Abstract
Heparin-induced thrombocytopaenia (HIT) is a rare life-threatening complication following exposure to heparin. It is mediated by immune-mediated thrombocytopaenia due to antibody against heparin platelet factor 4 complex. Early identification of the condition and the prompt administration of appropriate treatment are important to prevent morbidity and mortality from HIT. We report a case of HIT associated with open abdominal aortic aneurysm repair causing anterior tibial artery and peroneal artery thrombosis following prophylactic use of unfractionated heparin for prophylaxis of venous thromboembolism. Even though enoxaparin was switched to rivaroxaban after HIT was suspected, our patient unfortunately developed a minor cerebrovascular accident during the hospital stay as a consequence of HIT.
Introduction
Heparin-induced thrombocytopaenia (HIT) is a rare life-threatening complication following exposure to heparin. It is mediated by immune-mediated thrombocytopaenia due to antibody against heparin platelet factor 4 complex. Early identification of the condition and the prompt administration of appropriate treatment is important to prevent morbidity and mortality from HIT.
We report a case of HIT associated with open abdominal aortic aneurysm (AAA) repair causing anterior tibial artery and peroneal artery thrombosis following intraoperative and postoperative prophylactic use of heparin. Even though enoxaparin was switched to rivaroxaban after HIT was suspected, our patient unfortunately developed a minor cerebrovascular cerebrovascular accident (CVA) during the hospital stay as a consequence of HIT.
Case presentation
A 55-year-old gentleman with a significant past medical history of diabetes, chronic obstructive pulmonary disease, ankylosing spondylitis and peripheral vascular disease (for which he was treated by an aortobifemoral bypass in 2010 at another tertiary center) was noted to have large infrarenal AAA on follow-up with computed tomography (CT) angiograms. A serial CT angiogram showed an increase in the size of the known infrarenal aortic aneurysm, measuring 6.7 cm×4.7 cm, with a stable mural thrombus within the aneurysmal sac. He sought a second opinion at our tertiary referral center, and in view of the large infrarenal AAA, he was offered electively open AAA repair.
Intraoperatively after adhesiolysis and exposing the aorta, bifurcated aortic graft was used to repair the AAA with the left limb anastomosed end to end to the previous aortofemoral graft and the right limb anastomosed end to side to the distal right external iliac artery. Intraoperatively, unfractionated heparin was used. He was started on prophylactic clexane postoperatively.
His initial recovery was uncomplicated, but unfortunately he developed severe left shin pain on postoperative day (POD) 8. An urgent duplex vascular scan performed the same day showed thrombosis of the anterior tibial artery (ATA) and peroneal artery (Figure 1). In view of acute thrombosis, he was offered emergency left ATA and dorsalis pedis embolectomy and short segment long saphenous vein interposition grafting, which he underwent on POD 9. Preoperative bloods on POD 8 showed an acute drop in platelet count to 44 from 284×109/L (Figure 2). No other cause for the thrombocytopaenia was identified, and given the history of exposure to heparin, a provisional diagnosis of HIT was made. The 4T scoring system score was 5, and so an urgent referral was made to haematology. Clexane was stopped, and he was started on rivaroxaban 5 mg q12h for 21 days followed by 20 mg o.m. Anti-PF4 assay was strongly positive. His platelet count was monitored daily while he was continued on rivaroxaban. Calculated creatinine clearance was 76 mL/min. He was started on rivaroxaban 15 mg q12h. Rivaroxaban levels were measured and were noted to be within therapeutic levels.
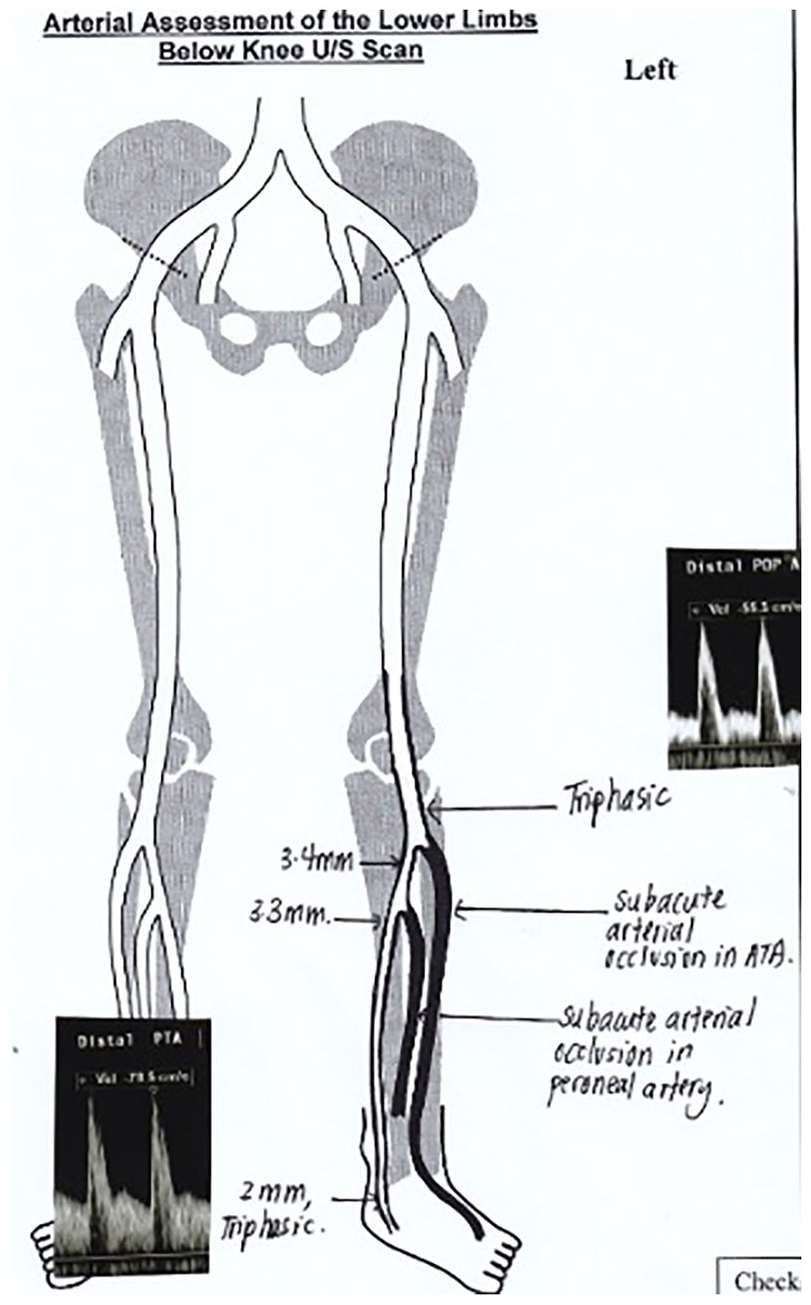
Arterial duplex scan showing anterior tibial artery thrombosis.
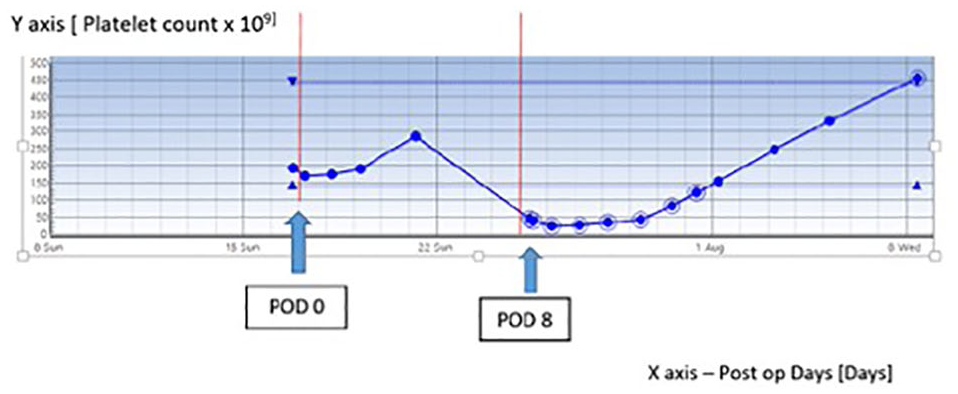
Trend of postoperative platelet count.
Unfortunately, he developed left arm weakness and left facial weakness on POD 14. Our suspicion of a left-sided CVA was confirmed by CT brain and subsequent magnetic resonance imaging of the brain. Further work-p failed to demonstrate any other cause for the CVA, and diagnosis of CVA secondary to failure of rivaroxaban on the background of HIT was made. Rivaroxaban was stopped, and the patient was counselled regarding alternative anticoagulants such as novel oral anti coagulants as well as warfarin. He decided on warfarin, which he was started on subsequently.
The patient went on to make an uneventful recovery. He was continued on aspirin, and warfarin was titrated to the international normalised ratio. Subsequently, his platelet count normalised. Neurological deficits continued to improve, and the patient was subsequently discharged well and stable, with minimal residual disability.
He has done well since and remains symptom free. Warfarin was discontinued after three months.
Discussion
HIT can be extremely subtle and needs a high index of suspicion to diagnose in the postoperative period. However, if this important diagnosis is overlooked, the consequences can be disastrous.
HIT has a very low incidence reported in the literature, typically between 0.5% and 5%. 1 Althought heparin was discovered in the 1930s, HIT was not reported till 1960, probably because it was largely unrecognised at that time and thrombocytopaenia was attributed to other causes. 2 In contrast to other immune-mediated thrombocytopaenias, HIT causes thrombosis instead of bleeding manifestations. Thromboembolic complications develop in about 50% of patients with confirmed HIT. The large vessels of the lower limbs are most frequently affected, causing venous thrombosis and pulmonary embolism, followed by peripheral arterial thrombosis and stroke. Although myocardial infarction has been reported, it is uncommon. 3
The typical onset of HIT occurs between 5 and 10 days after heparin is started, both in patients who receive heparin for the first time and in patients with re-exposure, 4 although this is variable, and rapid-onset HIT has been reported in individuals exposed to heparin in the preceding three months. This was the case in our patient, as he was exposed to heparin intraoperatively and to low molecular weight heparin postoperatively, developing thrombosis of the lower limb vessels on POD 8.
HIT can be diagnosed using both clinical and laboratory criteria. Two criteria are necessary to make a clinical diagnosis: (a) development of thrombocytopaenia/thrombosis following heparin treatment, and (b) exclusion of other causes for thrombocytopaenia/thrombosis. A commonly used scoring system for diagnosis of HIT is the 4T scoring system. 5 A score of 0, 1 or 2 is assigned for each of the following points: (a) timing of thrombocytopaenia, (b) severity of thrombocytopaenia, (c) presence of thrombosis and (d) exclusion of other causes of thrombocytopaenia. A score of six to eight points is considered as a high probability of HIT, while a score three or less is considered a low probability.
The pathogenesis of HIT has been extensively studied, and it is now clear that it is brought about by autoantibodies to platelet factor 4 complexed with heparins that initiate antibody-induced platelet activation. 6 This formed the basis of the current diagnosis of HIT using an enzyme-linked immunoassay. Other methods of diagnosing HIT include functional assays such as the platelet aggregation test and serotonin release assay. 7 Clinically, it may be suspected when there is a decrease in the platelet count of >50% or thrombosis, and confirmed by the above tests. Routine monitoring of platelet counts is not advocated due to the rarity of HIT. Such monitoring is advocated only when the risk of HIT is relatively high (>1%), such as patients who have undergone cardiac surgery or those receiving unfractionated heparin after major surgery. 8 In our patient, we noted the reduced platelet counts incidentally in preoperative bloods for the ATA embolectomy, but we presumed by that time the HIT had already established, and this was the underlying mechanism for the thrombosed ATA and peroneal arteries. An acute drop in platelet count was supportive, and the diagnosis was subsequently confirmed by antibody testing.
Once diagnosis is established, the mainstay of treatment is cessation of heparin or its derivatives and initiation of an alternate form of anticoagulation. Prophylactic dose anticoagulation is not sufficient to compensate for the massive prothombotic drive, even if the patient is clinically asymptomatic. 9 Currently, the most commonly used drugs approved for use in HIT include direct thrombin inhibitors such as argatroban and antithrombin-dependent factor Xa inhibitor danaparoid. 10 Similar case series have shown good outcomes in patients with HIT treated with fondaparinux 10 or bivalirudin. 8 Of note is the recommendation to avoid prophylactic platelet transfusions in patients with HIT. The risk of bleeding is negligible, and and such transfusions can increase the risk of thrombosis. 11 It seems that in our patient, there was treatment failure with rivaroxaban, and the patient suffered a minor CVA, with full recovery of function subsequently. Unfortunately, we did not have bivalirudin available. Hence, we switched to warfarin after consulting with haematology.
The lack of availability of alternative anticoagulating agents such as bivalirudin can certainly be a problem in cases similar to ours. We believe that hospitals dealing with cardiac vascular surgery with common use of unfractionated heparin may come across HIT, although its occurrence is extremely rare. It is important to have access to medications such as bivalirudin, as they are indicated in rare instances of treatment failure of rivaroxaban such as our case.
Conclusion
Our case is unique in the sense that to the best of our knowledge, there are no previous reports of HIT affecting AAA repair. Second, there was treatment failure of rivaroxaban, and despite being on anticoagulation, our patient went on to suffer a minor CVA. Our case illustrates the potential serious sequealae of HIT, the subtle nature of the presentation and the need for a high index of suspicion for diagnosis. Prompt diagnosis and treatment can reduce the associated morbidity, even though treatment failures do occur rarely.
Footnotes
Acknowledgements
NA.
Authors’ contributions
LBS conceptualised the project and prepared the initial draft with FC. YH and EC supervised the project. All authors read and approved the manuscript.
Availability of data and materials
NA.
Ethical approval
Singapore General Hospital does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient for his anonymised information to be published in this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
