Abstract
Keywords
Introduction
This paper introduces the laminar SAH of infant venous stroke or CVT (cerebral venous thrombosis aka cerebral venous sinus thrombosis). Neuroradiologic evidence for the laminar SAH derives from a case study series of 34 infants suffering from a venous stroke. These cases feature a chronic subdural hemorrhage (cSDH or hygroma). The laminar SAH may be seen in CT and MR imaging studies along the undersurface of the arachnoid membrane (AM) within the subarachnoid space (SAS). The radiologic appearance of the laminar SAH is correlated with published photographic images of autopsy brains of infants with CVT. The importance of distinguishing the laminar SAH from a subdural hemorrhage (SDH) over the brain convexity is discussed.
Radiologic evidence supports the existence of a laminar SAS, a potential space hypothetically located between the AM and an investing membrane that fills with blood leaking from a thrombosed cortical vein. The investing membrane may be formed by the trabeculae that cross the SAS and anchor to the AM. Surface cells of the trabeculae cover the cortical veins to form a bed through which a cortical vein travels adjacent to the AM while the trabeculae collagen terminate in the AM. Alternately, the recently reported 4th meningeal layer or SLYM (subarachnoid lymphatic-like membrane) divides the SAS into inner and outer compartments and may confine the laminar SAH. Another potential space between the AM and the neomembrane of the cSDH within the subdural space is also discussed.
A laminar SAH differs from a SDH and its recognition is important in establishing a differential diagnosis of intracranial hemorrhage. The cases of infant venous stroke support the association of a chronic SDH with venous stroke. 1
Method
A retrospective review of 34 cases of infant venous stroke in the period 2014–2025 is based on brain CT and MR imaging of CVT in infants with a cSDH. The cases are unsolicitous referrals by parents, attorneys defending caregivers, and physicians on a collegial consultation basis where the caregivers are accused of abusive head trauma. Excluded were 4 cases with a calvarial fracture, one with a layered (double chronic) cSDH, one short fall, and 2 with global hypoxic ischemic encephalopathy, the possible presumptive cause of CVT. The radiologic data is from collected DICOM (Digital Imaging and Communications in Medicine) files from different institutions including general and children’s hospitals. The radiologic analysis herein is based solely on the author’s interpretation of the studies using accepted neuroradiologic practices.
Results
32/34 cases had a CT head scan and 31/34 had an MRI brain scan. All but two initial exams were followed up with a second exam 0 to 10 days later: median 1 day. All 34 cases exhibit venous stroke defined as presence of blood clot in a venous sinus or cortical vein or SAH from a thrombosed cortical vein that may have recanalized. 33/34 cases exhibit a cSDH supporting a strong association of a cSDH with a venous stroke. The sole case without a cSDH had a depressed occipital bone at the lambda and an adjacent superior sagittal sinus (SSS) thrombus. The infant age ranged from 1 to 16 months with average/median age equal to 4/3 months. None of the cases selected for the study had overt evidence of appendicular/axial skeletal trauma or major organ injury. None exhibited calvarial fracture or cervical spine injury. 4/34 had scalp swelling without calvarial fracture; for one infant the scalp swelling first appeared on the second exam. 2/34 had a depressed lambda of the occipital bone with associated SSS thrombosis. One had a small AVM, and one had a few small cortical infarcts, probably embolic of indeterminate etiology.
34/34 studies show cortical vein thromboses based on SAS hyperdense foci in CT and/or black SWI signal in MRI studies. 28/34 cases show multifocal (<2 cm diameter) SAHs. 24/34 cases show laminar SAHs (>2 cm diameter); 10/24 thin (<3 mm) and 14/24 thick (>3 mm). 4/34 show intrasulcal hemorrhage in CT images and 11/34 in MR SWI images (note that it may be difficult to distinguish intrasulcal SAH from a normal cortical vein or a thrombosed cortical vein with or without leakage into its PVS).
24/33 show sinus thromboses based on initial CT studies; 14/33 in the SSS including one probably in an adjacent lacune; 13/33 in at least one transverse sinus, and 2/33 in the straight sinus (both with intraventricular hemorrhage seen in MR SWI).
Additional cases with intraventricular hemorrhage include a case with an AVM (no CT study done), and 4 cases with transverse sinus and/or SSS thrombi.
Cortical veins are located subjacent to the AM over a significant extent of their course in the SAS as seen in Figure 1. Coronal T2 weighted MRI of an infant with a cSDH shows cortical vein black foci (white arrows) located along the undersurface of the AM. A cSDH is present (white blunt arrows).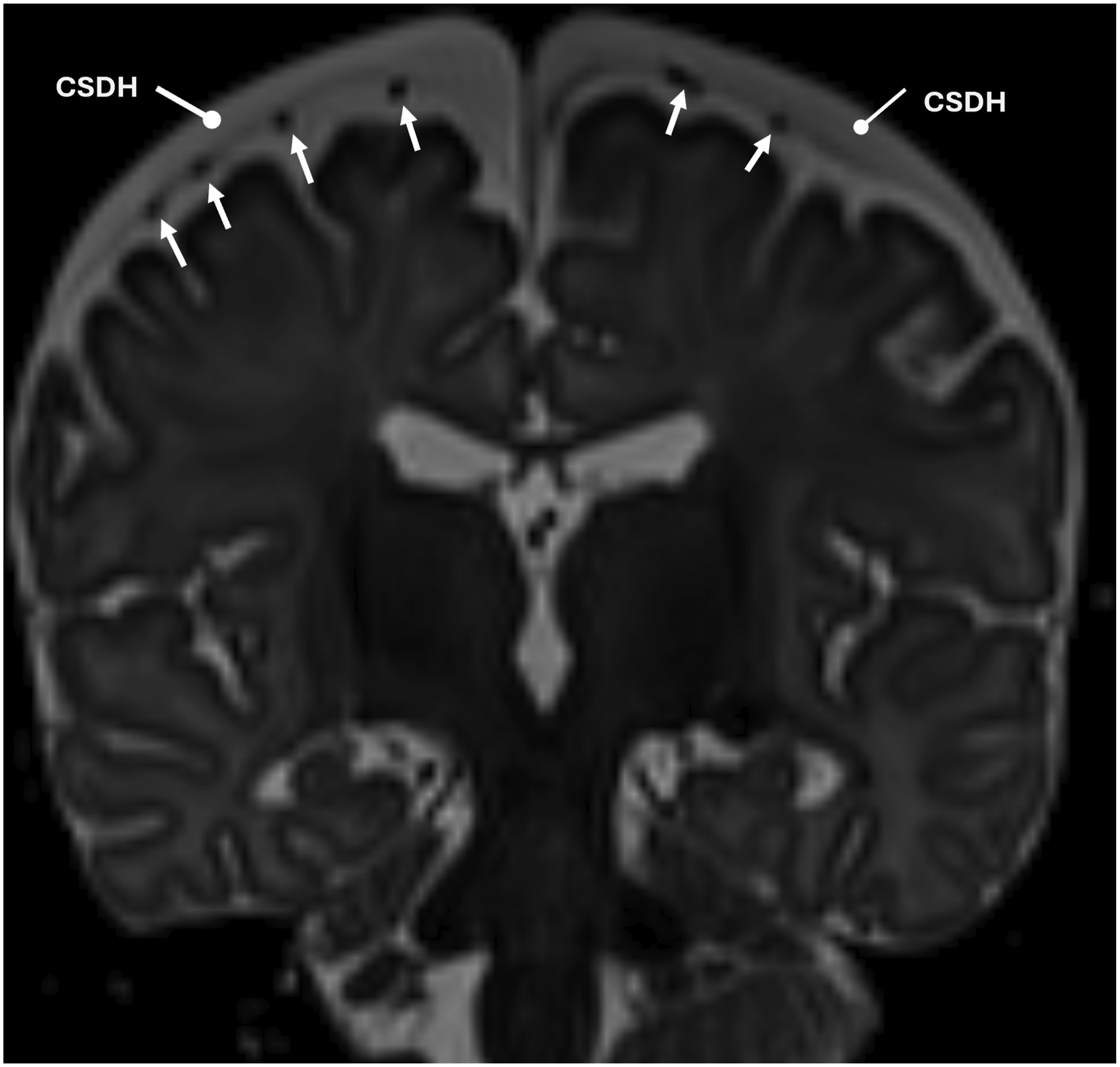
Cortical veins both within the SAS and those traversing a cSDH to the dura may exhibit a continuous perivascular space (PVS) (see Figure 2). MR T2 signal intensity differences between the fluid in the PVS and the SAS defines the PVS. The signal difference is likely the result of differences in CSF motion that causes T2 dephasing signal loss or fluid content (such as trabeculae in the SAS). Within the dura, the perivascular sheath is not visualized with clinical MRI. The globular perivascular thrombus (aka lollipop or tadpole) (white arrows) in the SAS and PVSs (higher T2 signal, black arrowheads) are shown. SWI image blooming obscures visualization of the perivascular space in B. Also note the bridging vein crossing the cSDH retains a perivascular sheath (pair of black arrowheads in A) (A-T2 axial, B-SWI axial, C-T2 sagittal, D-T2 coronal).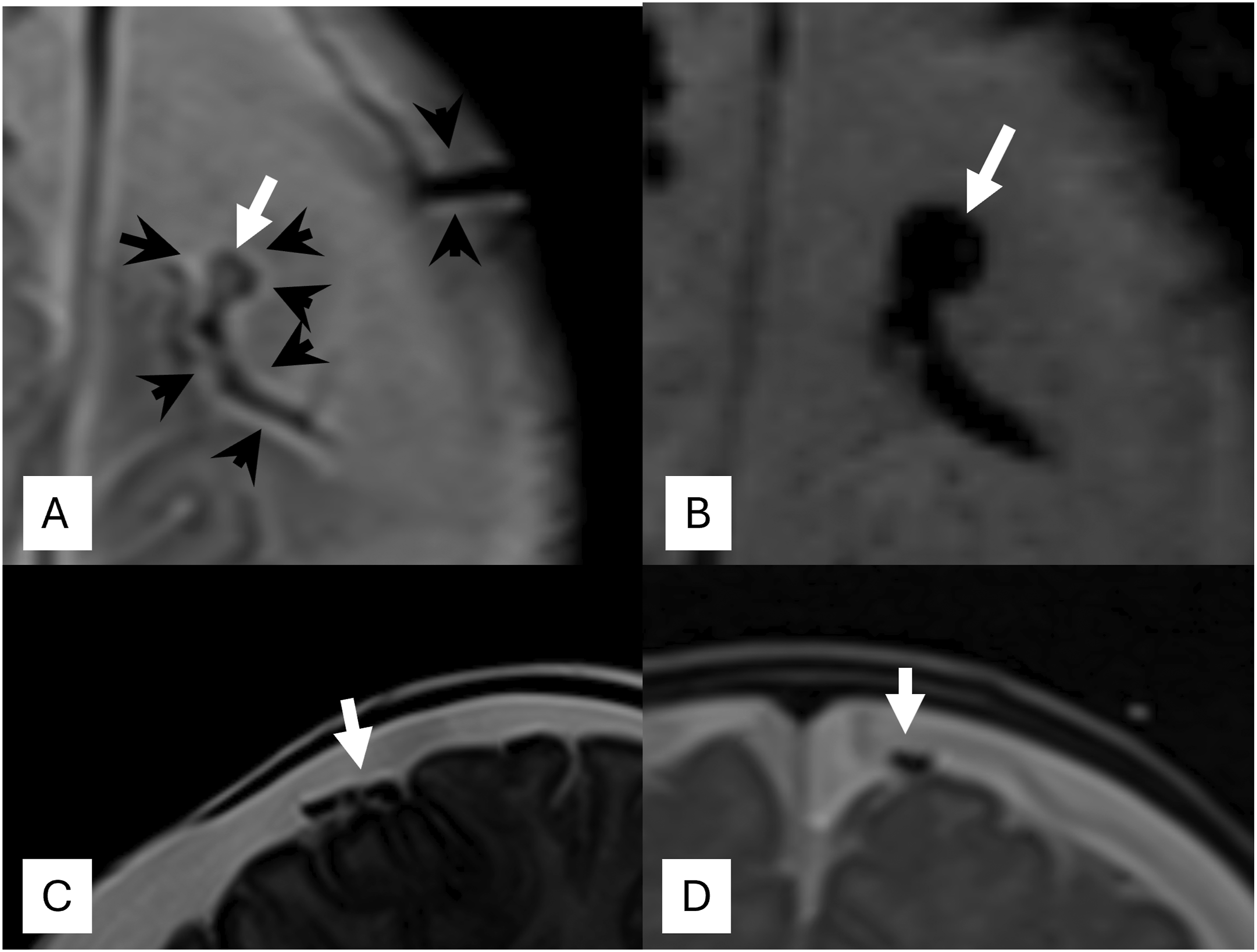
Figure 2 shows a cortical vein thrombus encased by an intact perivascular sheath that is designated the “globular perivascular thrombus sign” (see Figure 2). This is also known as a so-called “lollipop” or “tadpole.”2–5 Note there is no large volume of blood as would be expected from a traumatic rupture.
A globular perivascular thrombus typically resides in the SAS within its perivascular sheath. It may extend into the segment of the perivascular sheath crossing the cSDH. If the PVS ruptures, the leaked blood from a thrombosed cortical vein or globular perivascular thrombus spills outside the sheath and loses the smooth contour of the PVS. Many thrombosed cortical veins are not globular perivascular thrombi. Most show vein enlargement or focal hemorrhage with an irregular shape.
Blood leaking outside the PVS of a thrombosed cortical vein may remain focal or fan out as a layer in the SAS. A non-focal (planar diameter greater than 2 cm) SAH from a thrombosed cortical vein is described herein as a laminar SAH, either thick >3 mm or thin <3 mm.
The thick laminar SAH is illustrated in Figure 3. The CT study in Figure 3 has the appearance of a SDH, yet closer inspection shows a few intrasulcal SAHs. In the MRI note the continuity of hemorrhage extending from the subdural to the SAS along the expected course of a thrombosed bridging vein (BV). The leaked blood splits into intradural/subdural and subarachnoid components on either side of the cSDH. They are spanned by a bridge of hemorrhage along the BV either within its PVS or confined by the CSDH neomembrane. Note the intradural/subdural hemorrhage is “trapped” by the cSDH neomembrane. The intrasulcal SAH is absent in the 2 day later MR images indicating rapid resolution of intrasulcal SAH. In the SAS, the laminar blood obscures the thrombosed cortical vein. Thick laminar SAH. 6 month old infant presenting with seizures with SSS thrombosis and extra-axial hemorrhages. SSS thrombosis (density = 73 HU; bilateral cSDHs (blunt thick arrows), and right acute SDH and acute SAHs (white short arrows). Note the continuity of hemorrhage extending from the subdural to the SAS along the expected course of a thrombosed BV bounded by the cSDH neomembrane. The cSDH “traps” the intradural SDH. (A-CT axial, B-CT coronal, C-MR T2 axial, and D-MR T2 coronal).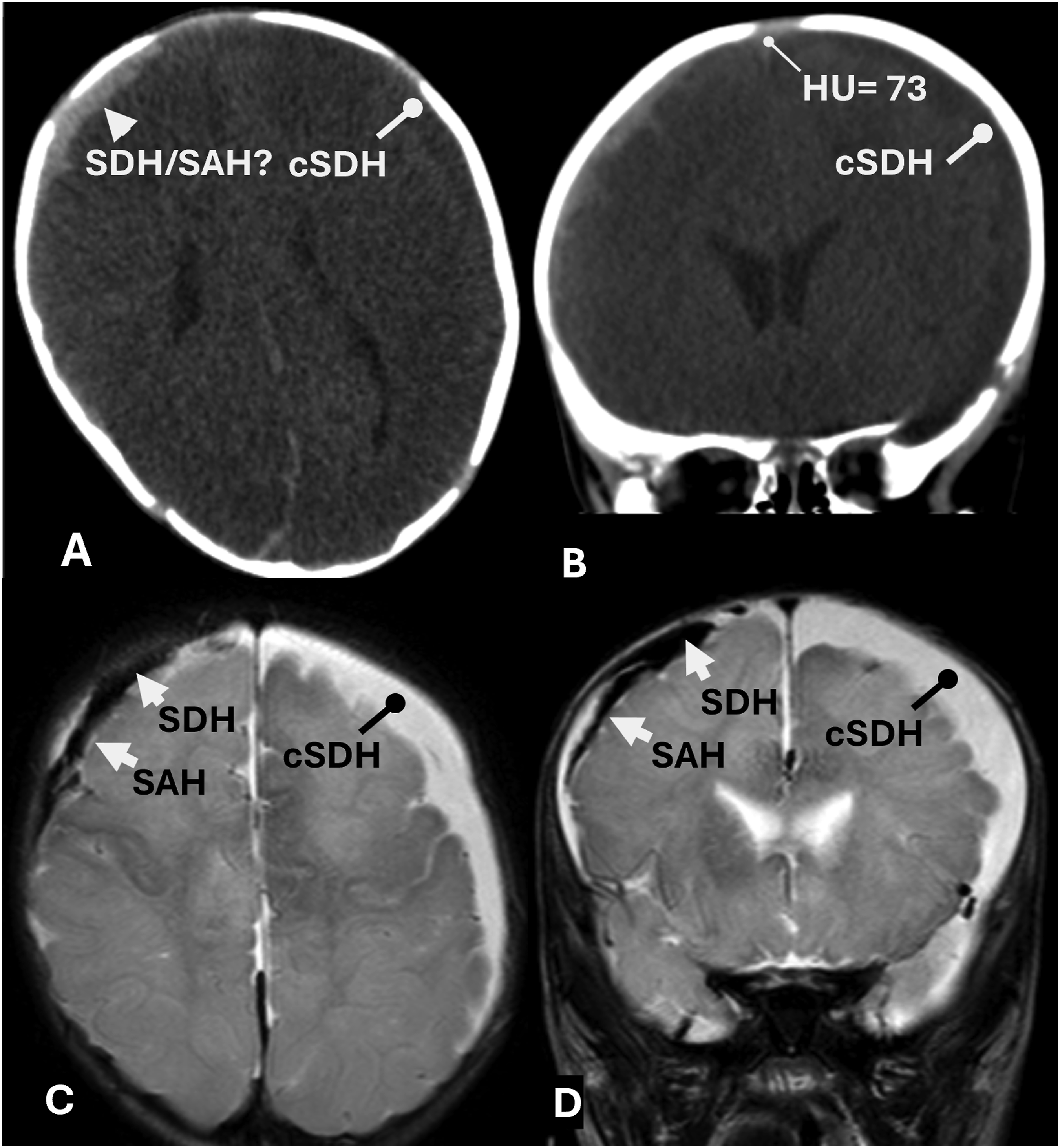
Subtle separation of a thin laminar SAH from the dura by a cSDH is an important clue as to its true location as seen in Figure 4. Often radiologists report this combination of an acute laminar SAH and a cSDH as “mixed density SDH” or “layered SDH.” The laminar SAH partially resolves in the follow-up MR images indicating its rapid resolution that helps differentiate a SAH from a SDH. Thin laminar SAH. 10 month old infant presenting with seizures with left transverse sinus and vein of Labbe thrombosis and extra-axial hemorrhages. Left cSDHs (white blunt arrows), and left thin laminar acute SAHs (white arrowheads). Note the 3 day significant resolution of the thin laminar SAHs in follow-up MR images in C and D. (A-CT coronal, B-CT axial, C-MR T2 coronal, and D-MR SWI axial).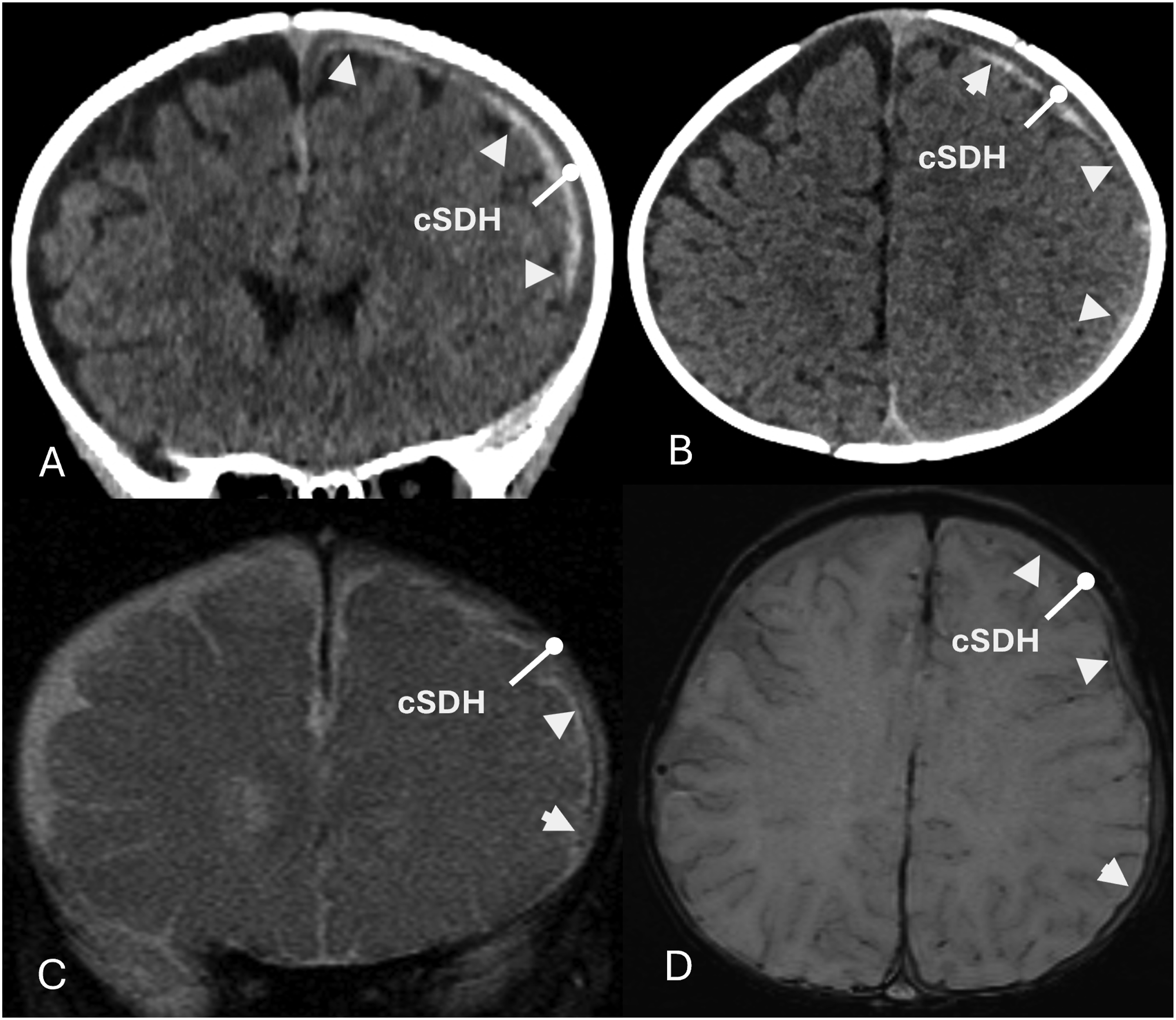
Discussion
Venous stroke (CVT)
Epidemiologically, venous strokes in infants are thought to be rare, and thus often are neglected in the differential diagnosis. On the other hand, the time of life of greatest incidence of venous stroke is the neonatal period, a consequence of the birthing process that includes compressive forces on the head and circulation alterations. This abrupt shift in the incidence of venous stroke between the neonate and infant has been challenged. 1
A pathophysiologic understanding of infant venous stroke contributes to proper diagnosis and patient care. Infant venous stroke is associated with medical conditions including dehydration from vomiting, diarrhea, and other causes; an infection located anywhere in the body; blood dyscrasia; venous pressure alterations such as from projectile vomiting and coughing/choking; and cSDH. 1 Awareness of the strong association of a venous stroke in infants with a cSDH should prompt the radiologist to inspect brain images for findings related to CVT whenever a cSDH is identified. When SAHs are detected in association with cSDH, the radiologist needs to raise awareness of a possible venous stroke. 1
Diagnosing a venous stroke in an infant is difficult. The sine quo non of the diagnosis is identifying a thrombus within a cerebral vein or sinus. Quite often the identification of a clot in a venous sinus is elusive because the clots resolve quickly in infants often within hours leaving behind difficult to identify remnants. Hence, the need for early imaging 6 and scrutiny of the initial scan, typically a CT of the head. Rapid clot resolution may be related to immature blood factors that protect the newborn from CVT at the time of greatest CVT incidence. Within the SSS, clots may be difficult to detect with high specificity. Normal folds within the SSS divide the lumen into compartments. 7 Hence, it is difficult to distinguish a web or adhesion of a recanalized thrombosed SSS from a normal anatomic fold. Thrombi in a lacune are difficult to distinguish from intradural hemorrhage in non-contrast images. Non-enhancing thrombi may be seen in lacunae and venous sinuses with post-contrast imaging which is infrequently done.
Clots within cortical veins over the surface of the brain, even if recanalized, leave behind identifiable blood leaked outside the vein wall, readily detected and localized with CT and MRI cross-section studies. 8 The morphology of the leaked blood is variable, either focal or fanned out, where the latter are characterized herein as thick or thin. In the dura both normal and thrombosed cortical veins and their PVSs are not readily seen. While SSS thrombosis detection is difficult, the presence of multiple easily detectable bilateral CVTs is a probable indication of SSS thrombosis that may have resolved.1,8,9
Detection of SAH
SAH is commonly observed after rupture of a brain aneurysm or traumatic disruption of small vessels in the SAS. A SAH is commonly recognized over the convexities of the brain by its location within the sulci of the cerebrum, sometimes referred to as a gyral pattern, 10 a feature that allows differentiation from an SDH. 11 Unfortunately, after a lapse of time, the intrasulcal sign is not useful because the hemoglobin disperses from CSF flow dynamics decreasing CT and MRI conspicuity. 12 The intrasulcal morphology leads to the common notion that a SAH exists only when observed within brain sulci. This is not true as demonstrated herein by the existence of the laminar SAH.
In sufficient quantity intrasulcal acute blood is hyperdense in CT images. When filled with lesser amounts of blood it may appear as “apparent effacement of sulci.” Conventional MR spin echo T1 and T2 MR images are insensitive to the detection of acute SAH in which these signals are nearly isointense to brain tissue. Higher signal in FLAIR images correlates with the cellularity and protein levels of acute SAH. SWI is the most sensitive sequence12,13 used to detect SAH, but the signal may be indistinguishable from bone or a normal vein.
Infant venous stroke: Acute SDH or SAH?
A major question facing the radiologist is whether blood superficial to the brain surface resides in either the SAS or the subdural space, an important distinction. Without other supportive evidence of trauma, neither is specific for trauma. Adult major trauma, such as dashboard injuries, involve rotational forces that cause BV ruptures, superior convexity intraparenchymal injuries, and mainly SAHs, not “noteworthy” SDHs. 14 Infant abusive head trauma is commonly associated with SDH, a component of the so-called triad, yet for congested and thrombosed cortical veins the pathologist sees predominantly SAHs, not SDHs. 2 As reported herein acute blood over the brain surface in alleged abusive head trauma without associated evidence of external head or neck injury is located mainly in the SAS, not the subdural space, is not associated with traumatic intraparenchymal injury, and originates from thrombosed, not mechanically ruptured cortical veins. The force is hydraulic, not mechanical.
The laminar SAH: Could it be mistaken for an SDH?
When the leaked blood fans out to form a laminar SAH, the location is questioned since it resembles a typical SDH covering the brain convexity. In the presence of a cSDH the laminar SAH is located alongside the cSDH and the AM, separated from and never adjacent to the dura. Typically, the laminar SAH features homogenous density, is immiscible with the cSDH, does not sediment like an acute SDH, 2 lacks inhomogeneous coagulation, and rapidly resolves unlike most acute SDHs.13,14 The cSDH serves as a barrier between the laminar SAH and the highly vascular dura. The combination of an acute laminar SAH and a cSDH is commonly, incorrectly described in clinical radiology reports as “layered or mixed density SDH.”
The rapid resolution of the laminar SAH may result from migration within the SAS to a location outside the SAS. Exit pathways are not readily identified in this study with the exception of a pathway between the SAS and the dura along the BV as may be seen in Figure 3 C and D. Hypothetically, blood may leak into the subdural space outside the cSDH through a rupture in the perivascular sheath of the BV crossing the cSDH. Figure 2 suggests the perivascular sheath remains intact, though this is inconclusive.
It is not unusual to visualize small amounts of fresh blood in the posterior extra-axial space. With the head in the supine position, visualized blood sandwiched between the posterior brain and the calvarium probably resides in the subdural space. The precise anatomic location, namely, within or outside the cSDH, is indeterminate. Fresh blood within the cSDH may represent a cSDH rebleed, either spontaneous or a response to the thrombosis; outside the cSDH may indicate a rupture of the perivascular sheath of the BV.
Some cases show spinal subdural hemorrhage likely derived from the blood engorged posterior falx/tentorium associated with increased intracranial pressure/venous stroke. 15
Autopsy photos of patches of blood viewed through the translucent AM over the brain surface in cases of infant venous stroke is sufficient evidence of laminar SAH.2,16–18 This is confirmed by radiologic imaging of the laminar SAH (Figures 3–5). Thin laminar SAH. 3 month old infant found unresponsive. Day 1 CT head images in A, B, and C show a thin patch of SAH subjacent to a cSDH that disappeared on day 2 (CT head study not shown). Note SSS focal thrombosis (HU = 36 in A and HU = 51 in B) On day 2 the SSS clots partially resolve (not shown). (A-CT axial, B-CT coronal, and C-CT sagittal).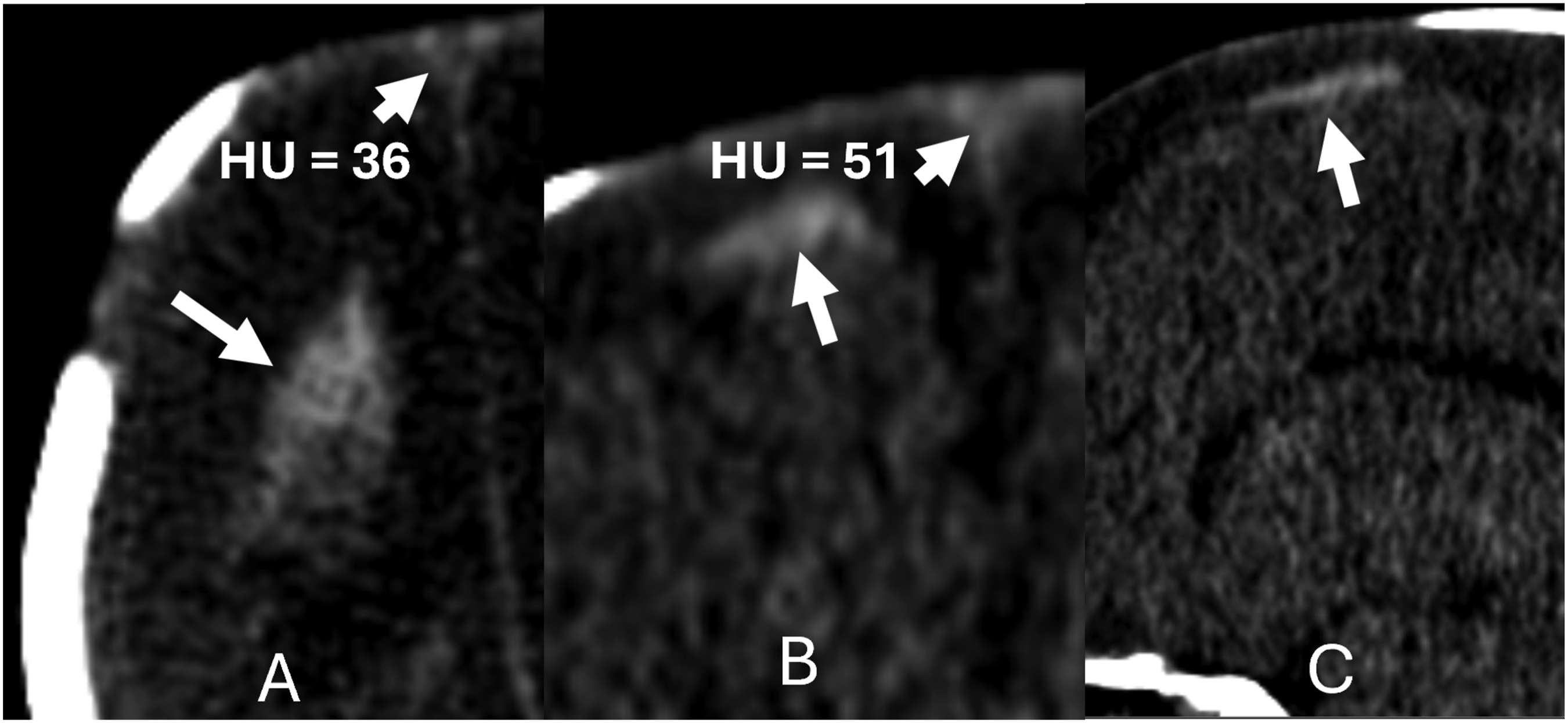
The laminar SAH: Could it be mistaken for a subpial hemorrhage (SPH)?
A SPH is usually distinguished from laminar SAH by neuroimaging, except when it has a gyral component. SPH occurs most frequently in the perinatal/neonatal period, may be post-traumatic underlying open sutures, often globular in shape, with adjacent brain mass effect, sulcal widening, intraparenchymal hemorrhage, abnormal MRI diffusion signal, and/or brain tissue edema. It is located most frequently in the temporal lobes and is associated with medullary vein congestion. 19
Cortical vein gross anatomy
The cortical veins of interest emerge from the brain, pass through the pia mater, course superomedially over the brain convexities within the SAS, enter the dura near the apex and terminate in the SSS.20,21 Cortical veins are valveless and have thin walls with a minimal muscular layer.22,23 They are not well seen in cross-sectional radiology imaging at their extremes, namely, in the brain parenchyma and at their dural termination in the SSS. They are best seen in MR T2 images and SWI in the SAS.
Within the SAS many of the cortical veins are closely applied to the AM (see Figure 1). Upon exiting the SAS, the BV penetrates the dura typically 1–2 cm from the SSS. The BV terminates in the SSS, often at a hairpin angle; it may pass through, under, or enter/terminate in lacunae. The lacunae are lateral projections of the SSS between layers of the dura and receive blood primarily from meningeal veins and drain dural channels containing fluid. As best seen in autopsy and venous angiogram studies, lacunae reside off the main channel of the SSS and are not reliably identified in MRV studies which rely on venous flow rates above a threshold value. Within the dura the cortical vein exhibits a ring of abundant smooth muscle cells “which acts as a sphincter regulating the flow of blood from the bridging veins, maintaining intravenous pressure when intracranial pressure rises.”2,7,18
The location of the cortical vein within the SAS closely applied to the undersurface of the AM is seen in radiologic studies (Figure 1). Also see referenced Figure 5. 24 In an autopsy, after removal of the calvarium and subdural blood, the rapid exit of CSF from the SAS shrinks the AM to the brain surface. The trabeculae in the SAS become matted and the relative location of the cortical veins cannot be readily discerned grossly. In published works on animal models, anatomists illustrate cortical veins randomly positioned in the SAS24–26 where the propensity to develop a laminar SAH may not exist. Such studies may not correlate with those in humans and are subject to site selection, processing errors, and small sample size. 26 The uncertainty in the position of the cortical veins and their perivascular sheaths calls for better pathologic correlation.
In the future it is likely that optical coherence tomography based on harmless backscatter of infra-red light will provide the cortical vein spatial location with 1–15 µm resolution. Optical coherence tomography is the first imaging technique to study brain microanatomy in vivo on dogs. 27 Injected saline expansion of the SAS in autopsied human specimens allows visualization of the SAS content with optical coherence tomography and has been used to quantify the volume fraction of trabeculae. 28 It has the capability of delineating cerebral vessel walls. 29 Intraoperative optical coherence tomography has the potential to correlate neuropathology studies of the deceased and neuroradiology studies of the living.
SAS microanatomy
The author’s depiction of the SAS microanatomy is shown in Figure 6. The pia mater rests on the basement membrane of the glial limitans of the brain, from which it is only separated by a subpial space which contains collagen bundles, fibroblast-like cells, and blood vessels.
30
Sketch of SAS over brain convexity. Sheet-like and filiform trabeculae (T) span the SAS, attached to the AM and the pia mater (PM). A cortical vein (CV) traveling along the undersurface of the AM is exposed in cross-section. It is covered by its perivascular sheath and a second layer, the leptomeninges of the trabeculae. (B = brain, D = dura, GL = glia limitans, and SPS = subpial space).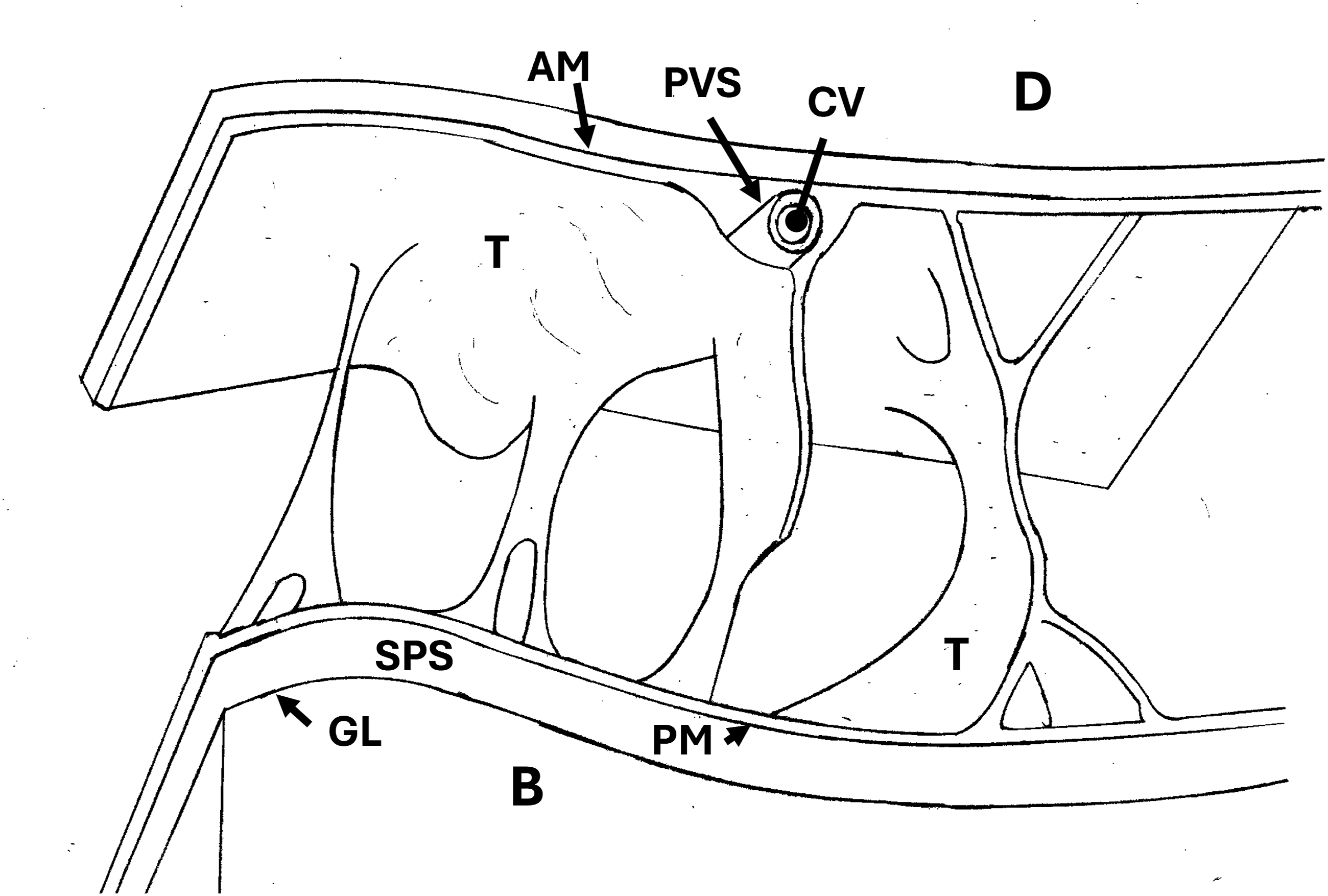
The SAS contains CSF, blood vessels, nerves, and trabecula. The trabeculae are delicate, sheet-like connective tissue that form a filamentous network containing CSF in the SAS. The trabeculae consist of a collagen core covered by a leptomeningeal cell layer. The collagen reinforced trabeculae extend through the SAS forming a loose fabric network that stitches the pia mater to the AM. The CSF within the SAS freely communicates subject to channeling by trabecular membranes.
The trabeculae play a role in the mechanical support of the brain and supportive framework for the arteries and veins. 24 A qualitative assessment of trabecular (and bridging vein) strength is illustrated in autopsy photos exposing the stretched bridging vein crossing from the AM to the dura across the subdural space noting that the AM is not pulled away from and remains attached to the brain surface by the trabeculae.2,31,32
Upon exit of the subpial space and entry into the SAS the veins acquire a leptomeningeal coat reflected from the pia mater that forms its PVS along its course in the SAS extending to the dura. Within the SAS the veins acquire another coat consisting of the trabeculae leptomeninges. The fenestrated trabecular coat encloses small veins, covers large veins, and joins the pia mater and AM outer layers.25,30,33,34
The perivascular canalicular system 35 of cortical arteries and veins plays a significant role in the clearance of waste products from the brain. 36 and may be seen in routine MRI studies. 37 The arterial PVSs or Virchow-Robins spaces may be seen in many specific locations in every day clinical practice,38,39 and in MRI studies where gadolinium contrast is injected in the lumbar SAS and imaged into the brain. 40
Normally, upon exiting the SAS, the BV penetrates the AM and passes through the contiguous deep dural layers before entering the sagittal sinus. 2 In the presence of a cSDH, the PVS of the exiting BV is continuous as it exits the SAS and crosses the AM and subdural space to the dura as shown in Figure 2, an observation that calls for further radiologic-pathologic study.
The laminar SAH anatomy
As shown in Figure 6 The cortical vein position is adjacent to the AM covered by the trabeculae and its PVS. As shown in Figure 7, as envisioned by the author, the leakage of blood from a thrombosed cortical vein fans out beyond the PVS into a space between the AM and the trabecular coat. This laminar SAH is limited in its extent by the amount of blood expressed and the adherence of the trabeculae to the AM. The blood clears via the trabeculae fenestrations. While the evidence presented herein and the autopsy photos of the SAH provide substantial support for this proposed anatomic explanation, further radiologic-pathologic correlation is needed. Same as Figure 6 except a layer of blood is shown surrounding a thrombosed cortical vein outside its ruptured PVS between the AM and a trabecular coat.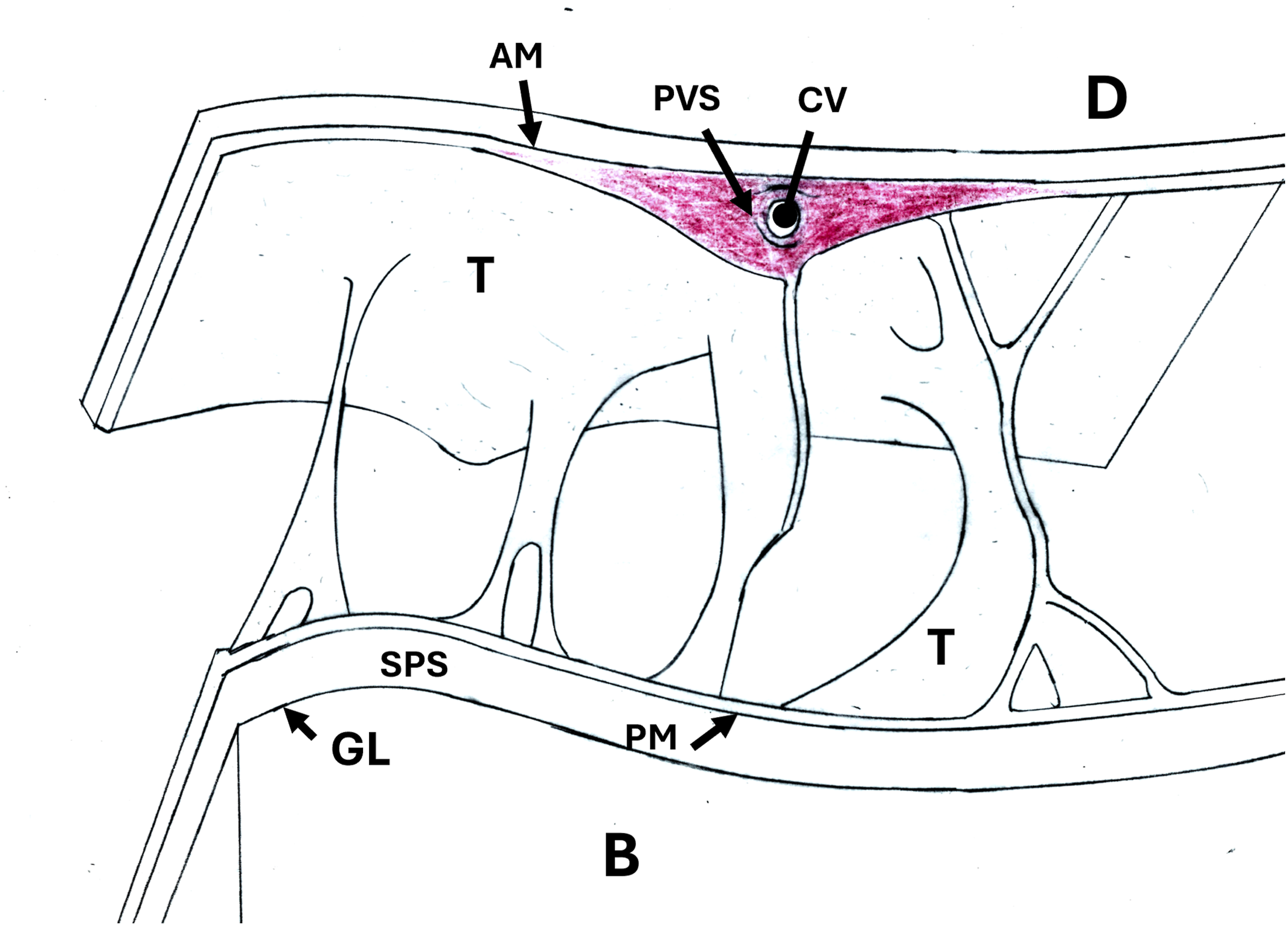
SAS structure: Is there a 4th meningeal layer?
Since laminar SAH does not disperse throughout the SAS, it suggests that the laminar SAH is confined to a space between tissue layers in the SAS. Hence, the existence of a 4th meningeal layer is speculative. Such a 4th meningeal membrane has been observed in studies based on in vivo two-photon microscopy in mice and analysis of immune-labeled human brain sections.41,42 It has been termed SLYM (subarachnoid lymphatic-like membrane); it divides the SAS into compartments.
Conclusion
Infants suffering a venous stroke show CVTs recognized as blood in the SAS. This paper provides evidence that SAH may exist in a laminar form. The existence of the laminar SAH is hypothesized to be located between the AM and a trabecular coat investing the cortical veins. Alternately, the laminar SAH is bounded by a 4th meningeal membrane. The presence of laminar SAH in an infant is evidence of a venous stroke.
Study limitations: The author acknowledges the study has limitations including lack of inter-reader validation, inconsistent imaging protocols and parameters from multiple hospitals, case selection bias, and speculative hypotheses.
Footnotes
Author note
The author is employed at the Harry S. Truman Veterans Administration Hospital, Columbia, MO as an Intermittent Radiologist since 2014.
Declaration of conflicting interests
The author declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Vaslow receives remuneration for consultations and legal testimony in infant alleged abusive head trauma cases.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
