the final analyses; 106 subjects received Protocol 1, and 86 received Protocol 2. Their baseline demographics, transplant characteristics, CMV risk status and prophylaxis are presented in Table 2. Subjects who received Protocol 1 were significantly younger (43.5 years (IQR 35.0–49.0 years) vs. 48.0 years (IQR 39.0–53.3 years),
Patient characteristics, transplant characteristics, CMV risk and CMV prophylaxis.
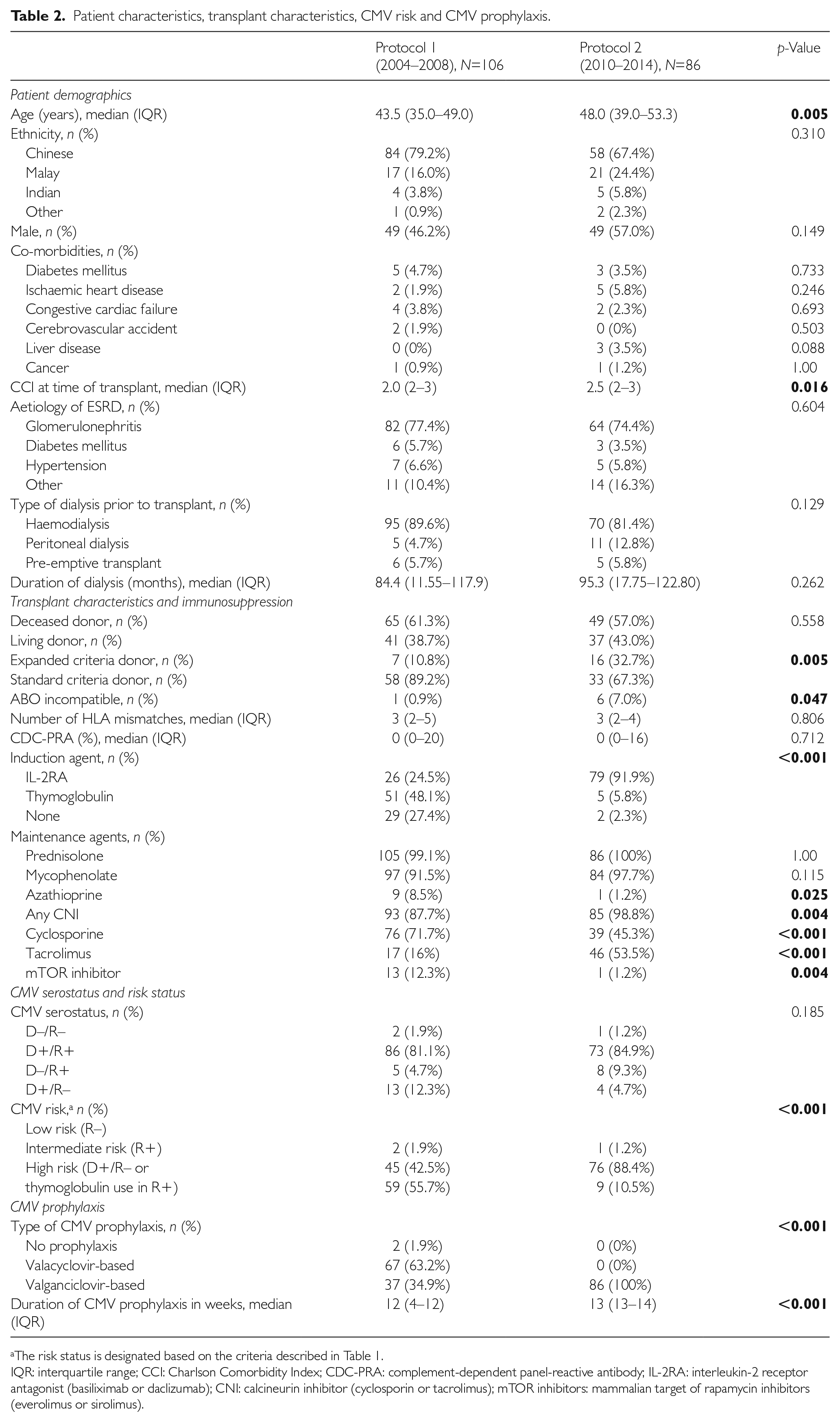
The risk status is designated based on the criteria described in Table 1.
IQR: interquartile range; CCI: Charlson Comorbidity Index; CDC-PRA: complement-dependent panel-reactive antibody; IL-2RA: interleukin-2 receptor antagonist (basiliximab or daclizumab); CNI: calcineurin inhibitor (cyclosporin or tacrolimus); mTOR inhibitors: mammalian target of rapamycin inhibitors (everolimus or sirolimus).
There was no difference in CMV serostatus in the two groups, but there were significantly more patients who were at high risk of CMV in those who received Protocol 1 (59 (55.7%) vs. 9 (10.5%),
It is important to appreciate the difference between the two protocols, as detailed in Table 1. Protocol 1 allowed use of either valacyclovir or valganciclovir; 67 (63.2%) patients received valacyclovir-based prophylaxis. Protocol 2 was exclusively valganciclovir based. The median duration of prophylaxis for patients receiving Protocol 1 was significantly shorter (12 weeks (IQR 4–12 weeks) vs. 13 weeks (IQR 13–14 weeks),
The major CMV outcomes in both groups are presented in Table 3. On univariate analysis, the incidence rates of CMV infections between the two groups were similar: 57 (53.8%) patients in Protocol 1 versus 48 (55.8%) in Protocol 2 (
Comparison of major outcomes between protocol 1 and protocol 2, and predictors of CMV infection.

Refers to any level of CMV antigen or polymerase chain reaction detected in blood.
Refers to both CMV syndrome and tissue-invasive CMV disease. For patients receiving Protocol 1, five developed CMV syndrome, and one developed CMV retinitis. Two patients in Protocol 2 had CMV syndrome; there were no cases of tissue-invasive disease.
OR: odds ratio; CI: confidence interval.
The type of CMV prophylaxis protocol was not significantly associated with CMV infection (OR=1.086, 95% confidence interval (CI) 0.613–1.924,
Amongst patients who developed CMV infection, the median time to development of CMV infection from time of transplant was significantly shorter in patients who received Protocol 1 (132 days (IQR 125–139 days) vs. 185 days (IQR 178–192 days),
Both prophylaxis regimens were relatively well tolerated. None of the patients who received high-dose valacyclovir developed neurotoxicity. Thirteen (12.7%) patients in Protocol 1 and nine (10.6%) in Protocol 2 (
Discussion
We describe a retrospective study comparing two different CMV prophylaxis regimens in renal transplant recipients. Protocol 1, used before 2009, included the use of valacyclovir for the intermediate- and high-risk groups, and duration of prophylaxis was in general shorter. Protocol 2, implemented after 2009, utilised the more potent valganciclovir exclusively, and prophylaxis duration was longer. This change in protocol was largely driven by evidence advocating longer CMV prophylaxis.10,15
Contrary to our hypothesis that an intensified prophylaxis regimen would reduce the incidence of CMV infection, this was not observed in our study; the rates of CMV infections were similar between both groups. This could be attributed to the heterogeneity of the patients’ baseline characteristics in the two groups. In general, patients who received Protocol 1 were at higher CMV risk, with a greater proportion receiving thymoglobulin, but the incidence of CMV was not increased compared to Protocol 2. The increased number of ABO incompatible transplants, use of ECD and older recipients in Protocol 2 – known risk factors for CMV infections16–18 – could have blunted the effects of the differences in prophylaxis regimen.
In our study, more than half of the subjects in both time periods (53.8% and 55.8% in Protocols 1 and 2, respectively) experienced at least one episode of CMV reactivation over a 24-month follow-up period post transplant. While this figure appears higher than the reported incidence of CMV infections (~20– 40%) at other centres, we recognise that follow-up duration, diagnostic thresholds and prophylaxis strategies used at different centres were variable and may therefore account for the differences observed.3,10,19–22 Another reason for the higher reported incidence of CMV infections in our study is attributed to the fact that we had defined CMV infection as the presence of any detectable CMV in body fluid or tissue specimen, and reflected CMV reactivation in the post-transplant recipient. It is important to note that the overall incidence of CMV disease was relatively low (5.7% and 2.3% in Protocols 1 and 2, respectively), and only one patient (who received Protocol 1) developed tissue-invasive disease with CMV retinitis. The majority of the CMV infections in both groups were asymptomatic antigenaemia/viraemia picked up through routine surveillance. The use of universal prophylaxis, regardless of prophylaxis regimen, was likely to have mitigated the risks of severe CMV infections in the two groups.
What we have described in this study is the classic finding of post-prophylaxis CMV reactivation, and this is reflected by significantly shorter time to CMV infection in patients who received Protocol 1. Post-prophylaxis CMV reactivation is not a new finding.1,14,16–18,23 Its specific pathophysiology is likely related to the complex interaction between CMV virus and the host, compounded by the use of immunosuppressants. Some experts believe that the lack of reconstitution of CMV-specific cellular immunity in transplant recipients could explain this phenomenon. 24 In our multivariate analyses, high CMV risk (determined by CMV D+/R– serostatus and receipt of thymoglobulin) and deceased donor transplant were independently associated with a higher risk of CMV infection, not choice of prophylaxis regimen, consistent with findings observed at other centres.19,20,25 Prophylaxis strategies alone are insufficient to reduce the incidence of post-transplant infections, and two key areas could be reviewed to improve CMV outcomes: (a) the role of evaluating CMV-specific immunity prior to the discontinuation of antiviral therapy, and (b) the need for individualised immunosuppression dosing as opposed to a protocol-driven approach.
CMV-specific immunoassays have been developed, and they are increasingly being evaluated for clinical use to identify patients who may be at high risk of CMV infection/disease. 26 The absence of documented CMV cell-mediated immunity (CMI) at the end of prophylaxis may suggest that the recipient is at higher risk of CMV reactivation/disease and therefore requires close surveillance. Similarly, weak or absent CMV CMI at the end of CMV treatment may suggest that the risk for recurrent disease is higher and may indicate a possible role for secondary prophylaxis. Conversely, in patients with documented CMV CMI, they may spontaneously clear low-level CMV reactivation without the need for systemic antiviral therapy. Serial monitoring of CMV CMI in the right clinical setting may guide clinical management in post-transplant care.27,28
We observed a high incidence of asymptomatic CMV reactivation upon discontinuation of prophylaxis, and this alludes to the possibility that the current immunosuppression dosing regimen (based on a protocol-driven approach) may be excessive for our local Asian population. The immunological and metabolic risk profile in our patients may be inherently different from the Caucasian population for which current immunosuppression dosing regimens are derived. 29 To date, except for the evaluation of trough levels of select immunosuppressants, there is lack of established clinical tools to guide individualisation of immunosuppression therapy for transplant recipients. 30 Strategies to reduce the depth of immunosuppression may include (a) the use of a steroid-sparing regimen, (b) reduction of CNI or (c) use of a CNI-sparing regimen.31–33 However, these strategies are often applied empirically after the diagnosis of CMV reactivation. There is an impetus to develop pharmacogenetic and pharmacogenomic strategies to guide selection of immunosuppression regimen in the era of individualised therapy in transplantation medicine.
This study has several limitations. Because of the retrospective nature of the study spanning over a decade and the evolving clinical practice of transplant medicine,32,34–37 our patient population in both groups was heterogenous, and there were baseline differences in recipient demographics, transplant and immunological risks, as well as maintenance immunosuppression. Therefore, we cannot be sure if the difference or lack thereof in CMV infections was due to the inherent differences between the two groups, or if there were truly no difference between the two different prophylaxis regimens. The small number of subjects in the study may limit our ability to detect statistically significant differences. Finally, excess immunosuppression, graft rejection or co-infections are factors that could potentially influence the incidence of CMV infections in our study, but these parameters could not be clearly ascertained in this retrospective study.
Within the limitations of this study, there are practical applications for some of our findings. We compared two different universal prophylaxis protocols: a shorter course of valacyclovir-/valganciclovir-based prophylaxis regimen versus a longer course of valganciclovir-based prophylaxis regimen with no demonstrable difference in the epidemiology and outcomes of CMV infections between the two groups. While a valganciclovir-based prophylaxis regimen remains the standard of care in intermediate- to high-risk kidney transplant recipients as advocated by major society guidelines, 35 a valacyclovir-based protocol may be a reasonable alternative, especially in resource-limited settings, given its comparable efficacy, tolerability and cost-effectiveness.9,36 In our study, breakthrough CMV reactivation while on valacyclovir prophylaxis was rare. Although neurotoxicity has been reported in patients receiving high-dose valacyclovir, 37 this was not observed in our study. Where available, we would recommend the use of CMV prophylaxis with either valganciclovir or valacyclovir, given the high rates of CMV infection and disease (~70% and 26–52%, respectively) in the absence of prophylaxis. 38
Post-prophylaxis CMV infections are not uncommon, regardless of valganciclovir or valacyclovir universal prophylaxis. Vigilance for CMV reactivation should be exercised, and we recommend a period of CMV surveillance after universal prophylaxis is discontinued, especially for patients at high risk of CMV.
Conclusion
Intensifying CMV prophylaxis by using valganciclovir exclusively and prolonging the course was associated with longer time to first CMV infection but not overall reduction in incidence of CMV infection at 24 months. Valacyclovir may be a reasonable alternative to valganciclovir, especially in resource-limited settings or in situations where valganciclovir cannot be tolerated. After prophylaxis is discontinued, CMV surveillance and careful titration of immunosuppression are adjunct strategies for the prevention of CMV infection/disease.
Footnotes
Acknowledgements
M.T. would like to express her thanks to J.C., T.K., S.T. and H.Q.Y. for their guidance and valuable feedback during the course of this project. The authors would like to express their thanks to the transplant coordinators for their help in data collection.
Authors’ contributions
T.K., M.T. and J.C. researched the literature and conceived the study. T.K. and J.C. were involved in protocol development. M.T. and J.C. did data collection. Data analysis was done by J.C. and H.Q.Y. M.T. wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Availability of data and materials
The data sets generated are available from the authors.
Ethical approval
The Singhealth Institutional Review Board approved this study (CIRB Ref No. 2018/2367).
Informed consent
Waiver of informed consent for this audit was approved by the SingHealth Centralised Institutional Review Board (CIRB Ref: 2018/2367), as this was a clinical audit of routine clinical care, where participants were not subjected to additional risks or burdens beyond usual clinical practice.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
