Abstract
Objective
Treatment strategies for osteochondral defects, for which particulated autologous cartilage transplantation (PACT) is an emerging treatment strategy, aim to restore the structure and function of the hyaline cartilage. Herein, we compared the efficacy of PACT with control or human transforming growth factor-β (rhTGF-β), and clarified the necessity of bone graft (BG) with PACT to treat shallow osteochondral defects in a porcine model.
Design
Two skeletally mature male micropigs received 4 osteochondral defects in each knee. The 16 defects were randomized to (1) empty control, (2) PACT, (3) PACT with BG, or (4) rhTGF-β. Animals were euthanized after 2 months and histomorphometry, immunofluorescence analysis, semiquantitative evaluation (O’Driscoll score), and magnetic resonance observation of cartilage repair tissue (MOCART) score were performed.
Results
Hyaline cartilages, glycosaminoglycan synthesis, and collagen type II staining were more abundant in the PACT than in the control and rhTGF-β groups. The O’Driscoll score was significantly different between groups (P < 0.001), with both PACT groups showing superiority (P = 0.002). PACT had the highest score (P = 0.002), with improved restoration of subchondral bone compared with PACT with BG. The MOCART score showed significant differences between groups (P = 0.021); MOCART and O’Driscoll scores showed high correlation (r = 0.847, P < 0.001).
Conclusion
Treatment of osteochondral defects with PACT improved tissue quality compared with that with control or rhTGF-β in a porcine model. BG, in addition to PACT, may be unnecessary for shallow osteochondral defects. Clinical Relevance. BG may not be necessary while performing PACT.
Keywords
Introduction
Osteochondral defect (OCD) is a common orthopedic problem. Although many treatment strategies for symptomatic OCDs, including bone marrow stimulation (BMS), osteochondral autograft transplantation, autogenous cancellous bone graft (BG), autologous chondrocyte implantation, and frozen osteochondral allograft transplantation, have advanced significantly in the past decade,1-4 these techniques are limited by their disadvantages. Complete regeneration of the articular cartilage is still debated and remains an unmet goal at present despite the achievement of good histological results. Furthermore, no technique has arisen as superior to the others.
Transforming growth factor-ß (TGF-ß) stimulates proteoglycan synthesis in immature cartilage and is a strong blocker of chondrocyte terminal differentiation. 5 In a prior study, TGF-ß could not prevent the loss of proteoglycan in adult cartilage; however, it blocked the upregulation of the Col10a1 gene when combined with compressive loading. 6
Surgical treatment of an OCD should restore the structure and function of hyaline cartilage and ultimately prevent osteoarthritis. 7 The quality of repaired tissue is known to be related to clinical outcomes.8,9 Particulated autologous cartilage transplantation (PACT) is an emerging treatment method in the repair of articular cartilage to surmount the current shortcomings of poor long-term outcome for BMS and cost for cell-based cartilage repair. This method was first introduced by Lu et al., 10 in 2006, in a study in which autologous cartilage was particulated and transplanted into an animal chondral defect model. The authors of this study reported consequent chondrocyte migration and outgrowth, and indicated that particulated cartilage could be an abundant source of cartilage for redistribution. The surge in interest in this novel strategy has been stimulated by subsequent convincing clinical and preclinical studies, and the need for a more cost-to-benefit treatment for articular cartilage lesions.11-15 This technique uses autologous hyaline cartilage harvested from the trochlear border or intercondylar notch in a knee joint for transplant to the curetted lesion site. In a subsequent study, Christensen et al. 14 compared PACT with BMS, and reported that PACT resulted in superior regeneration of hyaline cartilage, less fibrous tissue components, and improved histologic scores. The authors further compared the efficacy of PACT with that of PACT with BG and BG alone, and concluded that PACT with BG showed a significantly lower fraction of fibrous tissue and improved histological scores. 12
Despite its clinical promise, BG requires sacrifice of the donor site and is also cumbersome and difficult to implement clinically. In addition, successful subchondral bone integration following autologous chondrocyte implantation in shallow OCD without a BG has been recently reported.16,17 In this study, we compared the efficacy of PACT with control and recombinant human TGF-β (rhTGF-β) as a positive control, and elucidated the necessity of BG when performing PACT for the treatment of shallow OCDs in a large animal model. We hypothesized that PACT would be superior to controls and rhTGF-β treatment, and the outcome of PACT alone would be comparable to that of PACT with BG.
Methods
Two skeletally mature male micropigs were included in this study. On the day of surgery, the pigs weighed 37.0 and 49.2 kg and were aged 19.4 and 21.0 months, respectively. This study was approved by the Institutional Animal Care and Use Committee of the investigation. All procedures were conducted in compliance with the ARRIVE (Animal Research: Reporting of in vivo Experiments) guidelines (https://arriveguidelines.org/arrive-guidelines) and all possible efforts to minimize the number of animals used and their suffering were made. Four cylindrical OCDs, 6 mm in diameter and 8 mm in depth, were created in the medial and lateral trochlea of each knee, resulting in a total of 16 defects. The defects in each knee were treated with either (1) empty control, (2) PACT, (3) PACT with BG, or (4) recombinant human transforming growth factor-β3 (rhTGF-β3, CGbio, Seoul, Korea), and follow-up was performed at 2 months.
Surgery and Follow-Up
The 2 pigs were anesthetized by intramuscular injection of 5 mg/kg zolazepam (Zoletil 50, Virbac Korea, Seoul, Korea) and 2 mg/kg xylazine hydrochloride (Rompun, Bayer, Leverkusen, Germany) after fasting for 12 hours, and abstinence from water for 6 hours. After intubation, surgical monitoring was performed under general anesthesia with 1.5% to 2.0% isoflurane (Piramal Critical Care, Inc., Bethlehem, PA), using the Primus anesthesia machine (Drager Medical, Lubeck, Germany). Under general anesthesia, a medial parapatellar incision was made over the knee, and the patella was everted. The femoral trochlea was exposed, and 4 OCDs (2 longitudinal defects each in the medial and lateral trochlea) were created, using an Osteochondral Autograft Transfer System (Arthrex, Naples, FL). In the control group, the lesions received no further treatment. In the PACT group, the cartilage from the defect was particulated with a scalpel to pieces of approximately 0.5 mm3. Those particulated cartilages were embedded in the defect, flush with the adjacent cartilage surface. In the PACT with BG group, the harvested cancellous bone from the defect was chipped into a small piece and packed in the defect in alignment with the adjacent subchondral surface. Additional PACT was performed as previously described. In the TGF-β group, 0.05 ml (60 μg/defect) out of 1.23 mg/ml rhTGF-β3 was pipetted and inserted into the defect (
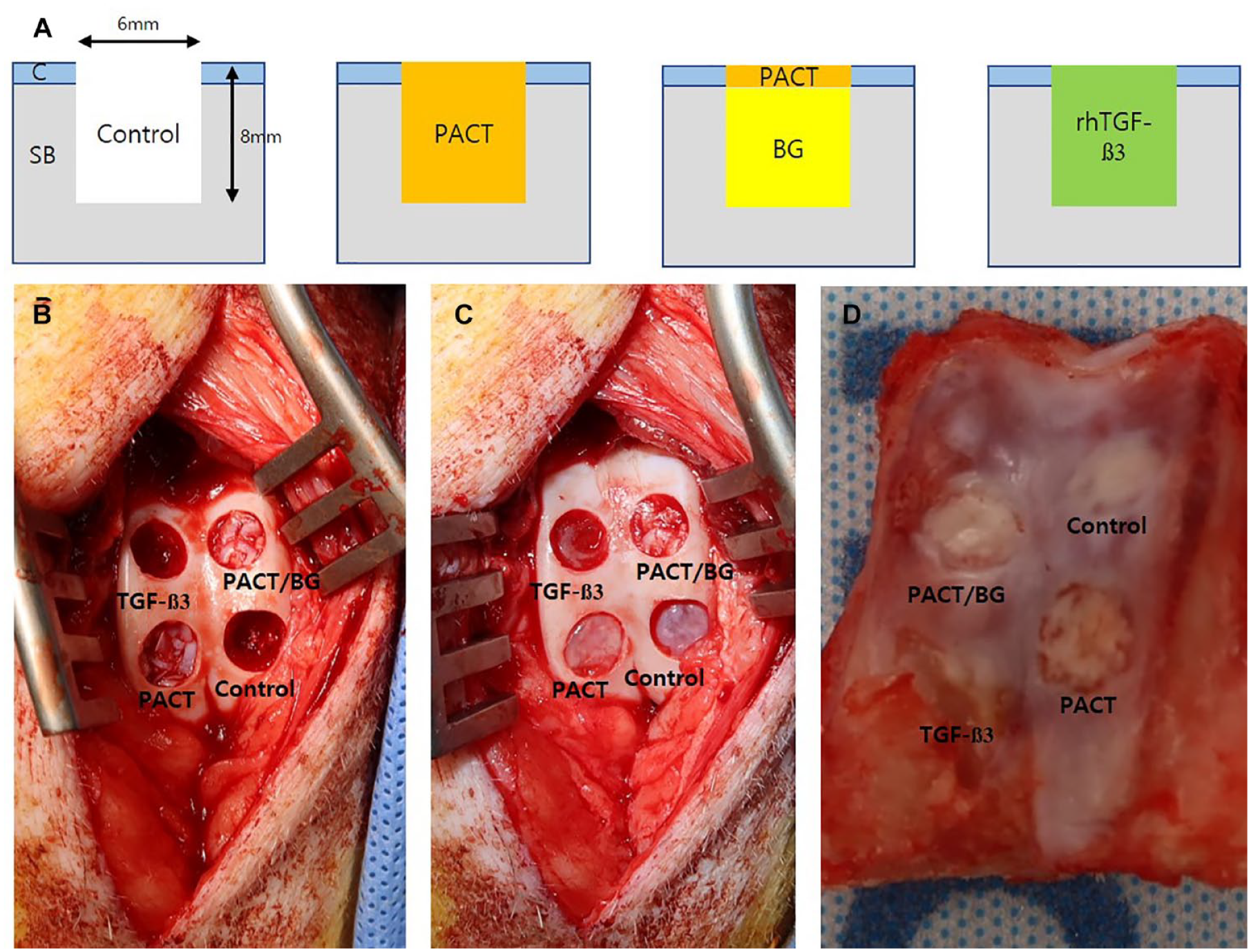
Cartilage regeneration in a porcine osteochondral defect model. (
Magnetic Resonance Imaging
To evaluate the quality of the repaired tissue, each pig was scanned by MRI before euthanasia under general anesthesia at 2 months, using a 3-T whole-body MRI scanner (Magnetom Skyra; Siemens, Erlangen, Germany) and a flexible 18-channel body coil (Siemens). T1-weighted and T2-weighted images were obtained using the following sequences: Turbo Spin Echo (TSE, field-of-view [FOV] 140 × 140 mm2; voxel size 0.2 × 0.2 × 3 mm3; 26 slices; TR 1190 ms; TE 11 ms; flip angle 150°; acquisition time 10 minutes 16 seconds) and TSE (FOV 140 × 140 mm2; voxel size 0.2 × 0.2 × 3 mm3; 26 slices; TR 7610 ms; TE 69 ms; flip angle 150°; acquisition time 8 minutes 24 seconds). In addition, T1 and T2 maps were measured from the following sequences: 3D Fast Low Angle Shot (FLASH, FOV 140 × 140 mm2; voxel size 0.2 × 0.2 × 2.5 [with gap 0.5 mm between 2 slices] mm3; 26 slices of 1 slab; TR 15 ms; TE 2.87 ms; 2 flip angles 5 and 26°; acquisition time 6 minutes 3 seconds) and 2D FLASH (FOV 140 × 140 mm2; voxel size 0.2 × 0.2 × 3 mm3; 26 slices; TR 1,060 ms; 5 TEs 4.36, 11.90, 19.44, 26.98 and 34.52 ms; flip angles 60°; acquisition time 13 minutes 36 seconds). An independent radiologist evaluated the images, and the magnetic resonance observation of cartilage repair tissue (MOCART) score was used (100, best; 0, worst) in the analysis. 18
Histology
Samples obtained from OCDs and micromass pellets were fixed for 14 days in 10% neutral formalin. After fixation, samples were decalcified in 0.5 M ethylenediaminetetraacetic acid (EDTA; pH 7.4) solution for 3 weeks at room temperature. The decalcified specimens were then embedded in paraffin and sectioned. Paraffin sections were deparaffinized, rehydrated, washed with phosphate-buffered saline, stained with hematoxylin-eosin (H&E) Safranin O (SafO) solution, and incubated with anti-collagen II (Abcam, ab34712) to evaluate the regeneration efficacy. The stained sections were investigated under a VS 120 virtual microscope (Olympus, Tokyo, Japan). Immunoreactive samples were visualized under an inverted fluorescence microscope (IX-71; Olympus, Tokyo, Japan). The O’Driscoll histological scoring system was used for histological assessment, and 3 independent experts assessed the quality of cartilage regeneration. 19
Statistical Analysis
All results are presented as the mean and standard deviations. Statistical analysis was performed using the commercial software SPSS (Version 22.0; IBM Corp, Armonk, NY). The Mann-Whitney test was used to detect differences between the 2 groups. Kruskal-Wallis analysis with subsequent Tukey’s post hoc analysis was performed using ranks. Correlations between MRI parameters and histology were further estimated using Spearman’s correlation coefficient (r) analysis. Spearman’s rho was interpreted as little (± <0.3), low (± 0.3-0.5), moderate (± 0.5-0.7), high (± 0.7-0.9), and very high (± >0.9). 20 Values were considered statistically significant at *P < 0.05 and **P < 0.01.
Results
Both model pigs completed the 2-month follow-up without any postoperative complications.
Magnetic Resonance Imaging
The postoperative MOCART scores ranged from 25 to 90 points, with a mean of 49.2 ± 18.4. The MOCART scores of each group were 41.7 ± 2.9, 66.7 ± 20.2, 58.3 ± 14.4, and 30.0 ± 5.0 in the control, PACT, PACT with BG, and rhTGF-β groups, respectively. A significant difference was found among the 4 groups on the Kruskal-Wallis analysis (P = 0.021) and post hoc analysis revealed significant differences between the PACT and TGF-β groups (P = 0.032;
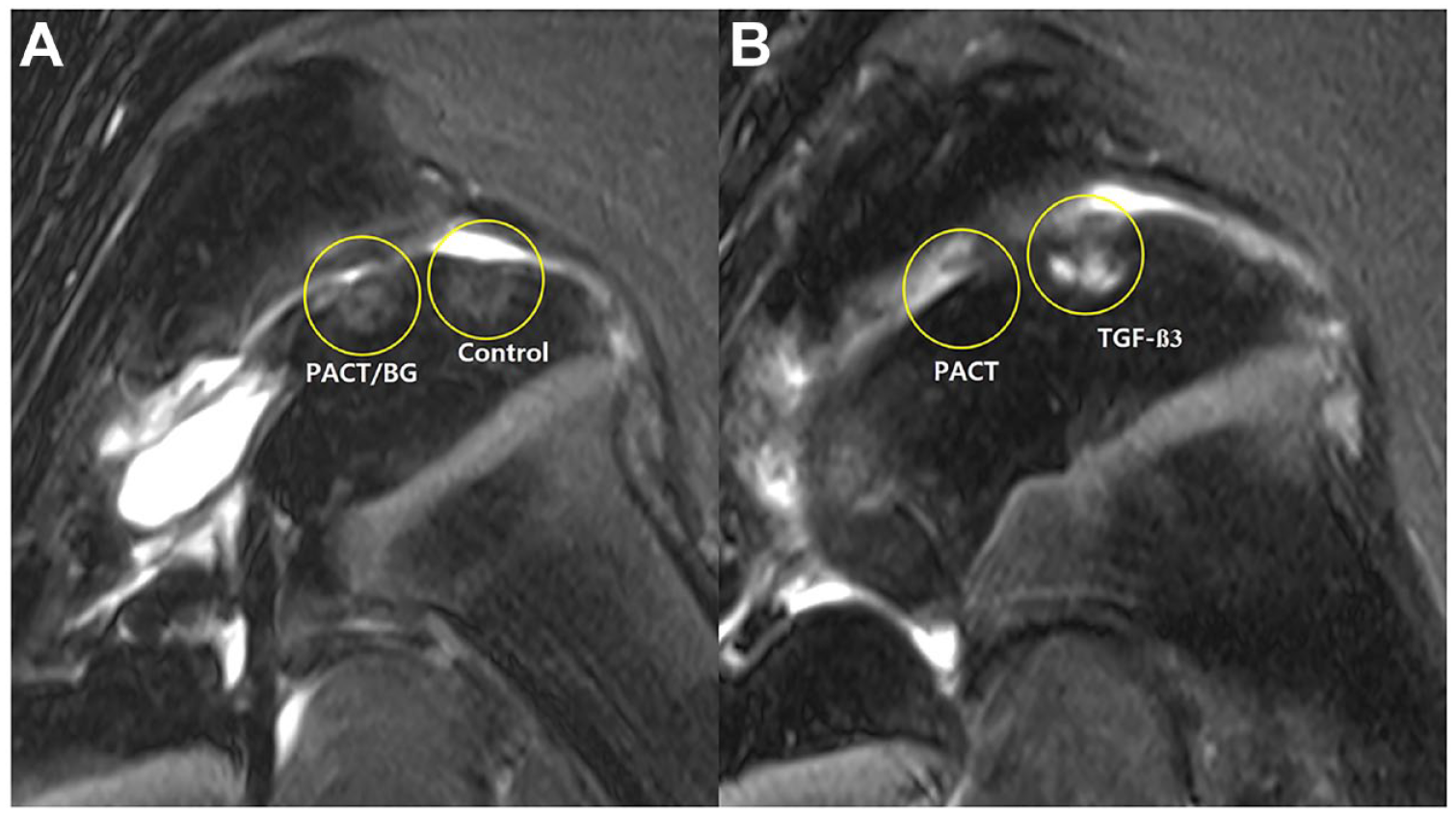
(
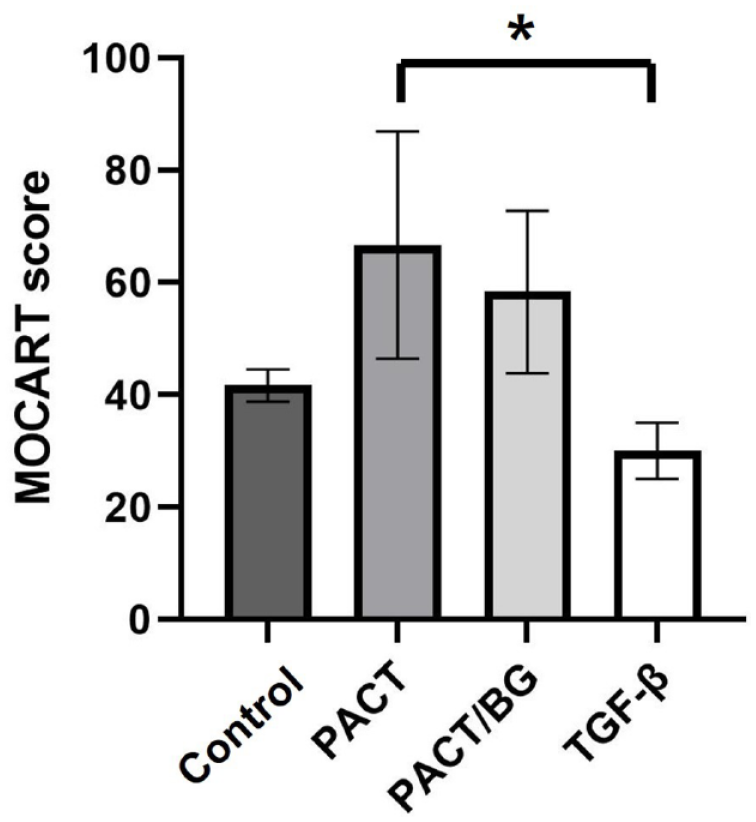
MOCART score (n = 4). Kruskal-Wallis analysis showed significant differences among the groups (P = 0.021). Post hoc analysis revealed significant differences between the PACT and TGF-β groups (P = 0.032). MOCART = magnetic resonance observation of cartilage repair tissue; PACT = particulated autologous cartilage transplantation; TGF = transforming growth factor; BG = bone graft.
Histologic Analysis
All groups showed good filling of the defects; however, regeneration of complete hyaline-like cartilage was limited in all treatment groups. H&E staining of the repaired tissue of the chondral defect further showed more fully integrated subchondral cancellous bone in the PACT with and without BG groups than in the control or rhTGF-β group (
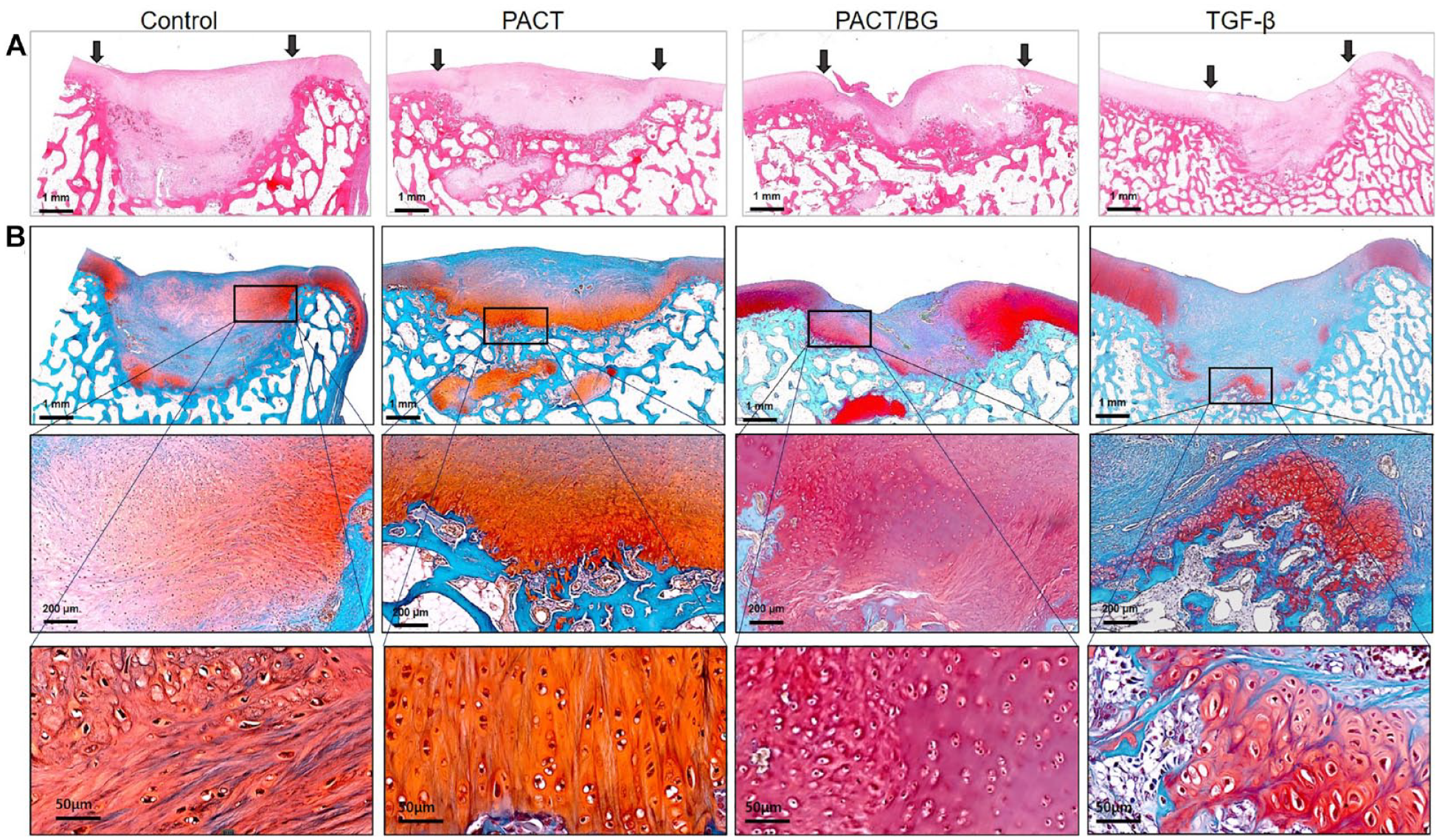
Histological examination of osteochondral defects (n = 4). (
The PACT with and without BG groups both showed a considerable increase in glycosaminoglycan (GAG) synthesis on SafO staining compared with the other groups. The quality of tissues was assessed with enlarged sections and chondrocytes in the PACT with and without BG groups were more consistent and uniform in shape than those in the other groups. Both groups showed chondrocyte-like cells on the surface of repaired cartilage (
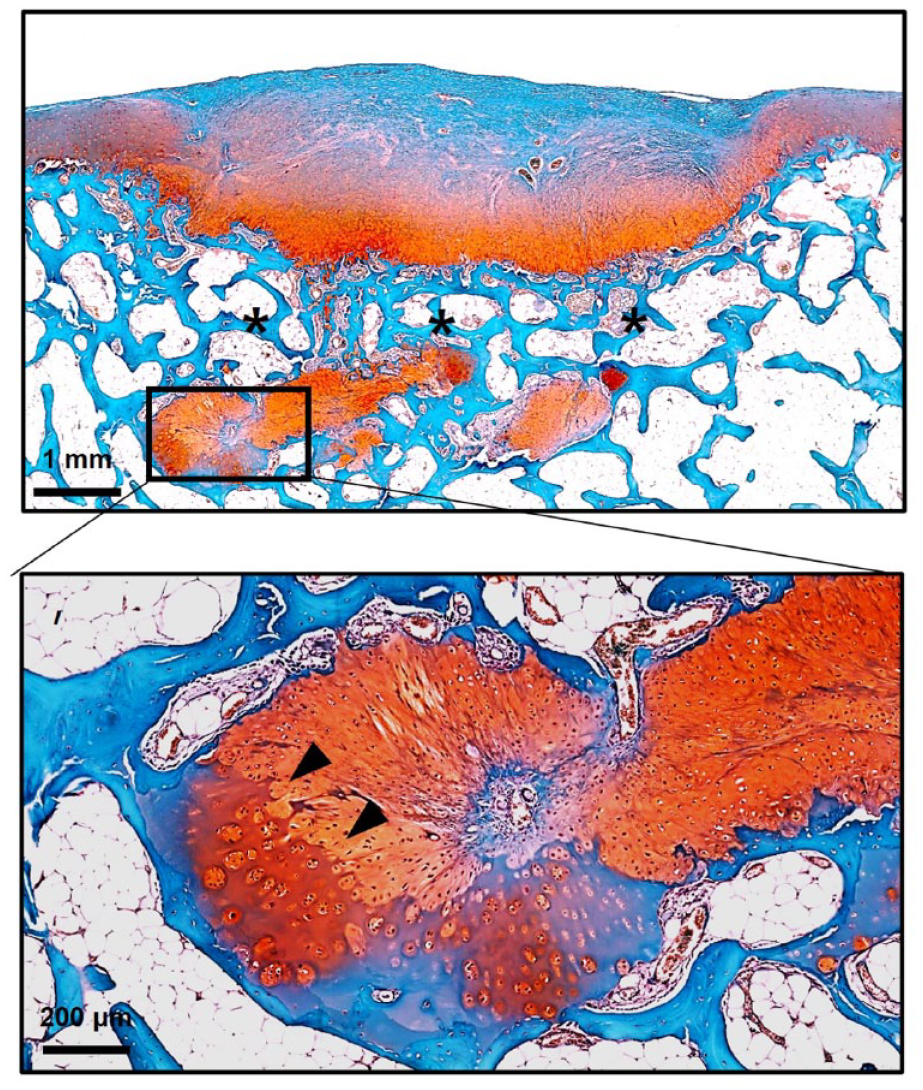
Safranin O staining. Histological section of the osteochondral defects treated with PACT. The cancellous bone was well-formed above the transplanted cartilage (asterisks). Chondrocytes at the border of the implanted particulated cartilage are hypertrophied (arrow heads). PACT = particulated autologous cartilage transplantation.
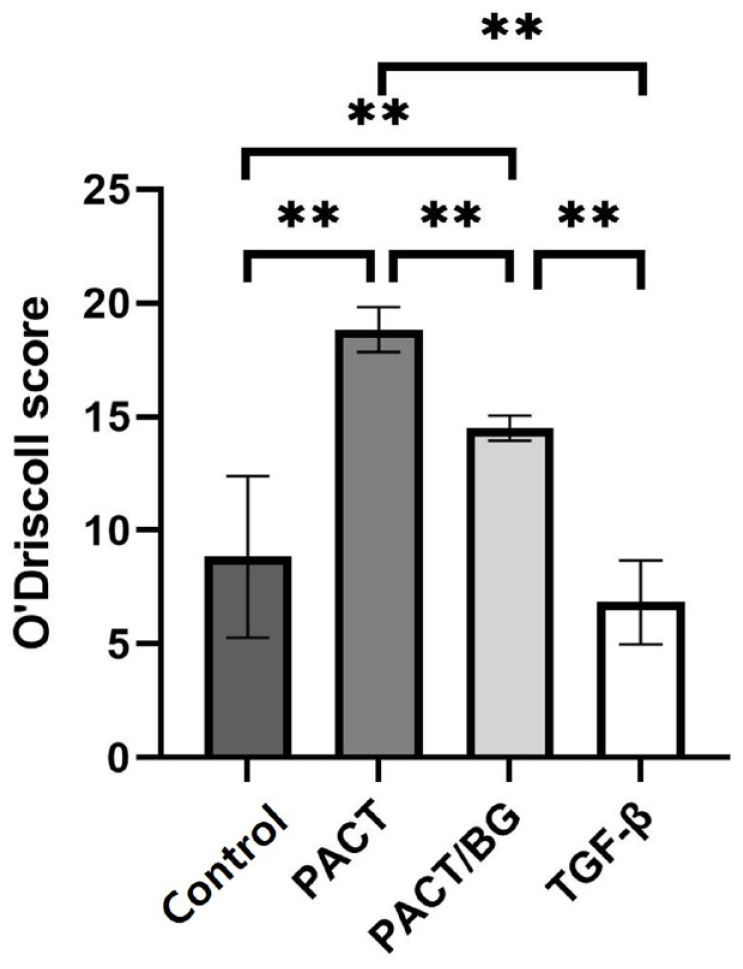
O’Driscoll histological score (n = 4). A quantitative analysis of articular cartilage regeneration was performed. Three blinded investigators scored the tissue quality of the repaired cartilage defects and the scores are shown as the mean value ± SD (P < 0.001, Kruskal-Wallis; **P < 0.008, post hoc). PACT = particulated autologous cartilage transplantation; BG = bone graft; TGF = transforming growth factor.
Expression of extracellular matrix proteins in the repaired cartilage was assessed using type II collagen by immunohistochemistry. The PACT without BG group showed a higher expression level of type II collagen than the other groups (
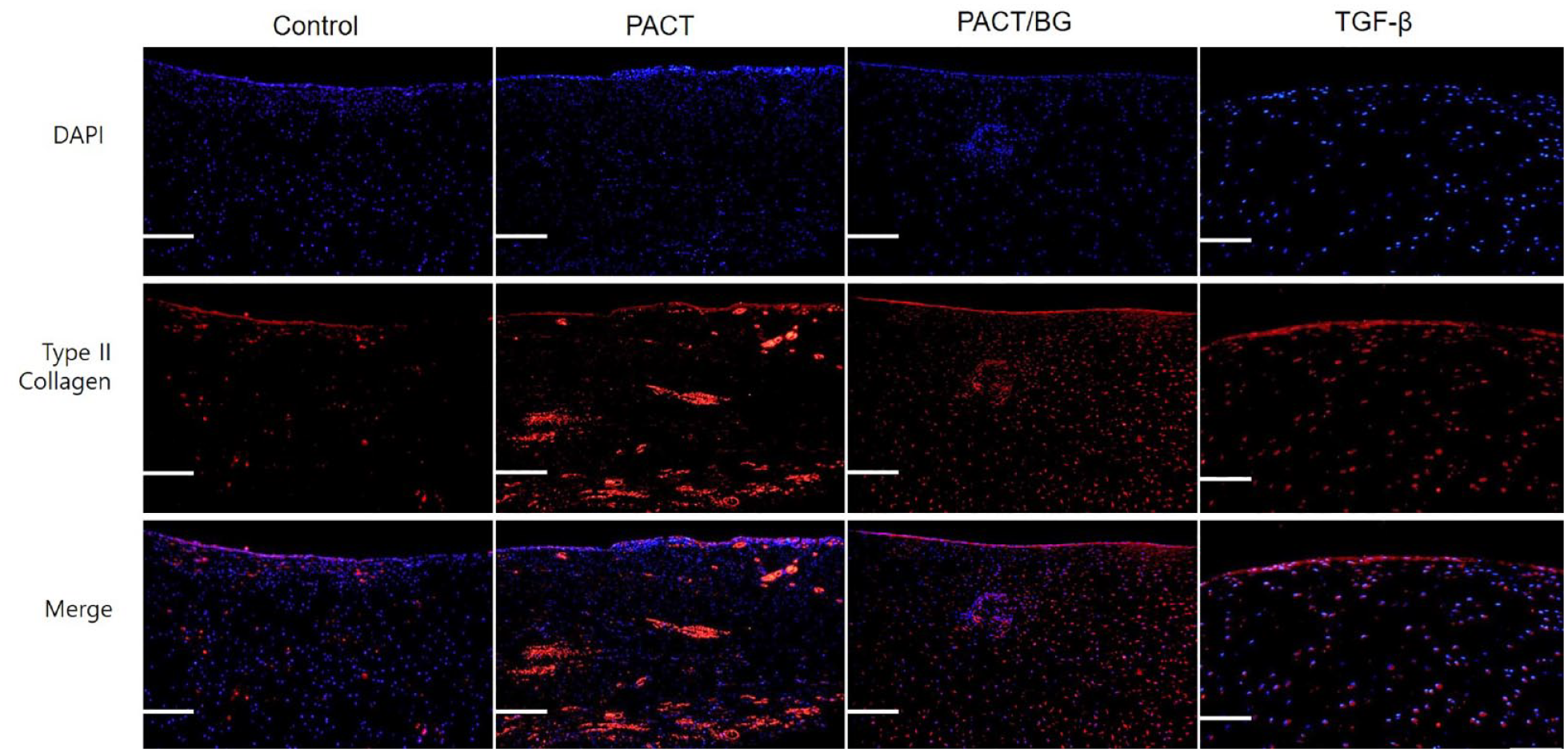
Immunofluorescence analysis of the repaired cartilage tissues; magnification, x20/ scale bar = 200 μm. Analysis was performed using antibodies of type II collagen (Abcam, ab34712: red fluorescence). Cell nuclei were stained with DAPI (blue fluorescence). DAPI = 4′,6-diamidino-2-phenylindole; PACT = particulated autologous cartilage transplantation; BG = bone graft; TGF = transforming growth factor.
Discussion
In this study, we observed improved early phase repair of OCDs driven by PACT with and without BG as compared with empty defects and defects treated with TGF-β. We believe that quite short-term follow-up could have revealed the hypertrophic chondrocytes on the border of the chip, indicating reactive differentiation to cancellous bone. This suggests the efficacy of PACT in driving an efficient repair even when the defect is filled by PACT without BG, due likely to the endochondral recapitulation of bone formation at the bottom of the defects starting from the particulated cartilage. As a consequence, it could be hypothesized that a better and more efficient final repair of OCDs could be achieved even when treated simply with PACT and fibrin glue without BG. Thus, PACT alone may represent a promising alternative in the treatment of cartilage lesions and OCDs up to 8 mm in depth.
The use of autologous particulated cartilage for repair was first introduced in 1983, and has recently gained significant attention.10,12,14,21 Prior preclinical investigations showed that articular chondrocytes from particulated cartilage are not significantly damaged if cut using a sharp scalpel and are able to migrate from their initial matrix to form a novel extracellular matrix.22-26 Chondrocytes that are brought to the surface of their marginal matrix start to proliferate again, which has been shown to spontaneously heal small cartilage lesions. 27 Moreover, the degree of mincing is also important; when particulated cartilages are too large, a limited number of chondrocytes are activated and large pieces may not harmonize well with repaired cartilage. Bonasia et al. 28 showed that a paste-like appearance of particulated cartilage would provide an optimal environment for the regeneration of high-quality tissue.
The mechanism underlying cartilage regeneration was initially thought to involve the outgrowth of chondrocytes from particulated cartilage and subsequent extracellular matrix production10,29; however, the relevance of cellular outgrowth from cultured adult cartilage tissue was not found in human cartilage samples in a recent study. 30 In this study, however, the authors explained that as the normal cartilage portion of a patient undergoing total joint replacement for knee arthritis was taken, the results may be different in normal young cartilage. Similarly, Christensen et al. 12 showed that the fraction of hyaline cartilage in the autologous BG combined with particulated cartilage group was 25.8% and 20.1% at 6 and 12 months, respectively, which was not significantly different. In this study, the authors concentrated on the unchanged or decreased hyaline cartilage fraction that might not be explained by chondrocyte outgrowth and cell proliferation, which would have increased the hyaline tissue fraction. Instead, they proposed a possible mechanism involving paracrine stimulation of the transplanted tissue.
In this study, the quality of repaired cartilage was significantly higher in the PACT than in the PACT with BG group, with comparable quality of subchondral bone regeneration. Bone defect reconstruction through endochondral ossification has been studied as a promising route.31-34 Hypertrophic cartilage tissue from human adipose tissue was implanted subcutaneously into mice and a bony shell was expressed around bone trabeculae inside after 12 weeks. 33 Kim et al. 34 utilized spheroid model reconstructed by prechondrocyte cells to recapitulate endochondral ossification and reported hypertrophic chondrocyte differentiation of the prechondrocyte cells especially in the surface region of the spheroid. Hypertrophied chondrocytes at the border of the implanted particulated cartilage in this study could achieve similar results to the aforementioned studies showing endochondral ossification. In addition, these results are believed to occur as an effect of short-term follow-up, which is both an advantage and disadvantage of this study.
We further found that the exogenous addition of TGF-β to the defect did not induce significantly superior cartilage quality compared with nontreatment. TGF-β family proteins are generally recognized as key initiators of chondrogenesis that are locally produced and stored in the cartilage. 35 TGF-ß in joint tissue is either low or load-activated under physiological conditions; however, it increases as inflammation arises, and cartilage degradation is expected to induce TGF-ß release. Furthermore, osteoblasts from the subchondral bone in damaged joints show increased TGF-ß expression, which is thought to increase the severity of osteoarthritis.36,37 Transgenic mice overexpressing TGF-ß, especially in osteoblasts, developed mandibular condyle cartilage degradation. 38 These high concentrations of TGF-β preferentially activate SMAD 1/5/8 phosphorylation, consequently triggering chondrocyte hypertrophy, synovial fibrosis, subchondral sclerosis, and osteophyte formation.39-41 Thus, damaged subchondral BGs in addition to PACT could have increased TGF-ß expression compared with PACT alone in this study, with negative effects on cartilage regeneration. Similarly, exogenous addition of rhTGF-β may have adversely affected cartilage and subchondral bone regeneration.
In this study, the quality of repaired cartilage was slightly higher with PACT alone than with BG according to the results of GAG and type II collagen synthesis assays, whereas the repaired subchondral bone structure of PACT was comparable to that of PACT with BG. The superior repaired cartilage quality with PACT is assumed to be due to the direct interaction between a sufficient number of transplanted autologous particulated cartilages and mesenchymal stem cells from the defect. The cancellous bone was well-formed above the transplanted particulated cartilage, and chondrocytes at the border of the implanted particulated cartilage were hypertrophied, suggesting recapitulation of endochondral ossification (
Our study was predominantly limited by its small sample size and short observation period. The complete reconstruction of the defect may further have been limited due to the short-term follow-up. Bone transplants most often undergo creeping substitution involving the appositional bone formation phase followed by a resorption phase. It may be that we may have been able to observe an even better repair if the defects had been followed for a longer time. However, the short observation period could have been a strong point to reveal the transitional recapitulation of endochondral ossification of PACT. The use of bilateral joints and multiple defects per knee was unavoidable due to the limitation of large animals; however, both elements are known to have no significant effect on the repair response or interactions between the lesions.13,47 Parametric statistical analysis was restricted by the small number of lesions in each treatment group; however, the consistency of outcome within groups appears to be significant. In addition, if the grafted bone particles and particulated cartilages were stratified strictly from each other, the quality of chondrogenesis in PACT with BGs could have been changed. The anti-chondrogenic effect of unseparated signals from the grafted cancellous bone could have further affected the quality of the repaired cartilage. Further studies regarding the stratification, size of the BG, and amount of PACT may be able to distinguish this difference. Third, different mechanical loadings on the medial and lateral trochlea may reflect variations in gene expression.48,49 In our study design, we randomized and applied each treatment to the defect alternately; however, differences in the characteristics between trochlea could have influenced consequent cartilage regeneration. Fourth, our study lacks an in vitro study that could directly support our assumptions about the possible roles of elevated TGF-β levels and chondrocyte differentiation. Nevertheless, the results coincide with those of previous studies that we have mentioned.
Conclusion
Treatment of OCDs with PACT improved repair tissue quality compared with the control or rhTGF-β groups 2 months after treatment in a porcine model. The short observation period is thought to have allowed us to observe the recapitulation of endochondral ossification driven by PACT and the possible interaction with resident mesenchymal stem cell. BG, in addition to PACT, might be unnecessary to treat shallow OCDs.
Supplemental Material
sj-pdf-1-car-10.1177_19476035231199442 – Supplemental material for Osteochondral Repair with Autologous Cartilage Transplantation with or without Bone Grafting: A Short Pilot Study in Mini-Pigs
Supplemental material, sj-pdf-1-car-10.1177_19476035231199442 for Osteochondral Repair with Autologous Cartilage Transplantation with or without Bone Grafting: A Short Pilot Study in Mini-Pigs by Dong Woo Shim, Kyoung-Mi Lee, Donghyun Lee, Jun Sik Kim, Yeon Seop Jung, Sung Suk Oh, Si Wook Lee, Jin Woo Lee and Bom Soo Kim in CARTILAGE
Supplemental Material
sj-pdf-2-car-10.1177_19476035231199442 – Supplemental material for Osteochondral Repair with Autologous Cartilage Transplantation with or without Bone Grafting: A Short Pilot Study in Mini-Pigs
Supplemental material, sj-pdf-2-car-10.1177_19476035231199442 for Osteochondral Repair with Autologous Cartilage Transplantation with or without Bone Grafting: A Short Pilot Study in Mini-Pigs by Dong Woo Shim, Kyoung-Mi Lee, Donghyun Lee, Jun Sik Kim, Yeon Seop Jung, Sung Suk Oh, Si Wook Lee, Jin Woo Lee and Bom Soo Kim in CARTILAGE
Footnotes
Author Contributions
D.W.S., J.W.L., and B.S.K. made substantial contributions to the design of the work; the acquisition, analysis, and interpretation of data; drafting the work; final approval; and agreement to be accountable for all aspects of the work. K.M.L. made substantial contributions to the analysis and interpretation of data, drafting the work, final approval, and agreement to be accountable for all aspects of the work. D.L., J.S.K., Y.S.J., and S.S.O. made substantial contributions to the acquisition and analysis of data, drafting the work, final approval, and agreement to be accountable for all aspects of the work. S.W.L. made substantial contributions to the design of the work and acquisition of data, drafting the work, final approval, and agreement to be accountable for all aspects of the work.
Clinical Relevance
Define the necessity of bone graft (BG) while performing particulated autologous cartilage transplantation (PACT).
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors feel grateful to CGbio (Seoul, Korea) for supporting recombinant human transforming growth factor-β3 (rhTGF-β3) and laboratory animals. One of the authors (D.W.S.) has received funding from National Research Foundation of Korea (NRF) grant funded by the Korea government (Ministry of Science and ICT [MSIT]; NRF-2022R1F1A1071373). Funding was utilized in the analysis, interpretation of data, and writing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All the authors have read and approved the final manuscript and the data being presented in the manuscript. This study was approved by the Institutional Animal Care and Use Committee of Daegu-Gyeongbuk Medical Innovation Foundation (Approval number: DGMIF-21091303-00).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
