Abstract
Introduction
It is unknown to what extent GLP-1 receptor agonists (GLP-1 RAs) mediated reductions emotional, uncontrolled eating affect weight loss in patients living with obesity (PwO), or which factors modify behavior responsiveness.
Methods
PwO completed a survey considering their state on vs. off GLP-1 RAs, which included the Three-Factor Eating Questionnaire Revised 18 item Version, assessing uncontrolled and emotional eating (UE and EE), as well as cognitive restraint of eating (CR).
Results
Responses were collected from 257 participants with a median age of 54 (44–64) and BMI 36.0 kg/m2 (33.1–41.7). Semaglutide use was reported by 81% of subjects taking a median dose of 1 mg, and liraglutide 3 mg use was reported by 19%. Treatment length was 14 months (6–25) with 12.3% (7.4–−19.7) weight loss. GLP-1 RA mediated weight loss was associated with reduced UE and EE (rs = 0.31 and rs = 0.29, p < 0.001 for both). Those who eventually discontinued therapy (31%) vs. those who persisted reported greater GLP-1 RA mediated reductions in UE and EE and less rises in CR. Females had greater reductions in UE and EEE. Age was inversely associated with changes in UE and EE (rs = −0.23, p < 0.001 and rs = −0.15, p = 0.02), with younger age correlated with greater improvements.
Conclusions
PwO with greater GLP-1 RA mediated reductions in uncontrolled, emotional eating experience more weight loss and are less likely to stop therapy. Modulatory effects on eating behaviors are age and sex dependent.
Keywords
Introduction
Emotional eating, most commonly defined as overeating in response to negative emotions rather than in response to physiological hunger, is more prevalent in people with obesity (PwO). 1 It has been postulated that this phenomenon is related to HPA axis dysregulation, 2 with blunting of the expected cortisol rise in response to stress in self-reported emotional eaters. Additionally, according to fMRI studies, PwO have heightened activation of mesocorticolimbic brain structures in response to food cues, reflecting increased reward sensitivity, such that stress-induced reward seeking behavior could lead to overeating. 3 Furthermore, PwO have decreased Insula functional connectivity which could reflect lower interoceptive awareness, impairing satiety signaling and thus inhibition of food intake. 4 These associations may have genetic underpinnings, as demonstrated in a cohort of Iranian adults, where the most significant MC4R polymorphism associated with obesity was also associated with emotional eating. 5
Emotional eating can interfere with behavioral weight loss interventions and caloric restriction,6,7 as well as weight maintenance after bariatric surgery. 8 According to a few studies, emotional eaters also appear to lose less weight with in response to GLP-1 RA treatment.9–11 On the other hand, mostly small clinical trials have demonstrated that GLP-1 RAs can both reduce weight and improve control of eating behaviors,10–12 including uncontrolled eating, frequently defined as a loss of control leading to overeating regardless of physiological hunger cues. 13
Although emotional eating and uncontrolled eating behaviors are not classified as distinct eating disorders in the Fifth Edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-5), these behaviors constitute components of recognized eating disorders. 14 Importantly, even in the absence of formally diagnosed eating disorders, emotional and uncontrolled eating behaviors are associated with multiple adverse outcomes. These include not only weight gain,15,16 but also significant psychological comorbidities, including anxiety, depression, and diminished self-esteem.17,18 Furthermore, validated assessment instruments for these behaviors are available and widely in use. The Three Factor Eating Questionnaire (TFEQ) is considered the gold standard for evaluating both emotional and uncontrolled eating behaviors, demonstrating high levels of internal consistency across hundreds of studies in diverse populations.13,19
Associations between reduction in emotional, uncontrolled eating and other factors have not been adequately studied. Furthermore, little is known regarding how GLP-1 RA treatment affects emotional eating among subpopulations in the real world, and how these effects relate to different outcomes. Therefore, we distributed a survey, which included the TFEQ, to determine how analogs affect emotional, uncontrolled eating in PwO, and how these effects interplay with demographic factors, weight loss and persistent medication use in a real-world setting.
Materials and methods
Respondents
On July 2 2023, following approval from the Sheba Medical Center IRB-Helsinki Committee (protocol number 0177-23-SMC), a survey was sent to 1879 adults followed in a hospital-based weight management clinic and posted to Hebrew Facebook groups “Ozempic® Israel” (then listing 59,000 members) and “Saxenda®-Ozempic®-Razin® Forum for Weight Loss Medications” (then listing 39,000 members). Respondents provided informed consent for study participation. Three hundred and nineteen responses were collected from adults through August 14 2023, and 62 were excluded since they indicated not receiving liraglutide or semaglutide for weight management over the past two years. Statistical analysis was conducted on the remaining 257 survey responses.
Regional availability of GLP-1 RA therapy for weight loss
At the time of survey completion, the sole incretin based therapy approved by the Israel Ministry of Health for obesity management was liraglutide 3 mg. Semaglutide 1 mg was approved for the treatment of type 2 diabetes (T2D), with off-label for weight management requiring application and approval by the Ministry of Health every 6 months. Semaglutide use was further limited by supply shortages during the study period. Neither medication was covered by national health insurance.
Study tools
The survey included question topics including demographics, anthropomorphic measures, medical history, and information regarding use of semaglutide and liraglutide for weight loss (Supplement). Eating behavior was assessed using a Hebrew translation of The Three-Factor Eating Questionnaire Revised 18-item Version (TFEQ-R18). The tool was validated with back-and-forth translation by native English and Hebrew speakers. Respondents were asked to complete the questionnaire twice, once recalling their state under maximal effect of GLP-1 RA and a second time considering their state being off therapy, either before initiation or after GLP-1 RA cessation.
The TFEQ-R18 is a widely used abbreviated, revised version of the original 51-item TFEQ which assesses eating behaviors in PwO. 20 The TFEQ-R18 consists of 18 items relying on a 4-point scale (definitely true/mostly true/mostly false/definitely false), where higher scores are indicative of more of each behavior. 21 Scores are summated to assess uncontrolled eating or “disinhibition” (eating more due to a loss of control over intake when feeling hungry), emotional eating or “hunger” (inability to resist emotional cues), and cognitive restraint of eating (restriction of food to control body weight). Raw scores are transformed to a 0–100 scale [((raw score – lowest possible raw score)/maximal raw score range) × 100]. In our study, internal consistency reliability coefficients (Cronbach's alpha) for overall TFEQ-R18 scores were above the 0.70 standard and below the 0.90 limit recommended for individual assessment, similar to the original instrument (0.86 off GLP-1 RA and 0.87 on GLP-1 RA). 13
Statistical analysis
Descriptive statistics are used to summarize all variables. Categorical variables are described asfrequencies and percentages. Continuous data is expressed as median with accompanying interquartile range (IQR). Error bars denote standard errors in figures. We tested for between-group differences in baseline characteristics using Chi-square test and Fisher's exact test. Continuous variables were tested for normalcy using the Kolmogorov-Smirnov test. As variables were not normally distributed, they were compared using the Mann-Whitney test. We calculated the difference between TFEQ-R18 scores on minus off treatment (ΔTFEQ-R18) and performed univariate analysis to assess associations between patient characteristics and ΔTFEQ-R18. These comparisons were made using Spearman's test for continuous variables. Data for patients using semaglutide and liraglutide were combined to increase power, given similar trends with these drugs. Univariate analysis was performed to evaluate associations between patient characteristics and GLP-1 RA mediated changes in weight. Variables significantly associated with weight loss in univariate analysis were included in multivariate analysis conducted as multiple linear regression. A forward selection method was applied, and the Wald test was used as criteria for variable removal (p > 0.1 as criteria for removal). All statistical analyses were performed in IBM SPSS version 29, with two-sided p < 0.05 considered statistically significant.
Results
Baseline characteristics
Median age was 54 (44–64) and 70% of respondents (n = 181) were female (Table 1). Baseline weight and BMI were 100.0 kg (89.0–115.0) and 36.0 kg/m2 (33.1–41.7), respectively. Forty-five respondents (18%) endorsed a history of bariatric surgery performed 9.3 years (4.9–13.5) prior to completing the survey, with only four reporting surgery within the past two years, and none within the past year. At least one comorbidity was present in 83% of respondents (n = 212), with 33% (n = 84) endorsing dyslipidemia, 31% (n = 79) hypertension, and 36% (n = 66) metabolic dysfunction-associated steatotic liver disease (MASLD).
Characteristics of 257 subjects.

Data is presented as median with interquartile range (IQR) or N with %. Abbreviations: HTN, hypertension; MASLD, metabolic dysfunction-associated steatotic liver disease; T2D, type 2 diabetes mellitus; OSA, obstructive sleep apnea; CAD, coronary artery disease; PCOS, polycystic ovarian syndrome; Δ change in Three Factor Eating Behavior – Revised 18 scores on minus off therapy; weight change (%), last weight on therapy minus baseline weight divided by baseline weight.
Footnotes:
bariatric surgeries (n = 11 had more than one surgery): gastric sleeve (n23) gastric bypass (n = 13) mini gastric bypass (n = 6) gastric band (n = 13) other (n = 5);
concomitant drugs for weight loss (n = 4 taking more than one) phenteremine (n = 5) lisdexamfetamine (n = 2) metformin (n = 4) topiramate (n = 3) other (n = 5);
length of treatment at time of answering survey or until drug cessation.
imissing data (n = 247).
Drug utilization, side effects and cessation
Semaglutide use was reported by 209 subjects (81%) taking 1.0 mg (1.0–1.0) weekly for 16 months (6–26), while liraglutide use was reported by 48 subjects (19%), taking 3.0 mg (2.4–3.0) for a significantly shorter period of 8 months (5–19) (p = 0.007) (Table 2). After a median of 6 months (3–12), 31% of patients (n = 80) stopped GLP-1 RA therapy, with a trend for a lower rate of drug cessation for semaglutide than liraglutide (29%, n = 60 vs. 42%, n = 20; p = 0.08) (Table 3). The most frequently endorsed causes for drug cessation in response to a multi-select question were cost (21%, n = 17) reduced efficacy and difficulty obtaining drug (34%, n = 27 for both), and side effects (55%, n = 44). The only significant difference between drugs regarding cause for cessation was difficulty obtaining the medication (42%, n = 25 vs. 10%, n = 2; p = 0.009 for semaglutide vs. liraglutide, respectively). There was a strong trend for discontinuation due to side effects occurring less often with semaglutide than liraglutide (48%, n = 29 vs. 65%, n = 13 p = 0.052). Among drug discontinuers, there were no differences between adherence to semaglutide and liraglutide, with 73% of respondents (n = 58) taking GLP-1 RA therapy 80–100% of the time. Overall, those who discontinued therapy most frequently endorsed gastrointestinal side effects (41%, n = 33), followed by fatigue (23%, n = 18), decline in mood (18%, n = 14), and headaches (16%, n = 13). There were no significant differences in side effects frequencies comparing semaglutide to liraglutide, but there was a trend for less fatigue with the former (17%, n = 10 vs 40%, n = 8; p = 0.06).
Comparison of subjects taking semaglutide vs. liraglutide.

Data is presented as median with interquartile range (IQR) or N with %. Abbreviations: HTN, hypertension; MASLD, metabolic dysfunction-associated steatotic liver disease; T2D, type 2 diabetes mellitus; OSA, obstructive sleep apnea; CAD, coronary artery disease; PCOS, polycystic ovarian syndrome; Δ change in Three Factor Eating Behavior- Revised 18 scores on minus off therapy; weight change (%), last weight on therapy minus baseline weight divided by baseline weight.
Footnotes:
bariatric surgeries listed by semaglutide users (11 had more than one): gastric sleeve (20) gastric bypass (11) mini gastric bypass (6) gastric band (12) other (5); bariatric surgeries listed by liragtlutide users: gastric sleeve (3) gastric bypass (2) gastric band (1);
concomitant drugs for weight loss for semaglutide (4 were taking more than one): phenteremine (n = 5) lisdexamfetamine (n = 2) metformin (n = 4) topiramate (n = 3) other (n = 4); concomitant drugs for weight loss for liraglutide: other (n = 1);
length of treatment at time of answering survey or until drug cessation;
weight change: baseline weight minus last weight on therapy;
missing data (n = 207);
missing data (n = 199).
Respondents endorsing GLP-1 RA therapy discontinuation.
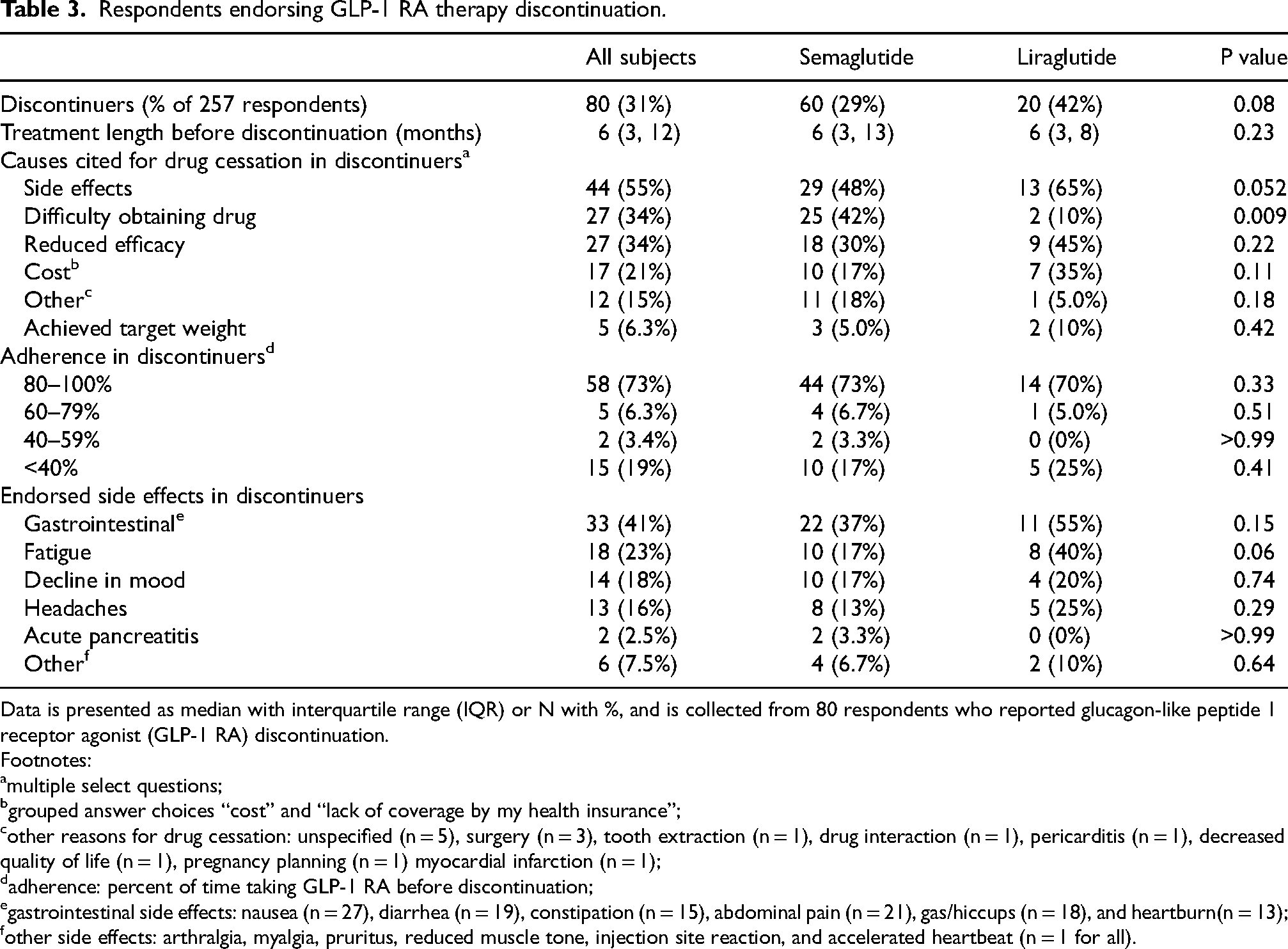
Data is presented as median with interquartile range (IQR) or N with %, and is collected from 80 respondents who reported glucagon-like peptide 1 receptor agonist (GLP-1 RA) discontinuation.
Footnotes:
multiple select questions;
grouped answer choices “cost” and “lack of coverage by my health insurance”;
other reasons for drug cessation: unspecified (n = 5), surgery (n = 3), tooth extraction (n = 1), drug interaction (n = 1), pericarditis (n = 1), decreased quality of life (n = 1), pregnancy planning (n = 1) myocardial infarction (n = 1);
adherence: percent of time taking GLP-1 RA before discontinuation;
gastrointestinal side effects: nausea (n = 27), diarrhea (n = 19), constipation (n = 15), abdominal pain (n = 21), gas/hiccups (n = 18), and heartburn(n = 13);
other side effects: arthralgia, myalgia, pruritus, reduced muscle tone, injection site reaction, and accelerated heartbeat (n = 1 for all).
Changes in weight and eating behaviors
Weight loss, defined as percent of weight reduction since initiating GLP-1 RAs as of last reported weight on therapy, was 12.3% (7.4–19.7) (Table 1), with more weight loss for semaglutide vs. liraglutide (13.4%, 7.9–21.3 vs. 9.8%, 4.6–14.6%; p = 0.003) (Table 2 ). Improvements in eating behaviors, as assessed by TFEQ-R18, were similar for semaglutide vs. liraglutide users, with pooled GLP-1 RA mediated declines in emotional eating and uncontrolled eating (11.1, 0–33.3 and 22.2, −3.7–44.4, respectively) and rises in cognitive restraint of eating (5.6, −5.6–22.2). Emotional eating scores were lower on vs. off GLP-1 RA (33.3, 55.6 vs. 55.6, 22.2 vs. 77.8; p < 0.001), as were uncontrolled eating scores (26.0, 1.1–37.0 vs. 51.9, 30.0–77.8, p < 0.001), while cognitive restraint scres were higher (50.0. 33.3–61.1 vs. 38.9, 27.8–55.6; p < 0.001) (Figure 1A).
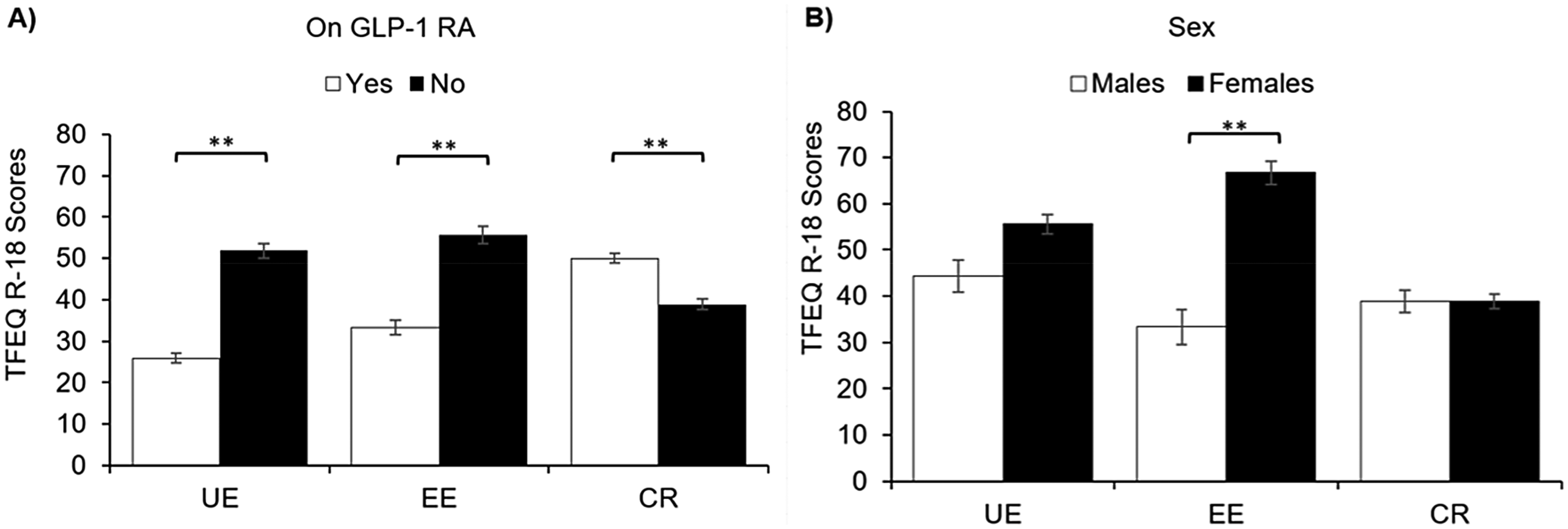
Eating behaviors according to GLP-1 RA usage and sex. A) Eating behaviors on vs. off GLP-1 RA at different time points (n = 257 for both) B) Eating behaviors for males (n = 76) vs. females (n = 181). Data presented as median and standard error. Abbreviations: UE, uncontrolled eating; EE, emotional eating; CR, cognitive restraint of eating; ** p < 0.001.
Correlations with uncontrolled and emotional eating
There was a moderate positive correlation between change in weight and uncontrolled eating and emotional eating on GLP-1 RA (rs = 0.31 and rs = 0.29, p < 0.001 for both), such that weight loss was associated with reductions in these behaviors (Figure 2A-B). There was a weak negative correlation between change in weight and cognitive restraint of eating (rs = −0.09, p = 0.02), so that this behavior increased with weight reduction (Figure 2C). Age was inversely associated with drug-induced changes in uncontrolled eating and emotional eating (rs = −0.23, p < 0.001 and rs = −0.15, p = 0.02), with younger age correlated with greater improvements in these eating behaviors (Figure 3A-B).
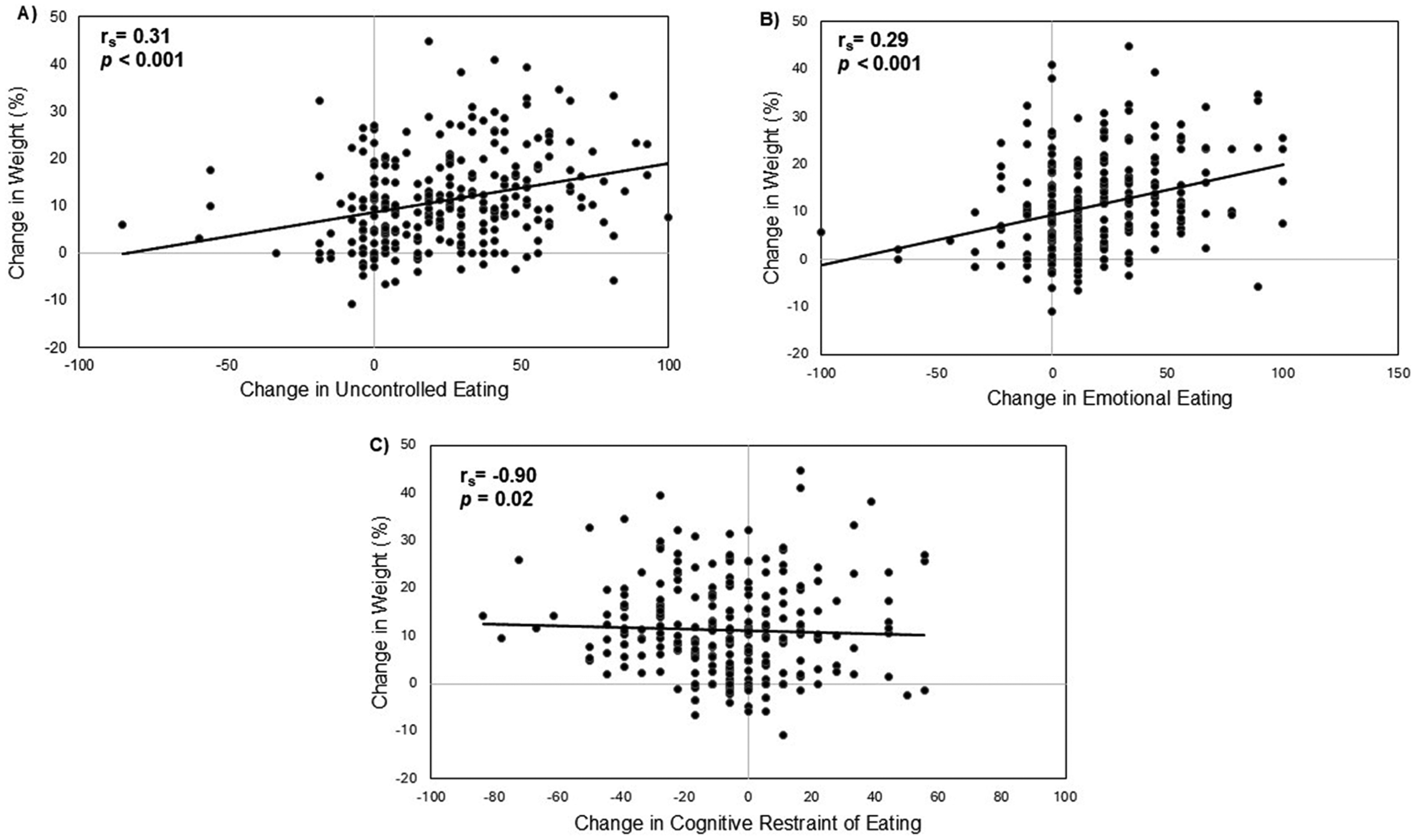
Correlations between GLP-1 RA mediated changes in eating behaviors and weight. A) Change in uncontrolled eating vs. change in weight B) Change in emotional eating vs. change in weight C) Change in cognitive restraint of eating vs. change in weight. Change in eating behavior scores reflect values off therapy minus value on therapy during perceived maximal effect; weight change reflects percent weight change from baseline to last weight on therapy; rs, Spearman's correlation coefficient; n = 257 for all panels.
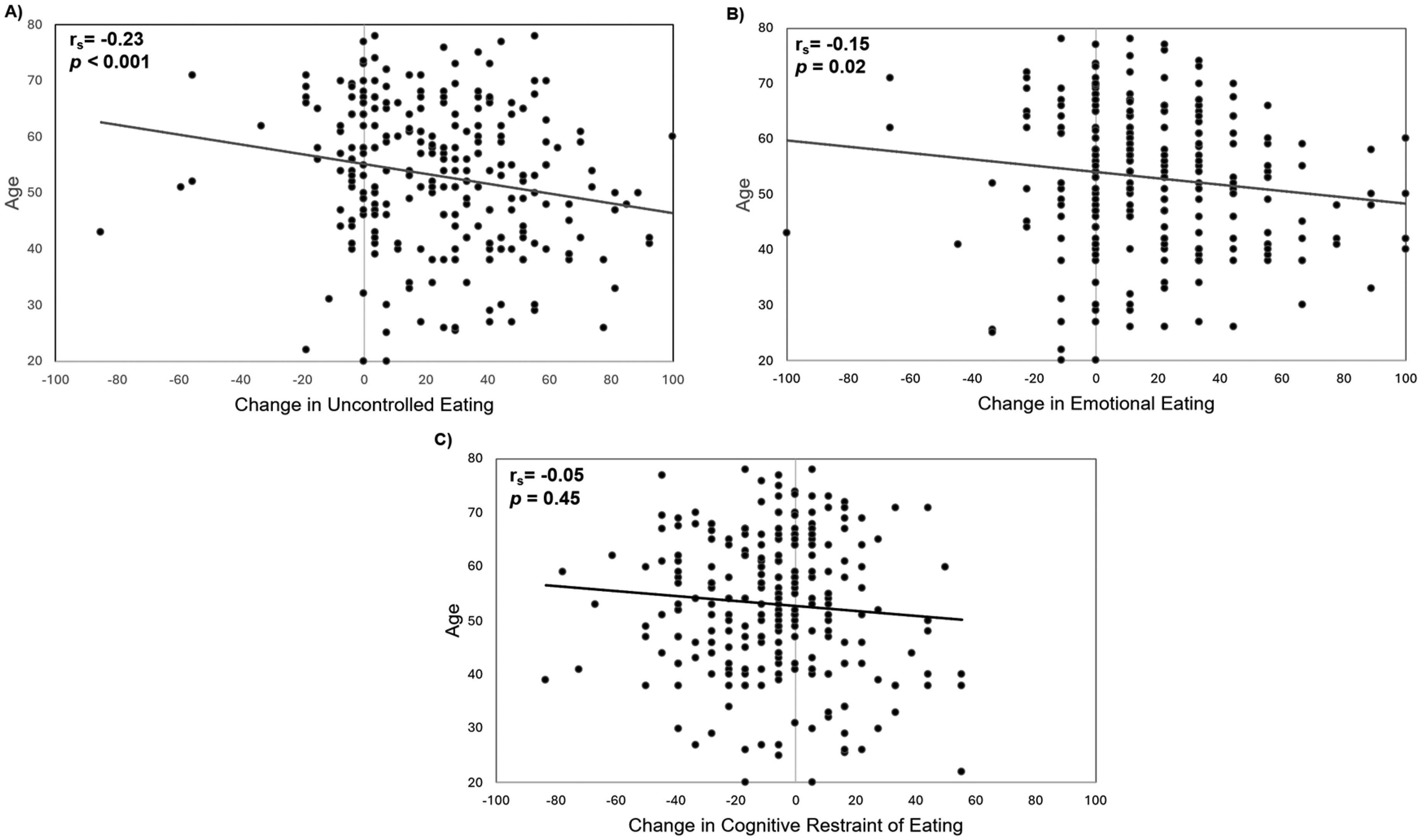
Correlations between GLP-1 RA mediated changes in eating behaviors and age. A) Change in uncontrolled eating vs. age B) Change in emotional eating vs. age C) Change in cognitive restraint of eating vs. age. Change in eating behavior scores reflect values off therapy minus value on therapy during perceived maximal effect; rs, Spearman's correlation coefficient; n = 257 for all panels.
Younger age was also associated with higher uncontrolled eating and emotional eating scores at baseline, off GLP-1 RA (rs = −0.30, p < 0.001 and rs = −0.23, p < 0.001, data not shown). No such correlations were identified for cognitive restraint of eating (Figure 3C). Baseline BMI did not exhibit a significant correlation with uncontrolled, emotional, or cognitive restraint of eating (p = 0.52, 0.21, and 0.14 respectively, data not shown). Among those who had undergone bariatric surgery (n = 45), there were no correlations in months since surgery and these eating behaviors at baseline. However, there was a trend for a weak correlation between months since surgery and reduction in uncontrolled eating (rs = 0.29, p = 0.07), but there were no such trends for other eating behaviors.
Changes in eating behaviors among subgroups
Those who persisted with GLP-1 RA therapy vs those with a history of drug discontinuation had significantly greater GLP-1 RA mediated reductions in uncontrolled eating (29.6, 7.4–46.2 vs. 7.4, 0–29.6; p < 0.001) and emotional eating (22.2, 0–33.3 vs. 0, 0–22.2); p < 0.001), as well a greater rise in cognitive restraint of eating (5.5, −5.6–27.8 vs. 0, −11.1–11.1; p = 0.002) (Figure 4A). Females had greater reductions in uncontrolled eating and emotional eating than males on therapy (25.9, 3.7–44.4 vs. 18.5, 0–37.0; p = 0.04 and 11.1, 0–33.3 vs. 0, 0–22.2; p = 0.005, respectively) (Figure 4B). Importantly, at baseline off therapy, females vs. males also had a trend for higher uncontrolled eating scores (55.6, 33.3–77.8 vs. 44.4, 22.2–73.1; p = 0.08) and significantly higher emotional eating scores (66.7, IQR 33.3–88.9 vs. 33.3, 0–66.7; p < 0.001) (Figure 1B). Respondents with T2D vs. without T2D endorsed less of a reduction in uncontrolled eating (14.8, 0–36.1 vs. 25.9, 3.7–44.4; p = 0.02) (Figure 4C). There were no differences in changes in uncontrolled eating, emotional eating or cognitive restraint of eating between subgroups classified according to psychiatric diagnoses or bariatric surgery (Figures 4D-E). Additionally, there were no differences in changes in these eating behaviors between those who underwent malabsorptive bariatric surgery (n = 27) vs. those who only underwent restrictive bariatric alone (n = 18) (data not shown).
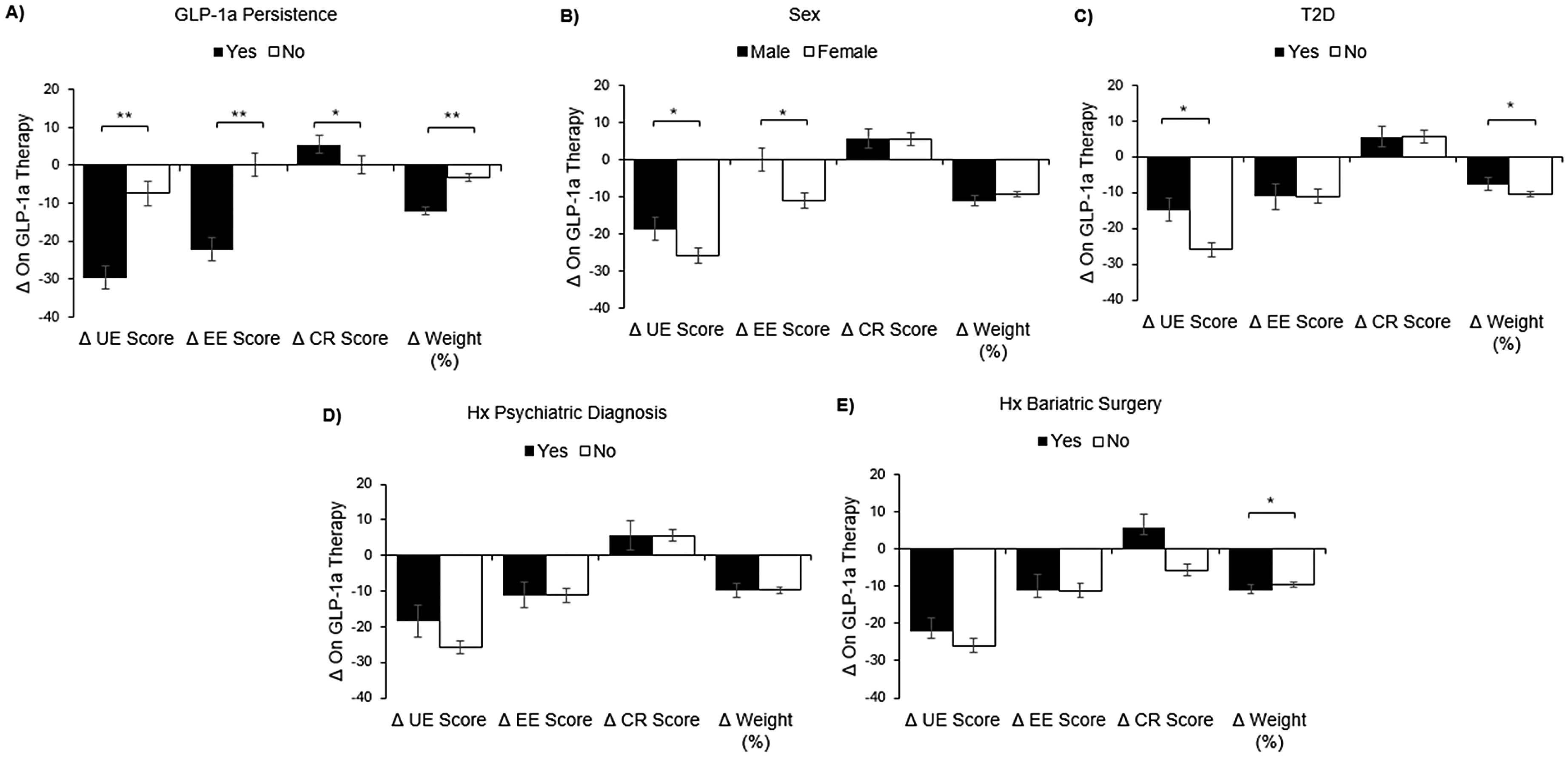
GLP-1 RA associated changes in eating behaviors and weight according to subgroups. A) Drug persistence (n = 80) vs. discontinuation (n = 177) B) Males (n = 76) vs. females (n = 181). C) T2D (n = 56) vs.no T2D (n = 201) D) Hx psychiatric diagnosis (n = 37) vs. no hx psychiatric diagnosis (n = 220) E) Hx bariatric surgery (n = 45) vs. no hx bariatric surgery (n = 212). Data is presented as median and standard error. Abbreviations: UE, uncontrolled eating; EE, emotional eating; CR, cognitive restraint ofeating; T2D, type 2 diabetes; Hx, history; Δ, change.
Change in weight among subgroups
The presence of T2D was associated with less weight loss (7.6, 2.6–15.1 vs. 10.6, 3.7–18.0; p = 0.04) (Figure 4C. Additionally, presence vs. absence of a history of bariatric surgery was associated with more weight loss (11.1%, 6.0–21.3 vs. 9.5%, 2.5–16.1; p = 0.04) (Figure 4E). Males and females experienced similar weight loss (Figure 4B). Those who eventually discontinued GLP-1 RA reported experiencing less weight loss than persistent users (3.3%, 0–9.9 vs. 12.1%, 7.5–19.6; p < 0.001) (Figure 3A). Similarly, there was a significant correlation between treatment length and weight loss (rs = 0.21, p < 0.001) (Table 4). While there were no correlations between baseline cognitive restraint of eating and emotional eating scores and GLP-1 RA mediated weight loss, baseline uncontrolled eating scores and weight loss were significantly correlated (rs = 0.148, p = 0.02). However, change in uncontrolled eating on GLP-1 RA was still correlated with weight loss, even when controlling for baseline uncontrolled eating (rs = 0.26, p < 0.001) (data not shown).
Additional correlations with weight loss on GLP-1 RA.
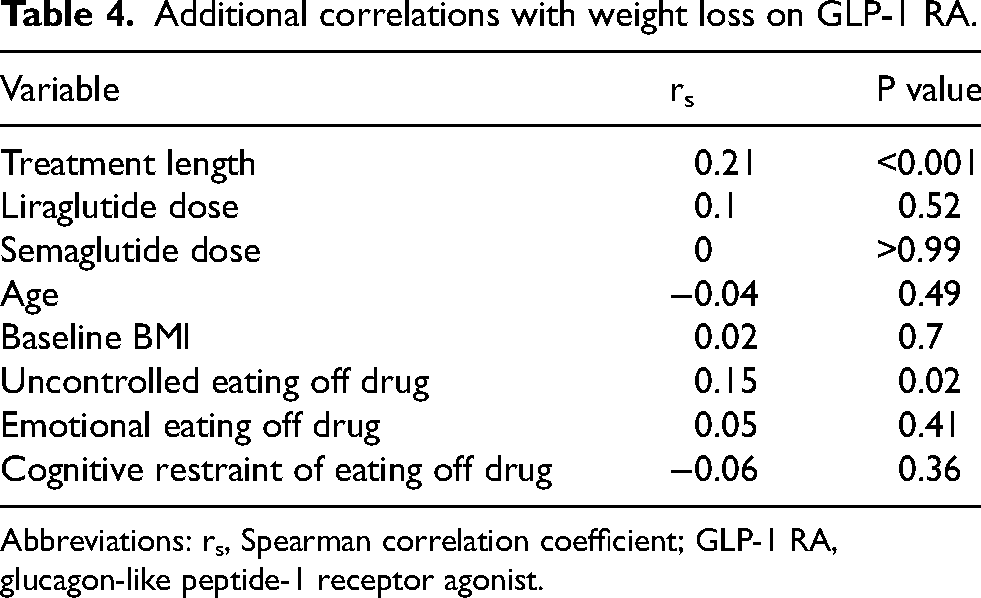
Abbreviations: rs, Spearman correlation coefficient; GLP-1 RA, glucagon-like peptide-1 receptor agonist.
Multiple linear regression analysis was performed to assess the predictive effects of variables that were significantly associated with weight loss in univariate analysis and non-significant variables were removed by forward selection (Table 5). The resulting model was significant (F = 14.8, p < 0.001). The model explains a significant portion (24%) of weight change on GLP-1 RA, with an adjusted R² of 0.24. VIF scores were below 4 for all predictors, indicating no multicollinearity, and the Durbin-Watson statistic of 2.012 ruled out autocorrelation. The strongest independent predictors of weight change in this model were a history of drug discontinuation and T2D, both negative predictors (β=-0.29 and −0.21, respectively). The next most prominent independent predictor was change in uncontrolled eating on drug (β=0.19). The weakest predictors of weight change in this model were GLP-1 RA type (liraglutide vs. semaglutide) and a history of bariatric surgery (β=-0.16 and 0.15, respectively).
Multiple linear regression analysis for predicting weight loss.
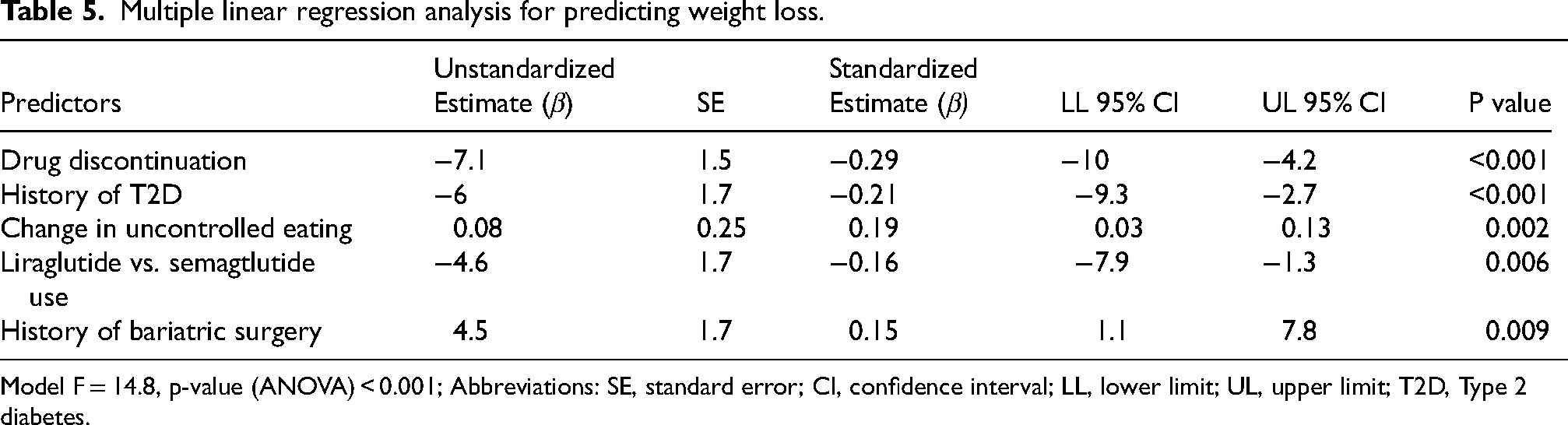
Model F = 14.8, p-value (ANOVA) < 0.001; Abbreviations: SE, standard error; CI, confidence interval; LL, lower limit; UL, upper limit; T2D, Type 2 diabetes.
Discussion
According to our study, GLP-1 RA therapy for PwO leads to reductions in emotional eating and uncontrolled eating and this effect directly correlates with weight loss and drug persistence, or continuation. Furthermore, females and younger adults endorse greater reductions in these eating behaviors. These are novel observations to the best of our knowledge, with broad implications for managing obesity. Many of the real-world findings described mirror results from previous studies regarding GLP-1 RA treatment for weight loss, further validating our overall findings. For example, the reported weight loss of 9.8% for liraglutide 3 mg is similar to that reported in randomized controlled trials.22,23 Weight loss of 13.4% on semaglutide 1 mg is comparable to that reported on a similar dose in people without T2D. 24 Our univariate analysis of predictors for weight loss is consistent with known variables, such as treatment length and the absence of T2D.25,26 While a history of bariatric surgery has previously been shown to not affect GLP-1 RA associated weight loss, 27 in our analysis bariatric surgery emerged as a predictor of greater drug-induced weight loss, raising the possibility of enhanced responsiveness due to changes in gut physiology.
Studies to date have demonstrated that GLP-1 RAs can regulate eating behaviors, and our analysis improves our understanding of this relationship and its clinical implications. Adding GLP-1 RAs to dietary restriction and behavioral therapy has not only a greater effect on weight reduction, but also on uncontrolled eating. 28 Similarly, in a trial of weight maintenance after weight loss, adding liraglutide to exercise increases cognitive restraint of eating compared with placebo. 29 Semaglutide administration was also shown to reduce emotional eating according to scales other than the TFEQ-R18.30,31 According to the Control of Eating Questionnaire, in a two-year study, semaglutide 2.4 mg use reduced cravings and improved control of eating in PwO [12]. In our study, GLP-1 RA therapy not only induced reductions in emotional eating and uncontrolled eating but these reductions also proved to be associated with weight loss.
Whereas previous studies also demonstrated that that females have more emotional eating or uncontrolled eating and that younger individuals have more uncontrolled eating,19,32 our study further demonstrates these high risk subgroups have greater reductions in these eating behaviors in response to GLP-1 RAs. This outcome may partially explain the finding that females lose more weight in response to GLP-1 RA based treatment.33,34 Such differences may be related to how sex hormones interact with other signals to regulate eating behaviors. 35 The fact that females did not lose significantly more weight in our study could be due to a small number of male respondents among other factors. Nonetheless, the observation that females have more improvements in emotional, uncontrolled eating and has ramifications even beyond weight loss.
While our study has several strengths as described, it also has limitations, including reliance on selfreporting and recall. Furthermore, response rates were low, which raises the possibility of self-selection bias and reduced generalizability. Additionally, outcomes for semaglutide and liraglutide users were pooled. There were a few significant differences between subjects treated with semaglutide and liraglutide, such as prevalence of T2D and psychiatric diagnoses, treatment length and weight loss. However, all other characteristics, including changes in TFEQ-R18, were similar and so data was combined to increase power.
Conclusions
In PwO treated with semaglutide or liraglutide weight loss is associated with reduced uncontrolled and emotional eating, and those with greater reductions are less likely to stop therapy. GLP-1 RA mediated changes in eating behaviors differ according to sex, age and history of T2D.
Our results imply modifications should be considered regarding the routine approach to GLP-1 RA treatment for obesity, with an assessment of emotional eating. A major barrier to successful therapy is drug discontinuation, which is associated with a lack of reduction in uncontrolled, emotional eating according to our analysis. This finding implies that it would be prudent to question patients about changes in emotional eating during follow-up visits and consider patient-related modulating factors, such as age and sex. It would be valuable to better understand if, to a certain extent, females or those without T2D lose more weight on GLP-1 RA treatment because of greater reductions in emotional, uncontrolled eating than their counterparts. Future prospective studies for newer GLP-1 RA therapies should include an assessment of emotional, uncontrolled eating to determine with greater certainty whether drug-induced reduction in these behaviors independently affect weight loss and drug persistence.
Supplemental Material
sj-docx-1-mnm-10.1177_1973798X251356434 - Supplemental material for Reduced emotional, uncontrolled eating with glucagon-like peptide 1 receptor agonists: Associations with demographics, weight loss and drug persistence
Supplemental material, sj-docx-1-mnm-10.1177_1973798X251356434 for Reduced emotional, uncontrolled eating with glucagon-like peptide 1 receptor agonists: Associations with demographics, weight loss and drug persistence by Noga C Minsky, Sara Kutzkel, Orly Tamir and Gabriella Segal-Lieberman in Mediterranean Journal of Nutrition and Metabolism
Footnotes
Acknowledgements
We would like to thank those who kindly took the time to complete our survey and the entire staff of our Center for Weight Management. We would like to thank Dr Jenny Cina who assisted with conceptualization of the research and registered dietician, Melanie Cunyo, who provided feedback on the questionnaire and considerable assistance with collection of responses.
Ethical considerations
Informed consent for study participation was obtained by all study subjects. Sheba Medical Center IRB issued approval 0177-23-SMC. Approval was obtained in 2023 to conduct this research from the Sheba Medical Center IRB (protocol number 0177-23-SMC) in accordance with the Declaration of Helsinki.
Author contributions
Noga Minsky: Conceptualization, Formal analysis, Data Validation, Investigation, Methodology, Project administration, Resources, Supervision, Visualization, Writing – original draft, Writing – review & editing. Sara Kutzkel: Conceptualization, Formal Analysis, Data curation, Data validation Investigation, Methodology, Project administration, Writing – original draft, Writing – review & editing. Orly Tamir: Writing – review & editing, Supervision, Methodology, Validation, Conceptualization. Gabriella Segal-Lieberman: Conceptualization, Investigation Methodology, Project administration, Resources, Supervision, Visualization, Writing – review & editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Gabriella Segal-Lieberman and Noga Minsky declare a relationship with Novo Nordisk that includes personal fees and support for attending conferences. All authors have declared that there are no other relationships or activities that could appear to have influenced the submitted work.
Data availability statement
Consent
Written informed consent for study participation was obtained.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
