Abstract
Background
Peritumoral edema is an important cause of morbidity and mortality in patients with breast cancer brain metastases (BCBM). The relationship between vasogenic edema and proliferation indices or cell density in BCBM remains poorly understood.
Purpose
To assess the association between tumor volume and peritumoral edema volume and histopathological and immunohistochemical parameters in BCBM.
Materials and Methods
Patients with confirmed BCBM were retrospectively identified. The tumor volume and peritumoral edema volume of each brain metastasis (BM) were semi-automatically calculated in axial T2w and axial T2-fluid attenuated inversion recovery (FLAIR) sequences using the software MIM (Cleveland, Ohio, USA). Edema volume was correlated with histological parameters, including cell count and Ki-67. Sub-analyses were conducted for luminal B, Her2-positive, and tripe negative subgroups.
Results
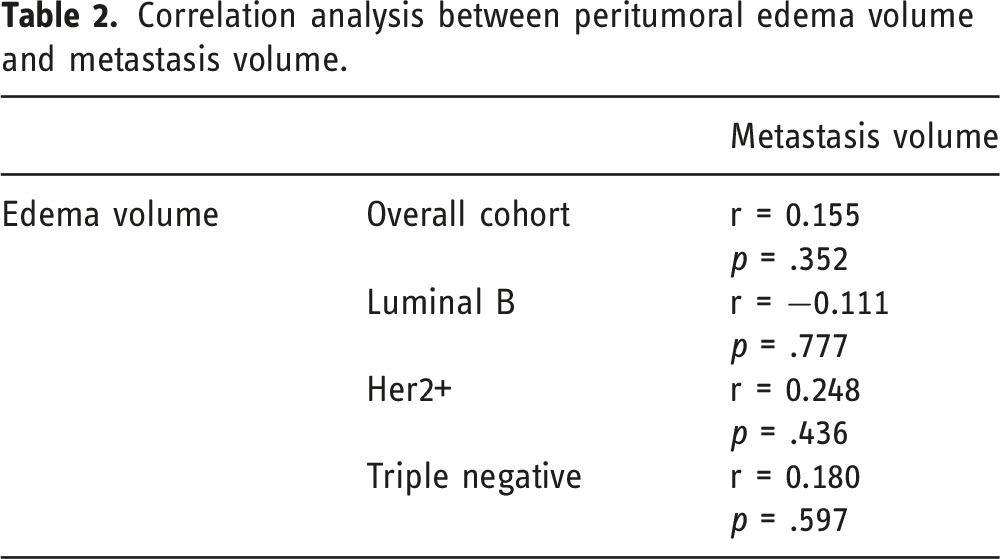
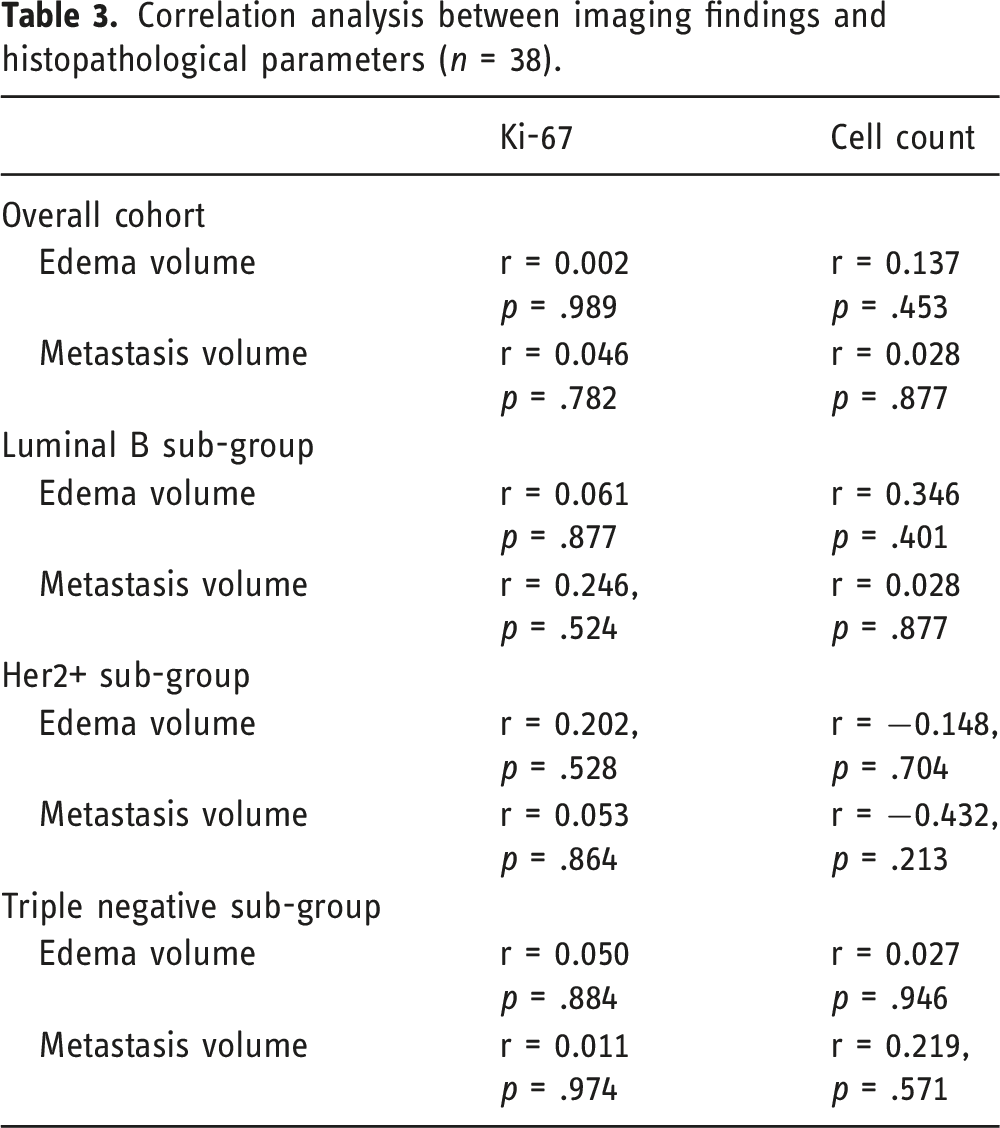
Thirty-eight patients were included in the study. There were 24 patients with a single BM. Mean metastasis volume was 31.40 ± 32.52 mL and mean perifocal edema volume was 72.75 ± 58.85 mL. In the overall cohort, no correlation was found between tumor volume and Ki-67 (r = 0.046, p = .782) or cellularity (r = 0.028, p = .877). Correlation between edema volume and Ki-67 was r = 0.002 (p = .989), correlation with cellularity was r = 0.137 (p = .453). No relevant correlation was identified in any subgroup analysis. There was no relevant correlation between BM volume and edema volume.
Conclusion
In patients with breast cancer brain metastases, we did not find linear associations between edema volumes and immunohistochemical features reflecting proliferation potential. Furthermore, there was no relevant correlation between metastasis volume and edema volume.
Introduction
Metastatic breast cancer ranks among the most prevalent cancer types associated with the occurrence of brain metastases (BM).1,2 BM occur in up to one-third of patients with metastatic breast cancer. 3 Despite advancements in diagnostic and multimodal treatment strategies, the prognosis for individuals with BM remains generally unfavorable, with only a minority surviving beyond 12 months. 4
In BM, vasogenic edema typically manifests due to the tumor-induced disruption of the blood-brain barrier, leading to the infiltration of protein-rich fluids into the interstitial space of the white matter.5,6 In patients with BM, peritumoral edema is an important cause of morbidity and mortality. 7 In clinical settings, magnetic resonance imaging (MRI), particularly T2-weighted sequences utilizing the fluid-inversion recovery technique (FLAIR), offers a highly accurate assessment of peritumoral edema.5,8
Our understanding of the relationship between vasogenic edema and proliferation indices or cell density in breast cancer patients with BM (BCBM) remains limited. Previous studies have proposed a positive correlation between tumor volume and the volume of edema across diverse primary tumors, although findings have been inconsistent.7,9,10 Notably, in cases of solitary metastases, studies suggest an association between the size of peritumoral edema, the degree of angiogenesis, and overall survival. 7 Additionally, peritumoral edema volume has been inversely correlated with survival. 7
The aim of this study was to analyze the relationship between perilesional edema in BCBM and histological and immunohistochemical features treated at our center.
Material and methods
Study cohort
Patients with histologically confirmed BCBM treated at our institution (level one university hospital) between 2010 and 2021 were retrospectively identified from the internal pathology report database. Inclusion criteria comprised patients who underwent pre-surgical cerebral MRI with available T1-weighted (T1w) and fluid-attenuated inversion recovery (T2-FLAIR) sequences and had histopathological specimens. The search identified 44 patients. Six patients were excluded from the study due to missing preoperative imaging (n = 5) and missing histopathological data (n = 1). Consequently, 38 patients constituted the final analysis. For patients with multiple metastases, only lesions with available histological analyses from the institute of neuropathology were considered.
Ethics committee approval for this retrospective study was waived.
Magnetic resonance imaging
Imaging studies were performed either on a 1.5 T or on a 3 T scanner (Achieva 3T, Philips, Best, The Netherlands; Philips, Intera 1.5 T, Philips, Best, The Netherlands; Sola 1.5 T, Siemens Healthineers, Erlangen, Germany) with a standard head coil. Studies were performed in clinical routine for staging or surgical planning purposes.
The imaging protocol included T1 3D sequences (1 mm or 4 mm slice thickness) before and after administration of contrast agent (0.1 mmol/kg Gadobutrol, Gadovist Bayer Schering Pharma, Leverkusen, Germany) and FLAIR sequence. T1- and T2-weighted sequences as well as T2* gradient echo sequences were utilized to exclude hemorrhage or necrosis of the metastasis.
Image analysis
All images were analyzed retrospectively by a radiologist with 6 years of experience in neuroradiologic imaging. All segmentations underwent review by a consultant radiologist with 17 years of experience. The preoperative MRI image data were co-registered and evaluated using the evaluation software MIM software (MIM, Cleveland, Ohio, USA, https://www.mimsoftware.com). The volume of the selected cerebral metastasis and of the peritumoral edema was calculated.
Magnetic resonance imaging analysis was done as follows: At first, total tumor volume was outlined by manually drawing the largest possible region of interest (ROI) around the contours of the metastasis in T1w post contrast. Subsequently, the tumor and peritumoral edema volumes were segmented by drawing ROIs along the perilesional edema contours in FLAIR sequences. These volumes were saved as “metastasis” and “metastasis and edema,” respectively.
The T1w and the T2/FLAIR sequence with the ROIs were then fused, transferred to the T1w sequence, and saved accordingly. The perifocal edema volume was calculated by subtracting the “metastasis” volume from the “metastasis and edema” volume (Figure 1). Representative case of a patient with breast cancer BM. (a) Axial T1w after intravenous contrast administration shows marginal contrast uptake. (b) The correlating axial T2 FLAIR shows a large perifocal edema. (c) Semi-automatic volumetry of tumor (green) and edema volume (red) with (d) maximum intensity projection in the sagittal and coronal plane. The volume of the tumor was 17–98 mL. The volume of the edema was 154.38 mL.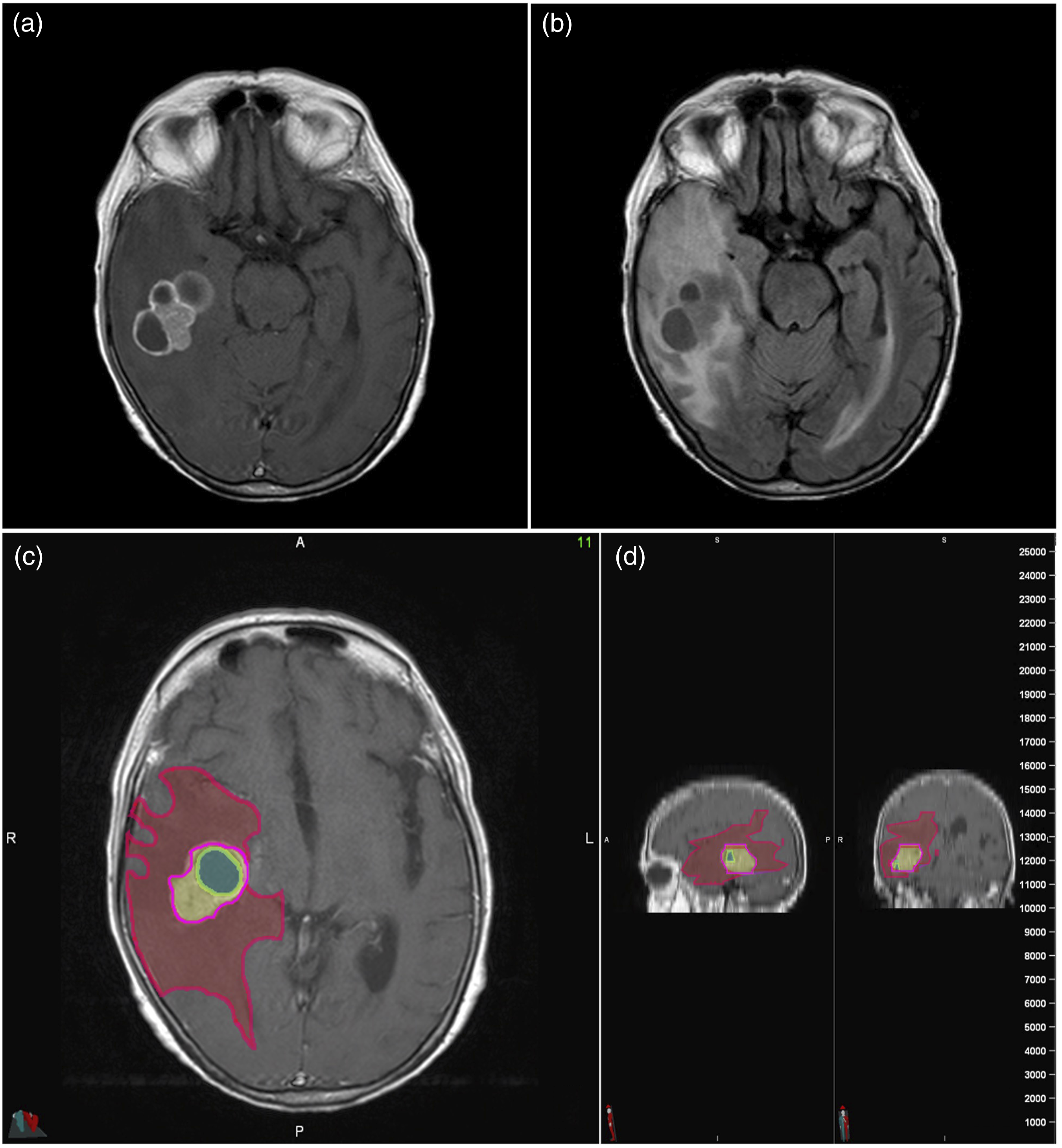
The region of interest (ROI) drawn in each slice was automatically combined with the volume of interest (VOI), incorporating voxel information regarding the metastatic portions and perifocal edema through processing using the MIM software. Subsequently, histogram parameters were automatically calculated from both the metastasis and the perifocal edema.
Histopathological analysis
All histopathologic assessments were conducted by an investigator blinded to the clinical and imaging data.
For the determination of the Ki67 tumor cell proliferation index, tissue sections were immunostained with the monoclonal antibody MIB1. Initially, the sections were scanned at low magnification to identify the area with the highest density of immunolabeled tumor cell nuclei, commonly referred to as the “hot spot” (Figure 2). Within this area, 500 tumor cell nuclei were systematically evaluated. The proportion of labeled nuclei per 500 tumor cell nuclei was determined through manual counting using an eye grid and expressed as a percentage. MIB1-stained specimen of a patient. The proliferating cells can be appreciated. The immunostained tissue section was first scanned at low magnification to identify the area with the highest density of immunolabeled tumor cell nuclei. The proportion of labeled nuclei per 500 tumor cell nuclei was determined by manual counting using an eye grid and expressed as a percentage.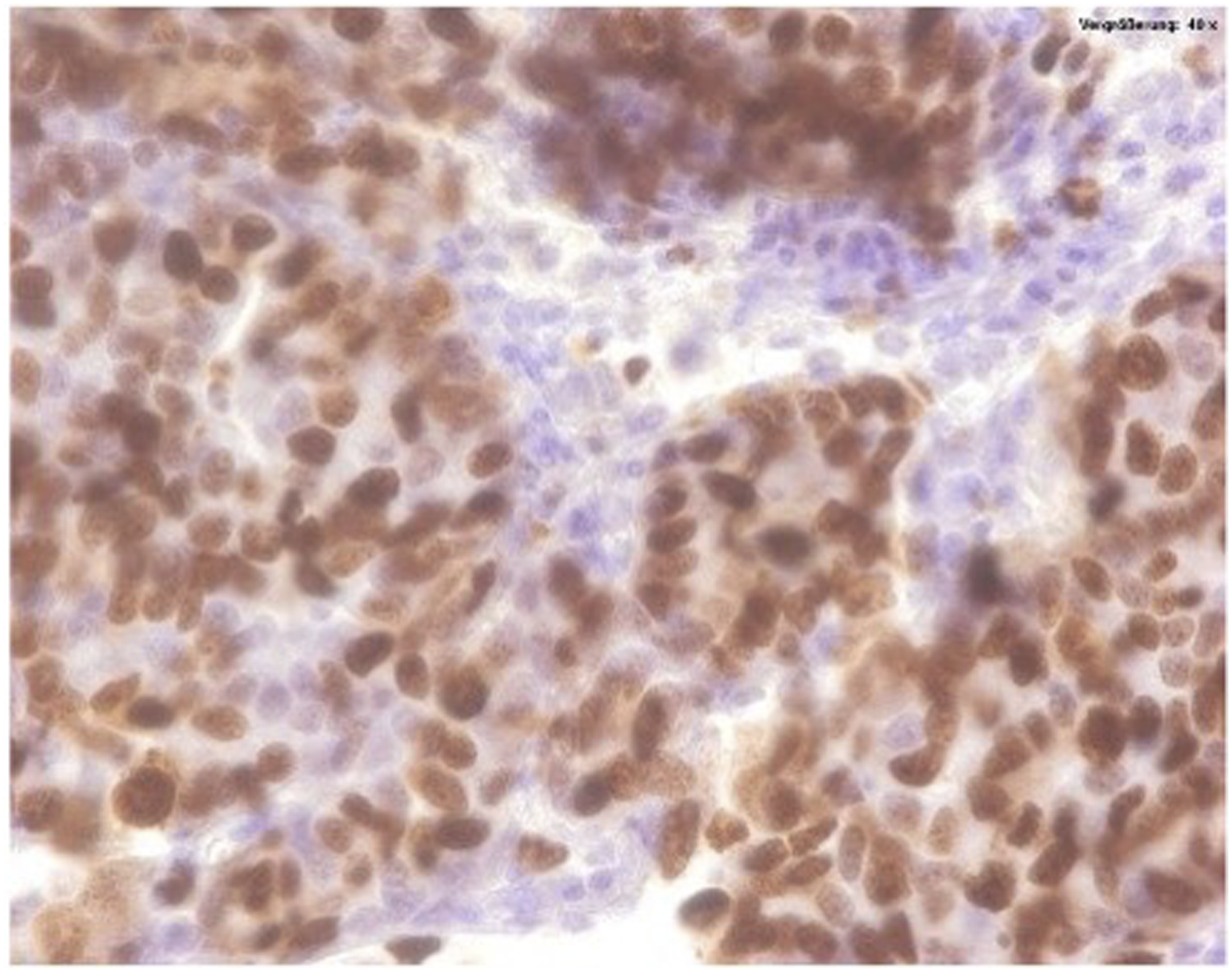
Cell density was calculated for each case on hematoxylin and eosin stained slides as the average number of cells per five main visual fields (0.16 mm 2 per field, ×400)/(40× magnification). All sections were examined using an Olympus BX41 examination microscope with UC90 digital microscope camera (Olympus Microscope Trinocular 4 U-CMAD3) and saved using the cellSens program (version 3.1).
The interpretation of the immunohistochemical reaction results regarding the hormone receptor status was performed according to the recommendations of the ASCO/CAP guidelines. 11
Statistical analysis
SPSS statistical software package was used for statistical analyses (SPSS 28.0, SPSS Inc., Chicago IL, USA). Numerical data are expressed as mean ± standard deviation and categorical data as frequency (%). The theoretically normally distributed continuous variables are expressed as mean ± standard deviation. The theoretically normally distributed continuous variables are conveyed as mean ± standard deviation, and for theoretically non-normally distributed continuous variables, median and interquartile range (IQR) are provided. Categorical data are presented as frequency in percent (%). Normal distribution was tested using the Kolmogorov–Smirnov test (KS test).
Differences in numerical data were presented according to the distribution of continuous variables with a two-tailed T-test/independent t-test. Variables that were not normally distributed were compared between two groups using Mann–Whitney U test. One-way analysis of variance (ANOVA) between three groups was evaluated. Correlation statistics between volume values of BM and perifocal edema and Ki-67 were performed using Spearman correlation analysis. A p-value of <0.05 was considered to indicate statistical significance.
Results
Baseline characteristics (n = 38).

M*, mean; SD, standard deviation.
Correlation analysis between peritumoral edema volume and metastasis volume.
Correlation analysis between imaging findings and histopathological parameters (n = 38).
Discussion
This study explores the correlation between peritumoral vasogenic edema volume and histological and immunohistochemical parameters in BCBM. Our analysis did not reveal a relevant correlation between the volume of peritumoral edema and the proliferation marker Ki-67 or cell density. Furthermore, no relevant correlation was observed between edema volume and BM volume. In contrast to previous studies that assessed peritumoral edema in the axial plane,7,12–14 our approach involved quantitative volumetry of tumors and the surrounding edema. This method is less susceptible to investigator bias and is not as reliant on irregularities in the edema. To the best of our knowledge, this study represents the largest investigation into the relationship between edema and BCBM to date, incorporating immunohistochemical markers.
Up to one third of breast cancer patients ultimately develop symptomatic BM. 15 Individuals with triple-negative tumors exhibit a heightened susceptibility to BM development in comparison to other subtypes. 16 The presence of BMs not only diminishes the overall quality of life but is also associated with increased mortality rates. 17 Neurological manifestations attributed to BMs commonly arise from perilesional edema rather than the tumor itself. The volume of edema is often several times larger than that of the actual tumor. 18 Consequently, the identification of prognostic factors associated with tumor aggressiveness holds significant potential for advancing patient management and treatment strategies.
Numerous histopathological factors exert an influence on BM. The proliferation index Ki-67, reflecting the amount of proliferating cells, is associated with prognosis and therapy response.19–21 Elevated Ki-67 levels in BMs correlate with diminished rates of survival. 21 Berghoff et al. reported an association between tumor infiltrating lymphocytes and perilesional edema in BM. 22
In a study including BM from several primaries, Spanberger et al. found a correlation between smaller peritumoral edema, shorter survival times, and specific characteristics, such as a more invasive growth pattern, lower expression of HIF1a, and reduced angiogenic activity. 7 No relevant correlation with proliferation markers was identified. Consistent with our study, patients underwent surgical resection, although cell density was not subject to examination.
Similarly, a study involving 110 patients with BMs originating from non-small cell lung cancer (NSCLC) revealed an inverse association between the extent of edema and survival. 23 An Italian study, comprising 42 patients with BM from NSCLC, reported that individuals with peritumoral edema measuring less than 10 mm exhibited a superior radiological response to radiosurgery and a reduced risk of new brain lesion onset. 13
Kiyose et al. identified an inverse correlation between the Ki-67 index and peritumoral edema in BM originating from both non-small cell lung cancer (NSCLC) and small cell lung cancer (SCLC). 12 The subset of BM with a high Ki-67 index also exhibited diffusion restriction, as indicated by lower apparent diffusion coefficient (ADC) values. The authors postulated that processes involving vascular remodeling or reduced time for angiogenesis might account for the limited extent of peritumoral edema in rapidly growing BM. The quantification of edema was conducted in the axial plane without volumetric assessment, and the volume of BM was not measured.
In concordance with findings from prior studies,6,7,10 this study did not identify a relationship between perilesional edema and BM size. Conversely, a recent study conducted by Nardone et al. demonstrated a statistically significant relationship between tumor volume and perilesional edema in patients with NSCLC. 24 Similarly, Tran et al. observed a weak correlation between these two parameters in patients with BM originating from melanoma and NSCLC. 18 Additional research is imperative to elucidate potential associations between tumor size and perilesional edema across various metastatic types.
Our study has some limitations. It was of retrospective design with data from a single center. Due to the study design, only resected metastases were analyzed, potentially leading to bias. Larger prospective studies are warranted to illuminate the relationships between perilesional edema and tumor characteristics. Notwithstanding these limitations, our study includes a large cohort of patients with breast cancer BMs. We applied strict inclusion criteria and a volumetric assessment approach of BMs. Two radiologists independently reviewed all imaging scans.
In conclusion, peritumoral vasogenic edema poses a substantial clinical challenge in individuals with brain metastases originating from breast cancer. Our study did not identify a relevant correlation between edema volume and histological or immunohistochemical parameters. Furthermore, no linear association was observed between the volume of edema and the volume of metastases.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
