Abstract
Background:
A recent systematic review alludes to the effectiveness of social media in facilitating smoking cessation. However, most interventions center on mobile applications in engaging smokers. A novel portable exhaled-carbon-monoxide self-monitoring device, the Smoke-analyzer to Track and Expedite Actions and Decisions to Eliminate Smoking (STEADES) linked to a specially-designed mobile phone application has been developed, which relays exhaled carbon monoxide-related data from the smokers to their selected quit supporters via social media.
Aim:
The study aimed to determine the precision and feasibility of using the STEADES device among smokers in primary care.
Method:
This pilot study recruited 15 Asian adult smokers from a primary care polyclinic in Singapore. Their exhaled carbon monoxide measurements were measured serially using STEADES, compared to a commercial smokerlyzer as reference. The subjects could use the device to transmit their exhaled carbon monoxide measurements via mobile application and social media to their acquaintances in the next two weeks. Data on the subjects’ demographic and clinical characteristics, their views on STEADES (from a questionnaire), and its utility (from the application) were collated on enrolment and at the end of study.
Results:
Measurements using STEADES were significantly correlated with those from a commercial smokerlyzer (Pearson correlation=0.882,
Conclusion:
The STEADES prototype had shown feasibility in this proof of concept study. Most subjects were in favor of using it to support their smoking cessation but its accuracy required enhancement.
Introduction
Tobacco use is the leading global cause of preventable deaths and a major threat to public health. Widespread efforts to curb its use are met with limited success. The expanding role of social media in healthcare creates an unprecedented platform to provide asynchronous behavior support to smokers. Social media facilitates verbal or social persuasion via personalized encouragement or prompts to energize the smoking cessation efforts of smokers. 1 A recent systematic review has alluded to the feasibility, acceptability, and preliminary effectiveness of social media interventions in smoking cessation. 2
With increasing expansion of the use of info-communication technology in healthcare, a mobile-phone based exhaled carbon monoxide (eCO) meter or analyzer to detect cigarette smoking has been shown to appeal to smokers in their attempts at quitting. 3 This self-monitoring device with the capability for reliable quantitative eCO measurements, if virtually supported by anti-smoking advocates over social media, provides opportunities for continuous behavioral support to smokers.
Hence, a portable smoke analyzer prototype, the Smoke-analyzer to Track and Expedite Actions and Decisions to Eliminate Smoking (STEADES), has been developed for pilot testing. It can measure and transmit eCO data via Bluetooth technology to a mobile-phone application (app) and this is subsequently transmitted to pre-identified recipients via social media (Facebook was the only option in this feasibility study). The STEADES device dimensions are 101 mm (length), 62 mm (breadth), 37 mm (thickness), and it weighs 120 g. Its sensor targets eCO specifically with very low cross-sensitivity to other gases in the environment, such as hydrogen sulphide (H2S) (H2S<0.1% measured gas @20 ppm), nitrous oxide (NO2) (NO2<0.1% measured gas @10 ppm), and hydrogen (H2) (at 200C H2<60% measured gas @400 ppm).
This pilot study aimed to determine the accuracy of STEADES in terms of eCO measurements in comparison with a commercial smokerlyzer, and to validate the proof of concept in evolving the utility of the device by smokers to support their quit efforts via social media.
Methods
The pilot study included assessments of (a) the accuracy of STEADES in measuring eCO, as compared to a commercial smokerlyzer (Bedfont Micro-Smokerlyzer B19522); and (b) smokers’ utility and feedback on STEADES in monitoring their quit efforts and transmitting their eCO data to their assigned recipients over a period of two weeks.
Adult multi-ethnic Asian smokers were recruited from a typical public primary care clinic (polyclinic) in eastern Singapore. The subjects were included if they were currently smoking cigarettes daily and were willing to use the STEADES device for a period of two weeks and to download the STEADES app in their smartphone for the transmission of data using social media. Those without smartphones were excluded. The stages of readiness to quit smoking, based on the Prochaska transtheoretical model, were not considerations for the enrolment into the study
The subjects were identified by physicians and the advanced practice nurse (APN) at the study site, who were then briefed on the study procedure. Informed consent was obtained after their doubts and queries were addressed. The subjects were instructed on the use of the STEADES device and mode of transmission of their eCO readings to their assigned recipients via a specially designed mobile phone app, which they downloaded into their personal smartphone (supported by Android models only).
In Part 1 of the study, the enrolled subjects were instructed to blow into the STEADES device to measure their eCO, followed by a similar maneuver using the Bedfont smokerlyzer at four time-points: commencement of study with about two hours of temporary abstinence from cigarette smoking, immediately after smoking a cigarette, 30 min, and 60 min post-smoking.
In Part 2 of the study, the same subjects were instructed to monitor their eCO using solely the STEADES device at least once daily for a period of two weeks, with the option to use it to relay the measurements to their assigned recipients using social media at their convenience. They also completed a questionnaire at the beginning and at the end of the study, which collected data on their demographic characteristics, existing medical conditions, nicotine dependency levels based on the Fagerström, 4 and stages of readiness for behavior change based on the Prochaska transtheoretical model. 5 An open-ended question in the questionnaire allowed the subjects to provide qualitative feedback on their use and perceptions of the STEADES device.
The subjects were reimbursed SGD20 and SGD30 upon completion of Parts 1 and 2 of the study respectively. The study protocol was reviewed and approved by the SingHealth Centralized Institutional Review Board (CIRB no: 2016/2025).
Statistical analysis
The reliability of the eCO measurements from STEADES device was assessed using intra-class correlation coefficients (ICCs). An ICC≥0.75 indicates excellent reliability; 0.4≤ ICC<0.75 shows fair to good reliability, and ICC<0.4 indicates poor reliability. 6 The Pearson correlation was used to test the correlation between the STEADES device and the Bedfont smokerlyzer. The eCO readings were classified as low (1–6 ppm), medium (7–10 ppm), and high (11-–30 ppm). The agreement between the three groupings of eCO was assessed using the Kappa statistic. Kappa values of ≤0 indicate no agreement; none to slight (0.01–0.20); fair (0.21–0.40); moderate (0.41–0.60); substantial (0.61–0.80) and almost perfect (0.81–1.00) agreement. 7 The differences in the smoking behavior on enrolment and at the end of the study were determined using McNemar’s test.
Results
Fifteen subjects completed the eCO measurements using both STEADES and the commercial smokerlyzer on enrolment. They included 13 males (86.7%); Malay (66.7%), Chinese (26.7%), and Indian (6.7%); single (46.7%), married (46.7%); and 70% had at least higher secondary education (Table 1). Their mean age was 29 years (25–48 years). All of them were employees and 93.3% lived in public housing. One-third had hypertension (33.3%), 26.7% had dyslipidemia, and 13.3% had type 2 diabetes mellitus. They smoked cigarettes for an average of eight pack-years, and only one subject (6.7%) had ever attended smoking cessation clinic and used medication to support smoking cessation.
Demographic characteristics of the study population (

COPD: chronic obstructive pulmonary disease.
Overall, the ICC of 0.847 showed excellent reliability in the eCO measurements using STEADES compared to the commercial smokerlyzer. The overall Pearson correlation coefficient of 0.883 and

Comparison between the exhaled carbon monoxide (eCO) levels (in ppm) using the Bedfont Micro-Smokerlyzer and the Smoke-analyzer to Track and Expedite Actions and Decisions to Eliminate Smoking (STEADES) device.
Thirteen subjects used the STEADES device with a median frequency of nine (range: 2–30) over the two weeks’ observation period. The most popular time of the day in the use of the STEADES device was 07:00–10:00 in the morning. Five subjects used Facebook to transmit the eCO data. The remaining eight subjects showed their eCO measurements from their smartphone directly to their family members (including spouse, parents, and siblings), their friends, and colleagues. Two of them relayed the data to the study team nurse, considering her as their “supporter” (Figure 2).
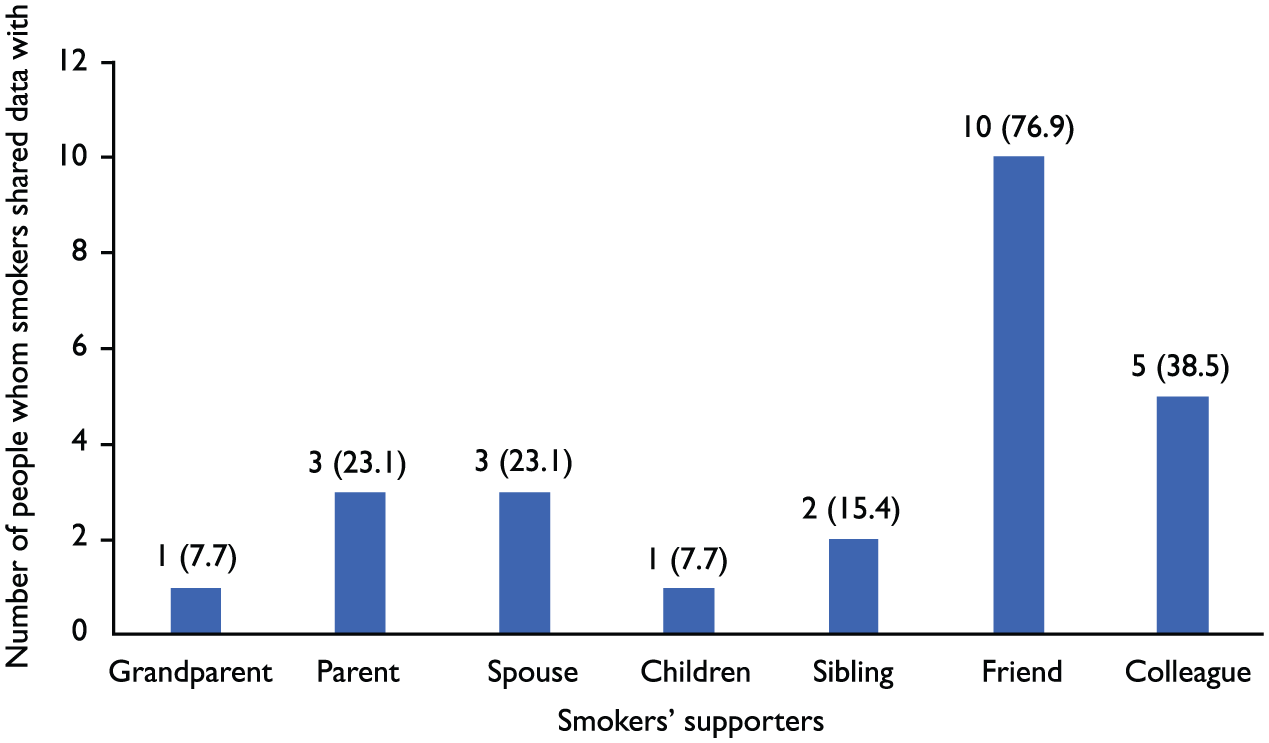
Persons with whom smokers shared their exhaled carbon monoxide (eCO) data.
Between the commencement and the end of the observation period, the subjects showed a shift from moderate (46.2% to 38.5%) to low nicotine dependence (7.7% to 23.1%) based on the Fagerström Test scores, although the differences were not statistically significant. For the stages of readiness for change, more subjects shifted from contemplation (from 53.8% to 30.8%), pre-contemplation (15.4 to 23.1) to action stage (15.4% to 30.8%), although none had quit smoking completely after two weeks (Table 2).
Fageström Test scores and Prochaska model of stages of readiness to quit smoking.

The subjects indicated that the STEADES helped them to understand the progress of their smoking cessation efforts (85.7%) all or most of the time; they felt more confident of success in smoking cessation (50%); felt comfortable (92.8%), and perceived receiving support (92.9%) in their quit-smoking attempt from the recipients of their eCO data and would recommend the STEADES device to other smokers (78.6%). Half of them (50%) reported that they would use the STEADES regularly as stipulated by the study protocol but only 38.5% would consider buying the device, assuming that the cost was about SGD30 or USD22.
While eight subjects (57.1%) indicated that the STEADES provided accurate information, four subjects highlighted a potential inaccuracy of the device. Two of them reported technical difficulties pertaining to connectivity to Facebook and downloading of the app. Other qualitative feedback remarks are encapsulated in Table 3.
Qualitative feedback on the use of Smoke-analyzer to Track and Expedite Actions and Decisions to Eliminate Smoking (STEADES) by the study participants.

Two subjects did not use the STEADES for the subsequent two weeks as instructed. One had a defective mobile phone which subsequently failed to connect STEADES via the app and was dropped from the study. The other subject sustained a severe lower limb injury which required surgery and hospitalization for two weeks and hence failed to participate during the stipulated observation period.
Discussion
This pilot study validates the proof of concept evolving around personal monitoring of smoking cessation efforts by a portable smoke analyzer and transmitting data to their assigned recipients over social media. The STEADES prototype demonstrated satisfactory accuracy, compatible with a similar commercially available instrument used in healthcare establishments, but with added functionality. Although its precision can be further improved at the higher eCO range with adjustment of the sensor, the STEADES prototype demonstrates the potential to track smoking cessation efforts and to garner support using social media. Its portability and usage at the convenience of the smoker are other advantages.
However, the intervention with the STEADES device will be most apt and timely when the smokers reach the contemplation stage and are ready to quit smoking. The device will complement their quit plan. Screening to identify a subset of the smoker population who are ready to quit smoking using the Prochaska stages of behavioral change may potentially increase their success rate. This was alluded to by a subject’s qualitative feedback on using the device only when he/she reached the contemplation or decision stage. Shie et al. 8 at Taipei Veterans General Hospital have shown that, among smokers with high intention to quit, an additional independent predictor for successful smoking cessation was the eCO level of less than 4.5 ppm at day eight of the smoking cessation program. Goldstein et al. 9 have also reported that the use of eCO measurement provided biomedical feedback on assessing smoking behavior.
Only five subjects (38%) used Facebook to convey their eCO readings. The rest were amenable to show their eCO readings directly to their family members, friends, and colleagues directly from their phones. One feasible measure to increase utility will be to expand the number of social media platforms that can be linked to the STEADES app.
Thus, targeting those who are active social media users and developing a ready pool of virtual anti-smoking advocates to receive the data and interact with the smokers may be crucial for successful implementation of this social media intervention. In addition, recruitment and training of family members and friends as voluntary anti-smoking advocates will be a strategy to use, as the subjects largely targeted familiar acquaintances to convey their data from the app. Healthcare professionals and anti-smoking advocates from healthcare establishments can form another pool of back-up “supporters” to assist in the attempts to quit smoking (see Figure 3).

Uses of the Persons Smoke-analyzer to Track and Expedite Actions and Decisions to Eliminate Smoking (STEADES) device.
Half of the subjects were willing to use the device once daily as per the study protocol. The data showed its utility almost once daily on average over the two weeks’ period. The lack of interactive features, gamification, and other incentives in the app design could discourage more intensive, sustained use of the prototype. A dashboard which highlights the progress of the quit efforts in an animated, pictorial, or interactive fashion for easy reference could further enhance the app design.
Only one in three subjects showed willingness to pay for the device. Nevertheless, the manufacturing cost of the device can be potentially reduced by mass production due to economy of scale. Its design and accuracy can be improved in the next version of the prototype to enhance the visual perception of consumers and to lower their purchasing threshold. Another option is to loan the devices to selected subjects at more affordable rates.
Limitations and strength
The number of subjects was small in this pilot study, as the primary intent was to validate the proof of concept. The next phase will be a randomized controlled trial (RCT) to determine the proof of value of STEADES and the effectiveness of associated social media intervention. The results will allow a more realistic computation of the sample size for a subsequent RCT. Assessing the quit rate, enabling factors, and cost-effectiveness of the successful social media intervention will be the main outcomes of the RCT.
Conclusion
This pilot study shows the compatible accuracy between the STEADES device and a commercial smokerlyzer. Most subjects seemed receptive to use the STEADES to monitor their eCO to track their smoking cessation efforts, and about 40% of them were willing to convey their data to their preferred recipients via social media.
Footnotes
Acknowledgements
The investigators appreciate the sponsorship by the Singapore Totes Board and SingHealth Polyclinics. They would like to thank Caris, Patricia, and Marissa for their administrative support during the study execution.
Author contributions
NC Tan conceptualized the development of the portable smoke analyzer, its linkage to social media and the design of the pilot study. ZM Mohtar executed the pilot study to collect the relevant data and feedback from the subjects. U Sankari audited, rectified, and de-identified the data. NC Tan and EYL Koh analyzed and interpreted the data. DHC Tay, S Yu, WBW Tan designed and developed the STEADES prototype and the app. NC Tan, U Sankari and EYL Koh drafted the manuscript. All authors reviewed and finalized the manuscript.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The Singapore Tote Board sponsored the development of the STEADES device. The pilot study was funded by the SingHealth Polyclinics POC fund.
