Abstract
Objective
To investigate the effect of higher cumulative defined daily dose per year (cDDD/y) compared with lower cDDD/y of statin use in the incidence of any joint osteoarthritis (OA).
Design
In this population-based retrospective cohort study, patients who were aged ≥40 years were newly initiated on statin therapy between 2002 and 2011, and had a statin prescription for ≥90 days in the first year of treatment were identified from the 2000 Longitudinal Generation Tracking Database. All patients were separated into groups with higher cDDD/y (>120 cDDD/y) and lower cDDD/y (≤120 cDDD/y; as an active comparator) values. Propensity score matching was performed to balance potential confounders. All recruited patients were followed up for 8 years. Marginal Cox proportional hazard models were used to estimate time-to-event outcomes of OA.
Results
Compared with lower cDDD/y use, higher cDDD/y use did not reduce the risk of any joint OA (adjusted hazard ratio, 1.07; 95% confidence interval, 0.99-1.14). Dose-related analysis did not reveal any dose-dependent association. A series of sensitivity analyses showed similar results. Joint-specific analyses revealed that statin did not reduce the incidence of knee, hand, hip, and weight-bearing (knee or hip) OA.
Conclusions
Higher cDDD/y statin use did not reduce the risk of OA in this Taiwanese nationwide cohort study. The complexity of OA pathogenesis might contribute to the ineffectiveness of statin. Repurposing statin with its anti-inflammation properties might be ineffective for OA development, and balancing the catabolism and anabolism of cartilage might be a major strategy for OA prevention.
Introduction
Osteoarthritis (OA) is the most prevalent whole-joint degenerative disease and involves structural alterations in the articular cartilage, subchondral bone, ligaments, capsule, synovium, and periarticular muscles.1,2 OA has long been considered a “wear and tear” disease that is generated because of biomechanical overload on weight-bearing joints. Current research has revealed that systemic inflammation is involved in the development and progression of OA. 3 The pathophysiological process of OA is complex, and proinflammatory cytokines and matrix metalloproteinases cause joint tissue destruction.1,4 Adipokines secreted by periarticular fat or tissues also contribute to OA pathogenesis. 5 Various factors, including metabolic syndrome, innate immunity, inflammaging, and cell senescence, lead to low-grade systemic inflammation, which can exacerbate joint damage and pain in patients with OA. 3
Conventional treatments for OA temporarily alleviate symptoms. Currently, no regulatory agency–approved disease-modifying osteoarthritis drugs (DMOADs) are available for curing OA or halting its progression.6,7 This lack of DMOADs has led the US Food and Drug Administration to label OA as a “serious disease with an unmet medical need.” 2 Numerous drugs are being developed for targeting the balance of catabolism and anabolism in the cartilage, remodeling the subchondral bone, or alleviating synovial inflammation.2,8 However, because OA spans decades of patients’ life, 9 assessing the long-term efficacy and adverse effects of these drugs in clinical trials is challenging.
Cost-efficient treatment strategies, including the use of bundled payments, have been emphasized in clinical practice. 10 Medications that are cheap, are safe, and exhibit anti-inflammatory pleiotropic properties (e.g., statins)11,12 can be used for drug repurposing. Some preclinical studies have elucidated that statin exerts a protective effect on OA in cellular13 -15 and animal models.16,17 However, observational studies have obtained inconsistent results.18 -28 A systematic review and meta-analysis (SR-MA) of 11 observational studies revealed that statin use did not diminish OA incidence or progression. 29 However, some potential biases, including lack of comparability between study groups, confounding by indication, prevalent user bias, recall bias, and short follow-up period, were observed in each study, which might have biased the results of these studies. Furthermore, the populations of the aforementioned previous studies were distributed in Western countries, and the investigation of the association between statin use and the OA risk in Asian populations is lacking. Because the knees, hips, and hands are the most commonly affected appendicular joints in OA,8,9,30 we conducted a population-based retrospective cohort study in Taiwan to elucidate the effect of higher cumulative defined daily dose per year (cDDD/y) compared with lower cDDD/y of statin use in the incidence of any joint (knees, hips, or hands) OA.
Method
Data Sources
In Taiwan, the National Health Insurance (NHI) program covers more than 99.9% of the population. 31 The Health and Welfare Data Science Center (HWDC) of the Ministry of Health and Welfare collects the claims data of NHI beneficiaries into databases. This study used the 2000 Longitudinal Generation Tracking Database (LGTD2000), a randomly sampled data set containing the data of 2 million NHI beneficiaries. The LGTD2000 includes anonymized data on beneficiaries’ demographic information (e.g., sex and age), outpatient visit or inpatient care, disease diagnoses, and prescription details, and LGTD2000 is considered to be nationally representative of the 23 million inhabitants of Taiwan. 31 The Registry of Beneficiaries data set and Cause of Death Data were also applied in this study for the accurate calculation of age and the acquisition of the death dates of individuals. For privacy protection, the data in the databases of the HWDC are deidentified and encrypted. This study was approved by the Joint Institutional Review Board of Taipei Medical University, Taipei, Taiwan (TMU-JIRB No. 202107064), and the requirement of obtaining patients’ informed consent was waived. The reporting in this study followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guideline.
Study Design and Patients
This study was designed as a population-based retrospective cohort study. Using Anatomical Therapeutic Chemical (ATC) codes, we identified patients who were newly initiated on statins (ATC code C10AA) from LGTD2000. Subsequently, patients who were prescribed statins for at least 90 days in outpatient visits during the first year of treatment between January 1, 2002 and December 31, 2011 were enrolled. The index date was specified as the first prescription date of statin. When identifying an incident case, we adopted a washout period of at least 2 years before the index date. Follow-up was conducted from the index date until the date of OA diagnosis, death, meeting the exclusion criteria, the use of OA-related medications, or the end of 8-year follow-up, whichever occurred first.
Figure 1 illustrates the selection procedure for the study population. Patients aged less than 40 years were excluded. To ensure the enrollment of incident users, patients who were prescribed statins before the index date were excluded. We excluded patients with any OA diagnosis before the index date. Patients diagnosed as having OA in the 3-year exposure time window after the index date were excluded because earlier outcomes might cause the misclassification of prevalent OA as incident OA. 32 To avoid outcome misclassification 33 and unmeasured confounding effects, 20 patients who had joint replacement; those who were diagnosed as having secondary OA, rheumatoid arthritis, gout, ankylosing spondylitis, psoriatic arthritis, Paget disease, human immunodeficiency virus infection/acquired immune deficiency syndrome, substance abuse, and malignant cancer; or those who used OA-related medication (e.g., hyaluronic acid or glucosamine) before the index date and within the exposure time window were excluded. Moreover, patients who were prescribed the statin only once but did not use any statin within the exposure time window and with missing medication records in the follow-up period were excluded.
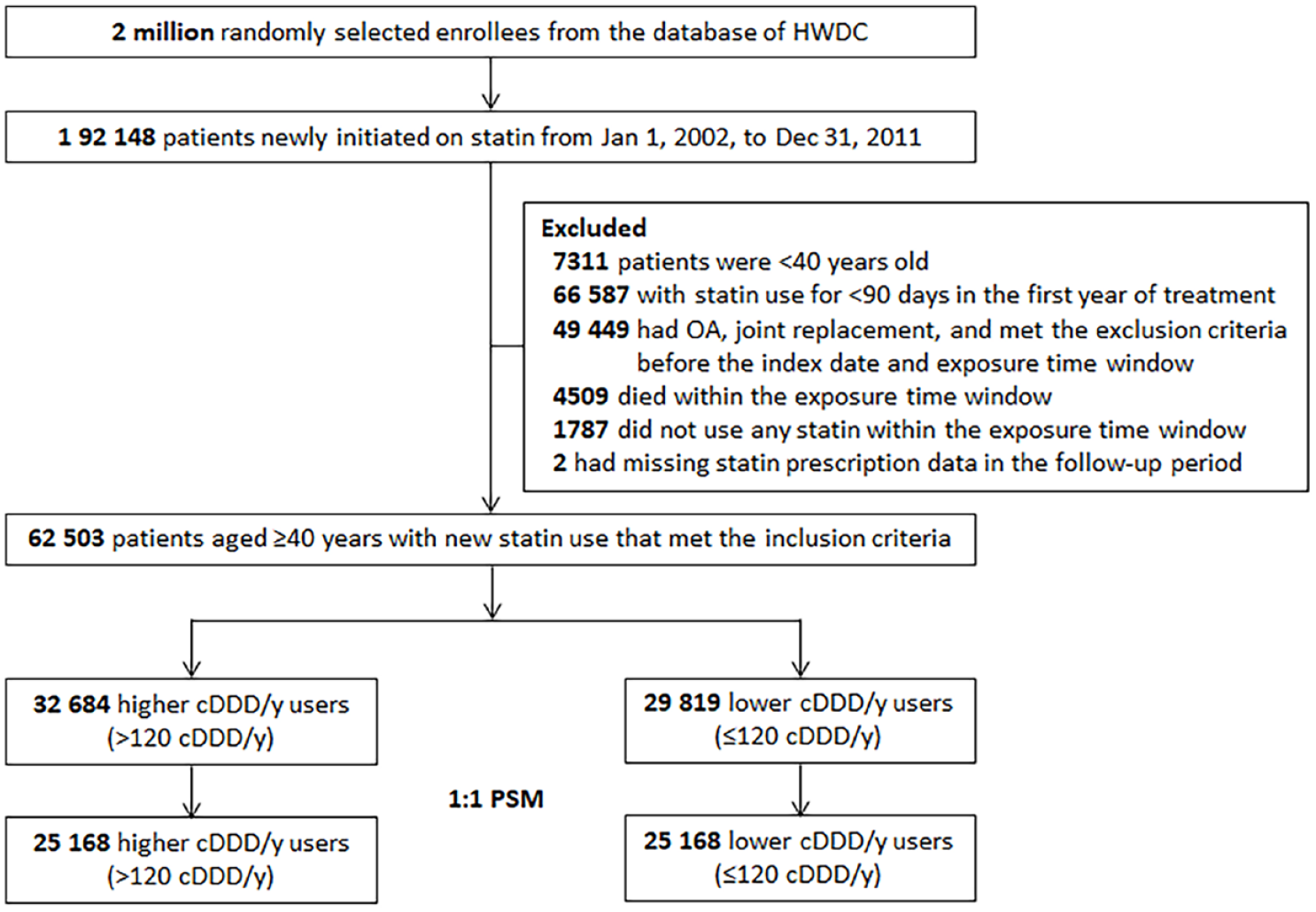
Flowchart for study cohort enrollment. HWDC = Health and Welfare Data Science Center; OA = osteoarthritis; cDDD/y = cumulative defined daily dose per year; PSM = propensity score matching.
To reduce the effect of confounding by indication, we adopted a study design involving the use of an active comparator.34,35 Moreover, the statin users were divided into a lower cDDD/y group and higher cDDD/y group (the comparator group). For increasing comparability, we utilized propensity score matching (PSM) to address the imbalance between the two groups by excluding dissimilar samples. 36 We adopted the greedy method with no replacement and a caliper width of 0.2; in this method, each patient from the higher cDDD/y group was matched with a patient from the lower cDDD/y group in a 1:1 ratio according to variables related to the likelihood of statin treatment (i.e., propensity score [PS]). 37 Furthermore, we exactly matched the two groups by age (10-year incremental), sex, and calendar year of the index date. The reason for matching the calendar year of the index date was to reduce the effect of secular trends. 38
Definition of Exposure
We applied the World Health Organization’s ATC/DDD Index to calculate statin use during the follow-up period. The DDD of each statin initiator was based on the assumed average maintenance dose per day for decreasing cholesterol levels in adults, and this index is similar to the system of statin equivalent doses. Usually, compared with the control group (or lower dose group), the intervention group (or higher dose group) has a longer follow-up time because of their higher statin exposure. In survival analysis, the risk might be underestimated (with a low hazard ratio [HR]) because of the longer person-time of the intervention group (in the denominator) compared with that of the control group. To address this problem, we used cDDD/y (calculated by dividing the summation of cDDD in the follow-up period by the total prescription days and then multiplying the quotient by 365) to classify our statin initiators. Higher cDDD/y users were defined as those who received >120 cDDD/y. By contrast, lower cDDD/y users were defined as those who received ≤120 cDDD/y. Dose-related analysis was also performed. We further divided the higher cDDD/y users into intermediate cDDD/y users (>120 to ≤240 cDDD/y) and the highest cDDD/y users (>240 cDDD/y), and these two groups were compared with the lower cDDD/y users or their matched individuals.
Covariates and Confounders
The covariates selected for adjustment were associated with the development of OA or items considered in the literature.20,27,28 They comprised demographic factors (age and sex), three lifestyle factors, seven comorbidities, six medications, and three other factors. We set a 2-year period (1 year each before and after the index date) as the baseline and collected the aforementioned covariates in this period. Comorbidities included hypertension (HTN), diabetes mellitus (DM), hyperlipidemia, cardiovascular disease (CVD), osteoporosis, chronic obstructive pulmonary disease (COPD), and previous joint injury. To avoid the duplicate identification of comorbidities, we did not include the diagnosis code of HTN in CVD because we had additionally accounted for HTN, an independent risk factor for OA. COPD was also included because it is not only a proxy of smoking but also a surrogate of physical activity. 27 We also considered previous joint injury because post-traumatic OA might be caused by joint damage, including fractures, cartilage damage, and ligamentous or meniscal injuries. 10 This study included comorbidities whose diagnosis codes were reported ≥3 times in the records of outpatient visits or once during hospitalization, except for previous joint injury. In contrast to the medical history of chronic diseases, that of joint injury is not consecutively recorded. To ensure those data regarding this variable were completely collected, we reset the baseline as 1 year after and an unlimited period before the index date, and we examined whether previous joint injury was recorded in this period. Medications included analgesics (including opioid, non-steroidal anti-inflammatory drugs, or corticosteroid), antidepressants, vitamin D, nitrates, other lipid-lowering agents, and hormone replacement therapy.20,27,28 We considered the use of analgesics or antidepressants because these two medications might mask or delay the diagnosis of OA. 28 Medication use was defined as a medication prescription for ≥90 days within the 2-year baseline period. Lifestyle factors (including obesity, alcohol, and tobacco use) are rarely recorded in the NHI database, and the presence of data on these factors indicates the high severity of these factors. Thus, the diagnosis codes of lifestyle factors were included in this study if recorded once or more. Three other factors, namely, Charlson comorbidity index (CCI) score, number of orthopedic visits, and initial statin intensity, were selected considering patients’ frailty, health user behavior, and severity of dyslipidemia, respectively.
Measurement of the Outcome
The main outcome was the incidence of any joint OA, which was defined as knee, hand, or hip OA; these types are the most prevalent types of OA. 9 The diagnosis of OA was identified on the basis of the ICD-9-CM (International Classification of Diseases, Ninth Revision, Clinical Modification) and ICD-10-CM (International Classification of Diseases, Tenth Revision, Clinical Modification) codes in outpatient and inpatient records, and the related codes are listed in Suppl. Table S1. To enhance the robustness of the outcome definition, the diagnosis codes of OA should be reported ≥3 times in the records of outpatient visits or once during hospitalization. If a patient developed more than one type of OA (each type still should meet the outcome definition), the date of the first diagnosis of OA was defined as the outcome date. Information on the date of death is recorded in the Cause of Death Database, which was utilized in this study to ensure that deaths were recorded. To avoid an outcome misclassification, death, the meeting of exclusion criteria, the use of OA-related medication (e.g., hyaluronic acid or glucosamine), or joint replacement developed in the follow-up period were censored.
Sensitivity and Other Analyses
For ensuring the robustness of the outcome estimation, we conducted a series of sensitivity analyses. In one analysis, we changed the follow-up period from 8 to 5 years because patient adherence to statin use might gradually decrease over time 39 ; therefore, the long-term effectiveness of statin might diminish. In another analysis, we changed the follow-up period from 8 to 10 years because OA development spans decades. 9 We limited OA diagnosis to that recorded by orthopedics for setting rigorous diagnosis criteria. For ensuring higher similarity among the study population, we trimmed the individuals whose PS were < 5% and >95% of the score distribution, and we also exactly matched covariates of CVD, nitrate, and CCI score, which were still imbalanced after the first PSM.
To consider patient adherence to statin use, we selected statin users who had CVD and atherosclerotic CVD from the original study population. This approach was adopted because we believe that patients with CVD exhibit higher adherence to statin therapy. However, the confounding by CVD indication might bias the aforementioned approach. Therefore, we additionally selected statin users who potentially received the therapy only for primary prevention (e.g., patients with dyslipidemia, DM, HTN or those with smoking habit).
For further analyzing the effect of statin in different types of OA, we performed a serial analysis of statin use and the risk of knee, hand, hip, and weight-bearing (knee or hip) OA.
Statistical Analysis
The data of continuous and categorical variables are described as mean ± standard deviation and number (%), respectively. After PSM, the standardized mean difference (SMD) was utilized to determine whether the covariates were balanced between two groups. An absolute SMD of >0.10 indicated significant imbalance. 36 The cumulative incidence curves of OA were generated using the Kaplan-Meier method. The log-rank test was applied to examine the differences between two curves. To explore the causal relation between statin use and the time-to-event outcome of OA, we adopted a marginal Cox proportional hazard model to estimate HRs and 95% confidence intervals (CIs). This model considered clustering based on the matching of variables of a sample and produced an unbiased estimation for the treatment effect with a precise standard error. 40 After the crude estimation, the remaining imbalanced variables and all variables were further adjusted in Cox regression models. Proportional hazard assumptions had been examined, and no violation was found. All analyses were performed using SAS (version 9.4, SAS Institute Inc. Cary, NC, USA). A statistically significant difference was defined as a two-tailed P value of < 0.05.
Results
Baseline Characteristics
The flow diagram of population enrollment is displayed in Figure 1 . We identified 62,503 patients who were aged ≥40 years with new statin initiation and the statin prescriptions for at least 90 days in the first year of statin use. These incident users were divided into 29,819 lower cDDD/y users and 32,684 higher cDDD/y users. After PSM, we obtained 25,168 users in each group. Table 1 lists the baseline characteristics of the enrollees. Before PSM, the mean ages of the two groups were similar. However, the proportion of men was higher in the higher cDDD/y group. CCI scores, statin intensity, HTN prevalence, CVD prevalence, and nitrate use were higher in the higher cDDD/y group. After PSM, all covariates except for CVD, nitrate, and CCI score were balanced. In the matched cohort, 1,503 (5.97%) and 1,557 (6.19%) lower and higher cDDD/y users developed OA, respectively. The mean follow-up time for the lower (7.35 ± 1.34 years) and higher (7.26 ± 1.42 years) cDDD/y users was significantly different (P < 0.05). The censoring rate was higher among the higher cDDD/y users (20.06%) than among the lower cDDD/y users (19.66%) (Suppl. Table 2).
Demographic Characteristics of the Full and PSM Cohorts.
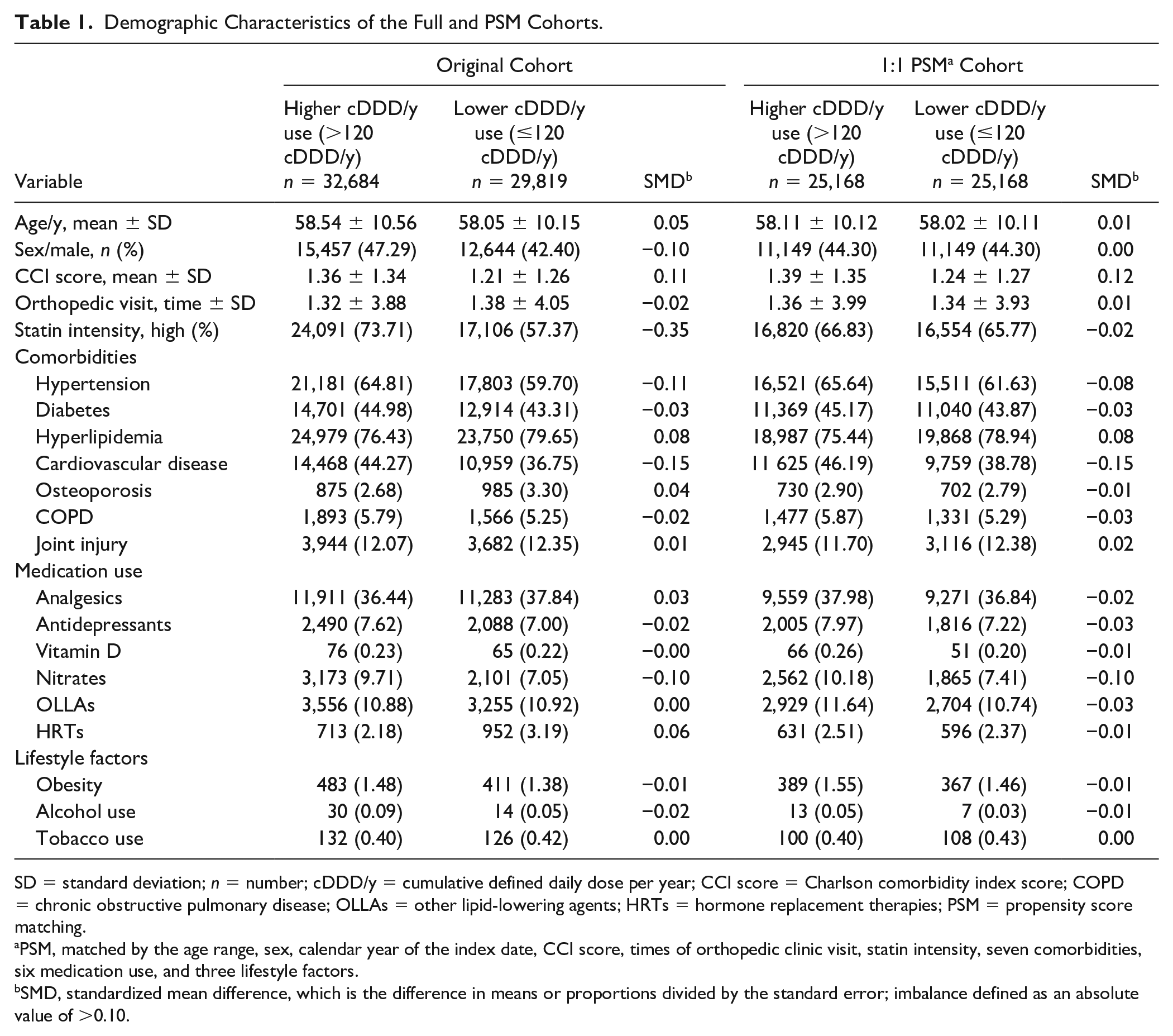
SD = standard deviation; n = number; cDDD/y = cumulative defined daily dose per year; CCI score = Charlson comorbidity index score; COPD = chronic obstructive pulmonary disease; OLLAs = other lipid-lowering agents; HRTs = hormone replacement therapies; PSM = propensity score matching.
PSM, matched by the age range, sex, calendar year of the index date, CCI score, times of orthopedic clinic visit, statin intensity, seven comorbidities, six medication use, and three lifestyle factors.
SMD, standardized mean difference, which is the difference in means or proportions divided by the standard error; imbalance defined as an absolute value of >0.10.
Risk and Dose Analyses
Figure 2 illustrates the Kaplan-Meier curves of the cumulative incidence of OA. The log-rank test revealed no significant difference between the higher and lower cDDD/y groups (P = 0.19). The incidence rate of OA was 8.52 per 1,000 person-years (95% CI, 8.11-8.96) and 8.12 per 1,000 person-years (95% CI, 7.72-8.55) among the higher and lower cDDD/y groups, respectively.
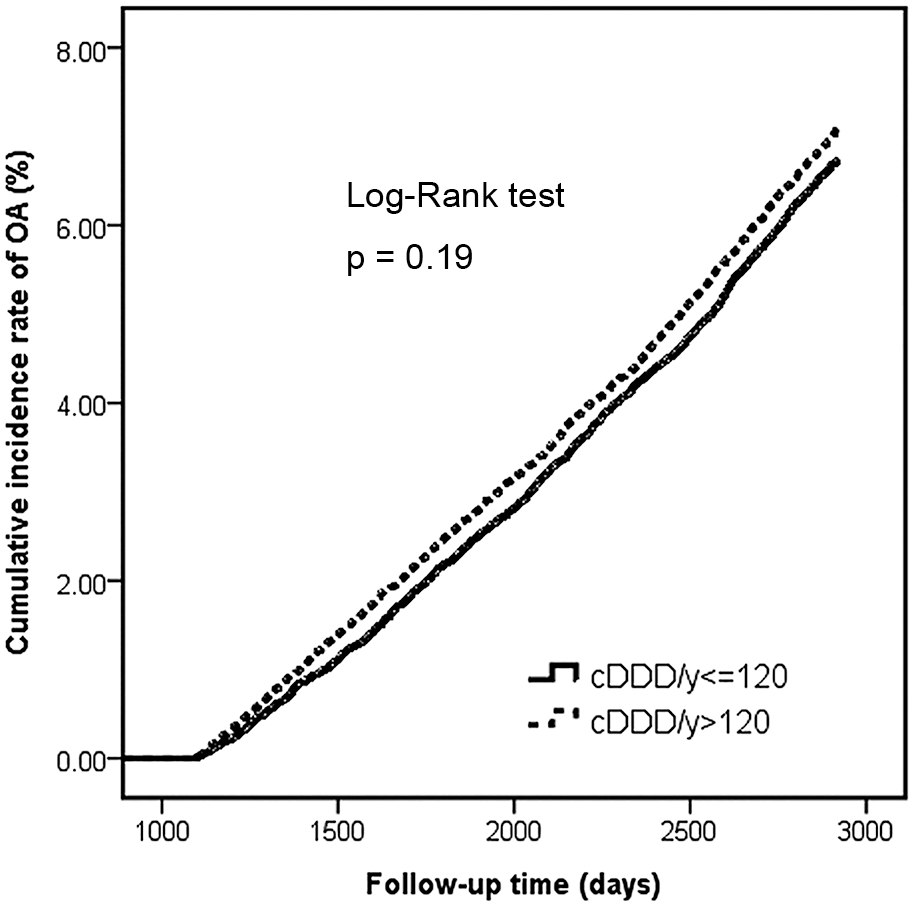
Cumulative incidence of OA in the groups with higher and lower cDDD/y use. OA = osteoarthritis; cDDD/y = cumulative defined daily dose per year.
In the risk analysis, a nonsignificant effect was observed in the crude model (HR, 1.06; 95% CI, 0.99-1.14). Although three remaining imbalanced covariates and all covariates were adjusted in the regression models, the adjusted HRs were similar to the crude HR (Table 2). We examined the dose-related association between statin use and the risk of OA. However, comparison of the highest (>240 cDDD/y) or intermediate (>120 to ≤240 cDDD/y) cDDD/y use with lower cDDD/y use revealed no dose-dependent association. Comparison of these two groups with their matched comparators also revealed the same pattern (Table 2).
Risk and Dose Analyses of Statin Use and Osteoarthritis Development in Crude and Adjusted Models.
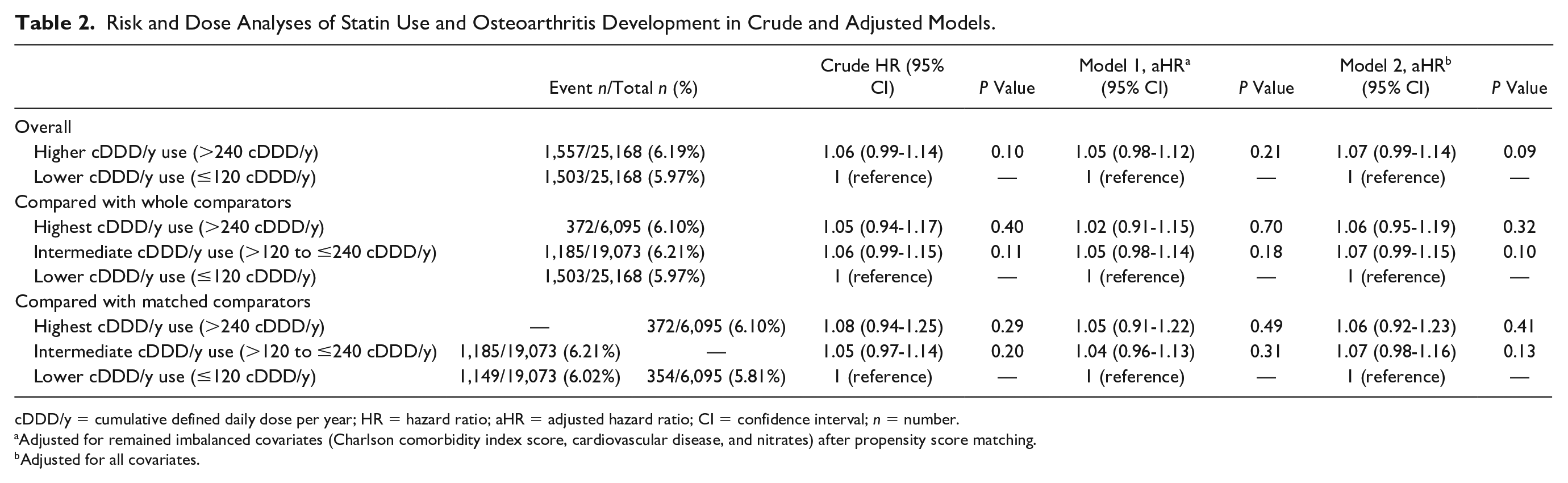
cDDD/y = cumulative defined daily dose per year; HR = hazard ratio; aHR = adjusted hazard ratio; CI = confidence interval; n = number.
Adjusted for remained imbalanced covariates (Charlson comorbidity index score, cardiovascular disease, and nitrates) after propensity score matching.
Adjusted for all covariates.
Sensitivity, CVD-Related, and Joint-Specific Analyses
In sensitivity analyses, we changed the follow-up time from 8 to 5 or 10 years, set more rigorous diagnosis criteria for OA, improved the similarity of the study population by trimming a part of individuals and exactly matched the covariates that were still imbalanced after the first PSM. Consistent results were observed regarding the HRs and 95% CIs in all analyses, including the results for the crude and adjusted models (Table 3). To consider the effects of statin adherence and confounding by CVD indication, three populations with different levels of CVD severity were created. The analyses provided similar results as those in the original risk analysis (Table 3). In joint-specific analyses, compared with lower cDDD/y use, higher cDDD/y use did not demonstrate any protective effect on the occurrence of knee, hand, hip, and weight-bearing OA. Although the crude and adjusted HR estimations for hip OA favored protective effects, the interval estimation did not reveal statistically significant differences (Table 3).
Sensitivity, CVD-Related, and Joint-Specific Analyses of Statin Use and Risk of OA in Crude and Adjusted Models.
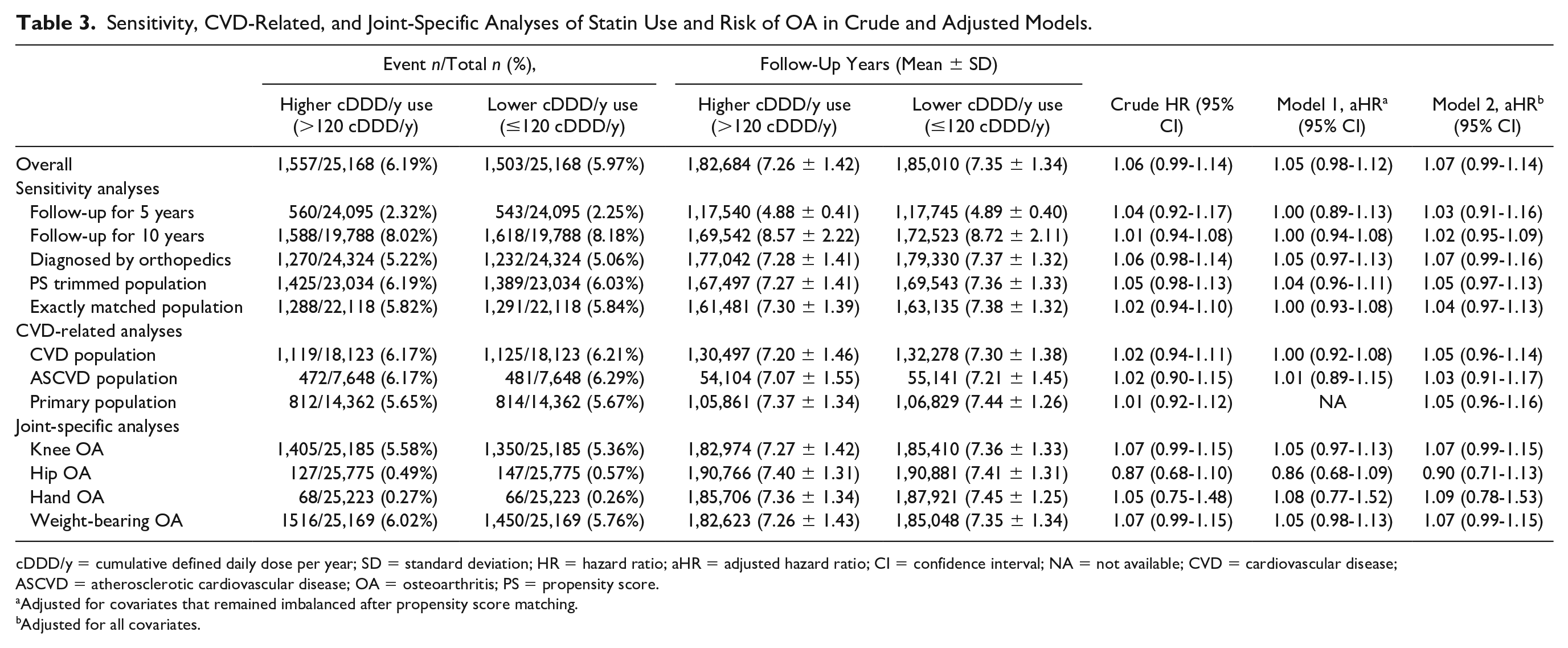
cDDD/y = cumulative defined daily dose per year; SD = standard deviation; HR = hazard ratio; aHR = adjusted hazard ratio; CI = confidence interval; NA = not available; CVD = cardiovascular disease; ASCVD = atherosclerotic cardiovascular disease; OA = osteoarthritis; PS = propensity score.
Adjusted for covariates that remained imbalanced after propensity score matching.
Adjusted for all covariates.
Discussion
To the best of our knowledge, this study is the first to adopt real-world data from an Asian country to examine the association between statin use and the risk of OA. In this nationwide retrospective cohort study, higher cDDD/y use did not reduce the incidence of any joint OA compared with lower cDDD/y use. The same result was obtained in dose analyses. Moreover, in sensitivity and CVD-related analyses, higher cDDD/y use did not exert any protective effect on any joint OA development. Similarly, joint-specific analyses revealed that higher cDDD/y use did not diminish the risk of knee, hip, hand, and weight-bearing OA.
Many studies have indicated that systemic low-grade inflammation contributes to OA development. 3 The components of cartilage fragments, the extracellular matrix, or deposited crystals in the synovial fluid serve as danger-associated molecular patterns, which initiate the innate immune response, activate TLR/MyD88/NF-κb and NLRP3/IL-1β signaling (namely, two-signal pathways), and trigger an inflammatory response. 41 Monosodium urate crystal-induced and cholesterol crystal-induced inflammation are involved in the two-signal pathways. 42 Statins have been demonstrated to target the toll-like receptor (TLR) pathway, inhibit NLRP3 inflammasome assembly, and reduce inflammation in cholesterol crystal-induced atherosclerosis. 43 In our previous study, we found that a higher cumulative dose and the long-term use of statin reduced the risk of gout. 44 Whether statin can prevent OA development by inhibiting the aforementioned pathways is unknown.
An SR-MA of observational studies found that statin use may not be associated with a lower risk of any joint OA (odds ratio, 1.06; 95% CI, 0.97-1.09), with nonsignificant and low heterogeneity being observed. 29 Although the point and interval estimation of this SR-MA is similar to our main result (HR, 1.01; 95% CI, 0.97-1.06), this SR-MA analyzed studies that had diverse designs, employed different diagnosis methods, measured different outcomes, and examined different joints.
The protective effect of statin on OA risk was not observed in our study, which might be attributed to the higher censoring rate and shorter follow-up period among higher cDDD/y users (Suppl. Table 2). However, patients who were diagnosed with diseases similar to OA or had severe diseases in the follow-period were rigorously censored; thus, the biases of outcome misclassification and unmeasured confounding could be avoided.
Another explanation for our result is that the effectiveness of statin for reducing OA risk might be masked by the effect of confounding by indication, which has been demonstrated to lead to treatment ineffectiveness for the intervention group in an observational study. 35 Statin users (or higher dose users) have comorbidities, such as hyperlipidemia, HTN, DM, or CVD, all of which are risk factors for OA. 8 Thus, compared with the control group (or lower dose group), the intervention group might have a higher possibility of OA development. Although we matched the aforementioned comorbidities through PSM and adjusted imbalanced covariates in the regression models, the severity of CVD-related comorbidities between the two study groups might vary considerably. The effects of confounding by CVD indication might substantially mask the effectiveness of statin in our study.
Our result regarding the ineffectiveness of statin treatment might also be explained by the complexity of OA pathogenesis. 7 One review described pharmaceutical therapies that target the pathophysiological processes of OA, including therapies that focus on matrix-degrading proteases, senescent chondrocytes, promoting cartilage repair, limiting bone remodeling, local low-grade inflammation, and Wnt signaling. 2 Clinical trials of anti-inflammatory therapies, including those targeting anti-interleukin (IL)-1, anti-tumor necrosis factor (TNF), anti-IL-6R, and anti-granulocyte macrophage colony-stimulating factor (GM-CSF), did not display favorable outcomes for symptom relief, and only few anti-cytokine therapies improved long-term structural outcomes. 2 Therefore, targeting synovial inflammation is not a suitable strategy for delaying the development of clinically diagnosed OA. Reducing cartilage breakdown (catabolism) and promoting cartilage repair (anabolism) might play a pivotal role in OA management.
Observational studies that have investigated the association between statin use and the risk of OA have provided inconsistent results. Some potential biases were observed in these studies, which might have confounded their results. First, in some studies, the characteristics of the intervention and comparison groups were not matched, and only the imbalanced covariates were adjusted in regression models.18,19,21 -23,25,28 Low comparability between the study groups might confound the estimation of the results. Second, some studies used statin nonusers as the comparator,18 -22,24 -26,28 which might have led to the confounding by indication and exaggerated risk estimation. Third, some studies did not adopt a new-user design,18,19,21,22,24,28 and the prevalent user bias might have caused reverse causation. Fourth, some surveys applied interviews to query whether participants used statins or had certain diseases,18,22,24 -26 which might have caused a recall bias. Finally, the results of few studies might not have external validity because they selected specific populations (e.g., female only) for research,18,22,24 and the follow-up periods for some studies were too short (e.g., 3-4 years).19,24
In our studies, we used PSM and exactly matched age, sex, and the calendar year of the index date to increase comparability and avoid the effects of secular trends. Moreover, we utilized an active comparator design to decrease the effect of confounding by indications. 35 To avoid the immortal time bias and reverse causality induced by prevalent user bias, a new-user design was adopted.34,35 For accurately defining the study population, statin use, covariates, and outcome, we applied rigorous criteria to avoid diagnosis and exposure misclassification. In addition, we excluded and censored patients diagnosed with diseases similar to OA to avoid outcome misclassification. We also accounted for attrition bias by following up each patient for 8 years (10 years in sensitivity analysis), and this follow-up period covered the average span of OA development.
This study has some limitations. The lack of laboratory data, such as the low-density lipoprotein cholesterol level, in the NHI database is a limitation. However, we adopted the intensity of initial statin as a surrogate for the severity of hyperlipidemia. Although unmeasured confounding effects can be reduced by adopting an active comparator and conducting PSM, 35 the influences of these confounding effects on the research results are unknown. Therefore, we also collected the CCI score and COPD data to represent the frailty and physical activity of patients at the baseline. If patients purchased over-the-counter analgesics and nutritional supplements (e.g., glucosamine) or underwent self-paid injection (e.g., platelet-rich plasma or hyaluronic acid), the effects of these interventions could not be accounted for because data on these interventions are not recorded in the NHI database. Although data on intraarticular (IA) corticosteroids are recorded in this claims database, the injection site is not specified. Information on lifestyle factors, such as occupation (e.g., heavy work) or participation in high-impact sports (e.g., football), is lacking in this database; however, we adjusted for other lifestyle factors (e.g., obesity, alcohol, and tobacco use), which are associated with OA development, in analyses. Genetic variance and family history might contribute to OA development; however, related information is not recorded in this database. For patients with statin use but with low adherence or those with OA but who did not seek health-care services might also violate the study results. Finally, the generalizability of this study is limited because a part of patients was excluded.
In conclusion, this nationwide retrospective cohort study revealed that higher cDDD/y statin use did not reduce the risk of OA. Because of the complexity of OA pathogenesis, repurposing statin with its anti-inflammatory property for OA development might be ineffective. Instead, balancing the catabolism and anabolism of cartilage might be a suitable strategy for preventing the risk of OA. Continual and adherent use is strongly recommended for patients who are prescribed statins because restoring cholesterol to normal levels might reduce the risk of OA. Future investigation of the effect of IA statin is recommended because local treatment might exert fewer systemic side effects and more direct effects on joints. 45
Supplemental Material
sj-docx-1-car-10.1177_19476035241247700 – Supplemental material for Association Between Statin Use and the Incidence of Clinically Diagnosed Osteoarthritis: A Nationwide Retrospective Cohort Study in Taiwan
Supplemental material, sj-docx-1-car-10.1177_19476035241247700 for Association Between Statin Use and the Incidence of Clinically Diagnosed Osteoarthritis: A Nationwide Retrospective Cohort Study in Taiwan by Guan-Ling Lin, Joseph Jordan Keller and Li-Hsuan Wang in CARTILAGE
Footnotes
Acknowledgements and Funding
The authors acknowledge the research grant support provided by Taipei Medical University Hospital and thank Wallace Academic Editing for their professional academic editing. The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was supported by a research grant from Taipei Medical University Hospital (grant no. 112TMUH-P-05).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of Data and Materials
According to the regulation of Health and Welfare Data Science Center (HWDC), data sets or raw data generated or analyzed from National Health Insurance Research Database (NHIRD) could not be shared in public to protect patients’ confidentiality, which is used under license. If request of details is needed, formal requisition can be sent to the HWDC.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
