Abstract
Laboratory support for the organ transplant patient is the provision of the best testing technology for a specific and accurate determination of immunosuppressive drug level. This translates to better management with the therapeutic cocktail of immunosuppressive drugs used with a lesser incidence of organ rejection and side effects. Over the years, development of automated immunoassay/chemistry test platforms with standardization of test protocols has demonstrated great improvements. The present clinical laboratory services with tandem mass spectrometry in our hospital present more precise and specific therapeutic drug monitoring so necessary still for the patient. This article follows the evolving testing technologies over the years for immunosuppressive drug monitoring following the organ transplants programme (renal in general) in Singapore General Hospital.
Introduction
Organ transplants remain the only good treatment modality for the afflicted person with kidneys, liver, heart and pancreas being the failed organs. As an external entity, the organ transplanted is subject to host rejection. Graft survival and acute rejection can be mitigated with the use of various immunosuppressants. However, it was only with the introduction of better immunosuppressive drugs that the rate of organ transplantations rose steeply in the 1980s with the discovery and utilization of cyclosporine for the management of organ rejection. Since then, other immunosuppressive drugs have been introduced but cyclosporine remains first in the front line of powerful immunosuppressive drugs to be used.1–5 These drugs act primarily on the signal transduction pathways in activating T-cell immune response. Over the years, the availability of immunosuppressants has led to dramatically reduced rejection rates.
Today the Clinical Biochemistry Laboratories of the Department of Pathology in Singapore General Hospital (SGH) have test assays for most of the common immunosuppressive drugs available – cyclosporine, tacrolimus, sirolimus, everolimus and mycophenolic acid (MPA) – whose introductions in SGH followed soon after worldwide release (Table 1).
Immunosuppressant tests in Singapore General Hospital.

SGH: Singapore General Hospital; LC-MS/MS: liquid chromatography-tandem mass spectrometry
Laboratory services for immunosuppressants monitoring
The wide inter- and intra-individual variable pharmacokinetics observed, compliance issues and also the narrow drug therapeutic range for effectiveness have required regular drug monitoring. With this need for drug monitoring and with the advent of immunoassays in the 1980s, the in-vitro diagnostics market for immunosuppressive drugs monitoring grew.6,7
In 1989, Pathology, then a Ministry of Health department, joined SGH. In the same year, cyclosporine testing by a fluorescence polarization immunoassay (FPIA) with whole blood was made available in its Clinical Biochemistry Laboratories. 8 Workload increased rapidly from 752 (1989) to 5070 (1995). The next immunosuppressant test that followed was tacrolimus, or FK506, in 1998. For the next six years, these were the only two tests available. Sirolimus and MPA (through its pro-drug mycophenolate mofetil), introduced for use worldwide in 1999 and 2004 respectively, were offered for drug testing in SGH in 2004.9–11 Thenext breakthrough immunosuppressive drug, everolimus, released in 2004, joined the test repertoire in 2006. By this time, the annual combined workload (cyclosporine, tacrolimus, MPA and sirolimus) had reached 12,299 (Figure 1). Subsequent years saw a sharp decline attributed to a significant drop in cyclosporine testing in tandem with increase FK506 testing. Overall continued growth in immunosuppressants testing would likely be due to increase transplants being performed.

Annual workload of immunosuppressant testing. Test introduced: 1989 cyclosporine (CsA); 1998 tacrolimus (FK506); 2004 sirolimus (SIR), mycophenolic acid (MPA); 2006 everolimus (EVR).
Mass spectrometry for the better
All immunoassays are affected to some degree by cross-reactivity of similar compounds and especially metabolites of the parent drug. The cross-reactivity of metabolites can be sufficiently substantial to cause drug dosing inaccuracies. Hence, very early on, the metabolites of cyclosporine given to the patient were recognized and studied. This is also shown for tacrolimus, sirolimus and everolimus.12–26 Cyclosporine was particularly affected, with the major metabolites identified and widely known (Table 2).11,15,16,18,19,22–29 Trough levels of AM1, the major metabolite and pharmacologically active, can equal or exceed that of the parent drug level. The same limitation can also be demonstrated for tacrolimus, sirolimus and everolimus, albeit to a lesser degree as compared with cyclosporine.
Immunosuppressants and their metabolites.
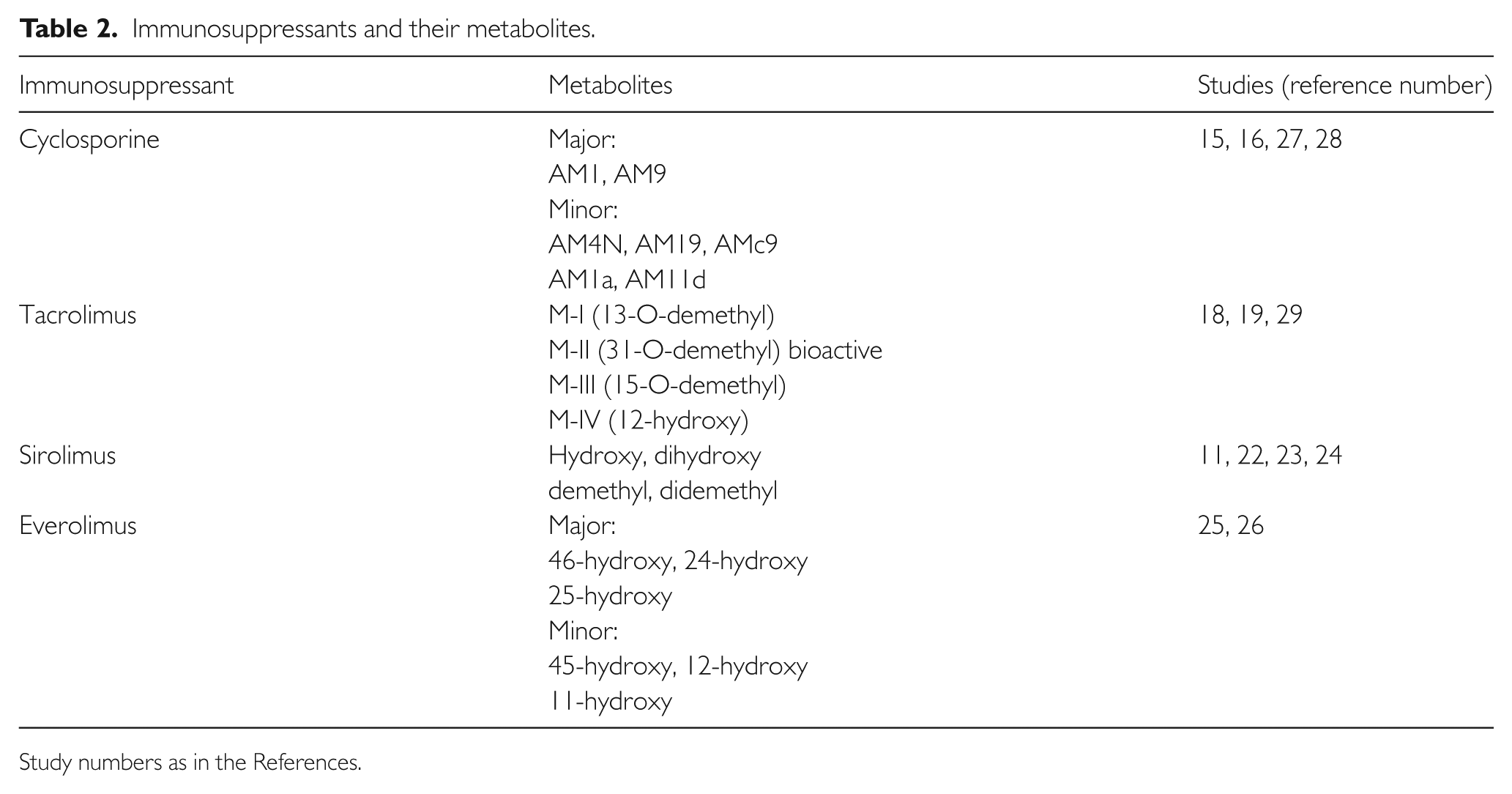
Study numbers as in the References.
Improved immunoassays in recent developments did not totally remove the issue of active metabolite cross-reactivity as shown for cyclosporine and tacrolimus immunoassays.27,29,30 The new improved assay for tacrolimus (a next generation assay on the Abbott ARCHITECT test system) showed a bias against LC-MS/MS, which can be accounted for by the two main metabolites, M-II and M-III, which can have 15% and 3% contribution respectively to results (in steady-state blood from renal patients) as the monoclonal antibodies used still have significant reactivity with tacrolimus M-II (94%) and M-III (45%). 29
Other shortcomings of immunoassays include effect of haematocrit on recoveries (tacrolimus, sirolimus), overestimation compared with high performance liquid chromatography (HPLC) (sirolimus) and a recent note on matrix-comparability for QC material (everolimus).31–39 Hence, it was noteworthy to consider more specific test methods for measuring the immunosuppressive drugs 28 and the idea of liquid chromatography-tandem mass spectrometry (LC-MS/MS) for immunosuppressant measurements in clinical practice took off.40–43
Clinical practice and standardization efforts
In 2007, we brought in an Agilent Technologies G6410 QQQ triple quadrupole system coupled with liquid chromatography to kick-start mass spectrometry for measuring immunosuppressants. A state-of-the-art technology then, the principle of measurement follows ionization of small molecules (as for the immunosuppressants) with the mass ions passing through three stages: the first to accurately select target analyte (precursor) mass ions, followed by fragmentation in a gas collision chamber and finally the expected fragment masses (product) identified and quantitated (Figure 2). What is novel about this technology is that for each pass of liquid chromatographically separated analytes, the instrument has the ability to perform ‘multiple reaction monitoring’ of various target ion pairs (precursor/product) with the system being quick enough to scan, identify and quantitate them. This was effectively a multiplexed analysis of each sample in one assay run. The subsequent quantitation technique follows the classic way by extrapolation of the unknown analyte level to a calibration curve of set values.
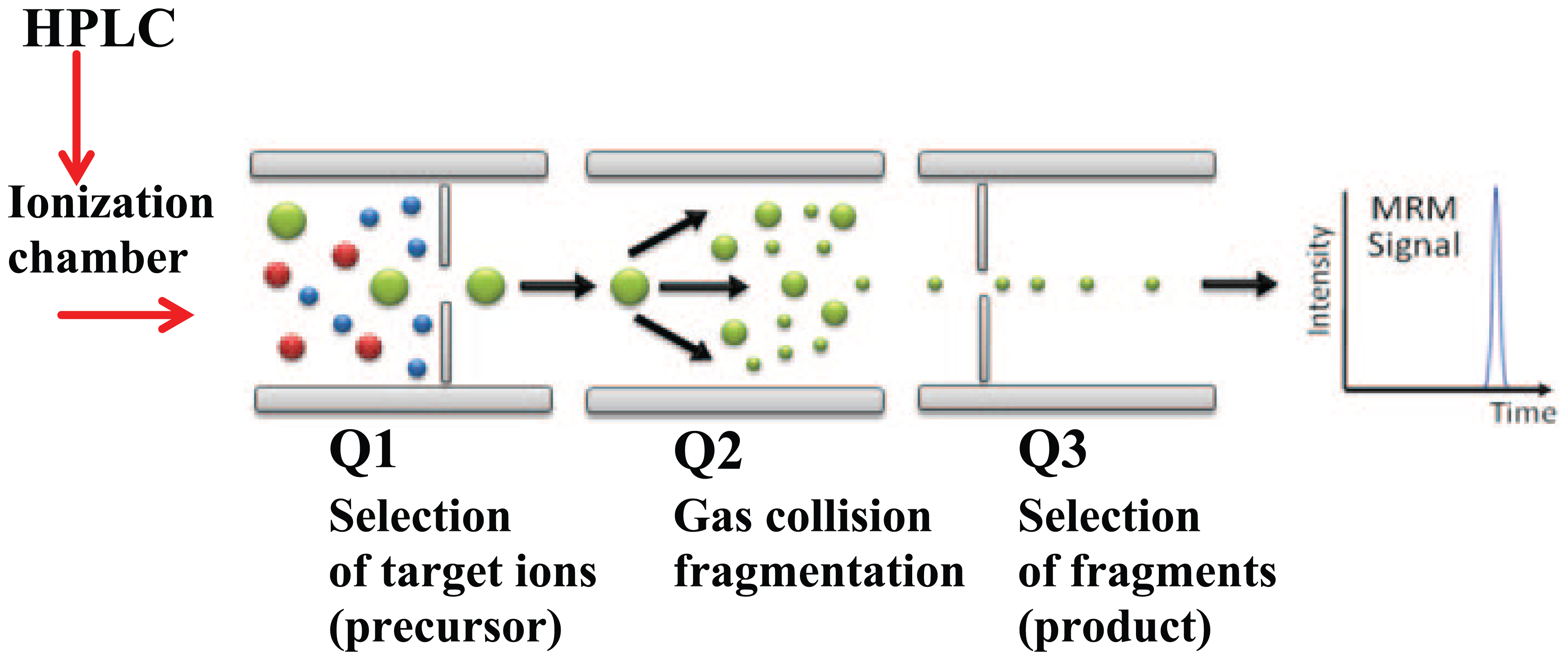
Tandem mass spectrometry schematics. Triple quadrupole segments (Q1–Q3) illustrating ion filtering and detection.
As no commercial assay kit was available at that time but based on published work on LC-MS/MS, our own development and establishment of a simultaneous measurement of all five current immunosuppressants (with commercially available calibrators/controls) on the LC-MS/MS test platform was presented at the 17th SGH Annual Scientific Meeting in April 2008. 44 A series of other studies – on measures taken for controlling assay variation, analyte stability and target therapeutic levels from proposed transition of immunoassay to tandem mass spectrometry – were later presented locally and abroad. In 2009 and 2010, we also worked with fellow transplant teams (pharmacists and renal physicians) on clinical correlation of test results by LC-MS/MS following the proposed transition from immunoassay to tandem mass spectrometry technique in determining the levels for everolimus, sirolimus and tacrolimus.45,46 The decision to move the tests to the LC-MS/MS platform was strengthened by many studies expounding the benefits of increase specificity and potential cost savings although negating factors were available staff skills, initial capital outlay and maintenance costs. Everolimus and sirolimus were ported over to the LC-MS/MS platform in November 2009 and April 2010 respectively. Currently tacrolimus and cyclosporine are slated to port over to LC-MS/MS within this year. During the in-between years, we built up staff experience and training.
Although we had also established testing for mycophenolic acid (MPA) on the LC-MS/MS with validation studies, the test remains on the immunoassay platform today in view of the specimen type (plasma instead of whole blood), workload and service requirements. The latest and current version of MPA assay in SGH is reported to align more with LC-MS/MS determinations and also less be impacted by the glucuronide form which earlier assays had cross-reactivity issues with. 47
Our laboratory performance amongst peer groups worldwide through the CAP (College of Pathologists) EQA (External Quality Assurance) Surveys have indicated satisfactory returns for the tests (sirolimus and everolimus) that have been ported over to the LC-MS/MS methods and also for tests (tacrolimus, cyclosporine and MPA) that have remained on immunoassay platforms. More laboratories have reported with LC-MS/MS technology over the years. Results from the 2015 CAP CSM-B challenges indicated there are now 83/543 laboratories reporting cyclosporine, 85/321 reporting sirolimus and 91/573 reporting tacrolimus with LC-MS/MS. There are also 43/63 laboratories reporting everolimus with LC-MS/MS, compared with its debut in the 2013 Survey with 23/35 laboratories. This is in contrast to five years ago, in 2010, when there were 51/465 laboratories (cyclosporine), 56/220 (sirolimus) and 53/434 (tacrolimus) with LC-MS/MS. Although there is increased adoption of LC-MS/MS, interlaboratory standardization is not yet within sight as most LC-MS/MS techniques are laboratory-developed. The recent availability of commercial kits is a development that could reduce this gap. Recent CAP Surveys have shown acceptable CVs for the LC-MS/MS group as compared with all-labs CVs, with the widest variation seen for everolimus (14.7% compared with 24.5% in all-labs – 2015) and a similar picture for the other three immunosuppressants. Higher CVs variation seen in the all-labs group is also a reflection of the dominant number of various immunoassay platforms in use. While it is still a difficult-to-acquire technology for any hospital, reports have marked LC-MS/MS as the right choice to institute for (at least) immunosuppressants.48,49 A difficulty with most research-originated techniques is that typically such rapidly developed procedures are in many cases ahead of the availability of reference materials. Standardization of reference material has yet to be available although calibrators/controls are commercially available. In addition, matrix effects are well-known caveats in any test assay and this issue is not fully resolved although the use of appropriate internal standards has mitigated this to a large extent. Several consensus meetings have also convened to establish standardization of drug monitoring mode, test optimizations, protocols and target levels and there is still much work to do at this moment.50–53
Challenges near and far
From a research setting to clinical practice, our technology quotient and the reliance of LC-MS/MS as a possibly disruptive technology in medical diagnostics will be put to the test. At the 2007 European Consensus Conference on Tacrolimus Optimization at the 10th IATDMCT (International Association of Therapeutic Drug Monitoring and Clinical Toxicology) meeting, a lower limit of quantitation (LOQ) was recommended to ideally be 1.0 ng/ml so as to allow confident measurements (down) to 3.0 ng/ml, important in low-dose tacrolimus treatment regimens. The LC-MS/MS technology in SGH reports levels down to 2.0 ng/ml, and with newer instruments the lower LOQ of 1.0 ng/ml would be achievable.
While test load growth can be gradual and concomitant with the organ transplant programme as in most centres, the expectations of test requests are likely to increase especially with enhanced use of drug monitoring in the outpatient clinics. For the physicians, their clinical expectation for test turnaround time (TAT) is one to consider. The current LC-MS/MS run dates are twice a week with same day TAT. There are occasions when the laboratory performs ‘urgent’ requests by appointment on other weekdays and weekends. A potential rate limiting step in providing STAT TAT is that sample preparation for both the LC-MS/MS technique and current immunoassays requires pre-treatment before analysis. Reducing the whole work process further to 1–2 h TAT (for all four immunosuppressive drugs) would stretch the laboratory’s capability as on a typical busy morning clinic day 50–70 specimens are received. To overcome this potential bottleneck, one approach is to increase capacity. As the local medical fraternity pushes for all things in a ‘hub-’n’-spoke’ model, it can only mean a more demanding need for test results from this hub to be ready in short times. Will the test be available 24/7 for a hospital with an active transplant programme? Can the average medical technologist in turn be able to handle the complexity of ‘research-grade’ instruments round the clock? The desired outcome is a ‘Yes’ to these expectations.
Conclusions
Better management of immunosuppressant therapy has depended to a large extent on accurate measurement of the effective parent drug while minimizing measurement of its metabolites. In the quest for tight titration norms, it is generally accepted that mass spectrometry is the way to proceed for drug testing. In a high volume demand situation like a central transplant hub in a tertiary acute hospital (such as SGH), operational challenges to the laboratory are in terms of both hardware availability and also skilled technologists to be able to provide full seven-day service and report the results in a timely manner. It will require full management support and encouragement to meet future expectations.
Drug developments such as the biologics present the next frontier for immunosuppression advances. Being antibody-based, manufactured in a customized form to reduce host (patient) reaction and more effective in stopping the immune transduction network, these biologically engineered bio-agents will be a challenge to measure by the clinical laboratory. Although available for many years, the number of biologics is also increasing, for example, alemtuzumab (CD52; Campath-IH), muromoab (CD30, OKT3), basiliximab and diachizumab (IL2R antagonist), and they are now heading for mainstream use. Will they ever need to be specifically monitored and what metric is best to measure their effectiveness?54–56 The laboratory will need to define the metric for measurement and the technology to determine it and report results that are appropriate for clinical management and monitoring purposes.
Footnotes
Conflict of interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
