Abstract
Background:
Critically ill patients require sedation for patient comfort and ventilator synchrony. Despite the extensive use of sedation, to date there is no consensus on the best sedation practices. We attempt to investigate our local sedation practices.
Method:
This was a single-centre prospective, observation cohort study in medical and surgical intensive care unit (ICU) patients who were ventilated and sedated for more than 24 hours. Baseline demographics were obtained and patients followed-up for 28 days or to ICU discharge. Details on sedatives, ventilation duration, vasopressors and renal replacement therapy use, hospital/ICU length of stay, mortality, delirium, and sedation depth were collected and analysed.
Results:
From March to July 2012, 58 patients were recruited with a mean Acute Physiology and Chronic Health Evaluation II (APACHE II) score of 20.2 ±8.5. Hospital mortality rates were 32.8%. Patients were followed-up for 387 ICU patient-days. In the early period (first 48 h), the most popular sedative used was propofol (74.1%), followed by morphine (29.3%). In the subsequent period (>48 h), most patients were not sedated (47.6%); morphine became the most popular sedation drug (32.5%) followed by propofol (31%). Ketamine, haloperidol and diazepam were not given. In total, 1994 Richmond Agitation and Sedation Score (RASS) assessments were performed over 387 ICU patient-days; 11.1% of RASS assessments were prescribed a sedation target, and 86% of them met the prescribed targets. Delirium was observed in 22.4% of patients. Compared with medical patients, surgical patients were more likely to be prescribed a sedation target (14.2% vs. 7.4%, p<0.01), require lower doses of sedation, have a RASS score of between −2 to 1 (84.8% vs. 72.3%, p<0.01) and have fewer incidences of delirium (4.1% vs. 12.1%, p=0.01).
Conclusion:
Propofol and morphine were the most commonly prescribed sedatives. Different sedation practices between units may contribute to a reduction in delirium incidence.
Introduction
Sedation is commonly prescribed in the intensive care to minimize ventilator dyssynchrony, enhance patients’ comfort and to aid in the intensive care unit (ICU) procedures. Jackson et al., in their systematic review of sedation practices, showed that suboptimal sedation was present in most trials, with a tendency towards over-sedation. 1 While there is currently no consensus on the best sedation practices, the Society of Critical Care Medicine published a guideline for sedation and analgesia in the ICU in 2013. 2 Despite this, compliance has been low. 1 Light sedation has been shown in recent years to be associated with a shorter ICU length of stay, to reduce the incidence of delirium, and to improve survival rates and duration of mechanical ventilation.3-7 However, light sedation also increases the stress response, although this has not led to an increased incidence of myocardial ischaemia. 2
Delirium is an important complication in the critically ill patient, with an incidence ranging from 16% to 29%.8,9 It is associated with increased mortality, prolonged ICU and hospital length of stay, greater costs and resource utilization and the development of post-ICU cognitive impairment. 2 The Richmond Agitation-Sedation Scale (RASS) is a score used to provide a reliable and reproducible diagnosis of delirium. 10
The ANZ SPICE study 3 reported that early sedation depth predicts mortality and ventilator time. The SPICE SG study was a prospective, observational cohort study conducted in Singapore in 2012 to investigate sedation practices in public institutions. We report a nested-cohort study involving the Surgical and Medical ICUs in our institution with the aim to define our institution’s sedation practices in terms of drug use, sedation depth and incidence of delirium, with the potential to use this as a basis for further research and quality improvement initiatives.
Methods
Study design and process
From March to July 2012, SPICE SG conducted a prospective, observational, longitudinal multi-centre cohort study, involving seven ICUs (four surgical, three medical) in four major public hospitals in Singapore using the ANZ SPICE protocol. 3 We extracted and report on our local institution data which included a medical and a surgical ICU. Our institution’s ICUs follow a closed model of care, led by a full-time intensivist. In the surgical ICU (SICU), the ICU is managed by intensivists who trained from anaesthesia, while in the medical ICU (MICU) they are trained from internal medicine/respiratory and critical care backgrounds. There is an on-duty specialist registrar at all times, and the nursing ratio was 1:1–2.
Ethics approval was obtained and the requirement for informed consent was waived due to the observational nature of the study.
Inclusion and exclusion criteria
All sedated, intubated and mechanically ventilated adult ICU patients who were expected to remain for more than 24 hours were eligible. Patients with suspected or proven dementia, neurological impairment, psychiatric illnesses or who were unable to communicate with the investigators were excluded. No centres recruited more than 30 patients.
Study logistics
Prior to commencement of the study, the investigators, research staff and ICU nurses were trained in the ANZ SPICE protocol, including assessment of patients using the RASS 10 and the Confusion Assessment Method for Intensive Care (CAM–ICU). 11 The local languages (English, Mandarin, Malay, Tamil, local dialects) were used to communicate with the patients.
Definitions and data collection methods
We used the ANZ SPICE study protocol 3 with a streamlined standardized case report form for data entry.
Upon enrolment into the study, relevant demographic data including age, sex, weight, Acute Physiology and Chronic Health Evaluation II (APACHE II) 12 and admission source were collected.
RASS and pain scores were assessed at baseline and every 4 hours by trained nurses. Patients who were in the RASS range of −2 to +1 were considered lightly sedated. Those in the RASS range of −3 to −5 were considered deeply sedated. Patients with RASS greater than +2 were agitated. Daily CAM–ICU assessments were only performed for lightly sedated patients. A patient was diagnosed with delirium if CAM–ICU was positive.
The first 48 h of ICU admission was considered the early period. A patient was considered to have early deep sedation if a RASS score of −3 to −5 was recorded during this period. The period after was considered the subsequent period.
All cumulative sedative medication infusions and doses were collected. Daily sedation cessations and indications were noted. Adjunct therapies, such as use of physical restraints, renal replacement therapy and vasopressors, were recorded. The patients were followed-up for up to 28 day or until ICU discharge.
We recorded outcomes of development of delirium, tracheostomy performed, ventilation duration, vasopressor and renal replacement therapy use, ICU/hospital mortality and ICU/hospital length of stay. A subgroup analysis comparing patients in the MICU and SICU were also performed.
Statistical analysis
Statistical analysis was performed using SPSS 17. Comparisons of categorical data and proportion were performed using the chi-squared or Fisher’s Exact Test when appropriate. Parametric continuous variables were compared using the Student’s t-test. Non-parametric variables were compared using the Wilcoxon Rank-sum test.
Results
Baseline characteristics
In total, 58 patients were enrolled over 5 months. Their baseline demographics are presented in Table 1. We assessed a total of 387 ICU patient-days, of which 348 were ventilated days; 116 were in the early (first 48 h) period and 271 were in the subsequent (after 48 h) period.
Study cohort characteristics.
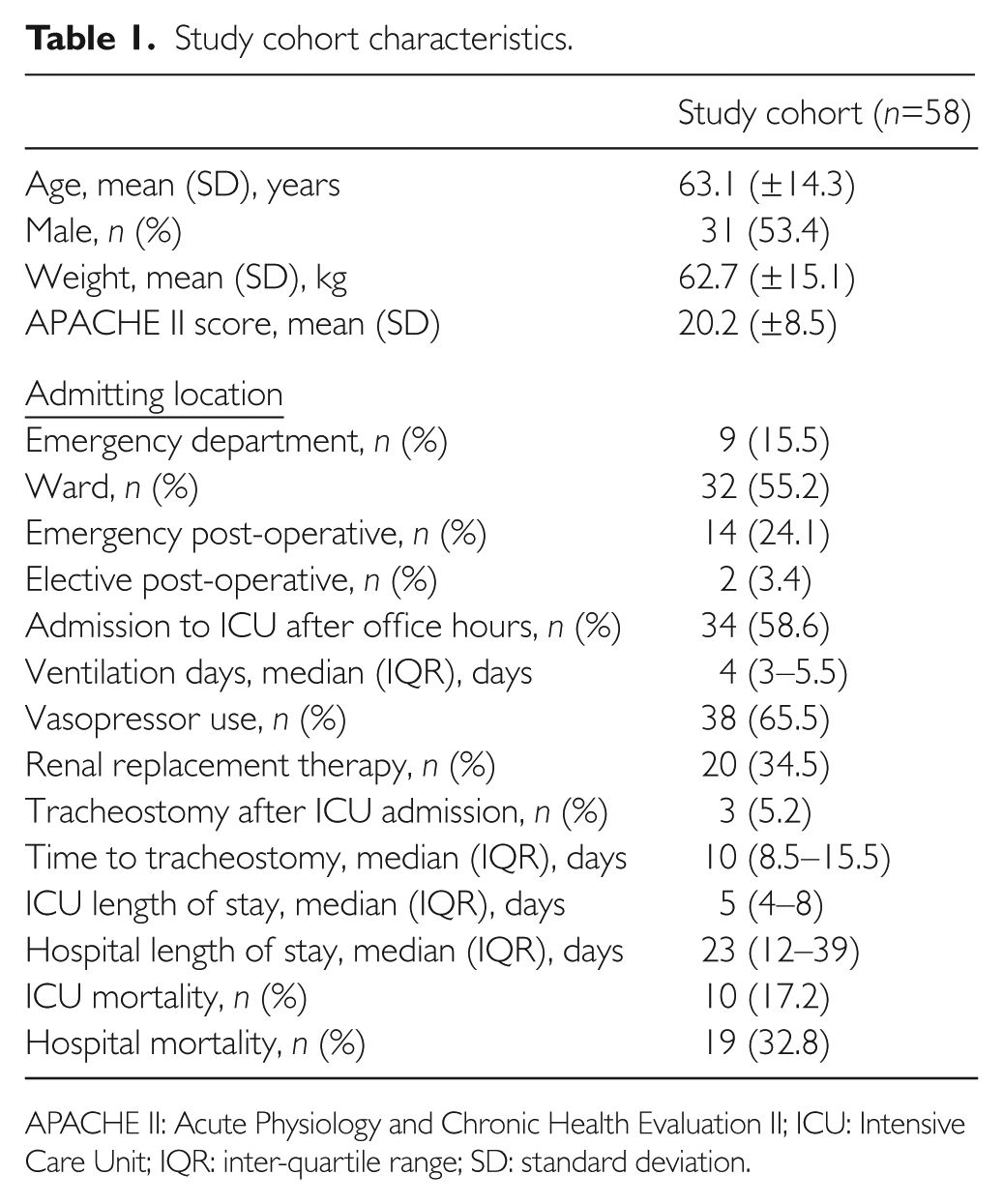
APACHE II: Acute Physiology and Chronic Health Evaluation II; ICU: Intensive Care Unit; IQR: inter-quartile range; SD: standard deviation.
31.0% of patients were lightly sedated within the first 48 h. Patients who were deeply sedated in the early period were more likely to be males (67.5% vs. 22.2%).
Sedation in the ICU
In the early period the most popular sedation drug used was propofol, given for 86 patient-days (74.1%), followed by morphine (34 patient-days, 29.3%). In the subsequent period, most patients were not sedated (129 patient-days, 47.6%); morphine became the most popular sedation drug (88 patient-days, 32.5%), followed by propofol (84 patient-days, 31%). Patients were not given haloperidol, diazepam or ketamine.
Sedatives were generally preferred over the use of analgesics, with 87.9% of patients sedated with propofol and 50% with morphine during the entire study period. The overall drug regimen and the comparison of type and dose of sedatives in lightly sedated versus deeply sedated patients in the first 48 hours are presented in Table 2.
Sedatives used and their doses.

mean ±standard deviation.
p-value calculated based on the mean dose of sedative used per day.
Note: light sedation refers to RASS −2 to 1; deep sedation refers to RASS ≥ (−3).
Sedation cessation refers to the deliberate intent by the clinician to stop sedation. This was done on 83 patient-days (21.4%), with the main indication being for extubation. This is presented in Table 3. In the five patients where routine sedation cessation was performed, there was no difference in ventilator days (6.4 days vs. 6.0 days where no routine sedation cessation was done) nor ICU length of stay (9.8 days vs. 6.8 days).
Incidence of deliberate sedation cessation and their reasons.

Note: deliberate sedation cessation refers to when sedation was intentionally stopped by the intensivist.
Clinical outcomes
There were 1994 RASS assessments performed over 387 ICU patient-days. In 221 RASS assessments (11.1%), the ICU team had a prescribed sedation target. Patients who were prescribed a sedation target met the prescribed targets 86% of the time.
Overall, 1577 (79.1%) of the sedation were in the light sedation range. This occurred more often in the subsequent compared with the early period (84.7% vs. 64.8%). There were more deep sedations in the early compared with late periods (30% vs. 13.8%). The range of RASS scores over the two periods are presented in Figure 1.

Daily sedation level scores in all ventilated patients.
Deep sedation occurred in 32 (55.2%) patients within 4 hours of commencing ventilation and in 41 (70.7%) patients at 48 hours.
In total, 13 patients (22.4%) had at least one episode of delirium during the ICU admission. There were 22 (5.9%) patient-days of CAM–ICU being positive with a median duration of 1 day (IQR 1–2), with more occurring in the subsequent period compared with the early period (60.1% vs. 39.1%).
In the cohort, 65.5% were given vasopressors, 34.5% underwent renal replacement therapy, and 5.2% received a tracheostomy. The median days on the ventilator was 4 (3–5.5). The overall ICU and hospital mortality were 17.2% and 32.8%, respectively. The median ICU and hospital length of stay were 5 (4–8) and 23 (12–39) days, respectively (see Table 1).
Patients who were lightly sedated within the first 48 hours of admission were less likely to require vasopressor use (eight patients, 44.4% vs. deep sedation 30 patients, 75%, p 0.04). There were no differences in ventilation days, renal replacement therapy, ICU and hospital length of stay and mortality between sedation depth.
Comparison between MICU and SICU
There was no difference in demographics between the patients from the two ICUs. In particular, the APACHE II scores did not vary significantly, suggesting that the severity of critical illness in both groups was similar (see Table 4).
Comparison between medical and surgical ICUs.
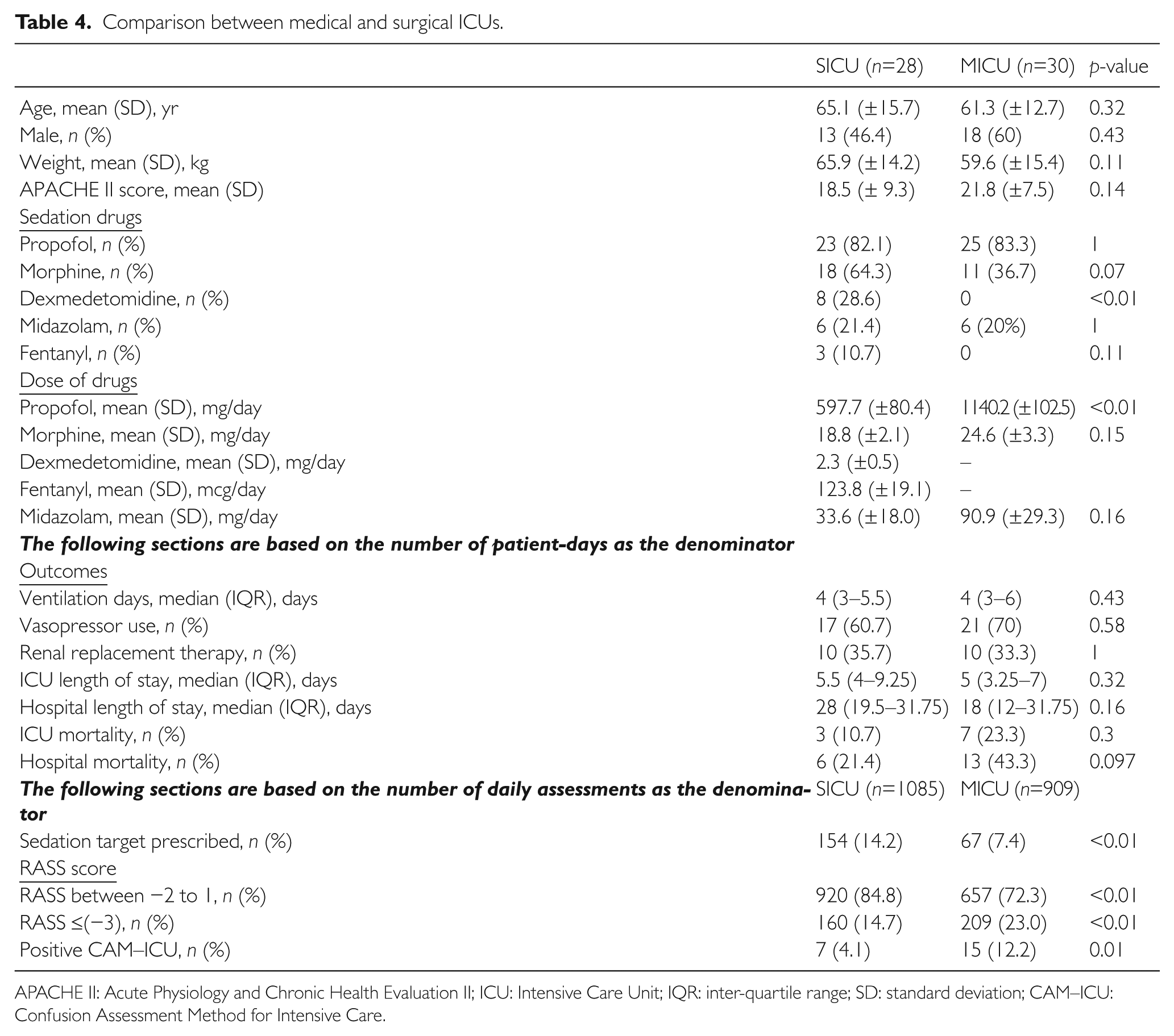
APACHE II: Acute Physiology and Chronic Health Evaluation II; ICU: Intensive Care Unit; IQR: inter-quartile range; SD: standard deviation; CAM–ICU: Confusion Assessment Method for Intensive Care.
While the choice of sedation drugs is similar, there was a significant difference in sedation practices between the two ICUs. In the SICU, 82.1% of patients were sedated with propofol, 64.3% with morphine and 28.6% with dexmedetomidine. In contrast, in the MICU, 83.3% of patients were sedated with propofol, 36.7% with morphine and 20% with midazolam. No patients in the MICU were given dexmedetomidine and fentanyl. For patients sedated with propofol, those in SICU were likely to receive a lower mean dose of drug per patient day compared with those in MICU (597.7 ±80.4mg/day vs. 1140.2 ±102.5 mg/day, p<0.01).
A significantly greater number of patients had a prescribed sedation target in the SICU compared with the MICU (14.2% vs. 7.4%, p<0.01). Correspondingly, a greater proportion of patients in the SICU had RASS scores of between −2 to 1 (920 RASS assessments, 84.8%) than the patients in the MICU (657 RASS assessments, 72.3%) (p<0.01). There were fewer delirious patients in the SICU with a positive CAM–ICU score 4.1% vs. 12.2% (p<0.01).
Despite these differences, the outcomes for both ICU patients in terms of median ventilation days, administration of vasopressors and renal replacement therapy, ICU/hospital length of stay and ICU/hospital mortality were similar.
Discussion
In our study, we found that lightly sedated patients were more likely to have been prescribed a sedation target. In addition, propofol and morphine were the most commonly used sedatives. Compared with MICU patients, surgical patients were more likely to be prescribed a sedation target, require lower doses of sedation, have a RASS score of between −2 to 1 and have fewer incidences of delirium. The demographics of our study patients resemble those in Shehabi et al. in both his Australia/New Zealand and Malaysian studies.3,13
Some 87.9% of our patients were sedated with propofol and 50% with morphine during the study period. To date, studies are mixed with regards to the most optimal sedation regime for use in critically ill patients. In a review of surveys done on sedative choices, midazolam and propofol were the top choices for sedation. 14 There were no patterns observed amongst the surveys, and the choice of drugs was primarily related to geography. European physicians were equally divided on the use of morphine or fentanyl (33% each), and propofol was more frequently used in SICUs. Americans tended to use lorazepam and midazolam. 14 There is a paucity of data on Asian practices of sedation, but one study from Malaysia identified the most frequently used sedatives as midazolam (39.6%) and morphine (31.7%). 13
Dexmedetomidine is an alpha-2 adrenergic agonist which causes sedation by inhibiting the release of noradrenaline at the locus coeruleus in the brainstem. The use of dexmedetomidine has been gaining popularity, as it is believed to result in a lower incidence of delirium (3%) compared with that of propofol and midazolam (50% each). 15 Dexmedetomidine can also reduce the ventilator time and provide analgesia (and hence reduces the need for more analgesic sedatives). 16 Another drug that was not used in our study but is a good alternative is remifentanil. Remifentanil has been shown by Rozendaal et al., 17 in a randomized controlled trial, to have shorter ventilator weaning time, earlier extubation, shorter ICU length of stay, and improved sedation-agitation scores and intensivist/ICU nurse satisfaction.
As many as 70% of critically ill patients who are admitted to the ICU are sedated. 18 Ways to minimize sedation include daily sedation breaks, avoiding sedation and protocolized sedation. However, the Cochrane review by Burry et al. found that there was no strong evidence that scheduled daily sedation breaks alter the duration of mechanical ventilation, mortality, length of ICU or hospital stay, adverse event rates, drug consumption or quality of life for critically ill patients. 19 The notion of no sedation in this group of patients was also studied by Strøm et al., who found that patients not sedated had more ventilation-free days and a shorter ICU stay. 6 Sedation strategies obtained from survey results indicate that about 28% of correspondents practised daily sedation breaks, and 41% protocolized sedation. In our study, similarly 21.4% of patients had sedation breaks, but the majority of them were for extubation (13.2%). The low frequency of practice for sedation breaks may be due to fear of respiratory compromise, lack of nursing support and inadvertent self-extubations. 20
Despite our nationwide intent for routine sedation targets, only 11.1% of all RASS assessments had a sedation target. This is similar to that found in Shehabi et al.’s study, where sedation targets were prescribed for 24.9% of patients. 3 Once a target was prescribed, patients were likely to meet their sedation targets and achieve optimal levels of sedation. Over-sedation has been found to occur in 6–62% of sedation assessments. 1 By the use of a sedation target as in our study, up to 86% of patients will be optimally sedated. The use of a sedation target is also associated with reduced costs of 22–94%. 21 RASS is a 10-point scale, with each point corresponding to the state of arousal. The assessments are easy to administer, with a high reliability and validity in ventilated and non-ventilated, sedated and non-sedated, and medical and surgical ICU patients. 8 As such, all patients should have a RASS sedation target, and this will henceforth lead to better control of sedation depth which has a positive impact on outcomes, with a potential reduction in healthcare costs.
Some of the positive impacts lighter sedation can lead to include a reduction in incidence of delirium.3-7 Delirium is defined as an acute, fluctuating change in consciousness and cognition, 22 with an incidence ranging from 16% to 29%. 8 It can be classified into hypoactive, hyperactive and mixed. In this study, CAM–ICU was only assessed when patients had a RASS score of between −2 and 1. Traditionally, delirium in the local setting is monitored clinically. Unfortunately, this alone underestimates delirium by 72%. 22 Early recognition and diagnosis of delirium is important, as delayed diagnosis is associated with increased mortality, poorer patient outcomes and greater resource utilization. 23 In our study 5.9% of patient-days were positive for delirium. We recognized that even in our study delirium may be underestimated, as it may be present in patients with a RASS more than 2 and less than −3. Once patients are identified to be delirious, potential underlying causes such as hypoxia, sepsis, organ dysfunction and electrolyte abnormalities must be quickly identified and treated. Non-pharmacological interventions such as early ambulation, improved communication, noise reduction and exposure to natural light can help to alleviate and treat the symptoms. Benzodiazepines, if administered, should be ceased. Although haloperidol is the current drug of choice for the treatment of delirium, due to the limited quality of studies in critically ill patients, no recommendation by the critical care societies have been made regarding the use of haloperidol to prevent or treat delirium in ICU patients. 2 Some studies have, however, shown that haloperidol use significantly reduces the incidence of delirium in the ICU (15.3% vs. 23%) and ICU length of stay (21 h vs. 23 h), 24 but the study population was not critically ill with a mean APACHE score of less than 9.
The importance of recognizing pain is so significant that it has been labelled as our fifth vital sign. Some 64% of ICU patients have been reported to have moderate to severe pain in the ICU. 25 In our study, 1.8% of patients reported (if they were able to communicate) or 0.2% appeared (if they were unable to communicate) to be in pain. The low incidence of pain could be due to the fact that 55% (138/251) of sedated patients were given an analgesic (morphine, fentanyl or dexmedetomidine) in their sedation regime. The impact of pain on the critically ill patient leads to sympathetic overstimulation and can affect the patient’s psychology and subsequent recovery.26,27 Pain is a subjective sensation, and the perception of pain to a standard stimulus can vary from patient to patient. Patients in the ICU are subjected to multiple procedures which may lead to pain such as intubation, surgery, and the insertion of invasive lines. The assessment of pain differs in the conscious and sedated patient. In the former, the Numeric Rating Scale or Visual Analogue scale are often used. However, in the latter, patients are unable to verbalize their pain and the Behavioural Pain Score tool can be utilized. This is an observational pain scale taking into account patient’s facial expression, upper limb movement and ventilator compliance, and has been shown to be reliable in sedated, mechanically ventilated patients. 28
Strengths and limitations
The strengths of our study are that it is a prospective longitudinal assessment of a combination of critically ill patients in both the medical and surgical ICUs. Daily CAM–ICU and RASS assessments were also performed by trained staff, and delirium assessment only performed if patients were lightly sedated.
The limitations of our study are that it has a small sample size and is a single-centre nested-cohort study. Data from the other centres were not included in our study. Patients who were sedated on oral medications were also excluded. In addition, as this is a cohort study, causality cannot be determined.
Conclusion
Propofol and morphine are the most commonly prescribed sedatives in our ICU, with sedatives being more frequently administered in the early period and analgesics during the subsequent period. Routine sedation goals using the RASS score should be prescribed to patients as this may result in lighter sedation for patients. Future trials may be needed to assess causality.
Footnotes
Acknowledgements
We thank Dr Shehabi for sharing with us the ANZ SPICE protocol and Dr Lee Tze Wee for his initial work on this study. The study has been approved by Singhealth CIRB (Ref: 2012/040/D). The author contributions were as follows. YL Lee: data analysis, manuscript preparation; K Ganesh: study design and data collection and manuscript review; LK Ti: study design and manuscript review; SY Ng: study design, data collection and manuscript review.
Conflict of Interest
The authors declare that there is no conflict of interest.
Funding
The study was funded by departmental resources.
