Abstract
Objective
To investigate the effect of the pre-administration with aminophylline on the occurrence of post-dural puncture headache (PDPH) in women undergoing caesarean section by combined spinal-epidural anaesthesia (CSEA).
Methods
The study enrolled women undergoing elective caesarean sections with CSEA and randomly allocated them into two groups; for 30 min immediately after the infant was delivered, group A received 250 mg aminophylline intravenously and group B received an equal volume of normal saline. Demographic data, operation time, intraoperative blood loss, intraoperative transfusion volume and the occurrence of PDPH during the first 7 days after the operation were recorded. Side-effects such as hypersensitivity, convulsion and arrhythmia were also recorded in the patients and infants in group A within 24 h after aminophylline administration.
Results
A total of 120 patients aged 24–38 years (pregnancy range, 38–42 weeks) were randomly allocated into two groups (
Conclusions
Intraoperative intravenous infusion of 250 mg aminophylline reduced the incidence of PDPH after caesarean section under CSEA with no side-effects.
Introduction
Combined spinal-epidural anaesthesia (CSEA) is widely used during caesarean sections and post-dural puncture headache (PDPH) is one of the most common anaesthetic complications. 1 According to research, the incidence of PDPH in patients after caesarean section was 10% to 30%, which was significantly higher than that in other CSEA patients.2,3 PDPH is often accompanied by nausea, vomiting, photophobia, tinnitus and other symptoms, which seriously affect the life quality of patients.4,5 Recently, it was shown that oral or intravenous administration of theophylline or aminophylline can be effective in the treatment of PDPH, and that early treatment with aminophylline was more effective. 6 However, there have been no reports on the prevention and treatment of PDPH in patients after caesarean section. Therefore, the objective of this current study was to investigate the effect of aminophylline on the occurrence of PDPH in patients undergoing caesarean section under CSEA and to provide reference information for the clinical prevention of PDPH.
Patients and methods
Trial registration and ethics
The Consolidated Standards of Reporting Trials recommendations for reporting randomized, controlled clinical trials were followed in this study. 7 This study was also registered with the Chinese Clinical Trial Registry (ID: ChiCTR1800016831) on 26 June 2018. Ethical approval for the study was provided by the Ethics Committee of Jiangning Hospital Affiliated to Nanjing Medical University, Nanjing, Jiangsu Province, China on 1 March 2018 (no. 2018-3-1). All patients involved provided written informed consent prior to the study.
Study participants
This randomized, double-blind, controlled study enrolled patients with American Society of Anesthesiologists (ASA) physical status I or II, aged 24–38 years, G1P0LOA (i.e. G1, pregnant once, so the first pregnancy; P0, produced 0 times, so has not given birth previously; LOA, left occipital position of the fetus), undergoing elective caesarean section by CSEA between 2 March 2018 and 3 June 2018 in the Department of Anaesthesiology, Jiangning Hospital Affiliated to Nanjing Medical University. The exclusion criteria were as follows: (i) a history of intraspinal puncture; (ii) contraindications, such as coagulation dysfunction, spinal malformation and infection of puncture site; (iii) abnormal liver or kidney function; (iv) cardiovascular system diseases, respiratory system diseases and diseases of the nervous system; (v) a history of drug hypersensitivity; (vi) intraoperative blood loss > 500 ml; (vii) failure of the spinal canal puncture (epidural or spinal needle puncture failure).
Study design
Patients were randomly assigned to one of two groups using a computer-generated table of random numbers. Patients in group A received 250 mg of aminophylline immediately after the infant was removed and those in group C received an equal volume of saline. Both patients and observers were blind to the identity of each group’s patients. Drugs were prepared using white 100-ml plastic bottles by an anaesthesiologist who remained independent of the study.
Interventions and outcome measures
To decrease upper airway secretions, 0.5 mg atropine was injected intramuscularly 30 min prior to the patient arriving at the operating room. Patients were punctured intravenously with a 20-gauge needle on the dorsum of both forearms for intraoperative fluid resuscitation and intravenous infusion of aminophylline. Electrocardiography, heart rate (HR), noninvasive blood pressure (BP) measurement and pulse oximetry were monitored throughout the procedure.
A 25G spinal anaesthesia needle was used to inject 0.5% bupivacaine 2 ml into the subarachnoid space in the L3–4 space within 30 sec. Those who failed to meet the surgical requirements during the operation were given 1.5% lidocaine supplementation 5–10 ml into the epidural space. Patients in group A were administered an intravenous infusion of 250 mg aminophylline (diluted in 100 ml saline; Yuncheng Shiyao Pharmaceutical, Shanxi, China) for 30 min immediately after the infant was removed. Group C received the same volume of normal saline. Haemodynamics were maintained smoothly during the operation. Ephedrine was injected intravenously at 6 mg/time when hypotension occurred (systolic BP < 90 mmHg); and uradil was injected intravenously 5 mg/time when hypertension occurred (systolic BP > 140 mmHg). Atropine was injected intravenously 0.5 mg/time when bradycardia occurred (HR < 50 beats/min); and esmolol was injected intravenously at 10 mg/time during tachycardia (HR > 100 beats/min).
Post-dural puncture headache, which was confirmed according to the third edition of the international classification of headache, 8 was evaluated daily for the first 7 days after the operation by an anaesthesiologist who was blinded to the pre-treatment drug and grouping of patients.
Side-effects in the patients and infants in group A were checked blindly by a separate anaesthesiologist within 24 h after administration of aminophylline and included allergy, convulsions and arrhythmia.
Statistical analyses
According to previous studies,2,3 the frequency of PDPH in the group treated with aminophylline was expected to be reduced by approximately 15%. Power analysis showed that based on a reduction rate of 15% with α = 0.05 (1-tailed) and a 15% dropout rate, a sample size of at least 54 per group was needed for 90% power. The study aimed to include 60 patients in each group. The statisticians did not participate in the clinical trial and did not know how the patients entered the groups. All statistical analyses were performed using the SPSS® statistical package, version 16.0 (SPSS Inc., Chicago, IL, USA) for Windows®. Continuous variables are presented as mean ± SD and differences between groups were analysed using mutual comparison after single factor variance analysis. Frequencies were considered as categorical variables and were analysed using χ2-test (Kruskal–Wallis test). A
Results
A total of 120 patients aged 24–38 years (pregnancy range, 38–42 weeks) were enrolled in this study. One participant was excluded from group A for intraoperative bleeding > 500 ml. Two patients were excluded from group C (one patient had intraoperative bleeding > 500 ml and one patient had a failed spinal canal puncture [spinal needle puncture failure]). The data from 117 patients were analysed (Figure 1). There were no significant differences with respect to sex distribution, age, body weight, ASA physical status, intraoperative blood loss and intraoperative transfusion volume between the two groups (Table 1).
Flow diagram showing the patient numbers are various stages of this randomized, double-blind, controlled study of the effects of aminophylline on post-dural puncture headache in patients (
Demographic and clinical characteristics of patients (

Data presented as mean ± SD or
ASA, American Society of Anesthesiologists.
No significant between-group differences (
The frequency of PDPH in patients from group A was significantly lower (two of 59 patients, 3.4%) compared with patients in group C (10 of 58 patients, 17.2%) (
Frequency of post-dural puncture headache in patients (
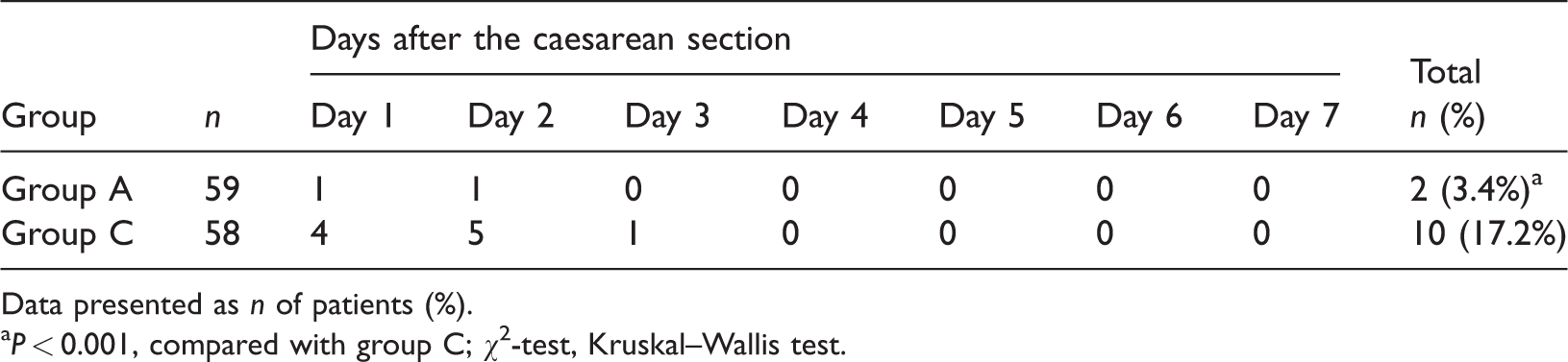
Data presented as
a
All participants showed stable cardiovascular profiles during their operations. There were no side-effects occurring in the patients and infants from group A with regard to allergy, convulsions and arrhythmia.
Discussion
Studies have shown that the incidence of PDPH was higher in women and obese patients compared with men of the same age and patients with a normal BMI, reaching 18% and 21%, respectively, which might be related to the greater flow of epidural or subarachnoid fluid in women and obese patients.9,10 Most expectant mothers meet these two conditions at the same time, resulting in a higher incidence of PDPH compared with other patients undergoing CSEA. 9
At present, most clinicians and researchers believe that the occurrence of PDPH is related to the loss of cerebrospinal fluid and the decrease of intracranial pressure after intraspinal puncture. 11 However, the exact mechanism remains unclear and might be related to the following factors: (i) decreased intracranial pressure leading to the compensatory expansion of the intracranial blood vessels and increased blood flow, resulting in PDPH; 12 (ii) reduced intracranial pressure directly activating adenosine receptors, stimulating intracranial pain sensitive structures and causing intracranial vascular dilatation, resulting in PDPH; 13 and (iii) decreased intracranial pressure causing the brain tissue to sink and shift, thus stretching the intracranial blood vessels and the trigeminal, vagus, glossopharyngeal nerve and other cerebral nerves, resulting in the occurrence of PDPH. 14
To date, the treatment of PDPH includes recumbent rest, fluid replacement, the administration of analgesic drugs and epidural saline or autologous blood filling.15–17 A previous study demonstrated that the success rate of autogenous blood filling using an epidural blood patch was about 90% after the initial treatment, which was the most effective method for the treatment of PDPH. 17 However, autologous blood filling is an invasive treatment method, which might cause subarachnoid adhesion and infection, and the procedure is complicated. 18 The incidence of PDPH decreased slightly with the improvement of the intraspinal puncture needle. 19 In this current study, the incidence of PDPH in the control group was 17.2%, with most patients experiencing symptoms within 3 days of the operation, which was consistent with previous findings. 15 This current study demonstrated that the pre-administration of aminophylline during an elective caesarean under combined spinal-epidural anaesthesia was simple and noninvasive, and it significantly reduced the incidence of PDPH and was not associated with any related side-effects. At present, the mechanism by which aminophylline prevents PDPH remains unclear, but it may be related to the inhibition of calcium uptake by the endoplasmic reticulum of endothelial cells, stimulation of sodium and potassium pumps, increased secretion of cerebrospinal fluid, contraction of intracranial blood vessels by blocking adenosine receptors and blocking the transmission of pain perception. 20
As a methylxanthine drug, clinical pharmacological studies showed that the adverse reactions of aminophylline were mainly allergy, arrhythmia and convulsions. Some studies suggested that 250 mg of aminophylline can be effective in the treatment of PDPH without significant side-effects and it can be used in parturients without affecting lactation.6,21 In this current study, no adverse reactions were observed in all pregnant women and infants. Therefore, the dosage of aminophylline used in the study was safe.
This current study had a number of limitations. First, there were no comparisons at different time-points within each group. Secondly, there was no comparison of pain scores between the two groups.
In conclusion, this study showed that an intraoperative intravenous injection of 250 mg aminophylline effectively prevented the occurrence of PDPH after caesarean section under combined spinal-epidural anaesthesia. These findings support the use of aminophylline in this setting in the future.
Footnotes
Acknowledgments
Chao-Jie Yang and Wei Wang designed the study. Wan-You Yu supervised the practical carrying out of the clinical trial. Chao-Jie Yang and Tao Chen analysed the data. Chao-Jie and Xin Ni wrote the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
