Abstract
Evaluation of a failing renal allograft is a complex and challenging diagnostic problem. While ultrasonography with colour Doppler is usually the first approach for evaluation of graft dysfunction, radionuclide imaging is an excellent modality which provides complementary information regarding the perfusion and function of the allograft without any deleterious effect on the precious allograft. In this article, we review the imaging techniques of the nuclear medicine studies most commonly performed after renal transplant, discuss their roles and limitations in different clinical settings and illustrate with cases from our institution. Lastly, we will explore future development in the arena of nuclear imaging for renal transplant related complications.
Introduction
With the installation of the first Anger gamma camera in the early 1980s, the Department of Nuclear Medicine in Singapore General Hospital was newly formed and the first radionuclide renography was performed in Singapore for native and transplant kidneys alike. Fast forward more than 30 years, although the graft failure rate has significantly decreased, evaluation of a failing allograft remains a complex and challenging diagnostic problem. Radionuclide studies, which help to quantify perfusion and function without any deleterious effect on the precious allografts, have assumed an important role in the evaluation of possible complications following renal transplantation. While ultrasonography with colour Doppler is the most widely used imaging modality of the transplanted kidney and is usually the first approach for evaluation of graft dysfunction,1,2 radionuclide scintigraphy can provide complementary information. An advantage of scintigraphy is that it provides functional information at the time of the study.
Overview of technique
Follow-up strategies of kidney transplants vary between countries and between transplant centres within each country. Some centres routinely perform baseline studies immediately post-operation while others only when clinical problems arise. While the ideal timing, frequency and modality of radionuclide studies in the post-operative renal transplant patients have yet to be established, it is generally agreed that:
a baseline study performed during the first few days after transplantation not only provides a diagnosis for post-operative complications, but also assesses the function of the allograft; 3
subsequent monitoring should follow a consistent protocol;
the technique must not vary from one study to the next;
the activity of radiopharmaceutical administered should be optimised to achieve images of sufficient diagnostic quality, while minimising radiation exposure to the patient.
Technically, acquisition of scintigraphic images of an allograft is identical to that of a native kidney, except the gamma camera detector is positioned anteriorly to image the transplanted kidney over the iliac fossa as close as possible instead of posteriorly for native kidneys. In our centre, technetium-99m-labelled mercaptoacetyltriglycine (Tc-99m MAG3) renography is the most commonly performed nuclear medicine study for the evaluation of post renal transplant complications. Tc-99m MAG3 is protein bound and is cleared predominantly by the proximal tubules (95%) with minimal filtration; 4 hence it is considered a tubular secretion agent, suitable for assessment of the functional status of the kidney. No fasting is required. The patient should be well hydrated prior to the procedure, since sub-optimal hydration may produce a scan result that mimics allograft dysfunction or obstruction. After a bolus injection of 5–10 mCi of Tc-99m MAG3 through a peripheral vein, the images are acquired in 15 second frames for 30 minutes to evaluate parenchymal radiotracer uptake and clearance. Semi-quantitative analysis is performed by generating a time activity curve based on a region of interest drawn around the kidney. Interpretation of a post-transplant renal scintigram begins with careful scrutiny of the images for prompt, homogeneous radiotracer uptake in the allograft. Subsequent radiotracer excretion into the pelvicalyceal system and the ureter should be prompt with no evidence of radiotracer retention at the end of the study. If no spontaneous excretion is observed, diuretic, in the form of intravenous frusemide, may be administered 10 minutes after the injection of radiotracer to exclude an outflow tract obstruction. A normal renogram curve demonstrates a rapid upslope which reflects the initial arrival of the radiotracer in the kidney followed by the cortical concentration phase which contains the peak of the curve. The subsequent downslope marks the excretion phase (Figure 1). 5 The technique for a transplanted kidney perfusion study is described in the following section.

Evaluation of complications following renal transplantation
The main indications for renal transplant scintigraphy are listed in Table 1.
Main indications for renal transplant scintigraphy.

Vascular occlusion
In our centre, renal perfusion study is usually performed immediately after the transplant surgery on the same day. The radiotracer used is technetium-99m pertechnetate. Prior to scanning, extreme care is taken to ensure the allograft is within the field of view of the gamma camera detector. Dynamic images are acquired (2–3 seconds per frame) for 60 seconds while 15–20 mCi of the radiotracer is given as a fast bolus through a large vein. The images and time-activity curves are interpreted by visual assessment. In a normal transplant perfusion study, the radioactive bolus reaches the transplanted kidney at almost the exact time it is seen in the iliac vessel adjacent to it. 5 Non-visualization of the allograft in the immediate post-operative setting is suggestive of arterial thrombosis, which is almost always the result of vascular kinking or technical difficulty at the time of surgery. 6 Prompt re-exploration is warranted to restore the vascular supply to the kidney (Figure 2).

Acute tubular necrosis, rejection and drug toxicity
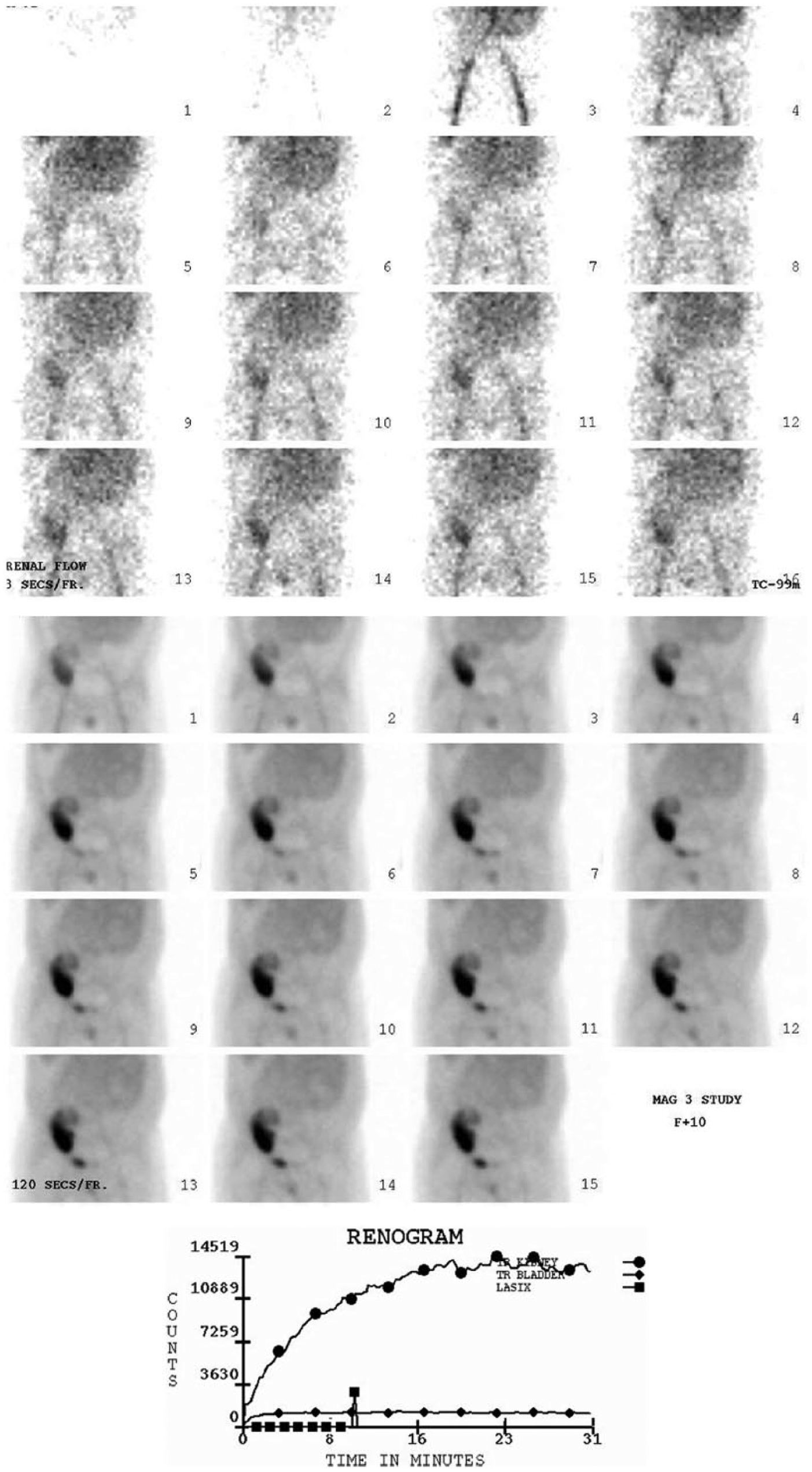
Acute tubular necrosis (ATN) and acute allograft rejection (AR) are the most common causes of impaired renal function in the early post-operative period. 1 ATN is encountered in up to 15% of patients after renal transplantation and occurs more commonly among cadaveric transplants. While it may cause delayed graft function and necessitate haemodialysis initially, overall it has no impact on patient or graft survival as it is expected to resolve in several weeks. 7 The classical scintigraphic findings of ATN include preserved or only mildly reduced perfusion but delayed uptake and excretion of tubular secretion agent, such as Tc-99m MAG3, with progressive accumulation of the radiotracer in the renal cortex (Figure 3).5,8

Renal transplant rejection is usually classified according to its time of onset (hyperacute, acute or chronic), each driven by different mechanism. In rejection, renography typically shows decreased perfusion, diminished uptake and delayed excretion (Figure 4). 5 Scintigraphic perfusion criteria have been advocated to aid in the differential diagnosis between ATN and AR, since perfusion is less affected in grafts affected by ATN compared to those affected by AR. Also, on serial studies, progressive decline in function and poor perfusion are thought to favour AR. 3 Unfortunately, considerable overlap exists, for example in cell-mediated AR; the scan findings are often indistinguishable from ATN. 3 Severe ATN may result in diminished perfusion, giving a scan pattern similar to that of AR. 9 Various perfusion and parenchymal indices derived from renogram studies have been used to assess graft dysfunction, but none of these mathematical strategies can reliably differentiate between ATN and AR,10,11 and allograft biopsy remains the gold standard to establish the correct diagnosis.
Similarly there is no specific imaging pattern for drug toxicity. The scan findings seen in cyclosporine nephrotoxicity have been reported to mimic both ATN and AR,2,3 though this condition can be distinguished from ATN by correlating with cyclosporine level and its time of presentation as it characteristically occurs several weeks after renal transplant, by which time acute tubular necrosis should have resolved. Nevertheless, in spite of its limited ability to discriminate the various parenchymal pathologies, in the context of declining graft function, radionuclide scintigraphy is useful to establish the presence of blood flow to the graft, and the absence of a urine leak or obstruction. 1
Post-transplant fluid collections
Post-transplant fluid collections can present at any point in time from the immediate post-transplant period to several months post transplantation and can be incidental or cause significant transplant dysfunction due to their potential to exert mass effect. 12 A post-transplant fluid collection, usually first identified on ultrasonography, may represent a urinoma, lymphocele, seroma, abscess or hematoma. Often the clinical picture or time of presentation may give a clue. A patient with an abscess may present with fever and raised inflammatory markers; hematoma, seroma and urinoma are often seen within the first few days following surgery while a lymphocele typically develops several months after surgery. 13 A post-transplant fluid collection appears as a peri-renal photopenic area on scintigraphy. A lymphocele presents as a persistent photopenic area, although a mild degree of filling-in on delayed imaging has been observed (Figure 5). 14 A photopenic area which gradually fills in with intense radiotracer during the scan or the presence of excreted activity outside the urinary tract is strongly suggestive of a urinary leak (Figure 6). 15 However if the leak is slow, it may not be possible to distinguish a urinoma from other fluid collection on scintigraphy.


Urologic complications
Vesicoureteral reflux is a common complication which can take place early after graft surgery.16,17 As symptoms of reflux are non-specific, it is often a diagnosis made incidentally on Tc-99m MAG3 renography while investigating for a cause of graft failure. When reflux occurs, renography classically demonstrates a ‘double peak’ seen on the time-activity curve. The first peak reflects the start of the normal excretory phase while the second peak is caused by re-entrance of radiotracer into the kidney as a result of reflux from the bladder. Visually, the radiotracer is excreted from the renal parenchyma and cleared from the collecting system initially. At the time of reflux, the kidney appears to fill with radiotracer again, sometimes preceded by radiotracer activity seen in the distal ureter (Figure 7). In patients with suspected vesico-ureteric reflux, an indirect micturating cystogram can be performed to confirm the diagnosis. After the administration of Tc-99m MAG3, anterior images are obtained over 30 minutes, followed by a second acquisition of dynamic images while the patient voids, standing upright in between the two gamma camera detectors. The images are scrutinised for re-accumulation of tracer activity in the pelvicalyceal system of the allograft. Regions of interest are drawn around the kidney and bladder to generate a time-activity curve for the voiding phase. As the bladder empties, tracer reflux into the kidney manifests as a single peak in the time-activity curve (Figure 8).
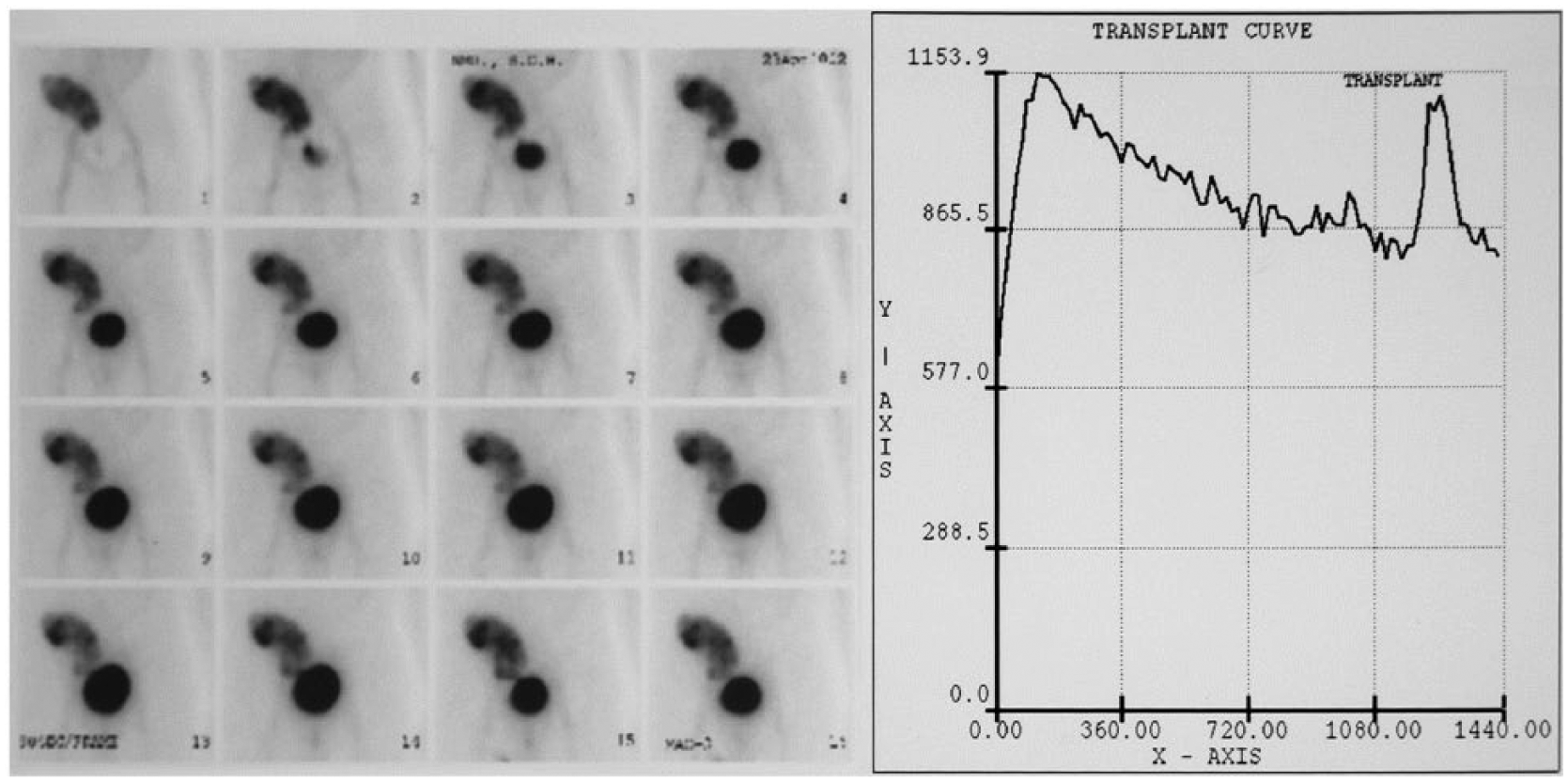

Obstruction of the transplanted kidney may occur at any location but is most frequent at the site of ureteric anastomosis with the bladder. 1 Patients are usually referred for renography to assess the patency of the urinary tract on the basis of pelvicalyceal dilatation detected on ultrasonography, or a rising serum creatinine level.
Prognostic information
In addition to its role in evaluating graft perfusion in the immediate post-operative setting, baseline scintigraphy performed in the early post-operative period may have a role in the prediction of long term allograft function. 8 Various quantitative indices of perfusion and/or uptake derived from Tc-99m DTPA and Tc-99m MAG3 renography performed between 1–4 days after transplantation have been reported to correlate with graft survival;18–20 however, there is a lack of consensus as to which of these indices are most appropriate to use for the purpose of prognostication. A recent study found that quantitative assessment of Tc-99m DTPA perfusion scintigraphy performed within 2 days after transplantation was useful in the prediction of long-term graft function up to 5 years and was superior to measurement of intra-renal resistance index by Doppler ultrasonography. 21
Future development
The field of nuclear medicine is rapidly evolving and ongoing development of new radiotracers and imaging techniques hold the hope of increasing the sensitivity and specificities of nuclear medicine studies in detecting transplant related complications. Anti-CD3 monoclonal antibody has been used as a therapy for treating acute renal rejection; studies have labelled anti-CD3 monoclonal antibody with technetium-99m for the imaging and detection of acute renal rejection. 22 A more biocompatible radiolabelled CD3 antibody Tc-99m SHNH-visilizumab is being evaluated. 23 Positron emission tomography (PET) tracers such as 13N-ammonia and H215O, which are currently unavailable in our centre, are being investigated for mapping of local renal blood flow in renal transplant grafts, including assessing the response of local blood flow to drug therapies. 24 Recently, PET with 18F-FDG labelled human cytotoxic T lymphocytes has been used to assess acute rejection in an allogeneic rat renal transplant model. 25 18 F-FDG PET might also assist in distinguishing acute rejection from other differential diagnoses such as cyclosporine toxicity and acute tubular necrosis in animal models.26,27
Conclusion
As a non-invasive study which provides valuable information on the functional status of an allograft, nuclear medicine renography plays an important role as an adjunct tool in the evaluation of possible complications following renal transplant. Interpretation of the scintigraphic findings must be correlated with clinical presentation and other available imaging modality. Ongoing development of targeted radiotracer and imaging technique is encouraging and the hope is to detect renal transplant related complication early and accurately for timely intervention.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
