Abstract
Objective
The cartilage regeneration field has not yet overcome the issue of effective “shaping”: growing regenerated cartilage in the desired shape, and maintaining that shape, is problematic. This study reports on a new method of cartilage regeneration in which the cartilage is shaped in three dimensions. Since cartilage is composed only of cartilage cells and an abundant extracellular matrix with no blood circulation, once it is damaged, the lack of nutrient supply means that it is difficult to repair. Scaffold-free cell sheet technology plays an important role in cartilage regeneration, avoiding inflammation and immune response caused by scaffold materials. However, cartilage regenerated from the cell sheet needs to be sculpted and shaped before it can be used for cartilage defect transplantation.
Design
In this study, we used a new ultra-strong magnetic-responsive Fe3O4 nanoparticle (MNP) to shape the cartilage in vitro. Super-magnetic Fe3O4 microspheres are manufactured by co-assembling negatively charged Cetyltrimethylammonium bromide (CTAB) and positively charged Fe3+ under solvothermal conditions.
Results
The Fe3O4 MNPs are swallowed by chondrocytes, and the MNP-labeled chondrocytes are acted upon by the magnetic field. The predetermined magnetic force makes the tissues coalesce to form a multilayer cell sheet with a predetermined shape. The shaped cartilage tissue is regenerated in the transplanted body, and the nano magnetic control particles do not affect cell viability. The nanoparticles in this study improve the efficiency of cell interaction through super-magnetic modification, and to a certain extent change the way the cells absorb magnetic iron nanoparticles. This phenomenon allows a more orderly and compact alignment of the cartilage cell extracellular matrix, promotes ECM precipitation and cartilage tissue maturation, and improves the efficiency of cartilage regeneration.
Conclusion
The magnetic bionic structure, which contains specific magnetic particle-labeled cells, is deposited layer by layer to generate a three-dimensional structure with repair function, and further induce the production of cartilage. This study describes a new method for the regeneration of tissue engineered cartilage which has broad application prospects in regenerative medicine.
Introduction
Scaffold-free cell sheet technology is considered a promising method for cartilage regeneration.1,2 Since cartilage is composed only of cartilage cells and an abundant extracellular matrix with no blood circulation, once it is damaged, the lack of nutrient supply means that it is difficult to repair. Tissue engineered cartilage is not limited by the defects of scaffold materials. It is mainly composed of cell and cell connections and extracellular secretion matrix (ECM) protein 3 and produces cartilage tissue similar to natural components. Compared with traditional scaffold-cell composite materials, autologous cell sheet technology shows higher regeneration ability, 4 and obviates material-induced problems such as inflammation, immune response, degradation rate, and biocompatibility. However, it is difficult to precisely regulate and control the thickness and shape of the sheet when using this method, and it is impossible to imitate the complex tissue structure in vitro. By contrast, the magnetic manipulation method based on magnetic nanoparticles (MNPs) is a new technique in this field and has a number of advantages for in vitro shaping. Controllable MNPs have been used in biomedical applications of external magnetic fields in vivo and in vitro in the past few decades5,6; indeed, nanotechnology plays an increasingly important role in many aspects of human life. MNPs have unique biomedical applications. The absorption of nano-level particles by cells is a fast and easy process. 7 In addition, super-strong magnetism means that cells can be stacked layer by layer to form more cell sheets than in ordinary cell sheets.
ECM is a natural scaffold material that is secreted and precipitated by cells. 8 It provides an ideal microenvironment for the tissue-specific physical and molecular structures of differentiated cells, and for the regulation and transmission of cell information. 9 Cartilage is composed of abundant ECM and chondrocytes distributed around it. Cartilage ECM is mainly composed of type II collagen and large proteoglycan networks, including glycosaminoglycans (GAGs), which play a key role in regulating chondrocyte function.10,11
In this system, the cells in each layer of the cell sheet can control ECM deposition through mediated magnetic force. According to the morphological characteristics of the defect tissue, we can construct and preserve cell 3D distribution and their ECM proteins in this artificial microenvironment to maintain specific tissue morphology (Scheme 1). Owing to the good biocompatibility of Fe3O4 nanoparticles, their effects and functions in cells—and indeed in the entire organism—have drawn the attention of researchers. The new Fe3O4 super MNPs we developed have excellent biocompatibility and biological activity and can be widely used in biomedical research.
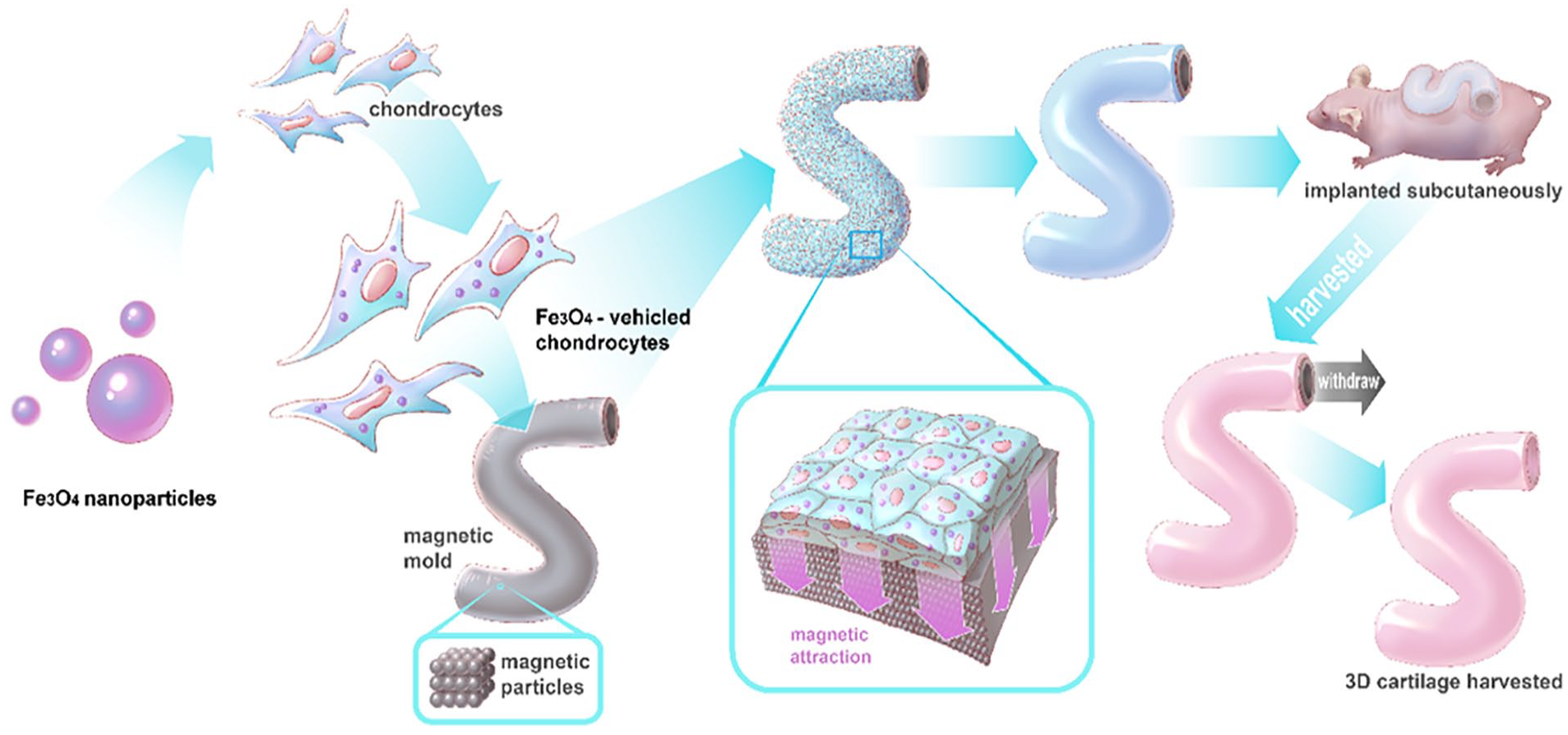
Magnetically controlled 3D-shape cartilage regeneration via chondrocytes endocytizing ultra-strong magnetic-responsive Fe3O4 nanoparticles and attracted to the 3D-shape magnetic mold surface.
Methods
All methods were carried out in accordance with the relevant guidelines and regulations.
Synthesis of Ultra-Strong Magnetic-Responsive Fe3O4 Microspheres
The ultra-strong magnetic-responsive Fe3O4 microspheres were prepared in the same way as in our previous work with slight modifications. In a typical procedure, 1.0 g of FeCl3, 4.0 g of NaAc, and 0.35 g of Cetyltrimethylammonium bromide (CTAB) was first dissolved in 40 ml of ethylene glycol (EG) and thoroughly mixed. Approximately 30 min later, a homogeneous brown solution was formed and delivered to a Teflon-lined autoclave where a temperature of 200°C was maintained for a whole day. After being cooled to room temperature, the products were collected, cleaned three times using deionized water and ethanol, and then dried at 60°C for 24 h.
Using Cu-Kα radiation (λ = 1.5406 Å) from 10° to 80° at a scanning speed of 0.33° min−1, a Bruker AXS micro-diffractometer (D8 ADVANCE; Germany) was applied to record the sample X-ray diffraction (XRD) patterns. A vibrating sample magnetometer (VSM; PPMS-9 T [EC-II], USA) at 300 K was used to measure the magnetization versus magnetic field curves. A field emission scanning electron microscope (FESEM; Germany) and a transmission electron microscope (TEM; JEM-2010, Japan) were separately applied to observe the surface structure and morphology.
Chondrocyte Acquisition and Culture
Under aseptic conditions, the excess human ear cartilage resulting from ear surgery was removed, cut into pieces, digested with type II collagenase overnight, neutralized with DMEM (Hyclone, Logan, USA) containing 10% fetal bovine serum (FBS, Gibco, USA) and centrifuged. The supernatant was discarded, and it was then inoculated in a petri dish and placed in a saturated incubator with 5% CO2, 37°C, and 100% humidity. The medium was changed for the first time on the second day, and then every 3 days thereafter. When the cells had reached about 80% of full size, they were digested, suspended with 0.25% trypsin and then subcultured in a 1:3 ratio. Cell growth status was dynamically observed with an inverted phase contrast microscope, and the second generation chondrocytes used for the experiments.
Characteristics After Cell + MNP Co-Culture
After the cells were digested, centrifuged, and put into suspension, the original supernatant was discarded, and the MNPs—suspended in DMEM containing 10% FBS—were added. Blank controls were set respectively for concentrations of 5, 10, 50, 100, 200, 500, and 1,000 μg/ml, and incubated with chondrocytes in the incubator for 24 hours. After washing with phosphate buffered saline (PBS), and centrifuging, the supernatant was removed and the cell morphology observed under a light microscope. The supernatant of the original medium was then discarded, and two new sterile PBS washes were performed. The samples were then fixed with 4% paraformaldehyde for 30 min, washed twice with PBS, and stained with Prussian blue staining solution to observe the phagocytosis of iron-containing particles in the cells. Visible blue stained particles count as Prussian blue staining positives. For transmission electron microscopic observation, 200 μg/ml Fe3O4 MNP was cultured and incubated with chondrocytes. After 24 hours, the samples were collected and scanned under the TEM and photographed.
Cell Proliferation Detection
We used the Cell Counting Kit-8 (CCK-8, Dojindo Laboratories, Kumamoto, Japan) method as previously described. 12 On Days 1, 3, 5, 7, and 9 of MNP co-cultivation with different concentrations of cell markers, 10 μl of CCK-8 solution was added to each well of a 96-well plate and incubated in a 37°C incubator for 1 hour. The optical density value was measured with a microplate reader at 450 nm wavelength and the cell proliferation curve drawn. A blank control was set up, and medium added to eliminate background. Each group had three samples and was tested in triplicate.
Apoptosis Detection
After the chondrocytes were co-cultured with MNPs for 24 hours, they were digested with 0.25% EDTA-free trypsin for 2 minutes, transferred to a flow tube and centrifuged to discard the supernatant. We added 100 μl of binding buffer to each tube to resuspend the cells, and then Annexin V and PI staining solutions. We incubated the samples for 15 minutes at room temperature in the dark, and finally added 100 μl of binding buffer, mixed thoroughly, and then tested on the machine. In the blank control group, normal chondrocytes were taken, and only the binding buffer was added. Each group had three samples and was tested in triplicate.
Live and Dead Cell Stain Detection
In order to determine cell viability and the growth status of chondrocytes and MNPs after 1 week of co-culture, a live/dead cell staining kit was used. We prepared a working staining solution in PBS (pH = 7.4). Calcein-AM stains live cells green, and propidium iodide (PI) stains dead cells red. The cells were incubated in a working solution at 37°C in an incubator for 15 min, then the samples were washed with PBS, and imaging was performed using fluorescence excitation under an inverted fluorescence microscope. Each group had three samples and was tested in triplicate.
Characteristics of Cells + MNPs + Mold Co-Culture
Magnetron is one of the advantages of using MNPs for cell control and labeling. Based on our experimental results and efficiency considerations, we used Fe3O4 MNPs with a concentration of 200 μg/ml to label the chondrocytes. They were seeded in a magnetically controlled shaping mold and used in subsequent experiments and in vivo studies. We collected the second-generation chondrocytes, labeled the cells with a green fluorescent proteins (GFP) immunofluorescence kit, and observed the growth and distribution of MNPs after phagocytosis and plating on the magnetic mold.
In vivo Experiment and Histological Examination
In this research, animal operations were performed strictly in accordance with the animal research guidelines: reporting of in vivo experiments guidelines; and ethical principles approved by the Ethics Committee of Shanghai Ninth People’s Hospital. We confirm that all experimental procedures are in accordance with ARRIVE guidelines for the reporting of animal experiments. The samples were cultured in vitro and transplanted subcutaneously into 18 8-week-old male nude mice. In an experimental protocol approved by the Animal Ethics Research Committee, using inhalation anesthesia, Fe3O4 MNP-labeled chondrocyte complexes were implanted subcutaneously under anesthesia. After 8 weeks of transplantation, specimens were taken, and histological HE was performed to observe chondrocyte morphology. Special cartilage staining with Masson and Safranin O was used to observe the secretion and expression of glycosaminoglycans in the extracellular matrix. Immunohistochemical staining was used to detect the secretion and expression of extracellular matrix type II collagen expression.
Statistical Analysis
All data are presented in mean ± standard deviation (SD) format. One-way analysis of variance was performed and Tukey’s post hoc test used for statistical analysis. P < .05 represents a statistically significant difference.
Results and Discussion
Characterization of Ultra-Strong Magnetic-Responsive Fe3O4 Microspheres and Chondrocyte Biocompatibility Tests
In order to improve the chondrocyte labeling rate and the response to the Fe3O4 nanoparticle magnetic field, we used a new method to prepare the ultra-strong magnetic-responsive Fe3O4 microspheres: they were co-assembled with negatively charged CTAB and positively charged Fe3+ under solvothermal conditions. The process is attracted by the surface mutual electrostatic interactions of CTAB and Fe3+, and is followed by EG reduction. Both TEM and scanning electron microscopy (SEM) were subsequently used to examine the sample morphology. As Figure 1A shows, the morphology of the as-obtained products, and the particles, is spherical. The average size was ca. 200 nm and they were highly dispersed. Each ca. 8-nm-sized Fe3O4 microsphere consists of numerous interconnected nanoparticles. The many voids between the nanoparticles make the entire microsphere structure porous, a hollow structure composed of many densely arranged subunits. The higher magnification SEM image Figure 1B gives a clear picture of the morphology of the microsphere surfaces.
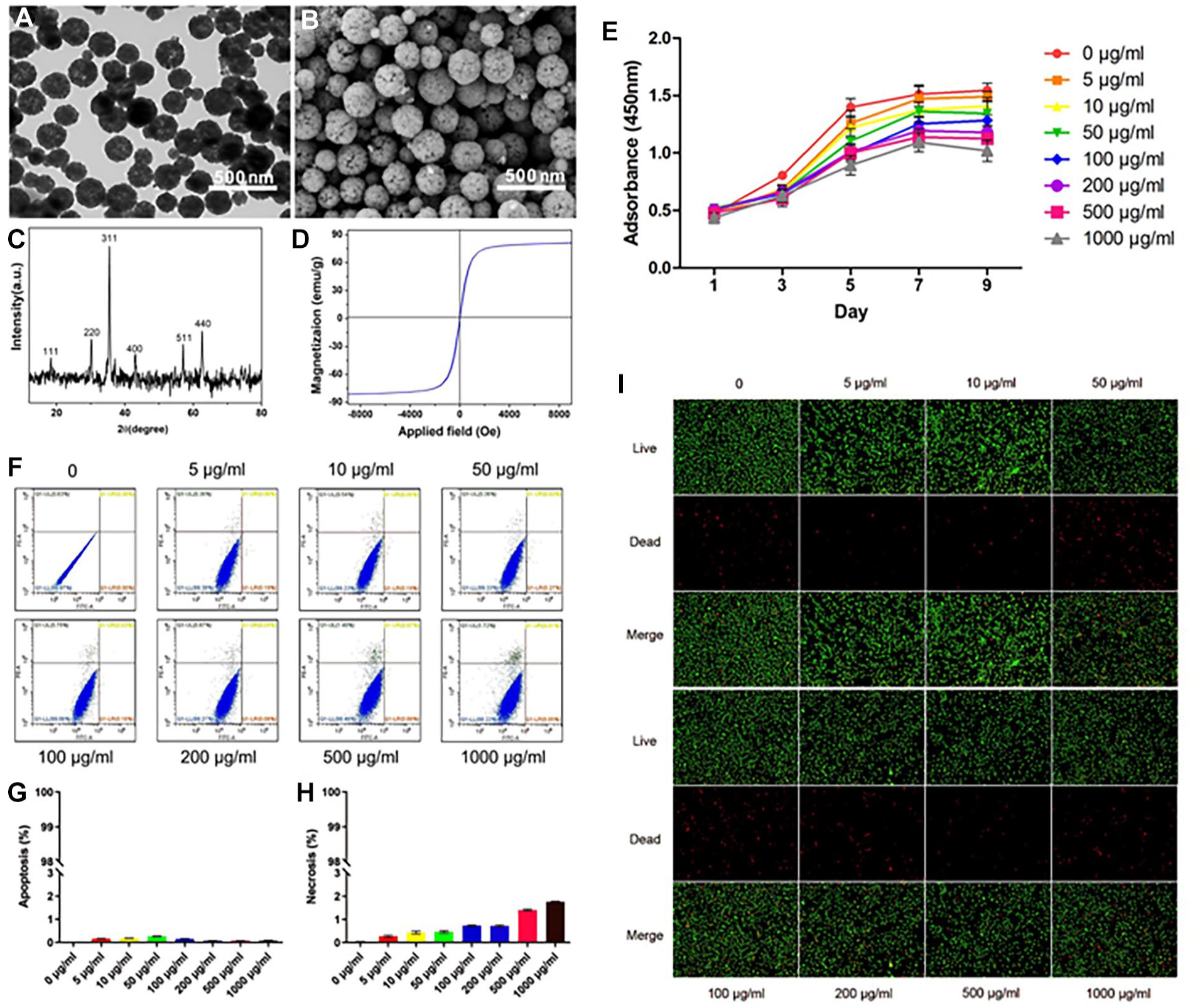
Synthesis and characterization of ultra-strong magnetic-responsive Fe3O4 microspheres and chondrocyte biocompatibility tests after culturing with Fe3O4 microspheres in different concentrations. (
The chemical composition and phase purity of the product were determined by XRD. As Figure 1C suggests, all diffraction peaks can be attributed to the Fe3O4 phase (JCPDS card no. 65-3107). No other impurities were observed. The crystalline size was estimated using the strongest diffraction peak (311). Calculated according to the Debye-Scherrer formula, the obtained grain size was about 8 nm, similar to the average grain size observed in the TEM image.
Figure 1D shows the magnetic curves of the prepared ultra-strong magnetic-responsive Fe3O4 microspheres. The results show that the Fe3O4 microspheres have typical superparamagnetic behavior without any hysteresis. Saturation magnetization reaches 83.4 emu·g−1, which is higher than other synthesized Fe3O4 microspheres. The strong magnetic performance of the Fe3O4 microspheres offers opportunities in nanomedicine and magnetic cell targeting. They are rightly considered to be an extremely promising application for the translation of cell-based therapies from the bench to the bedside.
In order to identify cell biocompatibility after the chondrocytes phagocytize the MNPs, cell proliferation, apoptosis, necrosis, cell live and dead tests were measured at different MNP concentrations. The CCK-8 method indicated that the chondrocytes gradually proliferated over time and grew well (
Annexin V-FITC was used in conjunction with PI to distinguish normal cells, necrotic cells, early apoptotic cells, and late apoptotic cells. Using this method of double staining combined with fluorescent labeling, flow cytometry was used to detect the cell state of chondrocytes and MNPs after 24 hours of treatment (
Apoptosis—a strictly controlled and programmed death method—is essential for maintaining cell homeostasis. Unlike cell necrosis, cell apoptosis is not accompanied by inflammation: cells are quickly cleared without affecting the overall function of the tissue. Excessive cell apoptosis, for chondrocytes, means cell necrosis, leading to the breakdown of the extracellular matrix and the loss of cartilage biological characteristics, which affects tissue remodeling. The more obvious the chondrocyte apoptosis, the worse the quality of cartilage regeneration. 13 Therefore, maintaining good cell viability is essential.
Unlike apoptosis, cell necrosis is a passive pathological death. We used direct fluorescent staining of live dead cells to see whether the environment caused by MNPs—and their own toxic effects on chondrocytes—lead to their death. What we observed was that the sheet permeability of dead cells increased and then ruptured. Irregular changes in cell appearance and cytoplasmic leakage can cause inflammation, destroying the microenvironment around the cells, and destroying tissue and degrading the cartilage matrix.14,15 Figure 1I shows the fluorescence images of live (green) and dead (red) cells after co-culture of chondrocytes and MNPs for 7 days. It can be seen that the cells were relatively evenly distributed in each concentration group, and only a small number of dead cells were observed. Moreover, the concentration groups differ only slightly from the control group, which has a concentration of 0 μg/ml.
We deduced that MNPs have no toxic nor side effects on cells and are biodegradable in the body. After being metabolized by cells, they enter the normal plasma iron pool and bind to red blood cell hemoglobins or are used in other metabolic processes. 16 Even if the labeled cells die and are swallowed by macrophages, the iron particles in them will be quickly metabolized. This means that MNPs have no long-term influence on cartilage functionality and metabolism.
Chondrocyte Mutual Magnetic Attraction After Being Labeled With Fe3O4 Microspheres
We used microscopic cell morphology and Prussian blue staining to measure different MNP concentrations after chondrocytes phagocytized the MNPs, identify changes to cell activity status and examine the effects on cell mutual magnetic attraction. The chondrocytes were polygonal with pseudopodia, and the polygons were closely arranged to grow; as the concentration increased, the MNP-labeled chondrocytes showed a tendency to aggregate, indicating mutual attraction between cells (
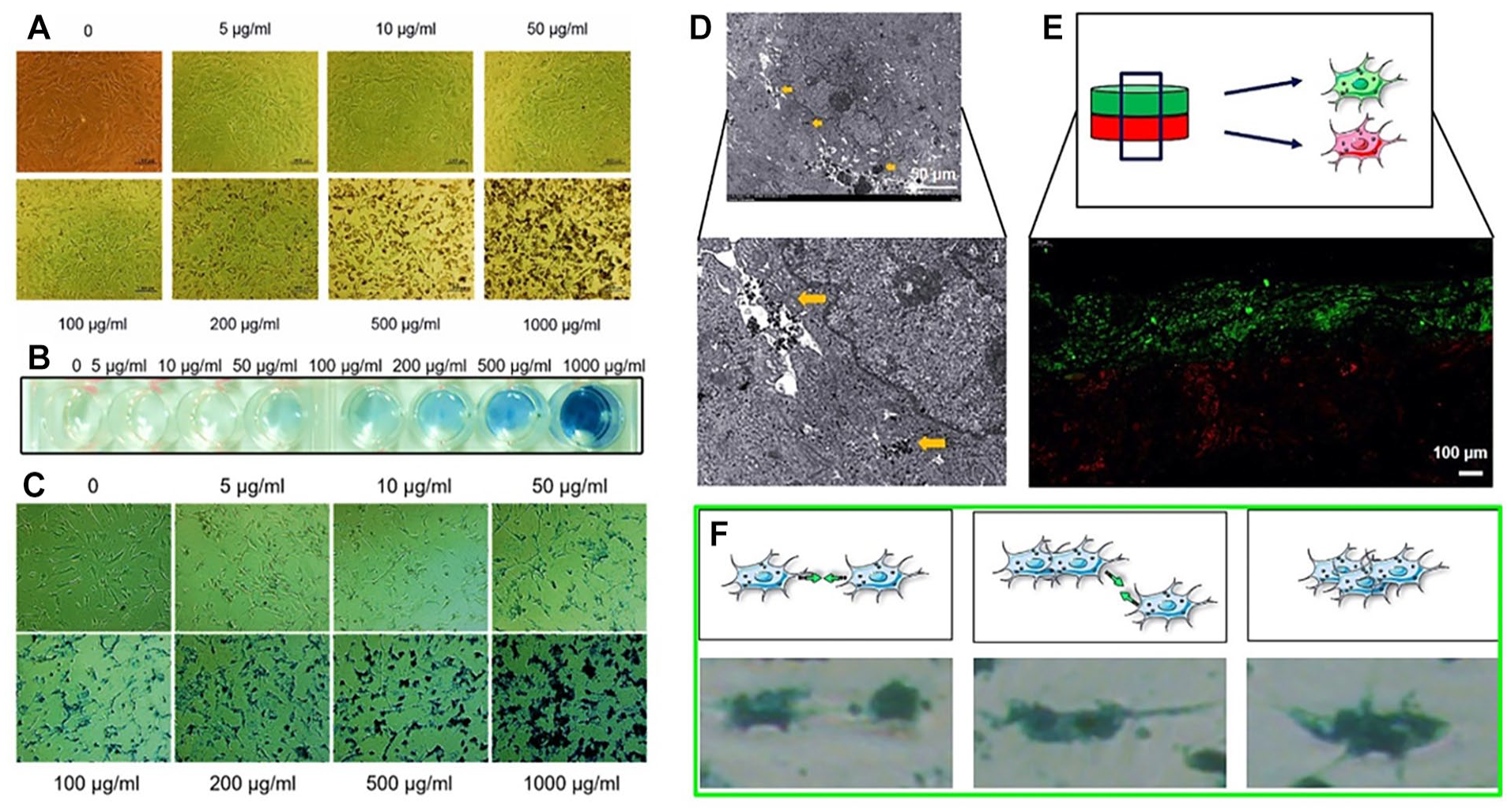
Characterization of chondrocytes cultured with Fe3O4 microspheres in different concentrations. (
To observe the microscopic condition of cells after phagocytosis of the MNPs, the cells were scanned by TEM. Electron microscopy showed that there were high-density scattered iron particles in the cytoplasm under the sheet of the labeled chondrocytes, and the small vesicle-like structure in the cytoplasm contained high-density iron particles (
Magnetic Mold Force on Fe3O4 Labeled Chondrocytes
As shown in
Figure 3A
, the staple was attracted by the “S” shape magnetic mold. The mold was hollow (
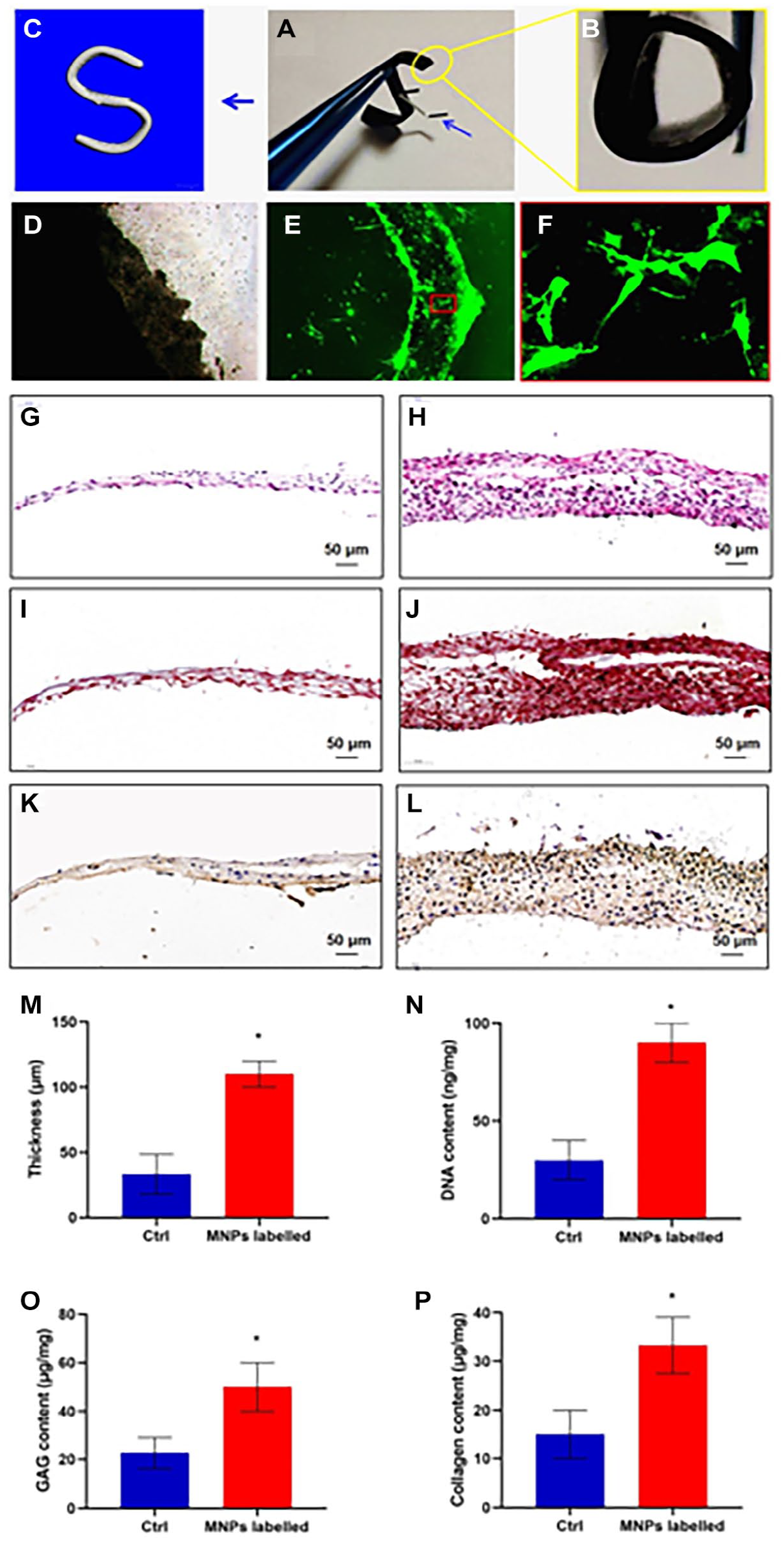
Magnetic mold force to Fe3O4 labeled chondrocytes. (
Because the super-strong magnetic force controls the attractive force, a smooth, dense cell sheet can be quickly built, containing multiple layers of cells and a large number of different mold forms. As shown in
Figure 3H
, after only 3 days culturing, we were able to build a strong, stable multilayer cell sheet. Compare this with the traditional cell sheet (
Cartilage tissue is composed of chondrocytes, collagen, proteoglycans, and so on. Repairing cartilage defects in the body is not easy.1,17 Tissue engineering technology provides a new way to repair cartilage defects. Autologous chondrocytes, which can be selected with little trauma and no immune rejection, are ideal seed cells for cartilage regeneration.
With the rapid development of tissue engineering technology, the use of cartilage regeneration to repair cartilage defects is not only a subject of research reports but also applied in clinical practice.3,18 Owing to the uncertain biological safety of stents for long-term use, 19 current cell sheet transplantation technology is used for cartilage regeneration. Its advantages of minimal trauma, easy operation, reproducibility, and fewer complications, make it an ideal tissue engineering cartilage regeneration method.
This study uses the paramagnetic properties and good biological safety of MNPs20,21 to introduce labeling technology into the field of cartilage tissue engineering and use it for 3D shaping cartilage regeneration. Not only do magnetic nano iron particles (Fe3O4) retain their nanomaterial biological activity, they possess magnetic responsiveness and superparamagnetism qualities, 22 in other words, they can be oriented and positioned under a constant magnetic field, allowing them to enter the cell and regulate cell functions.
Characteristics of Cells + MNPs + Mold Co-Culture
The schematic diagram of the entire research process (
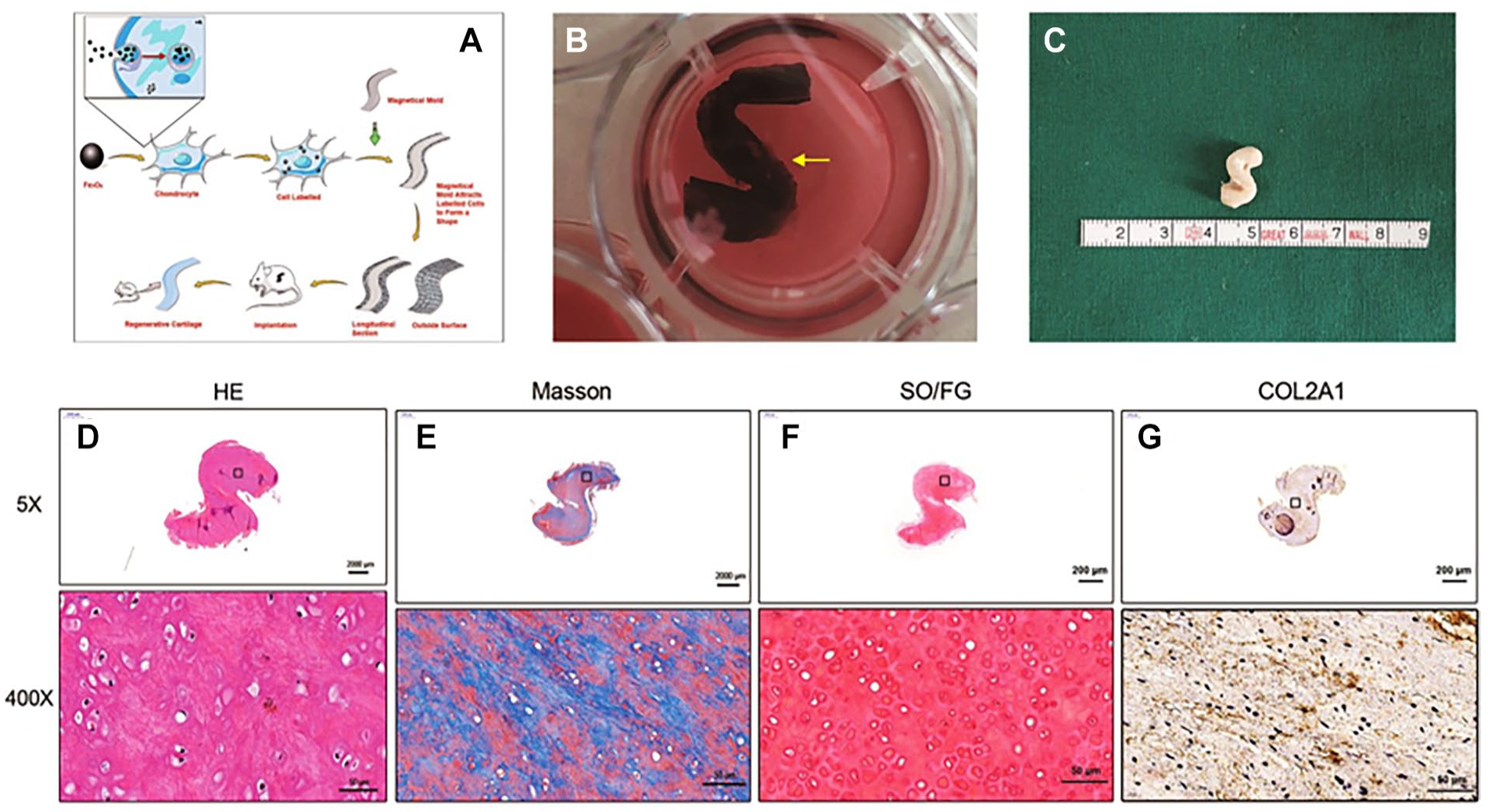
Chondrocytes labeled MNPs cultured with magnetic mold to shape a 3D structure in vivo. (
The unmagnetized cells can not adhere to grow on the magnetic mold, which means nonlabeled chondrocytes cannot be constructed into a specific shape cartilage. To achieve three-dimensional growth, an “S”-shaped three-dimensional magnetic mold was prepared for labeled chondrocytes to grow on. They formed a 3D “S” shape cell sheet (
The results confirmed that this method can specifically and efficiently target seed cells to the target location and use magnetic force to complete early attachment growth. After a period of magnetic assisted fixation, the cells can secrete extracellular matrix and adhesion molecules to reach the biological adhesion to achieve three-dimensional growth. In the complex physiological environment of the body, chondrocytes become increasingly mature, forming dense and stable cartilage tissue. This study provides an encouraging new idea for solving the three-dimensional shaping problem of cell transplantation in vivo, reducing complications and improving efficacy. It is worthy of further exploration and will doubtless be extended to other research and treatment fields.
The “S” shape is more complex than a line or circle shape, indeed it combines them. Achieving success with a three-dimensional “S” shape will make it easier to regenerate other cube or cylinder shape cartilage. We hope to simulate ear or trachea cartilage regeneration. Our intention in the future work is to regenerate more complex cartilage shapes that can be used to repair irregular shape cartilage that is deficient because of injury, tumor, or congenital malformation. This study is a first step and we will research more complex three-dimensional shapes in our forthcoming articles. Our desire is to move from experimental research right through to clinical application.
Conclusion
Magnetic iron nanoparticles—Fe3O4 nanoparticles in this study—have very broad potential applications in cells and indeed in the entire organism. MNP-labeled cells cluster together to form a multilayer chondrocyte sheet through magnetic force. Not only can the three-dimensional cartilage constructed by this magnetic control method be shaped, the regenerated cartilage tissue is also more efficient and of high quality. Furthermore, it can be easily developed from experimental research to clinical application.
Footnotes
Acknowledgments and Funding
The author(s) thank for Xiansong Wang and disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Fundamental research program funding of Ninth People’s Hospital affiliated to Shanghai Jiao Tong University School of Medicine (grant no. JYZZ143) and National Natural Science Foundation of China (grant no. 81974291).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study has been approved by the the Ethics Committee of Shanghai Ninth People’s Hospital (approval number: SH9H-2019-A257-1).
Data Availability
The data sets used and/or analyzed during the current study available from the corresponding author on reasonable request.
