Abstract
Objectives
Functions of mesenchymal stem/stromal cells (MSCs) are affected by patient-dependent factors such as age and health condition. To tackle this problem, we used the cellular reprogramming technique to epigenetically alter human MSCs derived from the synovial fluid of joints with osteoarthritis (OA) to explore the potential of reprogrammed MSCs for repairing articular cartilage.
Materials and Methods
MSCs isolated from the synovial fluid of three patients’ OA knees (Pa-MSCs) were reprogrammed through overexpression of pluripotency factors and then induced for differentiation to establish reprogrammed MSC (Re-MSC) lines. We compared the in vitro growth characteristics, chondrogenesis for articular cartilage chondrocytes, and immunomodulatory capacity. We also evaluated the capability of Re-MSCs to repair articular cartilage damage in an animal model with spontaneous OA.
Results
Our results showed that Re-MSCs increased the in vitro proliferative capacity and improved chondrogenic differentiation toward articular cartilage-like chondrocyte phenotypes with increased THBS4 and SIX1 and decreased ALPL and COL10A1, compared to Pa-MSCs. In addition, Re-MSC-derived chondrocytes expressing elevated COL2A and COL2B were more mature than parental cell-derived ones. The enhancement in chondrogenesis of Re-MSC involves the upregulation of sonic hedgehog signaling. Moreover, Re-MSCs improved the repair of articular cartilage in an animal model of spontaneous OA.
Conclusions
Epigenetic reprogramming promotes MSCs harvested from OA patients to increase phenotypic characteristics and gain robust functions. In addition, Re-MSCs acquire an enhanced potential for articular cartilage repair. Our study here demonstrates that the reprogramming strategy provides a potential solution to the challenge of variation in MSC quality.
Introduction
Human mesenchymal stem/stromal cells (MSCs) hold promise as a biologic agent for treating of orthopedic disorders due to their differentiation capacity and immunomodulatory potential.1,2 However, a critical challenge to be resolved is that properties of the cell associated with therapeutic effectiveness are affected mainly by the age and health condition of a donor.3,4 Studies show that human MSCs from aged donors exhibit significantly longer doubling time and increased senescence-associated markers, 3 and are subjected to age-related alterations, including increased oxidative damage, and higher levels of reactive oxygen species, resulting in loss of MSC “fitness.” 4 More recent research demonstrates that human MSCs from aged donors display a significant reduction in cell viability, proliferation, and differentiation during in vitro culture. 5 Moreover, an increase in the age of either recipients or donors attenuates the transplantation efficiency of MSCs. 6 Similarly, the therapeutic potential of MSCs for cartilage repair is decreased in elderly patients. 7
There have been several approaches, including the use of chemicals and hypoxic culture, developed to improve functional properties of the cell; however, most of them exert only transient effects.8-11 Recent studies suggest that cellular reprogramming using Yamanaka factors can rejuvenate MSCs through epigenetic modification to turn aged cells into young ones, providing a viable approach to mitigate cellular aging.12-14 For instance, the finding of cell rejuvenation via reprogramming prompts derivatives of induced pluripotent stem cells (iPSCs) as therapeutic agents for cartilage repair. 15 In a recent study using human iPSCs, Yamashita et al. generated scaffold-free hyaline cartilage subcutaneously in immunodeficient mice without tumor formation. 16 Furthermore, they found that iPSC-derived cartilage integrated within surgically created articular cartilage defects in minipigs. 16 Similarly, Kimura et al. showed that human iPSC-derived cartilage did not cause concerns of immune reaction, suggesting that undesirable immunologic responses do not limit the use of iPSC-based therapies for articular cartilage repair. 17
To demonstrate the effect of cellular reprogramming on altering MSC properties associated with donor’s pathophysiological conditions, we compared activities of human MSCs derived from synovial fluid of osteoarthritic joints before and after cellular reprogramming. Furthermore, we investigate the potential of reprogrammed MSCs for repairing articular cartilage damage in an animal model with spontaneous osteoarthritis (OA).
Materials and Methods
Preparation of MSCs from iPSC Lines
The Insitutional Review Board approved collecting human synovial fluid from patients for MSC isolation at the University of Wisconsin-Madison. MSCs used in this study were previously generated from independent iPSC lines created from three OA patients’ synovial fluid-derived MSCs as detailed in our published report. 18 Briefly, synovial fluid-derived MSCs were isolated from patients undergoing knee arthroscopy and transfected with episomal plasmids carrying OCT4, SOX2, KLF4, c-MYC, NANOG, and LIN28 (3 µg of pEP4 EO2S ET2K, 3 µg of pEP4 EO2S EN2K, and 2 µg of pCEP4-M2L) using Nucleofactor II with U-20 program (Amaxa, Walkersville, MD) and the Nucleofector kit R (Lonza, Hayward, CA) to generate iPSC lines. With the verification of pluripotency, each iPSC line was then individually differentiated into an independent MSC line using the STEMdiff mesenchymal progenitor kit (STEMCELL Technologies, Vancouver, British Columbia, Canada). This study characterized and evaluated three parental MSC (Pa-MSC) lines and their independently derived reprogrammed MSC (Re-MSC) lines.
Flow Cytometric Analysis
Cells were trypsinized and washed twice using a buffer solution made of ice-cold phosphate buffer saline (PBS), 0.1% sodium azide, and 1% bovine serum albumin (BSA; Sigma-Aldrich, Burlington, MA). Antibodies detecting surface markers CD73, CD90, CD105, and CD45, were used to identify MSCs. Detailed information of the antibodies used in this study is provided in Supplementary Table S1. 18 Briefly, cells were incubated with fluorochrome-conjugated mouse antibody for 30 minutes, washed with buffer three times to remove unbound antibody, and analyzed by flow cytometry. Expression of surface markers was analyzed by MACSQuant Analyzer 10 (Miltenyi Biotec, Bergisch Gladbach, Germany). Data were analyzed using the FlowJo software (TreeStar, Ashland, OR).
Assessment of Multi-Lineage Differentiation
Adipogenic, chondrogenic, and osteogenic differentiation of Pa-MSCs (patients’ MSCs) and Re-MSCs (reprogrammed MSCs) were induced as previously described. 19 Briefly, cells were cultured in adipogenic, chondrogenic, or osteogenic medium for differentiation. After 21 days of induction, adipogenic differentiation was analyzed by staining of Oil red O (Sigma-Aldrich). Chondrogenic differentiation was assessed by Alcian blue staining and glycosaminoglycan (GAG) quantification. Osteogenic differentiation was determined by staining of Alizarin red S (Rowley Biochemical, Danvers, MA), calcium quantification, and cytochemical staining and quantification of alkaline phosphatase (ALP) activity. In addition to histological and biochemical analyses, the transcript expression of fat-associated (PPARG2, LPL), cartilage-associated (SOX9, COL2, ACAN), and bone-associated markers (CBFA1, OC, ALP) were analyzed by quantitative reverse transcription-polymerase chain reaction (RT-PCR).
Immunofluorescence Analysis
Slides with sectioned cell pellets were first treated with 0.1% pepsin in 0.01 N HCl for 15 minutes, permeabilized with 0.1% Triton X-100 in PBS for 30 minutes, and blocked with 0.1% BSA in PBS for 30 minutes. They were then incubated with primary antibody goat anti-human collagen type 2 (Santa Cruz Biotechnology, Dallas, TX), collagen type 10 (Santa Cruz), or rabbit anti-human vimentin (Abcam, Cambridge, UK) for an hour at 4 °C. The slides were washed three times with PBS and then incubated with secondary antibody donkey anti-goat FITC (Santa Cruz) or donkey anti-rabbit Alexa Fluor 555 (Thermo Fisher Scientific, Waltham, MA) with 1:100 dilution in BSA/PBS buffer for 45 minutes. Staining of 4,6-diamidino-2-phenylindole dihydrochloride (DAPI; Vector, Burlingame, CA) was performed for 10 minutes to visualize nuclei for image analysis by a laser scanning confocal microscope (Nikon A1RS, Japan).
Cell Growth Analysis
Cells with the initial seeding density of 1 × 103 cells/cm2 were cultured and harvested at different time points. The Quant-iT PicoGreen dsDNA Assay kit (ThermoFisher Scientific) was used to detect the number of cells to determine the MSC growth in culture.
Immunomodulatory Assay
To evaluate differences in the immunomodulatory ability between Pa- and Re-MSCs, chondrocytes isolated from cartilage specimens of OA patients undergoing knee replacement surgery following a published protocol were seeded at the high density of 5 × 104 cells/cm2 in 12-well transwell plates (Corning Life Sciences, Lowell, MA) and co-cultured with MSCs in inserts at a 1:1 ratio for 14 days. 20 The cells were cultured in DMEM/F12 supplemented with 10% FBS (fetal bovine serum) and 1% antibiotic and stimulated by 10 µg/mL of IL1B. The medium was changed every three days. Chondrocytes cultured without MSCs or IL1B stimulation served as a negative control while those with IL1B but without MSCs served as a positive control. Proinflammatory factors in conditioned medium collected from the co-culture on day 14 were analyzed using the ProcartaPlex immunoassay (High Sensitivity 9-Plex Human Panel, Thermo Fisher Scientific).
Total RNA Extraction and Quantitative RT-PCR
Total RNA was extracted from cells using the Zymo Quick-RNA MicroPrep kit (Zymo Research, Irvine, CA) following the manufacturer’s instructions. One microgram of total RNA was used in each reaction with the High-Capacity cDNA Reverse Transcription kit (Applied Biosystems, Carlsbad, CA) to synthesize cDNA. These commercial kits allow efficient extraction of highly concentrated DNA-free RNA and conversion of cDNA for transcript analysis. 18 Quantitative RT-PCR was performed using the iQ SYBR Green Premix (Bio-Rad, Hercules, CA). Chondrogenesis of Pa- and Re-MSCs was evaluated by detecting expression levels of cartilage-associated markers, sex determining region Y-box 9 (SOX9), collagen type 2 alpha 1 (COL2A1), and aggrecan (ACAN). 18 The expression of COL2A1 splice variants A and B (COL2A1A and COL2A1B) indicates the maturity of differentiated chondrocytes.21,22 The detection of thrombospondin 4 (THBS4) and SIX homeobox 1 (SIX1) was to identify articular cartilage chondrocytes, and alkaline phosphatase (ALPL) and collagen type 10 alpha 1 (COL10A1) to identify growth plate chondrocytes. 23 Cartilage degradation was determined by the expression of matrix metalloproteases 3, 9, and 13 (MMP3, MMP9, MMP13) and aggrecanases 4 and 5 (ADAMTS4, ADAMTS5).24,25 Sequences of primers used for cDNA detection are listed in Supplementary Table S2. The 2−ΔCT method was used to determine relative expression levels of a target transcript to those of ubiquitin C (UBC) as an internal control.
Knockdown of Sonic Hedgehog Signaling Molecules
Cells cultured in T75 flasks until 80% confluent were transfected with 10 nM siSMO (Thermo Fisher Scientific) using the GenMute siRNA transfection reagent (SignaGen Laboratories, Rockville, MD) per the manufacturer’s instructions. Cells were then trypsinized, reconstituted, and seeded in 6-well plates with 2.0 × 107 cells/mL in a 15-µL droplet to establish micromass culture for chondrogenic differentiation.
Guinea Pig OA Model for Pilot Evaluation
Animal use and all procedures followed a protocol approved by the Institutional Animal Care and Use Committee at the University of Wisconsin-Madison, and the experimental data are kept at the Wisconsin Institute of Medical Research. To determine the effect of MSC injections on OA joints, we used Dunkin-Hartley strain 051 guinea pigs (Charles River, Wilmington, MA) as an animal model. This specific guinea pig strain develops OA phenotypes with severe cartilage damage naturally 26 without requiring induction with surgical manipulation or chemical treatment. The animal has been extensively used as a spontaneous OA model to study the disease or evaluate new therapies.27-29 A total of nine 12-month-old littermates with the mean weight of 1038.67 ± 94.68 g were randomly assigned to 3 injection groups (placebo, Pa-MSC, Re-MSC) with 3 animals (2 males and 1 female) per group. We chose the left stifle as the experimental side with MSC injection and the right as the control side without injection.
Intraarticular Injection of MSCs
A total of 1 × 106 MSCs per intraarticular injection, the dose commonly used by our group 30 and others, 31 were resuspended with 100 µL of PBS in a 1-mL sterile syringe (Monojet, Covidien, MA). Vehicle injection consisted of 100-µL sterile PBS only. Animals were anesthetized using an isoflurane chamber, stifles clipped and aseptically prepped, and injected with a 27-gauge needle using a suprapatellar approach under fluoroscopy (GE Healthcare; Suppl. Fig. S1).
Assessment of Serum Biomarkers
Sera were collected from anesthetized animals before the first intraarticular joint injection and sacrifice by pricking the lateral tarsal vein with a sterile 18-gauge needle for sample collection in microvette 500 serum collection tubes (Sarstedt Inc, Fischer Scientific). Samples were centrifuged until separated. Serum biomarkers associated with severity of guinea pig OA, including tumor necrosis factor alpha (Tnfa) and regulated on activation, normal T cell expressed and secreted (Rantes) as previously described, 27 were assessed using Multiplex ELISA (Bio-Plex Pro Mouse Cytokine 23-Plex, Group I Assay, Bio-Rad).
Stifle Joint Analysis
Stifle joint synovium was collected immediately after sacrifice, digested in Trizol, and analyzed for the expression of transcripts of interest. Stifles were fixed in 10% neutral buffer formalin and decalcified in 15% ethylenediaminetetraacetic acid. The stifles were bisected in the coronal plane 32 and paraffin-embedded. Four-micrometer paraffin sections were prepared and stained with hematoxylin and eosin, Alcian blue, and Safranin-O. Blinded histopathology evaluation of medial compartment OA used the Osteoarthritis Research Society International (OARSI) articular cartilage histopathology assessment. 29
Statistical Analysis
Quantitative data were expressed as mean ± standard deviation of three biological replicates aggregated in triplicate. Categorical data were expressed as counts and percentages. Student’s t-test was performed to compare means between 2 groups. Analysis of variance (ANOVA) was performed to compare means of more than two groups. A Tukey post hoc correction was used to adjust for multiple comparisons when the ANOVA was significant. Five blinded examiners assessed the degree of OA of each stifle. A mixed-effects linear regression model was used to compare the numeric OARSI assessment scores of each stifle’s medial compartment, with animal and rater as random effects, for cartilage structure and proteoglycan content. A P-value <0.05 was considered statistically significant.
Results
IPSC-Derived MSCs Obtained from Joint Synovial Fluid Exhibit MSC Characteristics
Pa-MSCs were isolated from synovial fluid and reprogrammed to generate iPSCs, then induced to differentiate into Re-MSCs ( Fig. 1A ). Re-MSC lines exhibited uniform, fibroblast-like morphology, similar to that of Pa-MSCs ( Fig. 1B ). The total DNA content, proportional to the cell number, revealed a higher proliferation rate in Re-MSCs, compared to that in Pa-MSCs ( Fig. 1C ). Pa-MSCs expressed CD73, CD90, and CD105 but not CD45, indicating the immunophenotype of MSCs ( Fig. 1D ). Re-MSCs exhibited the same surface markers with expression levels comparable with those on Pa-MSCs. Both cell types were multipotential and capable of differentiating into the adipogenic, osteogenic, and chondrogenic lineages ( Fig. 1E ). Notably, both qualitative staining and quantitative biochemical assays showed that Re-MSCs produced fewer fat droplets in adipogenic culture, more calcium content in osteogenic culture, and increased GAG in chondrogenic culture, compared to Pa-MSCs. Results of quantitative RT-PCR analysis showed consistent findings that bone-associated marker CBFA1, ALPL, and OC, fat-associated markers PPARG2 and LPL, as well as cartilage-associated markers SOX9, COL2, and ACAN significantly increase upon 21 day’s induction in both Pa-MSCs and Re-MSCs.
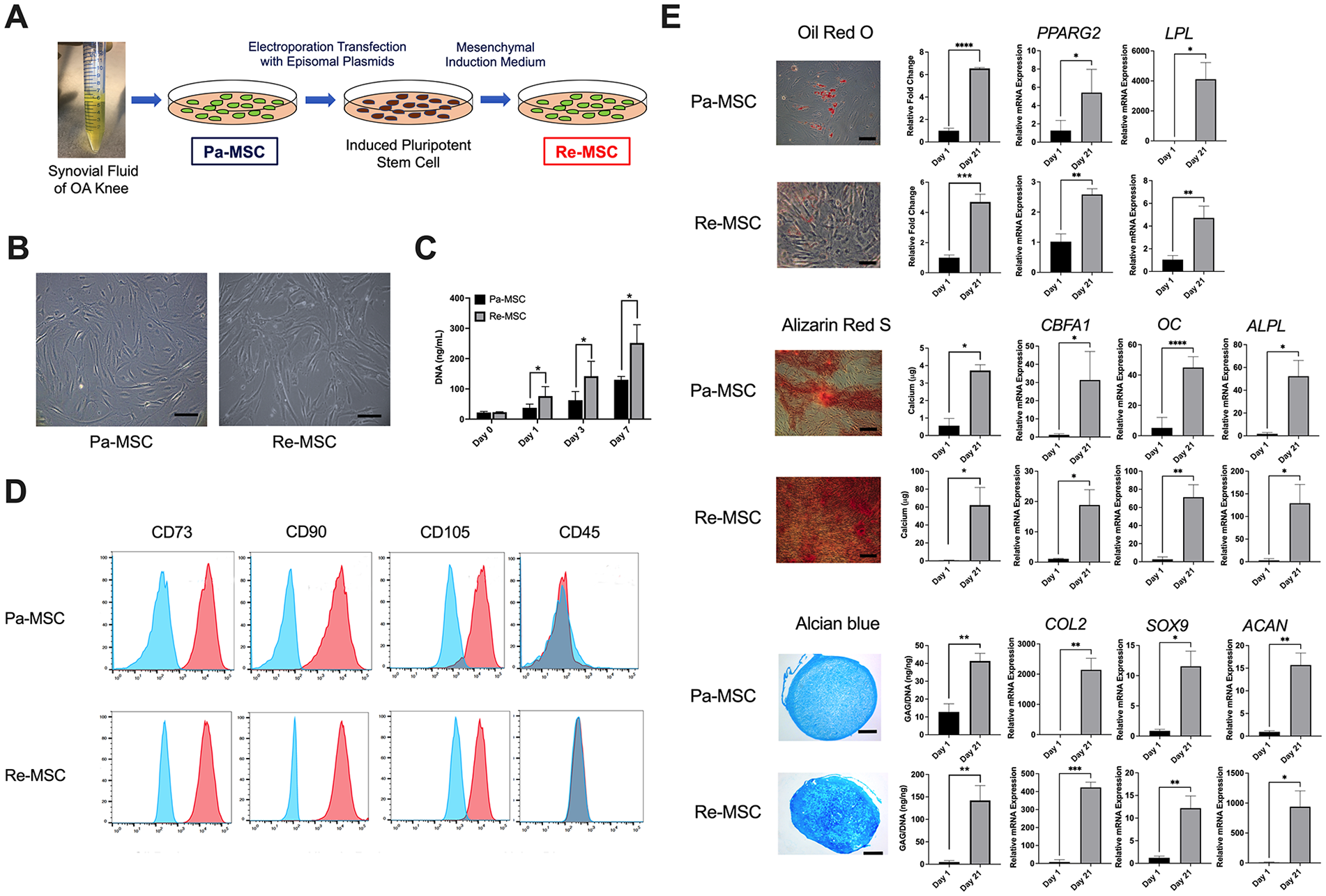
Characterization of synovial fluid-derived Pa-MSC and Re-MSC lines. (
Re-MSCs Are Capable of Differentiating Into Articular Cartilage Chondrocyte-Like Cells
The analysis of chondrogenesis showed that after 21 days of induction, cell pellets derived from Re-MSCs contained spherical chondrocyte-like cells in lacunae producing an increased number of sulfated proteoglycans, which appears to more closely resemble the cellular structure and extracellular matrix composition of articular cartilage than those from Pa-MSCs ( Fig. 2A ). In agreement with the results of Alcian blue staining, our quantitative data demonstrated that Re-MSC-derived pellets produced more sulfated glycosaminoglycans (GAGs) than pellets derived from Pa-MSCs ( Fig. 2B ). Quantitative RT-PCR analysis further demonstrated that mRNA levels of cartilage-associated markers COL2A1, SOX9, ACAN were significantly higher in pellets derived from Re-MSCs than those derived from Pa-MSCs ( Fig. 2C ). Immunofluorescence analysis showed that more COL2A1 and less COL10A1 were produced in Re-MSC pellets than those in Pa-MSC pellets ( Fig. 2D ), indicating that cells in Pa-MSC pellets are increasingly hypertrophic compared to those in Re-MSC pellets. Furthermore, our results demonstrated that expression levels of articular cartilage markers THBS4 and SIX1 23 were higher, whereas levels of growth plate cartilage markers ALPL and COL10A1 23 were lower in Re-MSC pellets than those in Pa-MSC pellets ( Fig. 2E ), suggesting that cells derived from Re-MSCs more closely resemble chondrocytes of articular cartilage, compared to those from Pa-MSCs. Given that chondroprogenitor cells produce COL2A and mature fully differentiated chondrocytes synthesize COL2B,21,22 our result further showed that both COL2A and COL2B were significantly increased in Re-MSC pellets, with a much larger difference in COL2B, compared to those in Pa-MSC pellets ( Fig. 2F ). Taken together, our data indicate chondrogenesis of Re-MSCs generates more mature, articular cartilage-like tissue compared to that of Pa-MSCs.
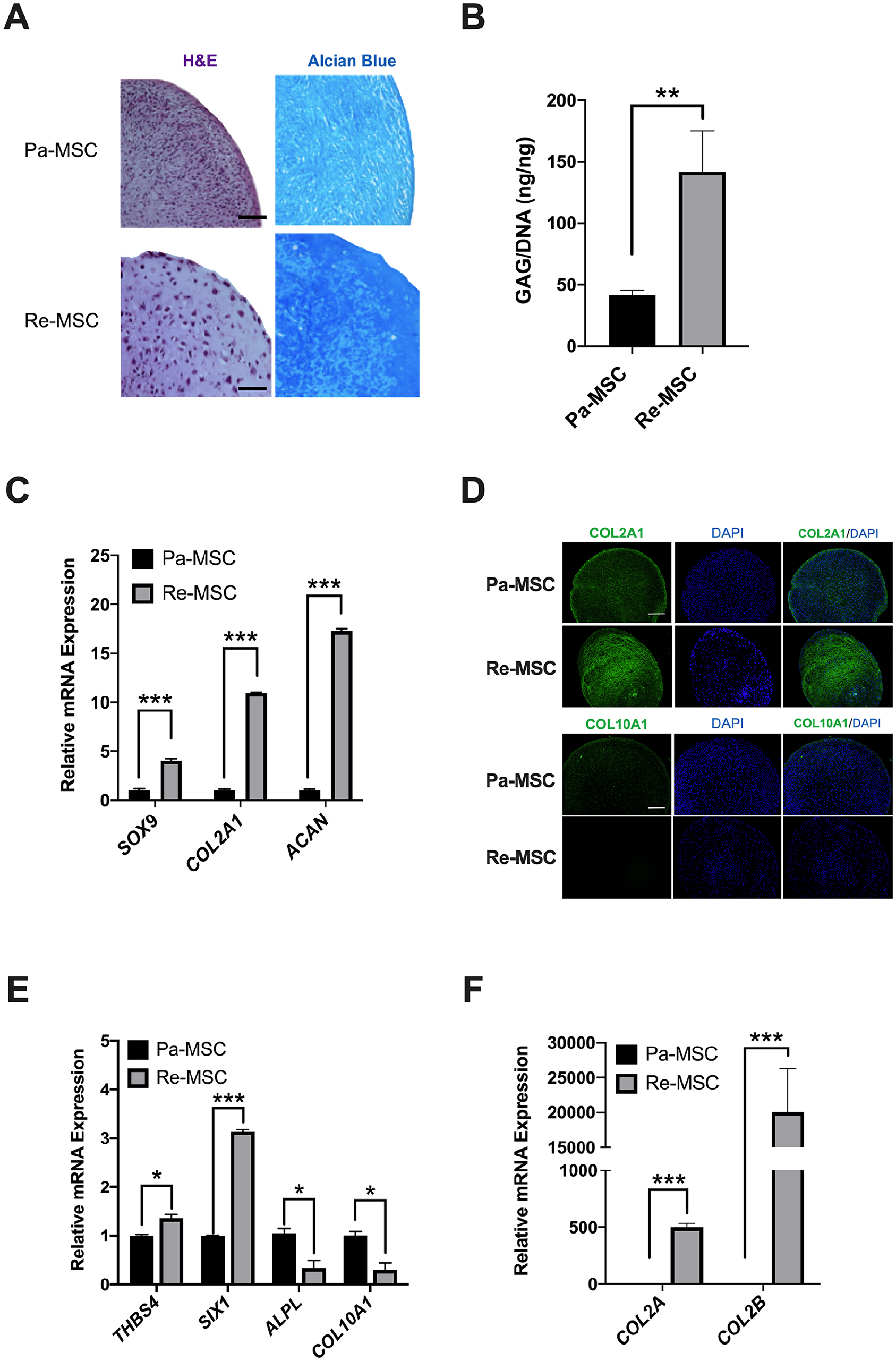
Comparison between chondrogenesis of Pa-MSCs and Re-MSCs. (
Sonic Hedgehog (SHH) Signaling Is Involved in Upregulated Chondrogenesis of Re-MSCs
Our recent study demonstrated that the SHH signaling pathway is increasingly activated in iPSC-MSCs compared to that of parental MSCs, 18 and other groups have also reported that the signaling pathway regulates chondrogenesis.33,34 Therefore, we hypothesized that differential regulation of SHH signaling results in enhanced chondrogenesis of Re-MSCs shown in Figure 2 . To test this hypothesis, we investigated effects of knockdown of the smoothened receptor (SMO), a key regulator of the SHH signaling pathway, on chondrogenesis of both Pa-MSCs and Re-MSCs. Successful knockdown of SMO was confirmed by the results demonstrating a significant reduction in SMO expression at both mRNA ( Fig. 3A ) and protein levels ( Fig. 3B ). SMO knockdown did not reduce the expression of the transmembrane receptor PTCH1 but significantly decreased the expression of downstream GLI1 at both mRNA and protein levels ( Fig. 3A and B ). After 7 days of chondrogenic induction in micromass culture, the SMO knockdown group showed less intense Alcian blue staining than the control group for both cell types. Quantification of Alcian blue staining further confirmed the qualitative results ( Fig. 3C ). Similarly, the expression of cartilage-associated markers, SOX9, COL2A1, and ACAN, in micromass of SMO-knockdown Pa-MSCs and Re-MSCs was significantly lower than that of control cells while levels of these markers in the knockdown group of Re-MSCs were significantly higher than those in the knockdown group of Pa-MSCs ( Fig. 3D ). These results indicate that increased activities of SHH signaling induced by cellular reprogramming led to enhanced chondrogenesis of MSCs.
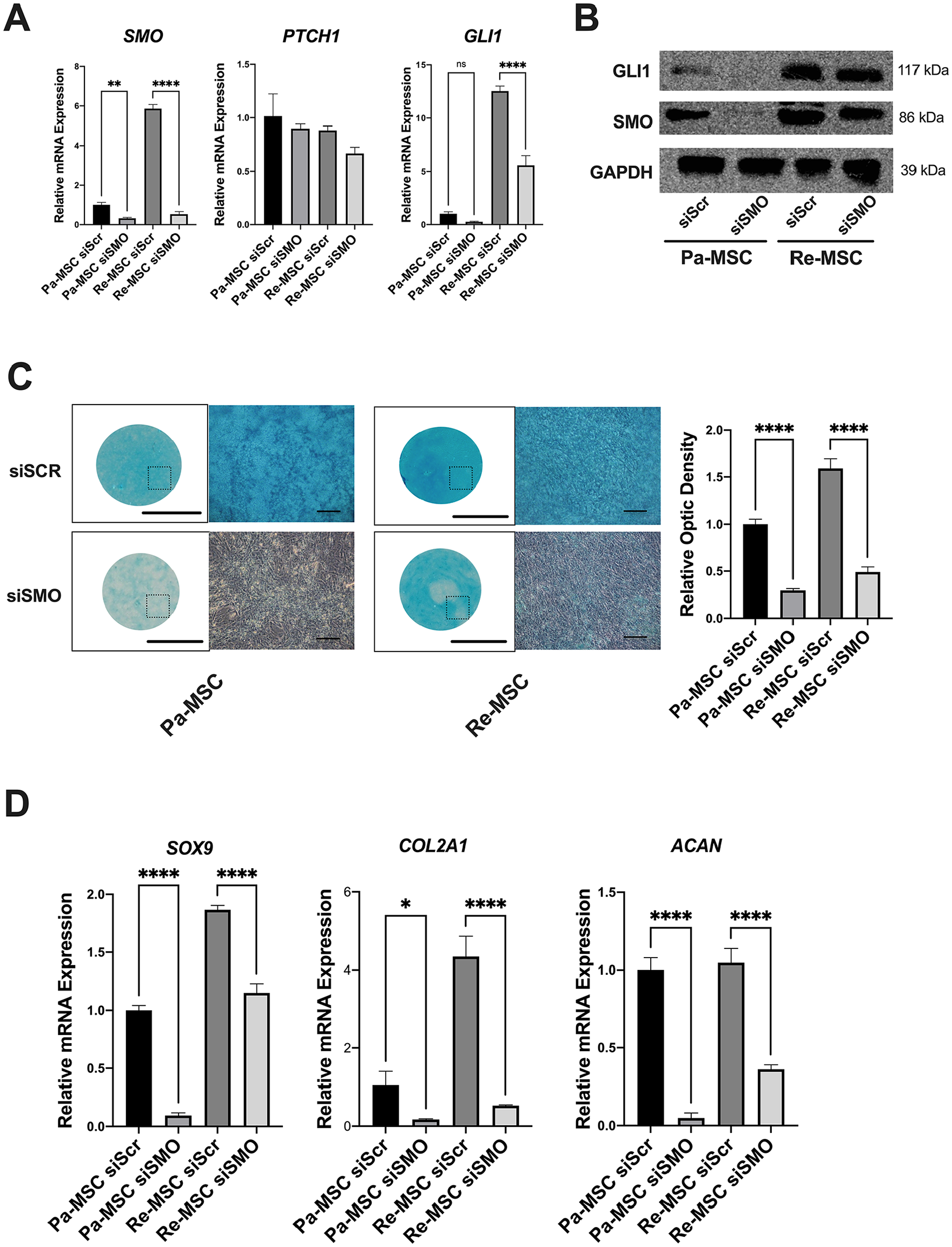
Effects of SMO knockdown on chondrogenesis of Pa-MSCs and Re-MSCs. (
Re-MSCs Possess Increased Anti-Inflammatory Capability
To determine differences in the capacity of inflammatory modulation between Pa-MSCs and Re-MSCs, chondrocytes treated with or without 10 µg/mL of IL1B were co-cultured with either of the cells in transwell plates. There was no significant difference in growth rates between Pa-MSCs and Re-MSCs in the co-culture over the 14-day period ( Fig. 4A ). As shown in Figure 4B , IL1B resulted in a significant increase in levels of the inflammation-related factors, IL1B, TNFA, IL10, and IL17A, in the medium of the chondrocyte-only culture compared to those in the control culture without IL1B treatment, and with MSC treatment levels of these factors were significantly downregulated. The reduction in IL1B and TNFA levels was greater in the Re-MSC-treated culture than in the Pa-MSC-treated culture, whereas the decrease in IL10 and IL17A levels was comparable between the 2 MSC-treated groups. Similarly, IL1B stimulation resulted in upregulated transcript expression of matrix metalloproteinases (MMP3, MMP9, and MMP13) and aggrecanases (ADAMTS4 and ADAMTS5) in the chondrocyte-only group compared to that in the control group ( Fig. 4C ). Modulated by co-cultured Pa-MSCs or Re-MSCs, mRNA expression levels of these catabolic factors in IL1B-stimulated chondrocytes were significantly downregulated. Notably, the group with co-cultured Re-MSCs showed significantly reduced expression of ADAMTS5 compared to that with co-cultured Pa-MSCs. Taken together, while both Pa-MSCs and Re-MSCs are capable of modulating the production of inflammatory cytokines and matrix proteases in IL1B-induced chondrocytes, it is shown that the modulation capability of Re-MSCs is greater than that of Pa-MSCs.
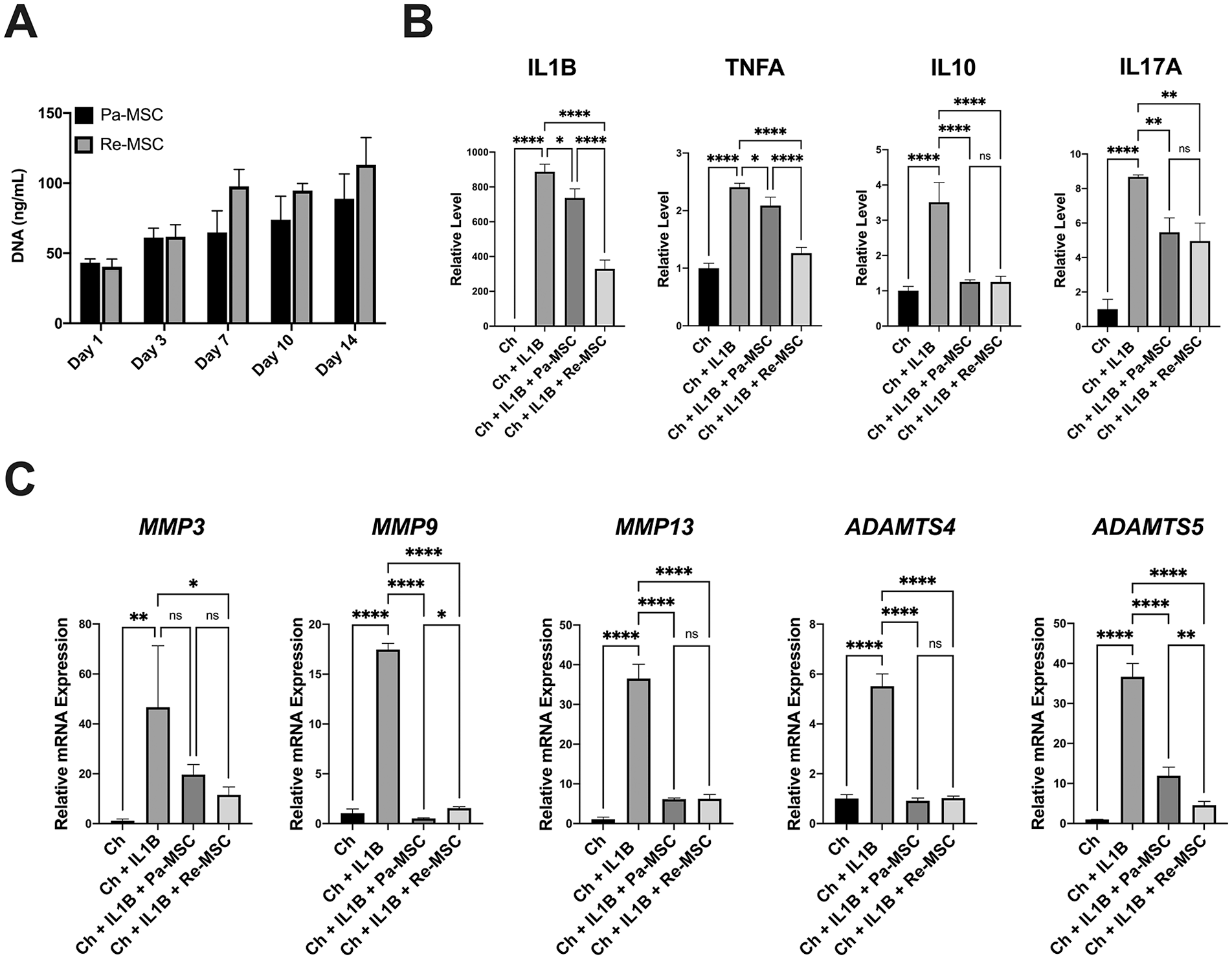
Expression levels of inflammatory cytokines and matrix proteases in IL1B-stimulated chondrocytes co-cultured with Pa-MSCs or Re-MSCs. (
Re-MSCs Preserve Articular Cartilage in a Spontaneous OA Model
The Duncan-Hartley guinea pig model of spontaneous OA was used to evaluate the potential of Pa-MSCs or Re-MSCs for the treatment of cartilage damage in this pilot investigation. Following randomization, stifle joints of animals were treated with vehicle alone, Pa-MSCs, or Re-MSCs, as presented in the schematic diagram of Figure 5A . All guinea pigs tolerated the injections well with no evidence of local inflammation, joint effusion, or observed unloading of the stifle. Fluoroscopic localization was utilized for each stifle injection. Iodinated contrast media was included allowing for confirmation of an entirely intraarticular injection and assessment of the distribution of fluid (Suppl. Fig. S1A). 35 MSC viability was found to be greater than 97% after needle passage (Suppl. Fig. S1B and C). There were no differences in the bodyweight of animals between groups (Suppl. Fig. S2). Serum biomarker analysis revealed that the serum levels of Tnfa in animals treated with Re-MSCs were significantly reduced compared to those in animals treated with either Pa-MSCs or saline alone, and the serum levels of Rantes in Re-MSC-treated animals were significantly lower than those in saline-treated animals ( Fig. 5B ). Furthermore, compared to vehicle-treated animals, Pa-MSC-treated animals showed a reduced mRNA expression of Tnfa in the synovium ( Fig. 5C ). Similarly, compared to vehicle- or Pa-MSC-treated animals, Re-MSC-treated animals showed a greater reduction in mRNA expression of Tnfa in the synovium.
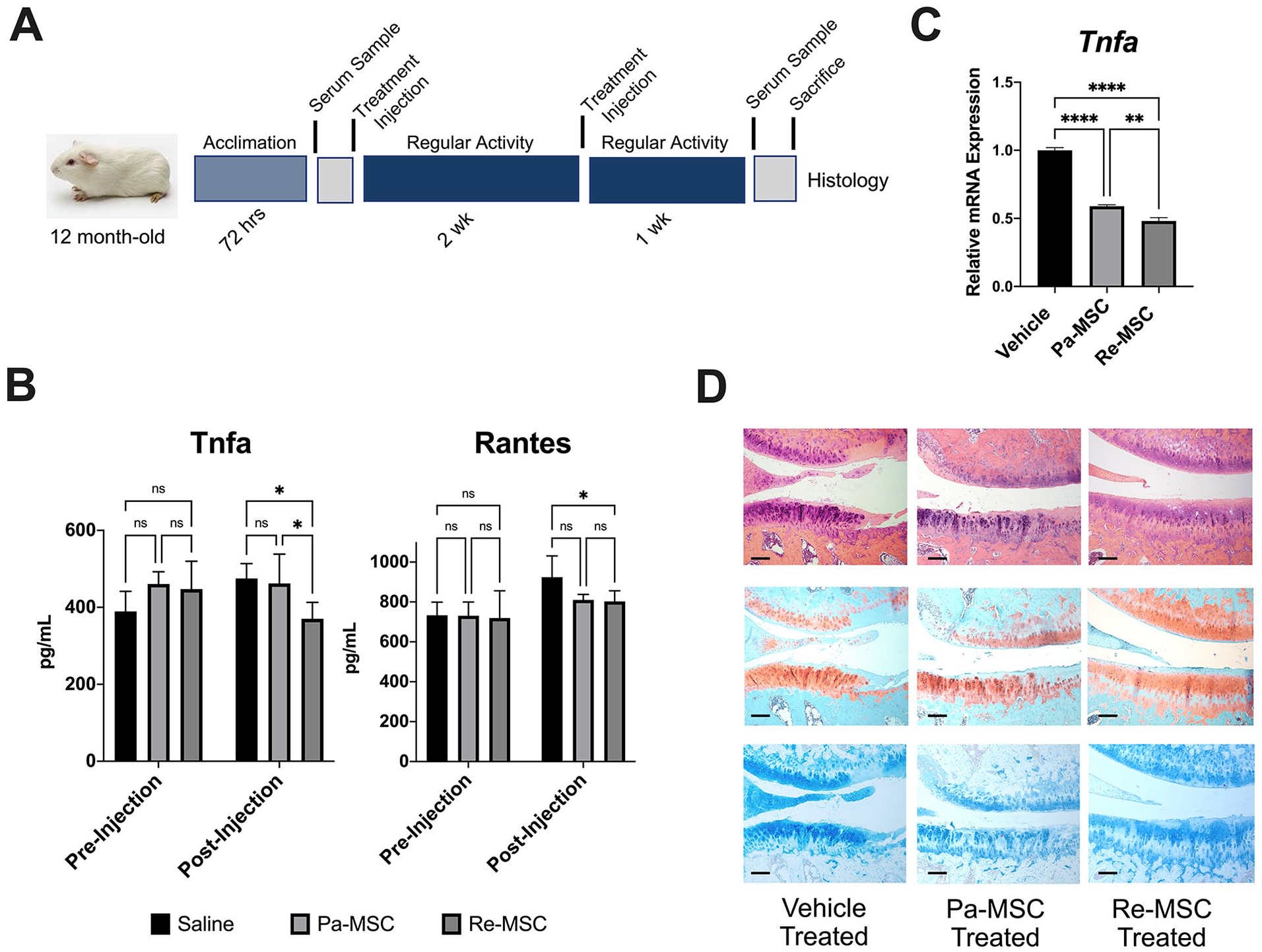
Joints of spontaneous OA model of guinea pigs. (
Histologic analysis, including hematoxylin-eosin, Safranin-O, and Alcian blue staining, was carried out to evaluate outcomes of Pa-MSC and Re-MSC injections ( Fig. 5D ). Qualitatively, animal joints treated with vehicle alone showed fissuring extending into the deep layer of articular cartilage, loss of chondrocyte cellularity, and reduced proteoglycan content across all layers of cartilage. Compared to the control vehicle group, joints treated with Pa-MSC injections showed increased cellularity and improved fissuring with less deep extension into cartilage but reduced proteoglycan content in the superior and middle layers. In contrast, joints receiving Re-MSC injections demonstrated that chondrocytes and rich proteoglycan were distributed across cartilage layers without fibrillation, suggesting cartilage structure and composition restoration.
Quantitatively, the effects of MSC injections on articular cartilage preservation of guinea pigs were evaluated using the standardized OARSI OA cartilage histopathology assessment system. 29 Based on histopathological grading, a higher OARSI score indicates a greater degree of OA. Differences in the structure and composition of articular cartilage between the experimental and control stifles were compared ( Table 1 ). In terms of cartilage structure, both the vehicle and Pa-MSC groups received a higher score for the experimental side compared to that for the control side. In contrast, animals treated with Re-MSC injections acquired a significantly lower OARSI histopathologic assessment score for the experimental side compared to that for the control side. In terms of proteoglycan content, there was no difference in the OARSI score between the experimental and control stifles in vehicle-treated animals. Animals treated with Pa-MSCs received a higher OARSI score for proteoglycan content on the experimental side than on the control side. Consistent with the result of cartilage structure scoring, the OARSI score for proteoglycan content was significantly lower in the experimental stifle than that in the control stifle of animals treated with Re-MSCs. Together, these data suggest that intraarticular injection of Re-MSCs in a spontaneous OA animal model promotes articular cartilage structure and composition preservation.
OARSI Articular Cartilage Histopathology Assessment a .
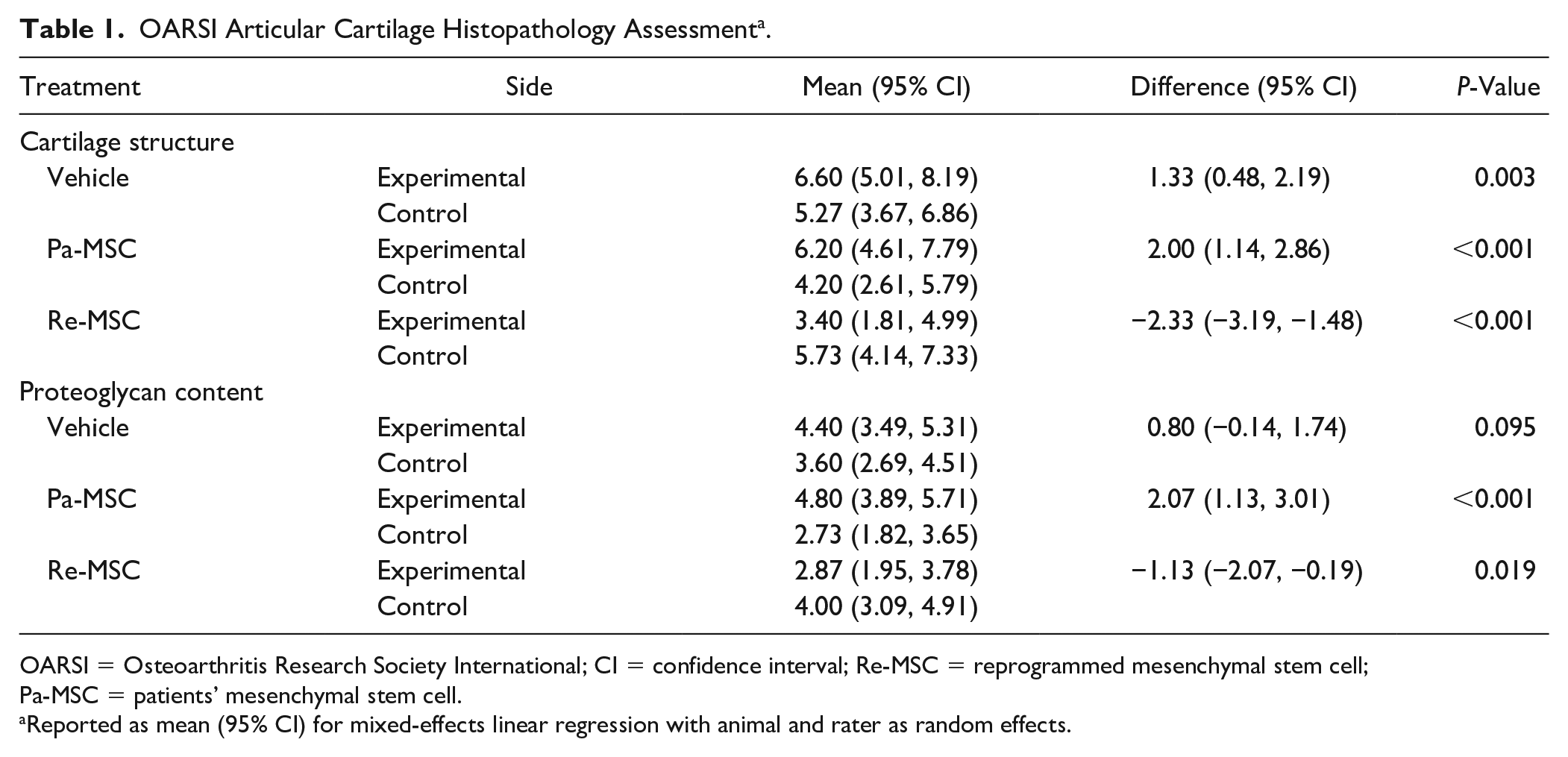
OARSI = Osteoarthritis Research Society International; CI = confidence interval; Re-MSC = reprogrammed mesenchymal stem cell; Pa-MSC = patients’ mesenchymal stem cell.
Reported as mean (95% CI) for mixed-effects linear regression with animal and rater as random effects.
Discussion
The current study demonstrates that cellular reprogramming of synovial fluid-derived MSCs from patients with OA successfully leads to the generation of hyaline-like cartilage with chondrocytes demonstrating the robust production of COL2A1 and GAG and upregulated expression of SOX9, COL2A1, and ACAN. Notably, our results also indicate that reprogrammed Pa-MSCs gain the capability toward articular chondrocyte differentiation and away from growth plate chondrocyte differentiation. Additionally, chondrocytes in Re-MSC pellets show a substantial increase in expression levels of COL2A and, to a greater extent, a mature chondrocyte-specific marker COL2B, compared to those in Pa-MSC pellets. Our findings are significant because they show that cellular reprogramming alters MSCs to increase the generation of mature articular chondrocytes through promoting effective chondrogenic induction.
Previous research has demonstrated that hedgehog signaling regulates activities of stem cells, 36 and is an essential pathway involved in early chondrogenesis. 37 We found that silencing SMO results in impaired chondrogenesis of Re-MSCs, as evidenced by a decrease in proteoglycan staining and decreased expression of cartilage-related transcripts, suggesting that upregulated chondrogenesis of the cell is mediated, at least in part, through increased activation of hedgehog signaling. The role of SHH signaling in the regulation of chondrogenesis has also been demonstrated in a previous study using bone marrow-derived MSCs, in which SHH overexpression leads to an increase in cartilage-related transcripts during chondrogenesis. 37 Moreover, Wu et al. found that inhibition of SHH with cyclopamine during chondrogenic induction results in a decrease in proteoglycan synthesis and COL2A1 and ACAN transcript expression. 38 Our results are consistent with previous findings that increased activation of hedgehog signaling results in enhanced chondrogenic differentiation and articular cartilage formation.34,39 Additionally, a recent study demonstrated that hedgehog signaling in osteoarthritic chondrocytes is upregulated compared to that in controls. 33 Using a surgically induced murine model, the study further indicates that increased activation of hedgehog signaling induces severe OA phenotypes, and conversely, inhibition mitigates the severity of OA. 33 Another study demonstrated that the expression of Indian hedgehog molecules in both cartilage and synovial fluid of OA patients is upregulated compared to that in control samples of non-OA patients. 40 These findings suggest that hedgehog signaling is important for regulation of chondrocyte activities, chondrogenic differentiation of MSCs, and OA development.
The induction of MMPs and ADAMTSs by proinflammatory cytokines, particularly IL1B and TNFA, results in structural destruction of osteoarthritic cartilage.24,25 For example, a previous study has shown that synovial macrophages secrete proinflammatory cytokines, IL1B and TNFA, to drive OA synovitis, and depleting OA synovial macrophages in culture reduces IL1B and TNFA. 41 Moreover, neutralizing both IL1B and TNFA results in the inhibition of proinflammatory cytokines. 41 Our result that Re-MSCs significantly reduce levels of OA-associated cytokines, particularly IL1B and TNFA, is consistent with the previously reported one that pluripotent stem cell-derived MSCs possess a robust capability of immunomodulation. 42 These findings suggest that through cellular reprogramming, MSCs acquire the increased capacity to regulate proinflammatory cytokines implicated in progressive cartilage destruction.
Previous studies have shown that MSC injections for the treatment of OA can mitigate articular cartilage destruction.43-45 OA models used in these studies are created by surgical manipulation43-46 or chemical inductions,47,48 which fall short of resembling spontaneous age-associated OA, most commonly found in humans. 49 In addition, both surgical and chemical approaches induce acute joint inflammation, representing a different proinflammatory response. 49 In this study, we have found that Re-MSC injections preserve the medial compartment of guinea pig stifle joints with significant improvement in maintaining both articular cartilage structure and proteoglycan content and reducing the expression of Tnfa transcripts in synovium and the production of serum Tnfa, compared to either saline control or Pa-MSC injections. Moreover, our findings that MSC injections lead to favorable outcomes of cartilage repair are in keeping with the findings of previous studies. For example, in a study using a collagenase-induced OA mouse model to evaluate effects of MSC injections, experimental results show that the injection reduces pain and generates a protective effect on damaged cartilage. 48 Another study also demonstrates that MSC treatment leads to amelioration of articular cartilage degeneration in a surgically induced-OA caprine model. 50
It is known that the injected dose of MSCs delivered to a joint affects the therapeutic outcome. While studies reveal a general trend that a greater MSC dose leads to more satisfactory tissue repair, it poses a practical challenge to efficiently prepare a large number of cultured MSCs needed for injection. A recent review systematically comparing previous studies using different doses of MSCs in various animal models has summarized that among the doses ranging from 2 × 104 to 2 × 107 cells per injection in these studies, the most commonly used ones are 1 × 106 and 1 × 107 cells. 31 Interestingly, several groups including ours have shown that a lower MSC dose delivered to a joint is more favorable for tissue repair than a higher one.51,52 For example, we have demonstrated that injecting the dose of 1 × 106 MSCs into a rat joint with ligament injury results in fewer M1 macrophages and stronger repaired tissue compared to the higher one of 4 × 106. 30 In this study, the outcome of cartilage preservation through injection of the selected MSC dose is shown promising; however, considering differences in joint size and repair tissue target between animal models, a future study will determine if a lower dose of MSCs can generate a similar or better repair outcome.
Here, we have shown that epigenetic reprogramming can alter the characteristics and activities of synovial-derived MSCs harvested from OA patients to enhance chondrogenic induction into hyaline cartilage chondrocytes and increase the immunomodulatory capacity to mitigate OA-associated inflammatory response. We have further demonstrated that injected Re-MSCs can maintain the structural retention and composition of articular cartilage in a spontaneous OA animal model. In summary, the findings of this study indicate that cellular reprogramming of MSCs presents a robust solution to address the current challenges that impact outcomes of MSC therapy.
Supplemental Material
sj-pdf-1-car-10.1177_19476035211040858 – Supplemental material for Reprogrammed Synovial Fluid-Derived Mesenchymal Stem/Stromal Cells Acquire Enhanced Therapeutic Potential for Articular Cartilage Repair
Supplemental material, sj-pdf-1-car-10.1177_19476035211040858 for Reprogrammed Synovial Fluid-Derived Mesenchymal Stem/Stromal Cells Acquire Enhanced Therapeutic Potential for Articular Cartilage Repair by Brian E. Walczak, Hongli Jiao, Ming-Song Lee and Wan-Ju Li in CARTILAGE
Supplemental Material
sj-pdf-2-car-10.1177_19476035211040858 – Supplemental material for Reprogrammed Synovial Fluid-Derived Mesenchymal Stem/Stromal Cells Acquire Enhanced Therapeutic Potential for Articular Cartilage Repair
Supplemental material, sj-pdf-2-car-10.1177_19476035211040858 for Reprogrammed Synovial Fluid-Derived Mesenchymal Stem/Stromal Cells Acquire Enhanced Therapeutic Potential for Articular Cartilage Repair by Brian E. Walczak, Hongli Jiao, Ming-Song Lee and Wan-Ju Li in CARTILAGE
Supplemental Material
sj-pdf-3-car-10.1177_19476035211040858 – Supplemental material for Reprogrammed Synovial Fluid-Derived Mesenchymal Stem/Stromal Cells Acquire Enhanced Therapeutic Potential for Articular Cartilage Repair
Supplemental material, sj-pdf-3-car-10.1177_19476035211040858 for Reprogrammed Synovial Fluid-Derived Mesenchymal Stem/Stromal Cells Acquire Enhanced Therapeutic Potential for Articular Cartilage Repair by Brian E. Walczak, Hongli Jiao, Ming-Song Lee and Wan-Ju Li in CARTILAGE
Supplemental Material
sj-tif-1-car-10.1177_19476035211040858 – Supplemental material for Reprogrammed Synovial Fluid-Derived Mesenchymal Stem/Stromal Cells Acquire Enhanced Therapeutic Potential for Articular Cartilage Repair
Supplemental material, sj-tif-1-car-10.1177_19476035211040858 for Reprogrammed Synovial Fluid-Derived Mesenchymal Stem/Stromal Cells Acquire Enhanced Therapeutic Potential for Articular Cartilage Repair by Brian E. Walczak, Hongli Jiao, Ming-Song Lee and Wan-Ju Li in CARTILAGE
Supplemental Material
sj-tif-2-car-10.1177_19476035211040858 – Supplemental material for Reprogrammed Synovial Fluid-Derived Mesenchymal Stem/Stromal Cells Acquire Enhanced Therapeutic Potential for Articular Cartilage Repair
Supplemental material, sj-tif-2-car-10.1177_19476035211040858 for Reprogrammed Synovial Fluid-Derived Mesenchymal Stem/Stromal Cells Acquire Enhanced Therapeutic Potential for Articular Cartilage Repair by Brian E. Walczak, Hongli Jiao, Ming-Song Lee and Wan-Ju Li in CARTILAGE
Footnotes
This work was completed at the University of Wisconsin-Madison, Wisconsin Institutes for Medical Research, Madison, WI, USA.
Author Contributions
BEW and W-JL developed the research concept and designed experiments. BEW, HJ, and M-SL acquired data. BEW, HJ, and W-JL analyzed and interpreted data. BEW drafted the manuscript. BEW, HJ, M-SL, and W-JL revised the manuscript. W-JL approved manuscript submission.
Acknowledgment and Funding
The author(s) disclosed receipt of the following financial support for research, authorshhip and/or publication of the article: This study was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health under Award Number R01 AR064803 and the Freedom of Movement Award (Department of Orthopedics and Rehabilitation, University of Wisconsin-Madison). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. Dr. Walczak was supported by NIH NIA T32AG000213-26 (Institute of Aging, University of Wisconsin-Madison), CTS UL1TR000427 (Institute of Clinical and Translational Research, University of Wisconsin-Madison), and CTS TL1TR000429 (Institute of Clinical and Translational Research, University of Wisconsin-Madison).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BEW is a consultant for AlloSource. HJ, M-SL, and W-JL have no competing interests to disclose.
Ethical Approval
Ethical approval for this study was obtained from Institutional Animal Care and Use Committee (M006043-R01) and Institutional Review Board (2016-1414) at the University of Wisconsin-Madison.
Informed Consent
Written informed consent was obtained from all subjects before the study.
Or
Verbal informed consent was obtained from all subjects before the study.
Or
Written informed consent was obtained from legally authorized representatives before the study.
Or
Verbal informed consent was obtained from legally authorized representatives before the study.
Or
Informed consent was not sought for the present study because it was exempt due to use of surgical waste.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
