Abstract
Objective
Articular cartilage defect causes joint pain and finally progresses to osteoarthritis. Although the subchondral bone condition affects clinical outcomes of cartilage defects, the natural course of changes in subchondral bone and associated pain in full-thickness cartilage defects remain unknown. Therefore, we investigated the natural course of histological changes in subchondral bone and joint pain in cartilage defects using a rat model.
Design
Full-thickness cartilage defects were created at the medial femoral condyle of 10-week-old male Sprague-Dawley rats. Rats were sacrificed at 3, 7, 14, 28, and 56 days postoperatively, and histological including immunohistochemistry and tartrate-resistant acid phosphatase (TRAP) staining and micro-computed tomography (μCT) analyses of their knees were performed. Pain was evaluated using behavioral analysis and immunofluorescence staining of the dorsal root ganglion (DRG).
Results
The contour of the subchondral bone plate was maintained until day 3, but it was absorbed just under the cartilage defect from day 7 to 14. Starting on day 28, sclerotic changes surrounding the bone absorption area were detected. In the subchondral bone, the number of TRAP-positive cells peaked on day 14. Osteocalcin-positive cells were observed at 7 days, and their number gradually increased till day 56. Behavioral analysis showed that the total distance and the number of getting up by hind legs decreased on day 14. The number of calcitonin gene-related peptide-positive fibers in the DRG increased and was the highest on day 14.
Conclusions
The subchondral bone condition under cartilage defects dynamically changes from bone resorption to sclerosis and is related to pain level.
Introduction
Articular cartilage provides smooth joint movement and weightbearing capabilities through its ability to absorb stress, and reduce friction, and its high resistance to wear. 1 In the clinical setting, articular cartilage injuries are often associated with joint injuries, such as ligament injury, joint dislocation, and fracture. It is difficult for articular cartilage to heal spontaneously as it is poorly vascularized and innervated, and the injury eventually progresses to osteoarthritis (OA), especially in lesions that are greater than 1.5 cm in diameter. 2 The homeostasis of articular cartilage is maintained by the subchondral bone, with interactions such as nutritional exchange and coordination of load distribution taking place. 3 Therefore, damage to the articular cartilage layer significantly affects the condition of the subchondral bone, and may introduce both sclerotic and porotic changes. Articular cartilage injury also induces joint pain, which causes functional disability of the joint and decreases daily activity. 4 Although pain originates from the synovium and subchondral bone, the experience of pain in the subchondral bone is crucial because it is directly affected by load-bearing. Thus, articular cartilage defects should be treated appropriately to improve activities of daily living by removing pain and inhibiting the progression of OA.
Appropriate treatment of articular cartilage injury is selected, according to the size of the lesion and the condition of the subchondral bone. 5 The bone marrow stimulation (BMS) technique is generally performed because of its simplicity. However, it has been reported to have poor long-term clinical outcomes due to fragile fibrocartilage tissue covering the cartilage defect. 6 Moreover, intralesional osteophytes and subchondral bone cysts have been reported as complications with this technique. 7 BMS induces endochondral ossification around the bone hole in the subchondral bone, altering bone metabolism and thus causing excessive osteogenesis and bone resorption in the subchondral bone, which affects clinical outcomes after surgery. 8 Autologous chondrocyte implantation may also be used to treat articular cartilage injury, although it was reported that the condition of the subchondral bone affects the clinical outcome. 7 As the condition of the subchondral bone influences the clinical outcomes, it is important to understand the changes in the subchondral bone after cartilage injury. However, the timeline of historical changes that take place in the subchondral bone after cartilage injury remains unclear. If the subchondral bone condition changes in the natural course of a cartilage injury and postoperative outcomes are affected by the condition of the subchondral bone, the timing of surgery and the selection of the surgical procedure are crucial. In addition to using imaging technology such as computed tomography (CT), to evaluate the condition of the bone, using joint pain as an indicator of the subchondral bone condition would be a useful technique for treating articular cartilage injury. Dynamic changes in osteoclast and osteoblast differentiation occur in the subchondral bone during the pathogenesis of osteochondral lesion (OCL) and OA.9,10 An increase in the activity of osteoclasts leads to a decrease in the threshold of the pain, and nociceptive nerve fibers, including neuropeptides, increase the subchondral bone in OA and OCL. Several studies also showed a significant relationship between joint pain and the subchondral bone condition in articular cartilage damage in the OA model. It is reported that inhibition of subchondral bone deterioration by bisphosphonate could alleviate joint pain in rat OA models. 11 Another report demonstrated that improving joint pain was reduced as the subchondral bone microarchitecture was improved by the treatment of the intermittent parathyroid hormone in the OA mice models. 12 Based on these 2 factors, we hypothesized that the subchondral bone condition would change dynamically after an injury to the articular cartilage and that the condition of the subchondral bone would affect pain at the joint. The purpose of this study was, therefore, to elucidate the histological changes in the subchondral bone after articular cartilage injury in the weightbearing area and to investigate the relationship between the subchondral bone condition and pain using a rat model.
Method
This study was performed in accordance with the Guide for Animal Experimentation and was approved by the Committee of Research Facilities for Laboratory Animal Science (A19-159).
Animals
Twenty-five male 10-week-old Sprague-Dawley rats were used in this study. The animals were housed 3 per cage in the animal experimentation facility under a normal 12-hour light-dark cycle with free access to food and water. Rats were anesthetized with an intraperitoneal injection of ketamine hydrochloride (1.4 ml/kg of body weight) and xylazine (0.4 ml/kg of body weight). Then right knee joints were exposed by a parapatellar approach, and 5.0 mm × 3.0 mm of full-thickness articular cartilage defects were created in the weightbearing area of the medial femoral condyle using a surgical knife without any damage to the subchondral bone plate according to the procedure described in a previous report. 13 The left knee joints were used without procedure as the control group. An additional 5 male 10-week-old rats were incised to the joint capsule using the same method and used as a sham group for behavioral analysis. After surgery, all rats were allowed to move freely inside the cages without load restriction. One week before sacrifice, 10 μl of 2% fluoro-gold (FG; Fluorochrome Inc., Denver, CO), a neuronal retrograde tracer, was injected into the knee joints. 14
Histological Assessment
Five rats were sacrificed at each time point of 3, 7, 14, 28, and 56 days after surgery, and both knee joints and bilateral L4 dorsal root ganglions (DRGs) were harvested according to a procedure described previously.
15
The knee joints were fixed in 4% paraformaldehyde phosphate-buffered saline (Wako Pure Chemical Industries Ltd.) for 24 hours at 4 °C and then decalcified in distilled water containing 10% ethylenediaminetetraacetic acid for 3 weeks. Each sample was embedded in paraffin and cut into 4.5-μm-thick sections along the sagittal plane, including the cartilage defect. Sections were stained using safranin-O/- fast green and hematoxylin-eosin (HE), according to standard protocols. Pathological changes in the subchondral bone below cartilage defects were graded by allocating a score ranging from 0 (best) to 3 (worst) to the subchondral bone using Aho et al.’s
16
subchondral bone grading score. A grade of 0 indicated that there was no evidence of subchondral bone sclerosis, and a grade of 3 that there was severe subchondral sclerosis and massively increased bone volume (
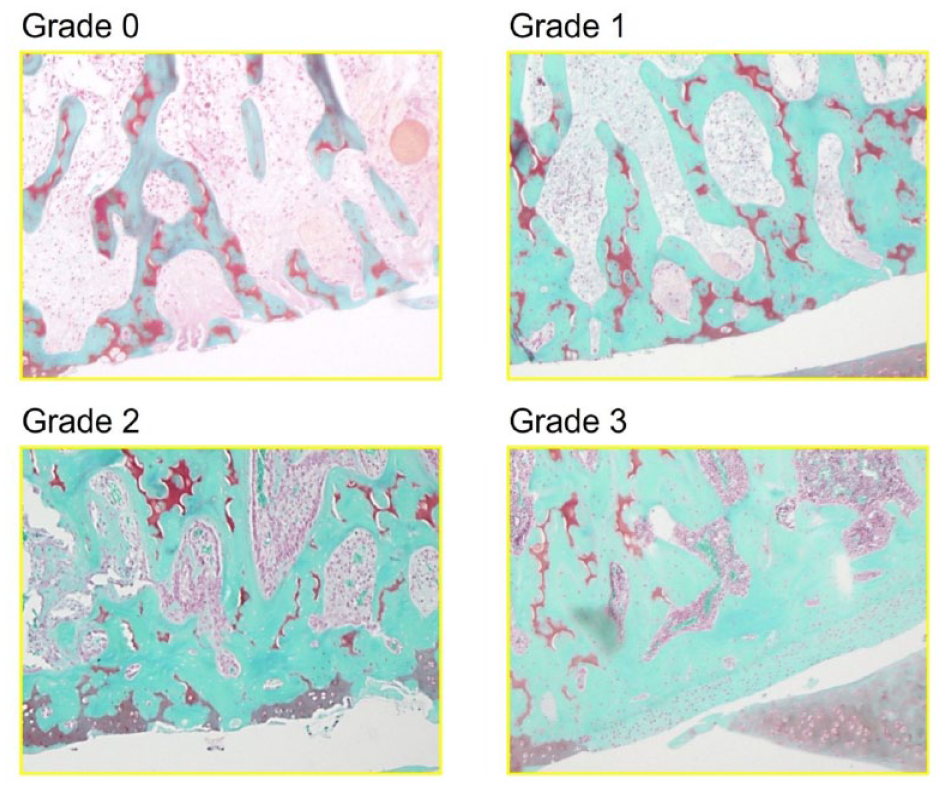
Aho’s subchondral bone grading score. Representative images of Grades 0 to 3 in the rat model. Grade 0: no evident subchondral bone sclerosis, thin subchondral bone plate, and trabecular. Grade 1: some subchondral sclerosis and bone volume are increased. Thickened bone trabeculae can be seen. Grade 2: a distinct increase subchondral sclerosis and bone volume. Grade 3: severe subchondral sclerosis and massively increased bone volume. Subchondral bone plate flattens.
Each section from the knee was immunostained with an anti-osteocalcin (1:100 dilution; Santa Cruz Biotechnology, Dallas, TX), anti-substance P (1:100 dilution; Santa Cruz Biotechnology, Dallas, TX), anti-calcitonin gene-related peptide (CGRP) (1:500 dilution; Abcam, Cambridge, MA), using a 3,3′-diaminobenzidine substrate (Vector Laboratories, Burlingame, CA). Osteocalcin-positive cells were evaluated at the trabecular edge, and the percentage of osteocalcin-positive cells was determined by the length of osteocalcin-positive cells divided by the length of the trabecular round, using Image J (National Institution of Health). TRAP staining was performed using a commercially available kit (Wako Pure Chemical Industries, Ltd., Osaka, Japan) according to the manufacturer’s protocol. TRAP-positive multinucleated cells containing more than 3 nuclei were identified as osteoclasts.
CGRP expression and FG were assessed using fluorescence microscopy. Sections of DRGs were incubated with rabbit anti-CGRP (1:500; Abcam, Cambridge, MA) overnight at 4 °C, and the second detection was performed with Alexa Fluor 488-conjugated goat anti-rabbit secondary antibody (1:500 dilution) for 1 hour at room temperature. CGRP-positive nerve fibers and FG-positive fibers were counted, and the ratio of the number of CGRP and FG-positive cells to the number of FG-positive cells, determined using Image J.
Micro-Computed Tomography
Samples were analyzed using high-resolution μ-CT (SkyScan1176, Toyo Corporation, Tokyo, Japan) using the following parameters: tube voltage of 70 kV/360 μA, AI 1-mm filter, and 18-μm isotropic resolution. Images were reconstructed (NRecon, Toyo Corporation, Tokyo, Japan) for analysis using a CT analyzer (Toyo Corporation, Tokyo, Japan). The volume of interest was 5.0 mm × 3.0 mm and 1.0 mm deep in the subchondral bone lesion just below the cartilage defect, with the bone volume/tissue volume (BV/TV, %) ratio measured as previously described. 17
Open-Field Test
The open-field test was performed twice for each rat in a square arena (100 cm long, 100 cm wide, 60 cm high), based on a previous experiment by Crumeyrolle-Arias et al., 18 just before surgery and sacrifice. Briefly, each rat was placed in one corner of an open-field arena lit in the center (500 lx) and allowed to freely explore the arena. The movements of the rats were monitored and recorded for 6 minutes. The total distance the rat traveled in the arena and the number of times of rearing, getting up by hind legs were calculated using specific devices (SMART, Panlab SL, Barcelona, Spain).
Statistical Analysis
All results are expressed as mean and standard deviation. The Mann-Whitney U test was used to compare the total distance traveled and number of times rearing occurred that were measured during the open-field test at each time point between the cartilage defect models and sham models. Tukey Kramer’s post hoc test was used to compare subchondral bone grading, TRAP-positive and CGRP-positive ratio of subchondral bone, CGRP-positive ratio of DRG, and BV/TV ratio of subchondral bone among 6 groups.
Results
Subchondral Bone Changes after Cartilage Injury
On day 3, the subchondral bone plate remained in all specimens, and there was no obvious change in the subchondral bone with cartilage defect. On day 7, bone resorption was observed in the subchondral bone, which progressed until day 14. From days 28 to 56, bone sclerosis gradually progressed in the subchondral bone (
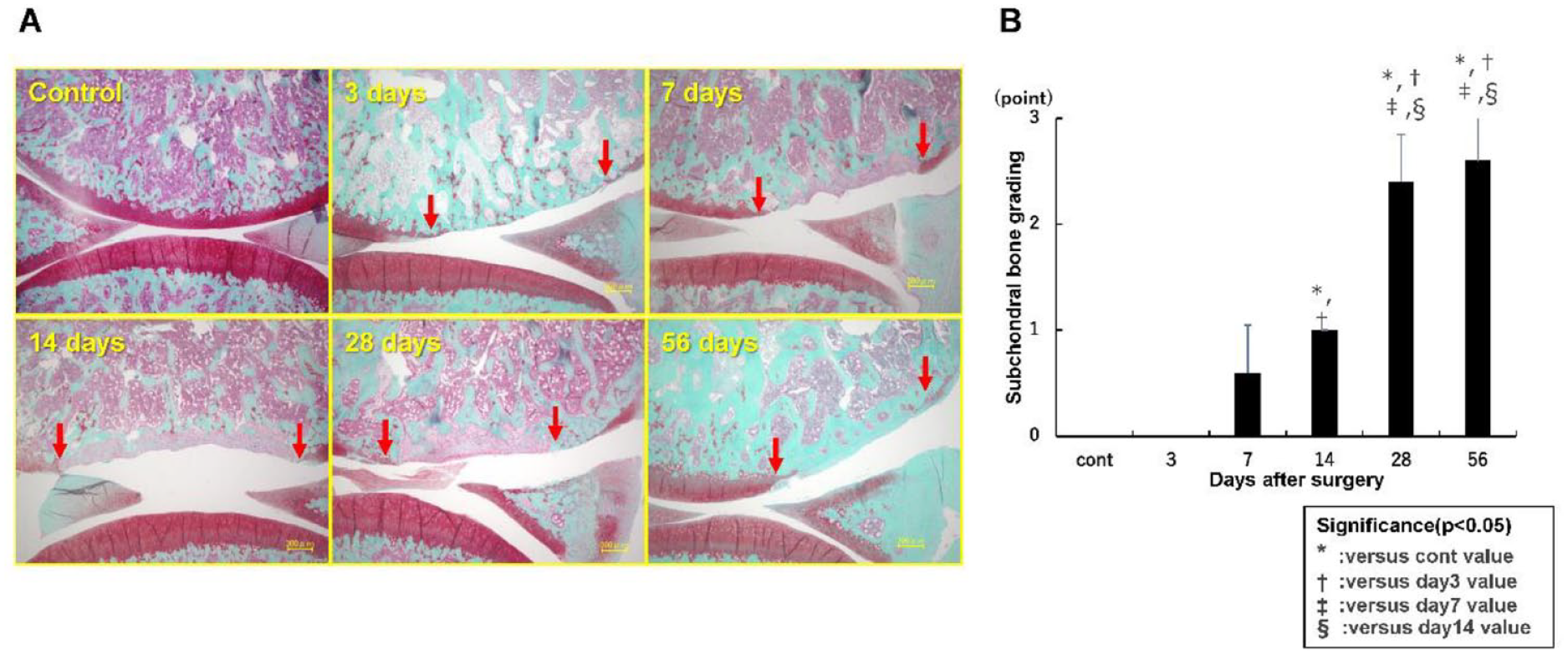
(
The μ-CT images showed that the contour of the subchondral bone plate was maintained until day 3, but it was absorbed in the area just under the cartilage defect on day 7. On day 14, the depth of the area of bone absorption expanded, and the subchondral bone plate disappeared, but there was no sclerotic change at the site of the cartilage defect. On day 28, osteogenesis in the subchondral bone defect filled toward the articular surface, with sclerotic changes surrounding the bone absorption area. On day 56, the subchondral bone defect was filled with sclerotic bone (
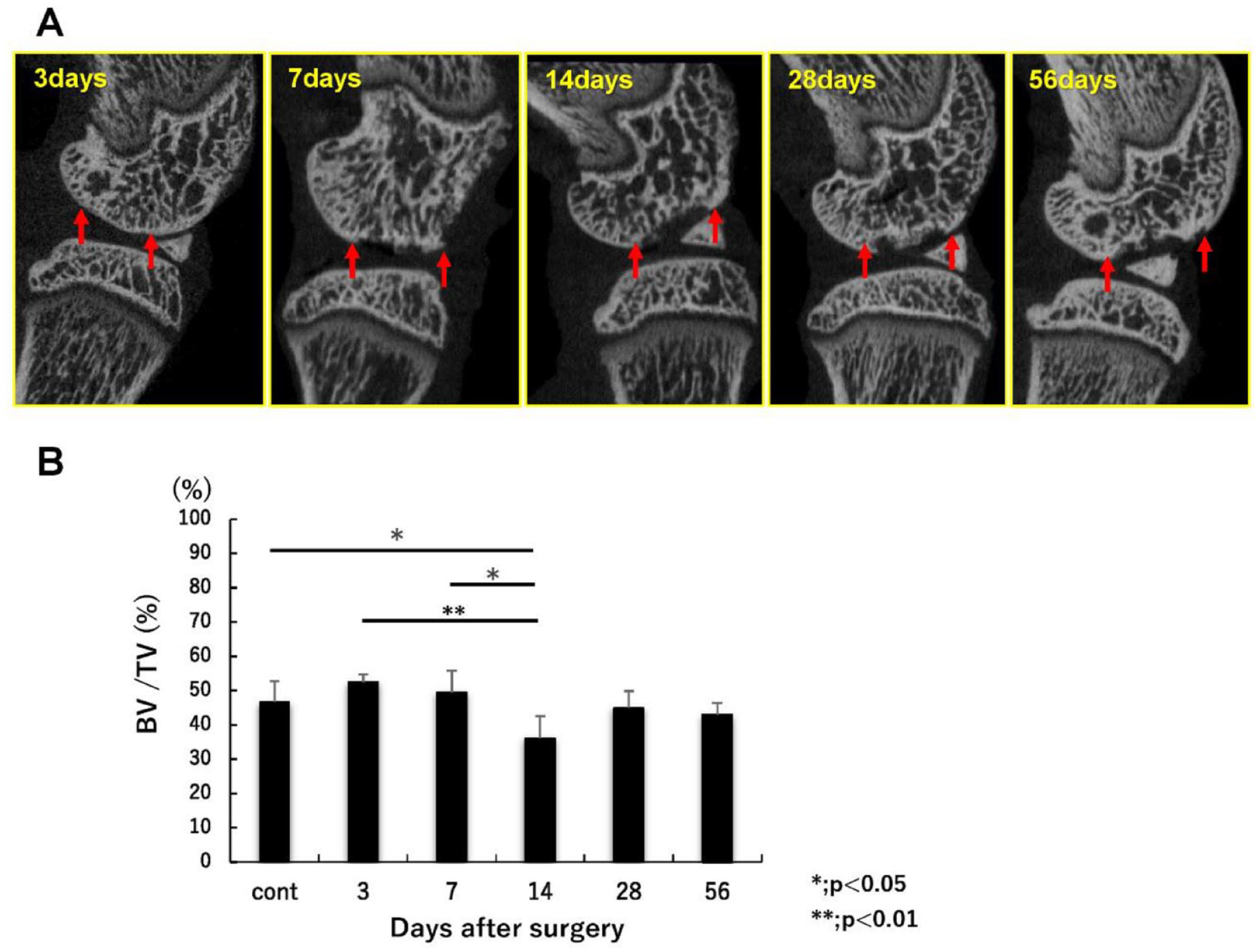
(
In the evaluation of the osteoclast activity in the subchondral bone, an increase in the number of osteoclasts in the subchondral bone just under the cartilage defect was observed from day 7, reaching a maximum on day 14, and then decreasing to levels similar to those observed in the control group by day 56. On day 14, the number was significantly higher than that in the control group on days 3, 7, and 56 (90.2 ± 23.9 vs. 20.4 ± 5.0, 13.8 ± 6.1, and 37.8 ± 14.8 for days 14, control group, days 3, 7, and 56, respectively) (
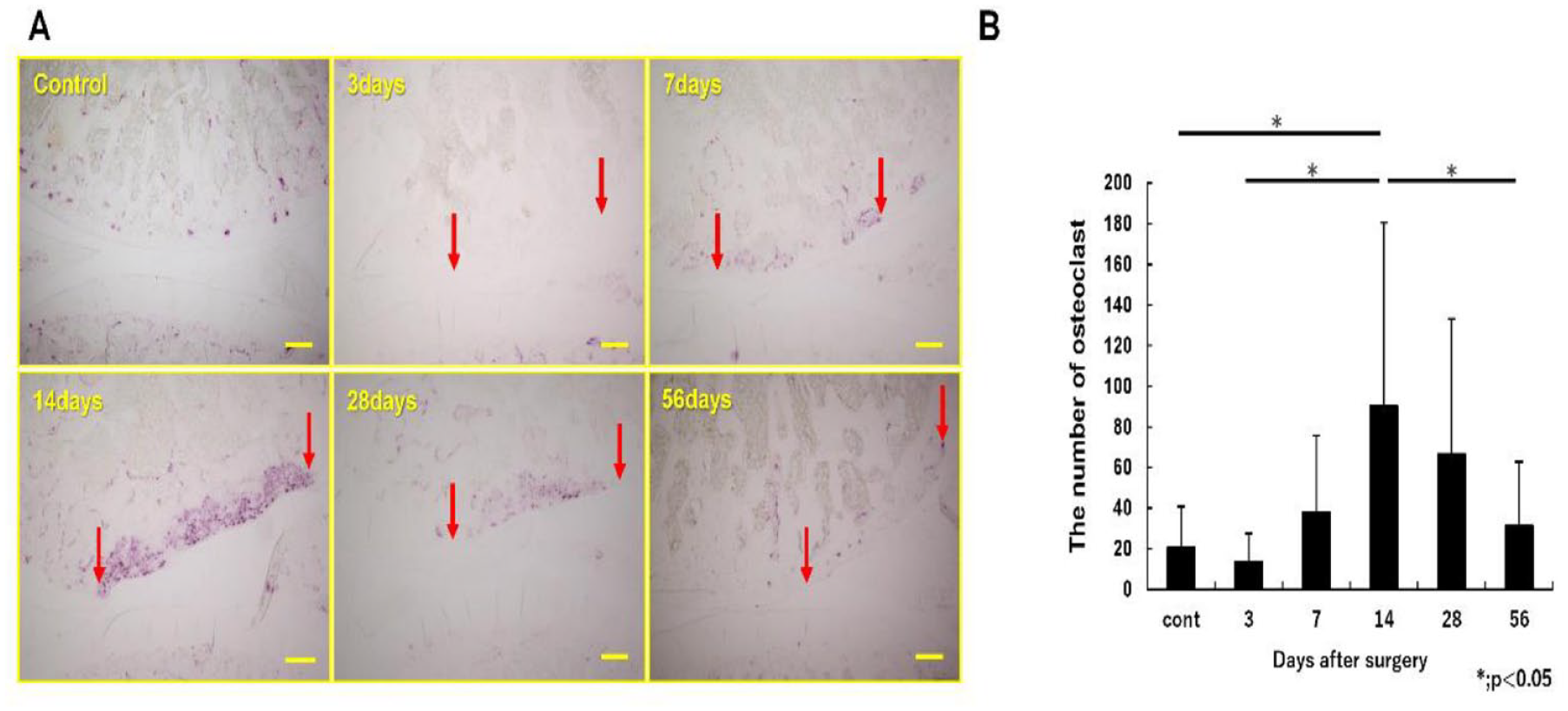
(
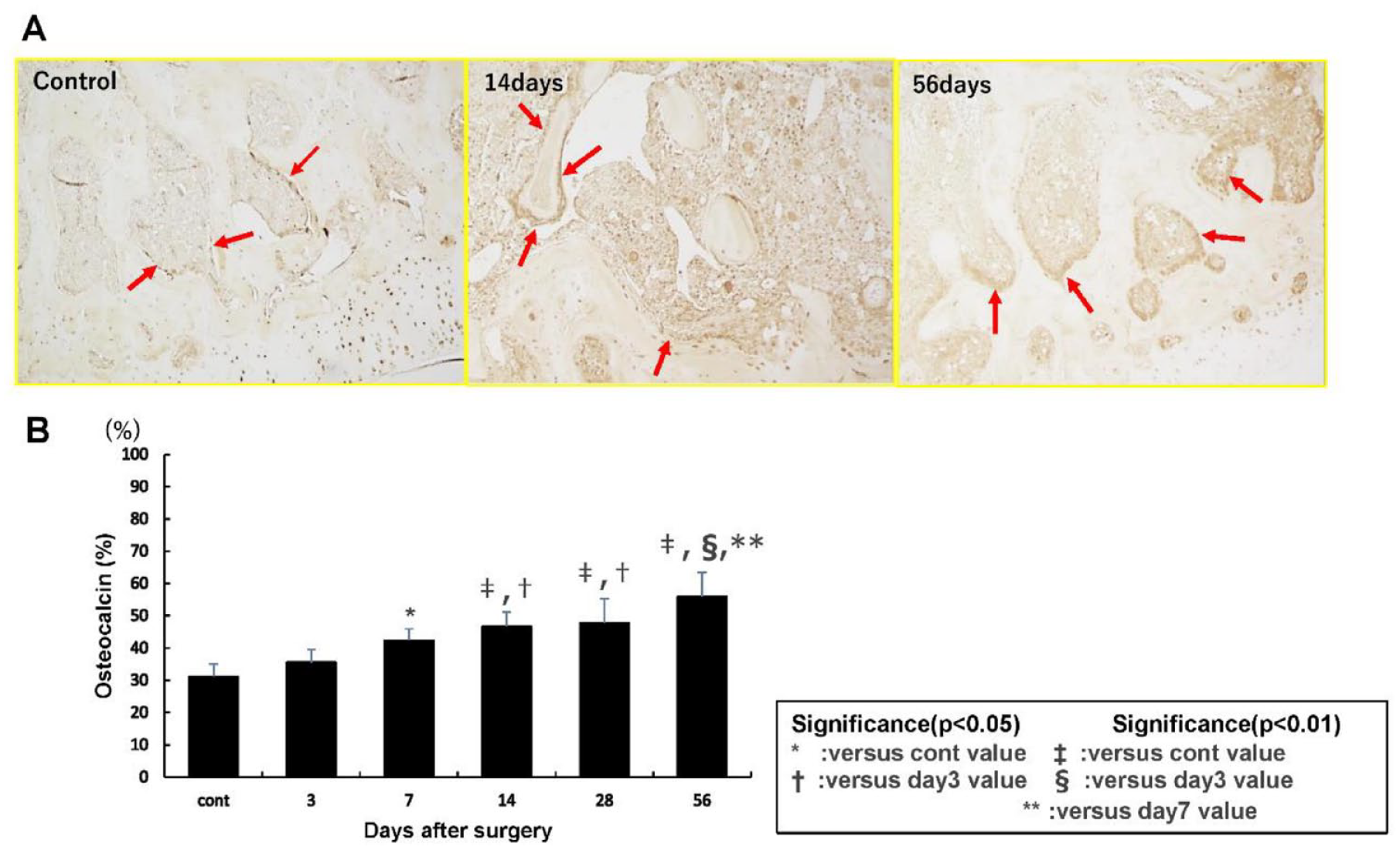
(
Pain Assessment
Compared with the sham models, cartilage defect models showed significantly reduced total distance traveled in the open-field test on days 14 (P = 0.008) and 56 (P = 0.00007) (
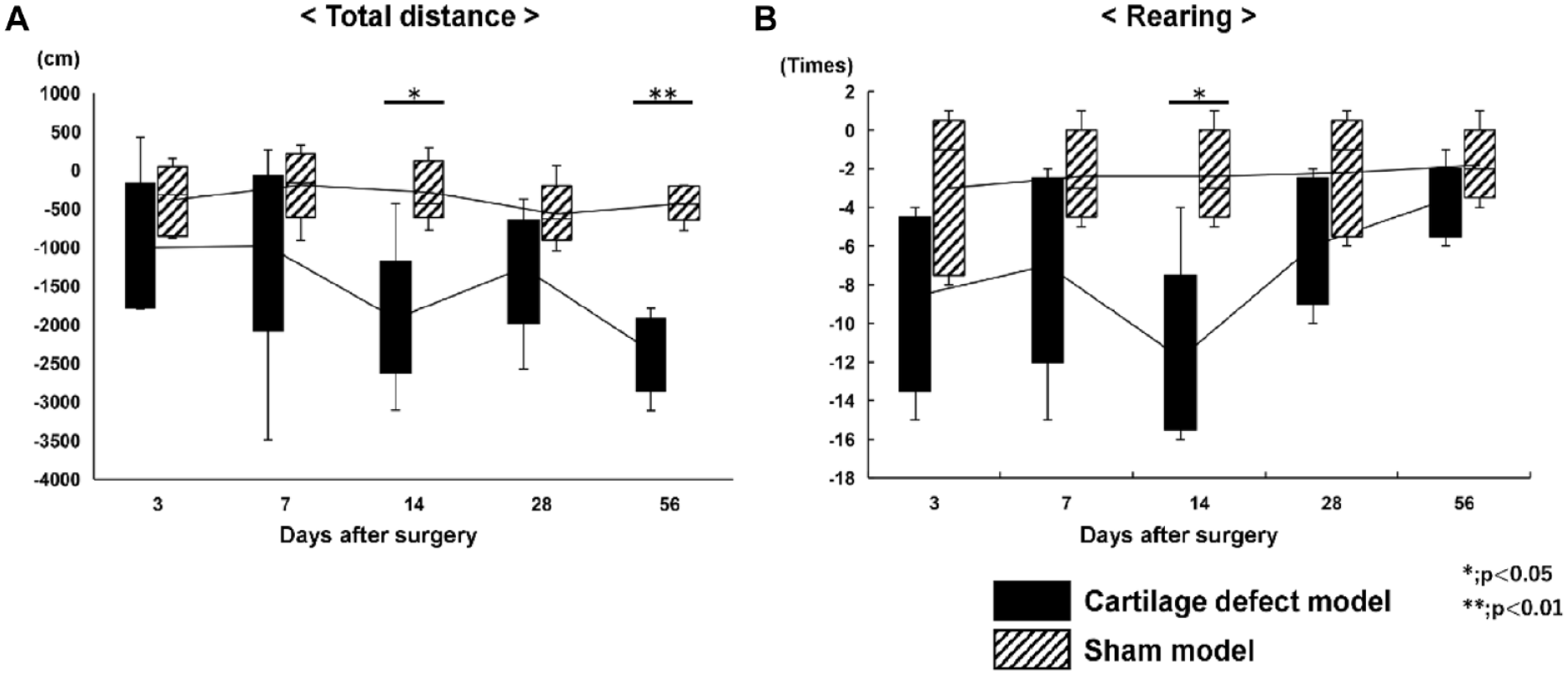
The results of the open-field test: (
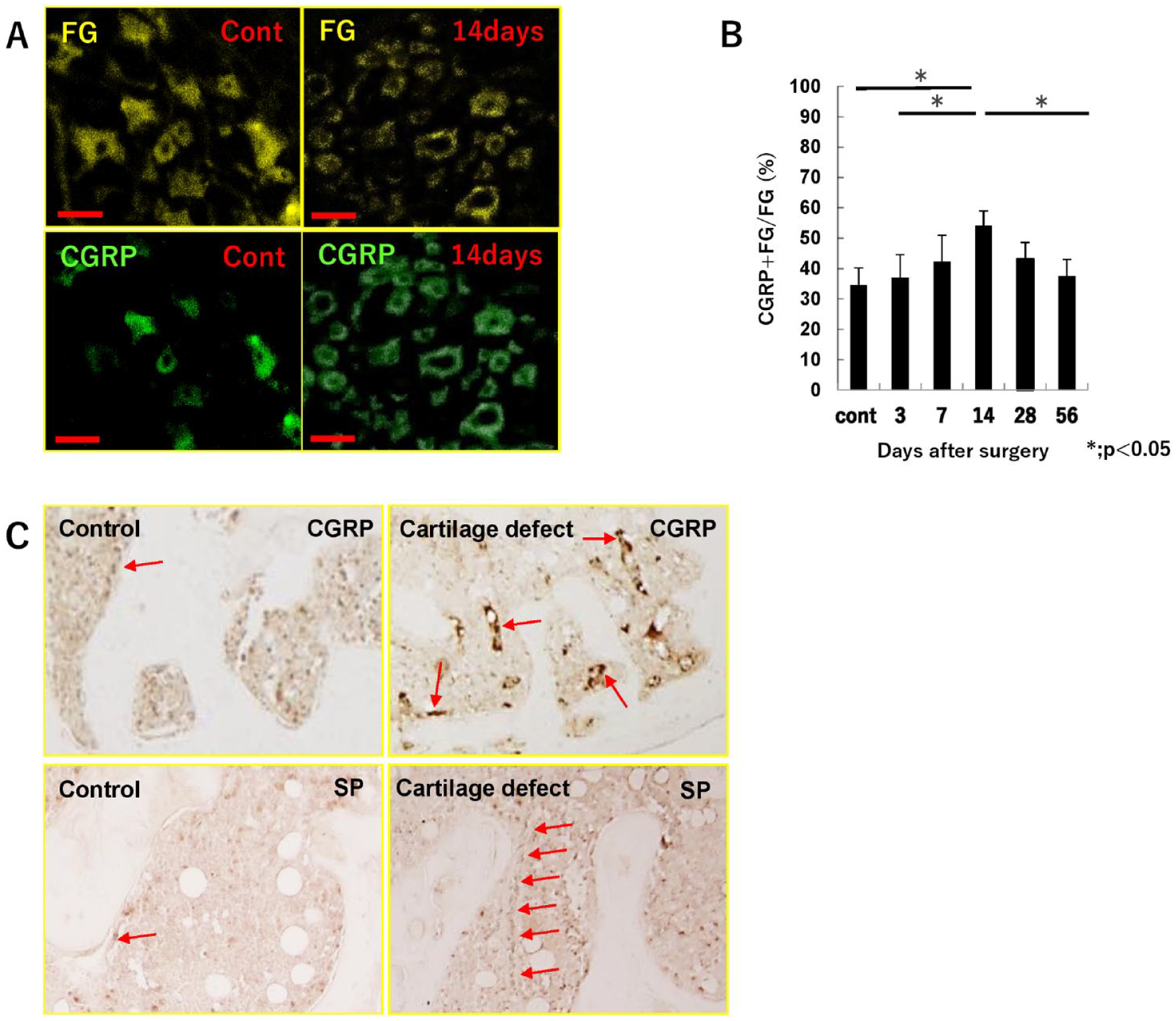
(
Discussion
The purpose of this study was to clarify the natural changes in the subchondral bone, and the relationship between the condition of the subchondral bone and pain in the early phase of articular cartilage defects in a rat model. Our results suggest that the activation of osteoclasts occurred from day 7, bone resorption of subchondral bone peaked on day 14, and subsequent bone sclerosis by osteoblast activation occurred from day 28. In addition, from the results of the open-field test and immunofluorescence staining of the DRGs and subchondral bone, the pain level was estimated to peak at day 14, concomitant with bone absorption in the subchondral bone. Thus, the natural course of an articular cartilage defect in a rat model was revealed, and it was associated with pain levels. The findings of this study suggest that the condition of the subchondral bone can be inferred from radiographic assessment and pain level, and this will contribute to the treatment of cartilage defects in clinical practice.
The pathogenesis of pain due to articular cartilage defects is not fully understood, although several factors are thought to be involved in the process. As articular cartilage does not have a nerve fiber, surrounding tissues such as the subchondral bone and synovium may cause pain. In particular, the subchondral bone under the cartilage defect is directly affected by the loading force, which may subsequently induce changes in the bone structure. 19 Also, the mechanism of pain experienced in OA and OCLs is better understood than that in cartilage defects. In OCLs, pain may develop from the pressurized fluid into the subchondral bone, which induces decreasing pH caused by osteoclasts. A low pH excites the nerve fibers present in the bone, inducing pain. 20 In OA, the subchondral bone has received much attention as a cause of pain. In previous reports, subchondral bone marrow edema-like lesions were visualized using magnetic resonance imaging, and were highly correlated with OA pain.21,22 It has also been reported that an increase in osteoclast-mediated bone resorption induces sensory innervation in the subchondral bone and hyperexcitability of DRG neurons, which induces OA pain. 23 In clinical practice, risedronate, an osteoclast inhibitor, has been tested for use in OA, and it has been shown to decrease subchondral bone marrow lesions, thus improving pain.24,25 In this study, osteoclast activity under the cartilage defect was highest on day 14, suggesting a correlation between the course of pain and activation of osteoclasts.
Bone remodeling is continuously maintained through a tight equilibrium between osteoblast activity which is responsible for bone formation through the synthesis of bone matrix and osteoclast activity which is responsible for degrading the bone microenvironment. 26 In this study, osteoclasts situated under the cartilage defects were activated from days 7 to 14, and osteoblasts were activated from day 7, suggesting that the metabolism of the subchondral bone was enhanced after the articular cartilage defect was created. It is well known that osteoclasts below the cartilage are activated due to the increased load, inducing subchondral bone loss in early stage OA, and subchondral bone becomes sclerotic at a later stage of OA.27,28 Our results suggest that subchondral bone remodeling after cartilage defect is similar to that observed in the pathogenesis of OA. Previous reports have shown that subchondral bone plays an important role in maintaining cartilage homeostasis, 3 and damage to the subchondral bone causes the progression of cartilage damage vice versa. 29 Furthermore, cartilage destruction in anterior cruciate ligament transection models is secondary to subchondral bone damage. However, the mechanism of the naturally occurring OA induced by collagenase injection is suggested that the absorption of subchondral bone is secondary to cartilage lesions. 30 In summary, the condition of the subchondral bone is related to the condition of the cartilage, and the condition of the subchondral bone affects the repair of cartilage.
Subchondral bone cysts are a major complication of the microfracture technique for cartilage defects. 7 Our results suggest that microfracture in an osteoclast-dominated situation, such as on day 14 after damage to the cartilage, may cause subchondral bone cysts to develop. After an acute traumatic articular cartilage injury, the cartilage lesion might best be treated within 2 weeks after injury to prevent subchondral bone deterioration due to osteoclast activation by careful weightbearing control to reduce the influx of joint fluid. Therefore, understanding the change in the condition of the subchondral bone will result in the successful treatment of damaged cartilage defects. Imaging examinations such as CT and MRI are used to understand the condition of the subchondral bone in clinical practice. In addition, the pain level can be a monitoring tool for subchondral bone conditions. It is reported that antiresorptive drugs such as bisphosphonates are well known to be useful for OA pain and OA progression.31,32 These findings, in addition to our results, support the hypothesis that drugs targeting osteoclasts can potentially inhibit pain and changes in the subchondral bone in the early stages after damage to the cartilage. Besides, further studies will be required to develop surgical procedures such as microfracture technique by analyses of the actual changes of the subchondral bone seen after microfracture and to investigate the feasibility of the application of biomaterials to improve the therapeutic repair outcomes including pain reduction.
This study has several limitations. First, we used rats as the animal model. As the rate of bone metabolism differs between humans and rats, the time course of changes in the subchondral bone after cartilage injury may be different from that in humans. Moreover, many factors affect subchondral bone changes, such as age, sex, and degree or location of, or intervention for the cartilage injury. Second, all the animals could move freely after the cartilage defect was created in their limb, which may have affected the rate of metabolism in the subchondral bone. 33 These factors may have affected the results of this study. Therefore, the bone resorption phase may start later than 2 weeks after cartilage injury. However, the process in which subchondral bone resorption occurs and then it changes toward bone formation as sclerotic change after cartilage injury remains unchanged, and cartilage injury should be appropriately treated according to the condition of the subchondral bone to achieve good clinical results.
In conclusion, the condition of the subchondral bone under an articular cartilage defect dynamically changes from bone resorption to sclerosis, and this process is related to the level of pain experienced. Appropriate intervention for the cartilage defect by taking into account the subchondral bone changes and their relationship with pain will enable patients with articular cartilage injury to be treated successfully.
Footnotes
Acknowledgments and Funding
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Animal experiments were approved by the Hiroshima University Animal Ethics Committee (A19-159).
