Abstract
Objective
Integrin α10β1-selected mesenchymal stem cells (integrin α10-MSCs) have previously shown potential in treating cartilage damage and osteoarthritis (OA) in vitro and in animal models in vivo. The aim of this study was to further investigate disease-modifying effects of integrin α10-MSCs.
Design
OA was surgically induced in 17 horses. Eighteen days after surgery, horses received 2 × 107 integrin α10-MSCs intra-articularly or were left untreated. Lameness and response to carpal flexion was assessed weekly along with synovial fluid (SF) analysis. On day 52 after treatment, horses were euthanized, and carpi were evaluated by computed tomography (CT), MRI, histology, and for macroscopic pathology and integrin α10-MSCs were traced in the joint tissues.
Results
Lameness and response to carpal flexion significantly improved over time following integrin α10-MSC treatment. Treated horses had milder macroscopic cartilage pathology and lower cartilage histology scores than the untreated group. Prostaglandin E2 and interleukin-10 increased in the SF after integrin α10-MSC injection. Integrin α10-MSCs were found in SF from treated horses up to day 17 after treatment, and in the articular cartilage and subchondral bone from 5 of 8 treated horses after euthanasia at 52 days after treatment. The integrin α10-MSC injection did not cause joint flare.
Conclusion
This study demonstrates that intra-articular (IA) injection of integrin α10-MSCs appears to be safe, alleviate pathological changes in the joint, and improve joint function in an equine post-traumatic osteoarthritis (PTOA) model. The results suggest that integrin α10-MSCs hold promise as a disease-modifying osteoarthritis drug (DMOAD).
Keywords
Introduction
Osteoarthritis (OA) is the most common joint disease and a major contributor to years lived with disability. 1 OA is characterized by inflammation of the synovium, progressive cartilage destruction, osteophyte formation, and subchondral bone sclerosis, resulting in joint-related pain.2,3 Post-traumatic osteoarthritis (PTOA) develops as a result of acute trauma to a joint. Not only untreated or unsuccessfully treated articular cartilage damage lead to PTOA, but also anterior cruciate ligament and meniscus injury often do due to trauma-related inflammation.4-8 Therefore, treatment should dampen inflammation, alleviate deterioration of the joint, and preferably aid cartilage regeneration. Surgical and injectable treatments have not proven effective in preventing PTOA.4,5,9,10
Mesenchymal stem cells (MSCs) are attractive as treatment for OA due to their immunomodulatory and regenerative properties. Studies on intra-articular (IA) MSC treatment for OA have shown beneficial, but varying effects, possibly due to heterogeneity in MSC preparations.11-18 To improve homogeneity and consistency, MSCs can be selected for beneficial attributes, such as a specific cell surface marker.15,19,20 Selection based on the cell surface marker integrin α10β1 resulted in consistent MSC preparations.21,22 Moreover, MSCs selected for the expression of integrin α10β1 (integrin α10-MSCs) showed a superior ability to adhere to damaged cartilage and subchondral bone compared with unselected MSCs, 22 likely facilitated by the binding of the collagen receptor integrin α10β1 to exposed collagen. In addition, integrin α10-MSCs also showed a superior chondrogenic differentiation ability and higher secretion of the immunomodulatory factor PGE2 in vitro compared with unselected MSCs. 22 Furthermore, treatment with integrin α10-MSCs allayed the progression of OA in an equine experimental model of PTOA resulting in significantly less cartilage fibrillation and less bone sclerosis compared with untreated OA joints. 23 The horse is a well-recognized model for human PTOA because of similar cartilage thickness and collagen composition as well as a natural tendency to develop OA. 24 Reliable equine PTOA models have been used to investigate the pathophysiology and effects of injectable treatments on PTOA development with great translational application for human PTOA.25,26
The aim of the present study was to further investigate the therapeutic effect, in terms of both joint function and tissue pathology, and changes in the inflammatory environment of the joint after IA injection of integrin α10-MSCs in horses with experimental PTOA. A secondary aim was to trace the integrin α10-MSCs in the joint.
Materials and Methods
Study Design
The study was approved by the Danish Animal Experiments Inspectorate (approval no. 2020-15-0201-00602 and 2017-15-0201-01314) as well as the local Ethical and Administrative body of the Department of Veterinary Clinical Sciences, University of Copenhagen (# 2020-016 and #2017-010). The horses were all purchased from a Standardbred sale and training stable.
PTOA was surgically induced using the carpal osteochondral fragment model (day 18).25,26 This model was chosen as it is a reliable PTOA model that allows for clinical examination, synovial fluid (SF) analysis and joint pathology determination. Treadmill exercise was initiated on day 4. Blood and SF were sampled weekly plus before MSC treatment (

Study overview of the post-traumatic osteoarthritis-model and data collection. Inclusion examinations were conducted, and horses were acclimatized to treadmill exercise 2 weeks prior to study start. Post-traumatic osteoarthritis was surgically induced on day 18. The horses were stall rested for 2 weeks before treadmill exercise was initiated. Clinical parameters (lameness, skin temperature, and joint circumference) and synovial fluid was collected weekly + on day 0 before integrin α10-MSC treatment. Radiographs were taken at inclusion and before study termination on day 52. MRI, CT, macroscopic examination, and histology were conducted postmortem. MSC = mesenchymal stem cell; CT = computed tomography; OA = osteoarthritis.
To evaluate effect of treatment, integrin α10-MSC treated horses were compared with an untreated group of horses from a previous study in which the exact same PTOA model was used, and data and samples had been collected using identical methods. Consequently, the study could not be randomized, and blinded observations were not possible for some parameters (lameness, pain behavior, carpal skin temperature, and joint circumference), but observers handling the integrin α10-MSC treated horses were blinded to the results from the group of untreated horses. Blinding was achieved for all postmortem analyses, including histology, macroscopic pathology, computed tomography (CT), MRI, and SF analyses.
Animals
Seventeen healthy Standardbred trotters aged 3-7 years, weight range 396-535 kg, 15 female and 2 castrated male horses were included in the study. There were 8 horses in the treated group, all female, and 9 horses in the untreated group. The horses were free from visual lameness, had no response to carpal flexion, did not have palpable joint effusion, and presented no abnormalities on radiographic examination. The horses were habituated to treadmill exercise before commencing the study.
Isolation and Selection of α10-MSCs
Equine MSCs were isolated from adipose tissue from a 7-year-old healthy male horse, culture expanded until P3, and MSCs with high expression of integrin α10β1 were selected as previously described (for details, see Supplementary File 1).22,23,27 Only integrin α10β1-selected MSCs (integrin α10-MSCs) were used in this study (Suppl. Fig. S1). The MSCs also expressed the standard equine stem cell markers CD44, CD90, and CD105, while being negative for CD45 and MHCII, and showed trilineage differentiation capacity. 22
OA Model
On study day 18, PTOA was induced by creating an osteochondral fragment in the third facet of the radial carpal bone (RCB) with arthroscopic surgery (
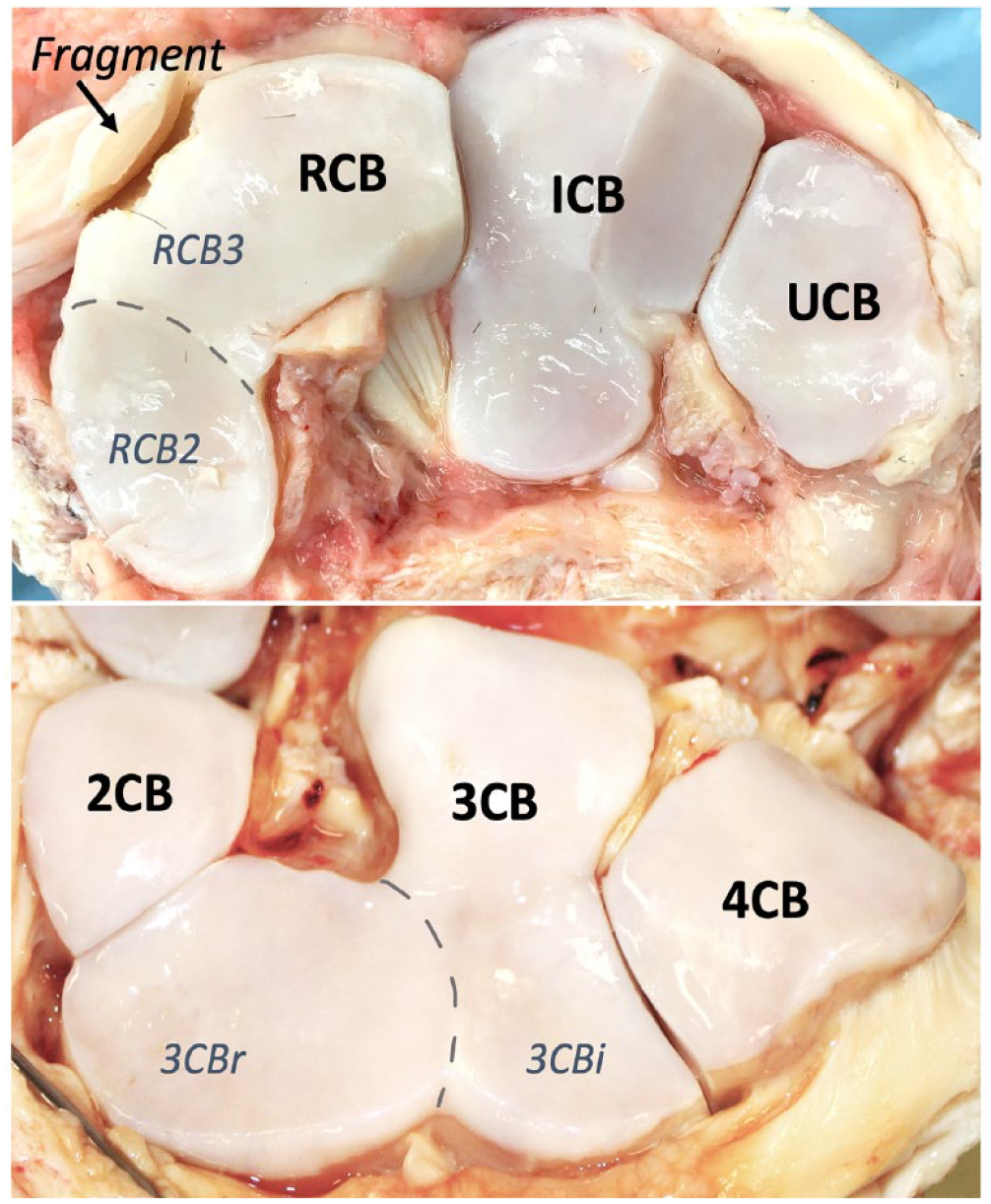
Overview image of the anatomy of the middle carpal joint with the proximal (top) and distal (bottom) rows of carpal bones. A label indicates the surgically created osteochondral fragment in the radial carpal bone. The image is taken of a cadaveric limb used for practice of the surgical procedure, not from a horse included in the study. ICB = intermediate carpal bone; RCB = radial carpal bone; RCB2 = second facet of the radial carpal bone; RCB3 = third facet of the radial carpal bone; UCB = ulnar carpal bone; 2CB = second carpal bone; 3CB = third carpal bone; 3CBi = intermediate facet of the third carpal bone; 3CBr = radial facet of the third carpal bone; 4CB = fourth carpal bone.
MSC Treatment
On study day 0, 2 × 107 equine allogeneic integrin α10-MSCs in 4 ml cryopreservation medium (Cryostor, BioLife Solutions) were administered by IA injection in the carpal joint (for details, see Supplementary File 1).
Clinical Assessment
On sampling days (
SF Analysis
SF was collected on sampling days (
Postmortem Assessment and Sampling
On study day 53 or 54, the horses were euthanized with an overdose of pentobarbital (Euthasol, Dechra).
Diagnostic imaging
Immediately after euthanasia, OA joints were subjected to MRI and CT (see parameters in Supplementary Tables S1 and S2 , Supplementary File 2). Images were evaluated by a veterinary imaging specialist (American College of Veterinary Radiology [ACVR] diplomate [JFG]) using a semi-quantitative scoring system adapted for use with a lower field strength (Supplementary Table S3, Supplementary File 2). 30 Images were analyzed blinded using Osirix MD (v.10.0.4, Pixmeo SARL).
Macroscopic pathology and histology
After diagnostic imaging, middle carpal joints were photographed to assess macroscopic pathology using a scoring system.
31
Synovial membrane (SM) samples were collected using sharp scissors. Osteochondral wedge sections (5 × 15 × 20 mm) were cut using an oscillating saw. Samples were collected from the RCB at the fragment site, the radial facet of the 3CB (3CBr), the 3CBi, and the ICB (
MSC Tracking by Polymerase Chain Reaction of the Y-Chromosome SRY-Gene
Polymerase chain reaction (PCR) analysis was used to detect the presence (not quantifiable) of an Y-chromosome-specific target gene, sex-determining region of the Y-chromosome (SRY), of the male is integrin α10-MSCs. 33 In the treated group, cell pellets from all SF samples, and SM, cartilage from the 3CBr, 2CB, the ICB, and from the fragment site in the RCB and subchondral bone at the healing fragment site were analyzed (for details, see Supplementary File 1).
Statistical Analyses
For repeated measurement of ordinal values, a binomial logistic regression model was used with 2 categories in each (lameness: grade 0-1 vs. grade 2-4; flexion test: grade 0 vs. ≥ 1) to estimate the odds ratio (OR) of being in one or the other category, including 95% confidence interval (95% CI).
A Shapiro-Wilk’s test, histograms, and qq-plots were used to assess normality of data. A Student’s t test was used for pairwise comparison of parametric data and Wilcoxon exact rank sum test to assess non-parametric or ordinal data. Multiple Mann-Whitney tests were used to analyze differences in for cytokine/chemokine levels.
A 5% level of significance was chosen, and raw P values are shown. All results with a P < 0.07 are mentioned in the “Results” section.
Results
Animals
There was no difference in age and weight of the horses between the groups. Two horses from the treatment group and one from the untreated group had a coronal bi-articular fracture of the radiocarpal bone after OA induction. These 3 horses were excluded from clinical assessments and postmortem analyses but included for SF analysis. The horse from the untreated group was euthanized on day 31, due to lameness at the walk.
Treatment with Integrin α10-MSCs Reduced Lameness and Response to Carpal Flexion
Lameness did not differ between groups prior to OA induction and prior to MSC treatment. The number of horses with moderate-severe lameness decreased over time after MSC treatment, resulting in more horses with no or minimal lameness in the treated compared with the untreated group: the OR of having AAEP grade 2 or higher decreased over time in the treated group (OR = 0.949/day; CI = 0.945-0.953, P = 0.02, median day 52 = 1.0), whereas the OR was unchanged over time in the untreated group (OR = 1.005/day; CI = 0.995-1.009; P = 0.051, median day 52 = 2.5) (
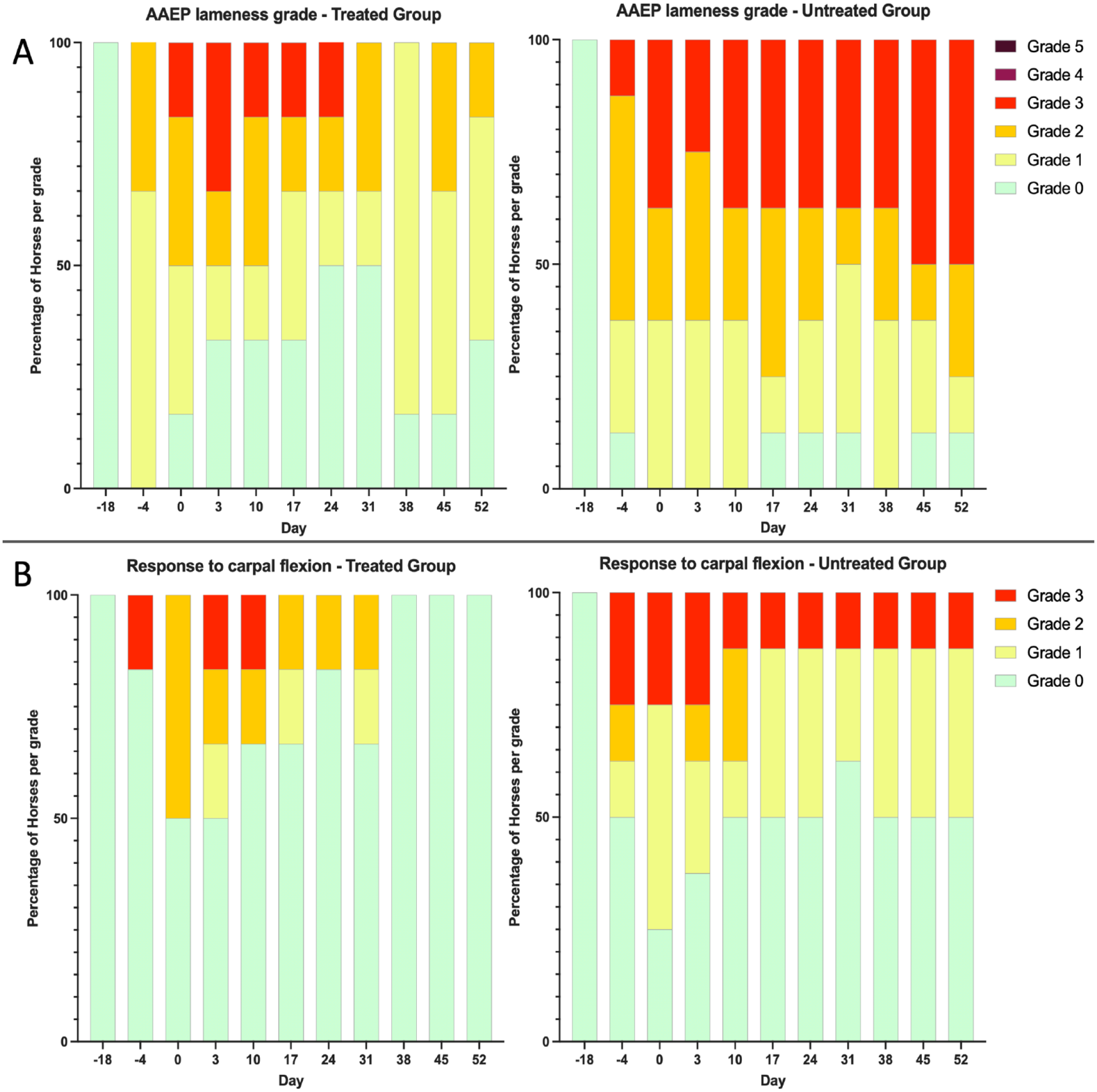
The development of lameness (AAEP score) (
There was no pain response to carpal flexion before OA induction and no difference between groups prior to MSC treatment. After treatment, there was less pain response to carpal flexion in the treated group compared with the untreated group: the odds of having a positive flexion test (score 1 or higher) decreased over time compared with day 0 in both the treated and untreated groups, but to a larger extent in the treated group (OR = 0.87/day; CI = 0.86-0.88, P = 0.004), compared with the untreated group (OR = 0.97/day; CI = 0.96-0.98; P = 0.03) (
No adverse effects were observed after MSC injection:, there was no increase in carpal skin temperature or carpal circumference for the first 3 days after injection (Supplementary File 3, Suppl. Fig. S3).
Integrin α10-MSCs Promoted Secretion of Anti-Inflammatory and Immunomodulatory Factors
PGE2 concentrations in the SF were increased on day 4 compared with day 18 in all horses (P = 0.0008), as a response to OA induction (
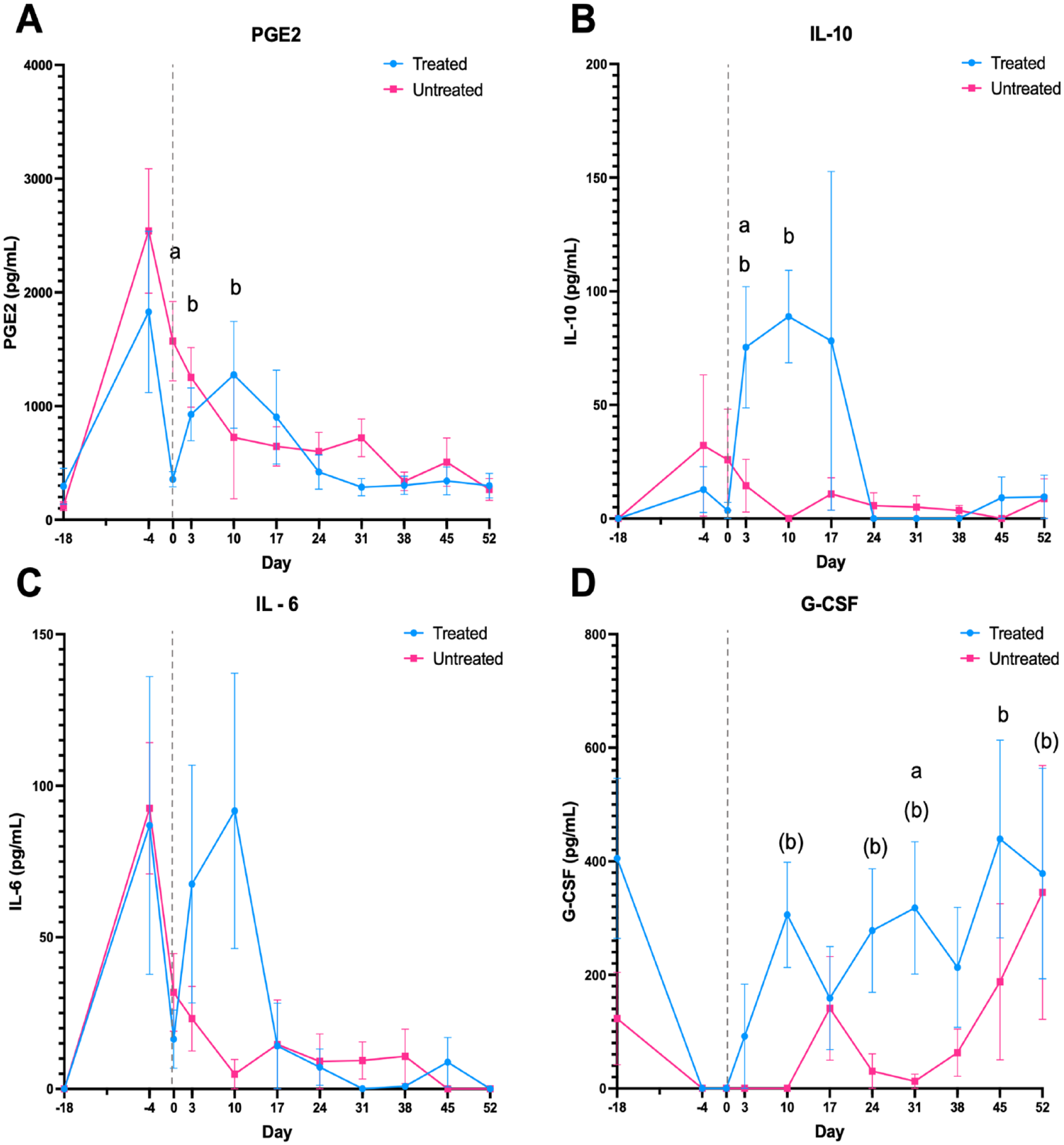
Concentrations of PGE2 (
Concentrations of interleukin (IL)-10 in the SF were higher in the treated group compared with the untreated group on day 3 (P = 0.036) and concentrations were increased in the treated group on days 3 (P = 0.047) and 10 (P = 0.016) compared with day 0 (
IL-6 followed the same pattern as PGE2, with higher levels after OA induction and a second peak in the treatment group at days 3 and 10, only IL-6 levels were not significantly increased (
The granulocyte colony-stimulating factor (G-CSF) concentration decreased in both groups on day 4 (P = 0.01) and day 0 (P = 0.03) compared with baseline (day 18), and concentrations subsequently increased over time in both groups throughout the study period. After treatment, concentrations of G-CSF were numerically higher at all timepoints in the treatment group compared with the untreated group with a significant difference on day 31 (P = 0.026). Compared with day 0, the concentration was significantly increased on day 45 (P = 0.031) in the treated group (
The results of the other cytokines and chemokines are described in supplementary File 1 and Suppl. Fig. S2, although most were below detection level at all sample points.
Integrin α10-MSC Treated Horses Showed Less Macroscopic Cartilage Pathology and a Lower Histologic Pathology Score in Postmortem Evaluations
Imaging
CT and MRI analyses revealed that both groups had cartilage lesions, osteophytes, and subchondral sclerosis. The size of the surgically created osteochondral fragment varied between 4.97 and 158.60 mm2, without difference between groups. There was no difference in overall OA score between groups (P = 0.26) or in any of the individual parameters: osteophytes (P = 0.79), sclerosis (P = 0.43), or cartilage erosions (P = 0.83). Furthermore, there was no difference between groups for synovial effusion (P = 0.54) or synovial proliferation (P = 0.63). There was a tendency toward improved fragment healing in the integrin α10-MSC treated group compared with the untreated group (P = 0.07).
Macroscopic evaluation
Macroscopic pathology scoring showed generalized cartilage erosions on day 52 in both groups (
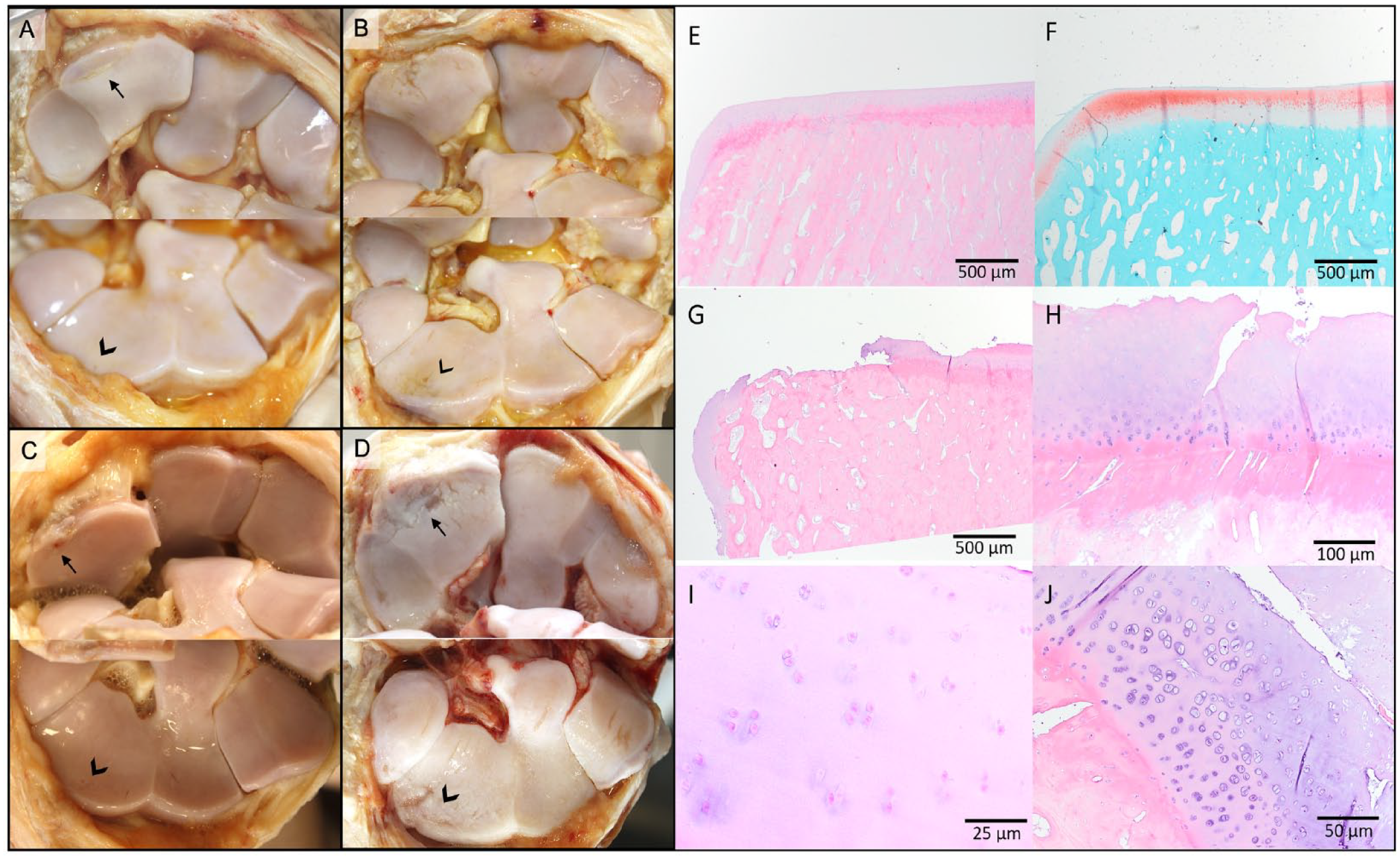
Photographs of the middle carpal joints (
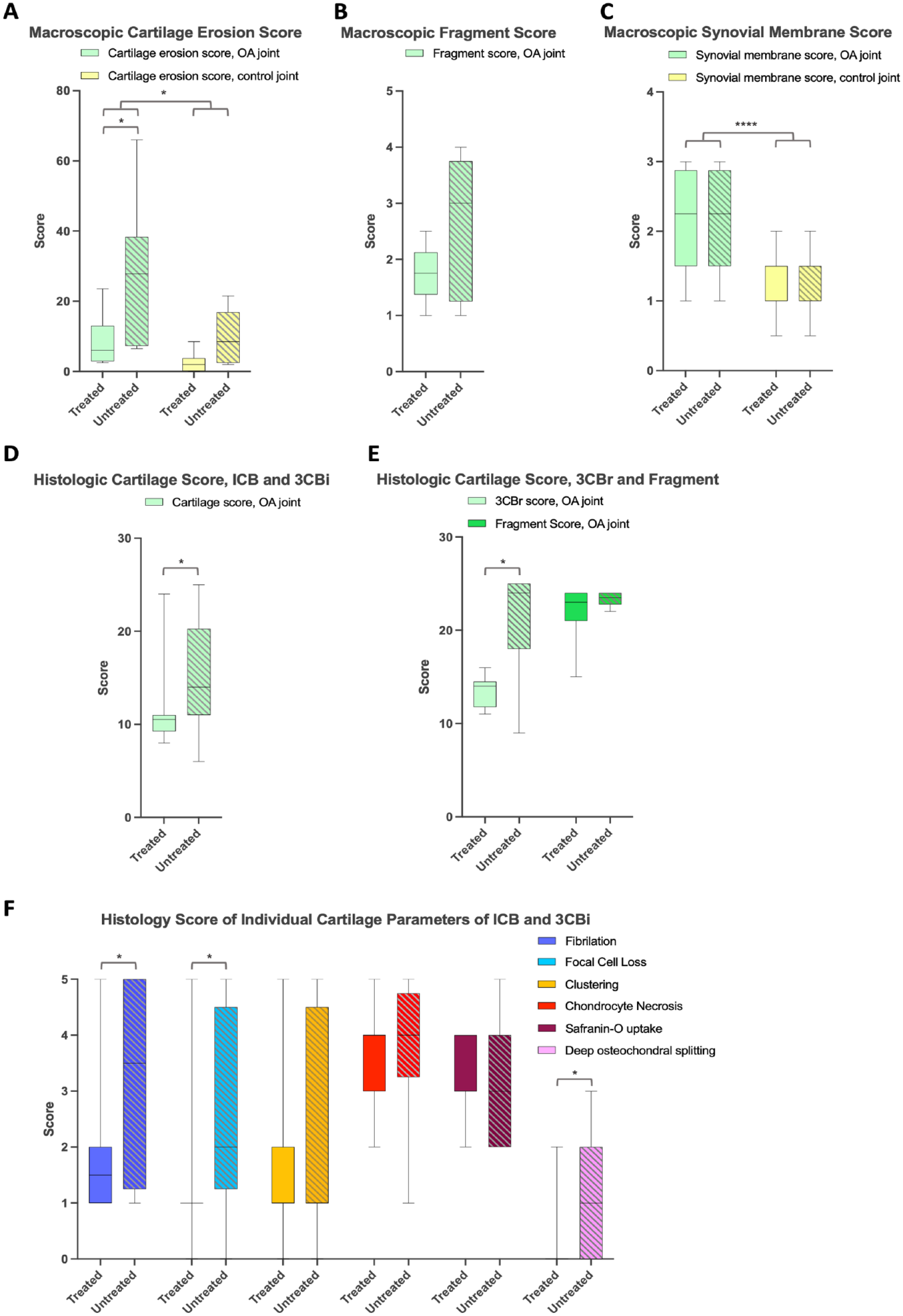
Macroscopic and histologic scoring was performed on the middle carpal joints of horses 70 days after experimentally induced post-traumatic osteoarthritis, either or not treated with integrin α10-MSCs 18 days after induction. Macroscopic cartilage erosions were scored on the carpal bones (
Macroscopic Pathology and Histopathology Score.
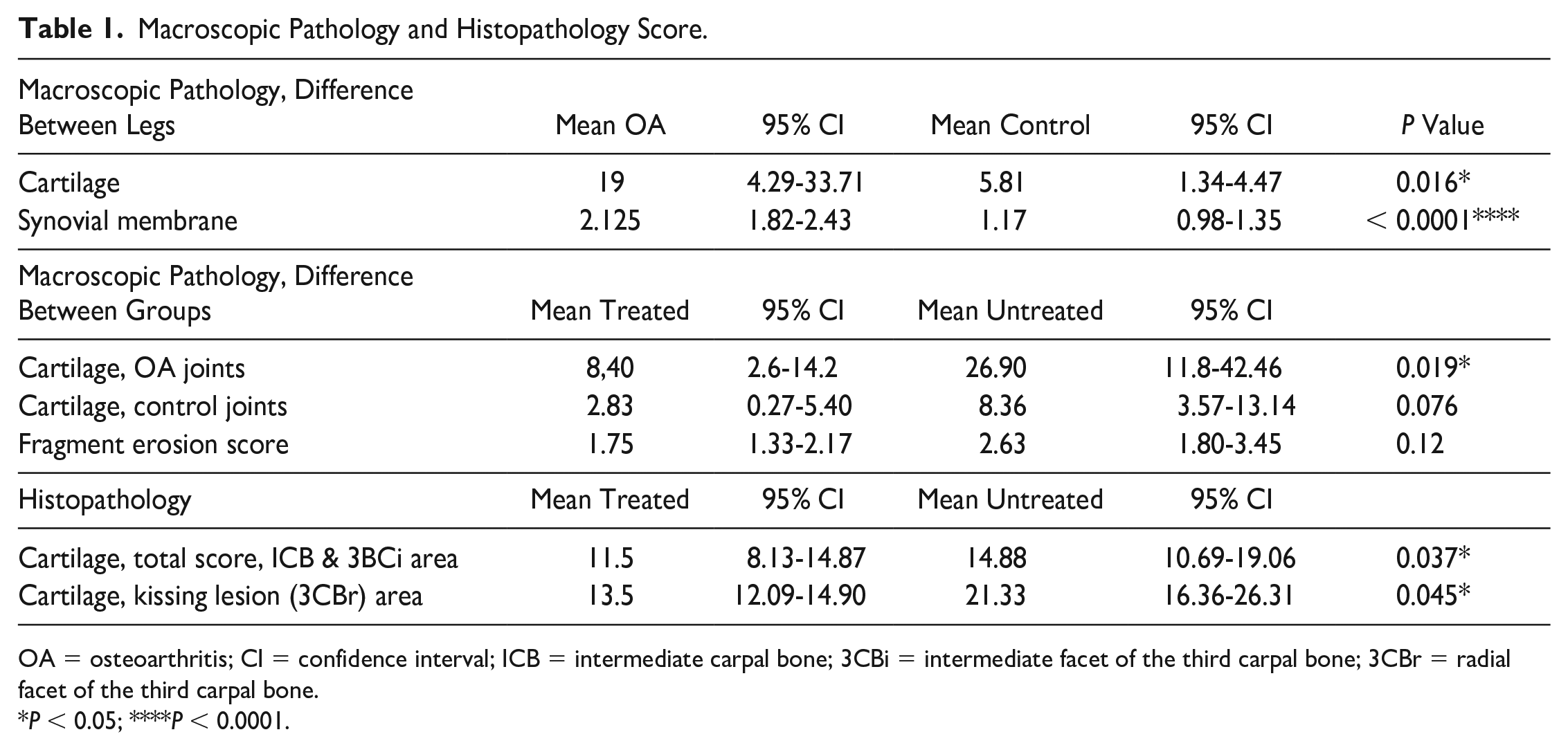
OA = osteoarthritis; CI = confidence interval; ICB = intermediate carpal bone; 3CBi = intermediate facet of the third carpal bone; 3CBr = radial facet of the third carpal bone.
P < 0.05;****P < 0.0001.
Histologic evaluation
Histopathologic cartilage lesions were apparent as fibrillation and fissuring, chondrocyte clustering and necrosis, focal cell loss, proteoglycan depletion (loss of safranin-O staining), and deep osteochondral splitting (
Histological analysis of the SM of the OA joints did not show any differences between the treated and untreated groups (P = 0.93).
Integrin α10-MSCs Were Detected in SF, Articular Cartilage, and Subchondral Bone
Male integrin α10-MSCs were traced using Y-chromosome-specific SRY gene expression. SRY gene expression was detected in SF cell pellets from all treated horses on day 3, day 10 (n = 1), and day 17 (n = 2) in the treated joints. SRY gene expression was detected in subchondral bone at the fragment site (n = 1), in the cartilage at the fragment site (n = 3), in the cartilage in the kissing lesion area (n = 2), and in the cartilage in the intermediate carpal bone area (n = 1) of the treated joints on day 52. No SRY gene expression was detected in the SM.
Discussion
IA treatment with integrin α10-MSCs reduced the development of PTOA in our equine model. There was significant reduction in lameness over time in the treated group compared with the untreated group. Response to joint flexion decreased over time in both groups, but more in the treated group. Less macroscopic and histopathologic changes were observed in the articular cartilage of the treated group compared with the untreated group.
Pain is the primary complaint in OA patients, and OA is one of the major causes of chronic pain worldwide.34,35 Lameness is an expression of orthopedic pain and impaired function in horses, 36 that can be assessed objectively. 37 The horses in our study developed moderate pain as a response to the induced trauma and the development of OA. The lameness decreased over time in the treated group, which corresponds to the reduced joint pathology found at the end of the study in this group. Similar positive effects of MSC treatment on lameness have been demonstrated previously, including in horses with naturally occurring OA in which MSC treatment resulted in a higher proportion of horses returning to their previous athletic performance.38-42 In human patients, MSC treatments improved patient reported outcome measures in clinical trials.11-14,43-47
MSCs are potentially a disease-modifying osteoarthritis drug (DMOAD), defined as a drug that inhibits or even reverses the progression of OA. 48 MSCs also allow for allogeneic transplantation as they are immune evasive and can therefore provide a true single-stage and off-the-shelf treatment. Chondroprotective effects of MSC treatment have been demonstrated previously in experimental equine23,49,50 and human clinical studies.12,13,51 More specifically, IA injection of integrin α10-MSCs in an equine tarsal PTOA resulted in significantly less histologic and macroscopic fibrillation of the cartilage and less severe histologic and radiographic subchondral bone sclerosis. 23
Paracrine effects are believed to be the main mechanism of action behind the DMOAD effect of MSCs. 52 In our study, integrin α10-MSC treatment resulted in an increased SF concentration of PGE2, IL-10, and G-CSF. This PGE2 increase was also observed after integrin α10-MSC treatment in the previous study by Delco et al. 23 PGE2 plays a diverse role in inflammation and is known to regulate both pro- and anti-inflammatory functions 53 with a biphasic regulation during the course of inflammation. 54 The initial PGE2 increase observed in treated and untreated horses was likely a pro-inflammatory response to the induced trauma. It is known that in a second wave of PGE2 induces secretion of the anti-inflammatory IL-10 and promotes anti-inflammatory effects in relation resolution of chronic inflammation, such as the repolarization of macrophages from a pro- to an anti-inflammatory state, which is one of the proposed mechanisms of MSCs.53,55-57 Such a second wave of PGE2, coinciding with a peak in IL-10, was observed after injection with integrin α10-MSCs in our study. Besides anti-inflammatory and anabolic effects,58,59 IL-10 can also play a more direct role in pain and nociception through direct neurogenic desensitization60,61 and by decreasing the expression of the proalgetic factor nerve growth factor (NGF),62,63 which is a major contributor to pain signaling,64,65 including in human66,67 and equine68,69 OA. In addition, it has been suggested that MSC treatment inhibits local inflammation through PGE2 only when IL-6 is present 70 and a tendency of accompanying increased IL-6 levels was noted after integrin α10-MSC treatment. Increased levels of G-CSF were also observed after integrin α10-MSC injection in the current study. G-CSF is secreted mainly by monocytes and macrophages, and also to some degree by MSCs.71,72 Both could be responsible for the increased levels of G-CSF in the SF, as an increase was observed over time in both groups, but more so in the treated group. MSCs are known to recruit resident progenitor cells or stem cells potentially through G-CSF secretion73,74-76 and in vivo studies have shown improved healing of cartilage defects in the presence of G-CSF.77-79 Taken together, increased levels of important paracrine signaling molecules were observed in the SF after integrin α10-MSC injection. These can be responsible for immunomodulatory and chondroprotective effects found in this study and possibly for the reduced pain and improved joint function.
In contrast to paracrine effects, the ability of MSCs to engraft and directly participate in cartilage regeneration is still debated.80-84 The allogeneic cells were detected in one or more joint tissues in 5 of the 8 treated horses 52 days after treatment. To our knowledge, this is the first time allogeneic MSCs have been detected in the cartilage of horses at any timepoint after injection. In a recent study, we demonstrated that IA injected human integrin α10-MSCs homed to cartilage defects in rabbit knees. The MSCs were integrated in all layers of the repair tissue, and the MSCs colocalized with aggrecan and type II collagen. 27 This suggested engraftment and differentiation of integrin α10-MSCs may also have occurred in this study, as the cells were still present in the osteochondral tissues of the treated joints.
There is a high level of evidence to support that MSC treatment is safe and very few adverse effects have been reported. The most common side effect is a self-limiting local inflammatory reaction in the joint after MSC injection known as “joint flare.”44,85 In horses, a number of studies have shown moderate or severe, transient inflammation in the joint in the first days after MSC injection.40,41 Therefore, IA injections of equine MSCs are often accompanied by a dose of non-steroidal anti-inflammatory drugs (NSAIDs).40,41,49,50 However, PGE2 secretion is one of the proposed paracrine signaling molecules.70,86-89 PGE2 is synthesized through the cyclooxygenases (COX) pathways, thus, it might be disadvantageous or even contraindicated to administer a COX-inhibitor simultaneously with MSC treatment. 90 In our study, integrin α10-MSC injection did not cause any clinical signs of joint flare and could therefore be administered without cotreatment with an NSAID.
The high prevalence of naturally occurring PTOA in horses 24 and the high resemblance between human and equine cartilage are the main reasons why the horse is a suitable translational model for human PTOA.91-96 The model used in this study is the most frequently used PTOA model in horses.25,26,97-99 The surgically created trauma initiates the inflammatory cascade leading to PTOA, which is further amplified by a treadmill exercise program throughout the study period. The results of this study are therefore believed to be translatable to human PTOA. There were several limitations to this study. The study groups were small. The small number of horses was in part due to unintended coronal bi-articular fracture of the RCB during OA induction surgery in 3 of 17 horses. There was also a noticeable variability in the data, such as the quantitative cytokine measurements and histological scoring, because of differences in the magnitude of responses between individual horses. All horses responded the same to OA induction and all treated horses responded in a similar way to integrin α10-MSC injection, but the degree of response varied between individual horses. The small number of horses did not allow for correlations between levels of response and overall outcome. Besides, blinding was not possible for many real-time observations, such as lameness evaluation and results of flexion tests. Observers were blinded as much as possible, including being blinded to results from the other group during data collection, and full blinding for all postmortem examinations was achieved. This model resembles a crude trauma followed by early return to exercise, which results in fast developing PTOA. Also, treatment is introduced at an early stage. As the underlying pathophysiology of PTOA remains largely unknown, results could be different with slower progressing PTOA or with treatment at a later stage.
Overall, this study demonstrated that IA injection of integrin α10-MSCs is safe and alleviates the progression of PTOA likely through the immunomodulatory factors IL-10 and PGE2, which results in less cartilage degradation and less joint pain. In addition, integrin α10-MSCs were traced back in cartilage and subchondral bone 52 days after injection. Together, this suggests that integrin α10-MSCs hold potential as a DMOAD.
Supplemental Material
sj-docx-1-car-10.1177_19476035231209402 – Supplemental material for Integrin α10β1-Selected Mesenchymal Stem Cells Reduce Pain and Cartilage Degradation and Increase Immunomodulation in an Equine Osteoarthritis Model
Supplemental material, sj-docx-1-car-10.1177_19476035231209402 for Integrin α10β1-Selected Mesenchymal Stem Cells Reduce Pain and Cartilage Degradation and Increase Immunomodulation in an Equine Osteoarthritis Model by Camilla Andersen, Stine Jacobsen, Kristina Uvebrant, John F. Griffin, Lucienne Angela Vonk, Marie Walters, Lise Charlotte Berg, Evy Lundgren-Åkerlund and Casper Lindegaard in CARTILAGE
Supplemental Material
sj-docx-2-car-10.1177_19476035231209402 – Supplemental material for Integrin α10β1-Selected Mesenchymal Stem Cells Reduce Pain and Cartilage Degradation and Increase Immunomodulation in an Equine Osteoarthritis Model
Supplemental material, sj-docx-2-car-10.1177_19476035231209402 for Integrin α10β1-Selected Mesenchymal Stem Cells Reduce Pain and Cartilage Degradation and Increase Immunomodulation in an Equine Osteoarthritis Model by Camilla Andersen, Stine Jacobsen, Kristina Uvebrant, John F. Griffin, Lucienne Angela Vonk, Marie Walters, Lise Charlotte Berg, Evy Lundgren-Åkerlund and Casper Lindegaard in CARTILAGE
Supplemental Material
sj-docx-3-car-10.1177_19476035231209402 – Supplemental material for Integrin α10β1-Selected Mesenchymal Stem Cells Reduce Pain and Cartilage Degradation and Increase Immunomodulation in an Equine Osteoarthritis Model
Supplemental material, sj-docx-3-car-10.1177_19476035231209402 for Integrin α10β1-Selected Mesenchymal Stem Cells Reduce Pain and Cartilage Degradation and Increase Immunomodulation in an Equine Osteoarthritis Model by Camilla Andersen, Stine Jacobsen, Kristina Uvebrant, John F. Griffin, Lucienne Angela Vonk, Marie Walters, Lise Charlotte Berg, Evy Lundgren-Åkerlund and Casper Lindegaard in CARTILAGE
Footnotes
Acknowledgments and Funding
The authors would like to acknowledge Lene Theil Skovgaard for her help with all statistical analyses. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Xintela AB, Torben og Alice Frimodt Foundation, the Frimodt Heineke Foundation, Direktør Emil C. Hertz og Hustru Inger Hertz’ Foundation, and Direktør Michael Hermann Nielsens Mindelegat.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: E.L.-Å. is the CEO and CSO of Xintela AB, holds stock in the company, and is an inventor of a patent related to this study. K.U. was an employee of and holds stock in Xintela AB. L.A.V was an employee of Xintela AB.
Ethical Approval
The study was approved by the Danish Animal Experiments Inspectorate (approval no 2020-15-0201-00602 and 2017-15-0201-01314) as well as the local Ethical and Administrative body of the Department of Veterinary Clinical Sciences, University of Copenhagen (approval no. 2020-016 and 2017-010).
Availability of Data and Materials
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
