Abstract
Objective
Subchondral insufficiency fracture of the knee (SIFK) is associated with high rates of osteoarthritis (OA) and arthroplasty. The implantable shock absorber (ISA) is an extra-capsular implant that unloads the medial knee compartment. This study compared the 2-year freedom from arthroplasty rates in subjects with medial knee OA and SIFK when treated with an ISA versus a matched cohort of patients treated non-surgically.
Design
This retrospective case-control study compared 2-year conversion rates to arthroplasty in SIFK score-, age-, and body mass index (BMI)–matched control subjects without prior surgical history with ISA-implanted subjects from an ongoing prospective study. Baseline and final radiographs, and MRIs were reviewed for evaluation of meniscus or ligament injuries, insufficiency fractures, and subchondral edema. Kaplan-Meier analysis assessed survival.
Results
Forty-two patients (21 Control: 21 ISA), mean age = 52.3 ± 8.7 years, BMI = 29.5 ± 3.9 kg/m2, 40% female were evaluated. Both ISA and Control arms had the same numbers of low (n = 4), medium (n = 11), and high-risk (n = 6) SIFK scores. One- and 2-year freedom-from-arthroplasty rates were both 100% for ISA subjects, and 76% and 55%, respectively, for Controls (P = 0.001 for cross-group comparison). Control knees with low, medium, and high-risk SIFK scores had respective 1- and 2-year survival rates of 100% and 100%, 90% and 68% (P = 0.07 vs. ISA), and 33% and 0% (P = 0.002 vs. ISA).
Conclusions
ISA intervention was strongly associated with avoidance of arthroplasty at a minimum 2 years, especially in patients with high-risk SIFK scores. SIFK severity scoring predicted relative risk of conversion to arthroplasty through at least 2 years in non-surgically treated subjects.
Keywords
Introduction
Spontaneous osteonecrosis of the knee (SONK) is associated with high rates of osteoarthritis (OA) and arthroplasty, 1 and bone marrow lesions predictive of potential early-stage SONK estimate disease prevalence to be as high as 9% in older persons aged >65 years. 2 Localized ischemia was originally thought to cause the necrosis associated with SONK. However, evolving evidence indicates that repetitive stress-induced subchondral insufficiency fractures of the knee (SIFKs), not ischemia, are the primary pathologic mechanism underlying necrosis.3 -7 Radial meniscal tears, including posterior medial meniscus root tears, result in diminished meniscal function and increased joint loading, leading to mechanical overloading of the subchondral bone. This results in insufficiency microfractures that cause fluid accumulation and edema within the bone marrow that induces focal ischemia and eventual necrosis, ultimately resulting in progressive functional loss. Thus, SIFK might be a more accurate descriptor of this disease than the term “SONK.”4,5
Clinical presentation of SIFK is usually acute onset of knee pain without predisposing trauma, commonly with tenderness localized over the medial femoral condyle (MFC). 8 Radiographs are often negative, especially in early SIFK disease, and therefore the diagnosis is frequently missed during early patient encounters. Later radiographs typically show subchondral radiolucent lesions in the weightbearing regions of the medial and occasionally lateral femoral condyle, although radiographs may remain unremarkable throughout the disease course in some patients. 5 More sensitive imaging technologies such as MRI can accelerate a SIFK diagnosis by identifying SIFK-specific characteristics of focal edema and possible fracture lines much earlier than radiographs. Differentiating SIFK from other joint pathologies early on is essential to initiating appropriate treatment to interrupt disease progression.
Management approaches for SIFK depend on disease severity and prognosis and range from non-surgical treatment focusing on load reduction and pharmacologic therapy aiming to mitigate SIFK progression to invasive surgical interventions such as posterior meniscal root repair (in early stages without bony changes) to high tibial osteotomy (HTO) or total knee arthroplasty (TKA).5 -7 Unloading strategies using braces and orthotics to reduce compressive tibiofemoral contact loads are frequently used for treating unicompartmental knee OA and are also part of the non-surgical treatment regimen for SIFK. 9 However, it is uncertain whether external bracing is sufficient to slow SIFK progression.10,11 If a patient’s SIFK progresses to subchondral collapse, arthroplasty is imminent.
An implantable shock absorber (ISA) has been developed to unload the knee. The ISA is placed subcutaneously, but outside the joint capsule and superficial to the medial collateral ligament (MCL), during an outpatient procedure ( Fig. 1 ).12,13 Immediate postoperative weightbearing is allowed as tolerated, and return to early full range of motion is encouraged. ISAs have been studied for the treatment of medial knee OA, where they appear to provide symptom relief while being both reversible and less invasive than arthroplasty.12,13
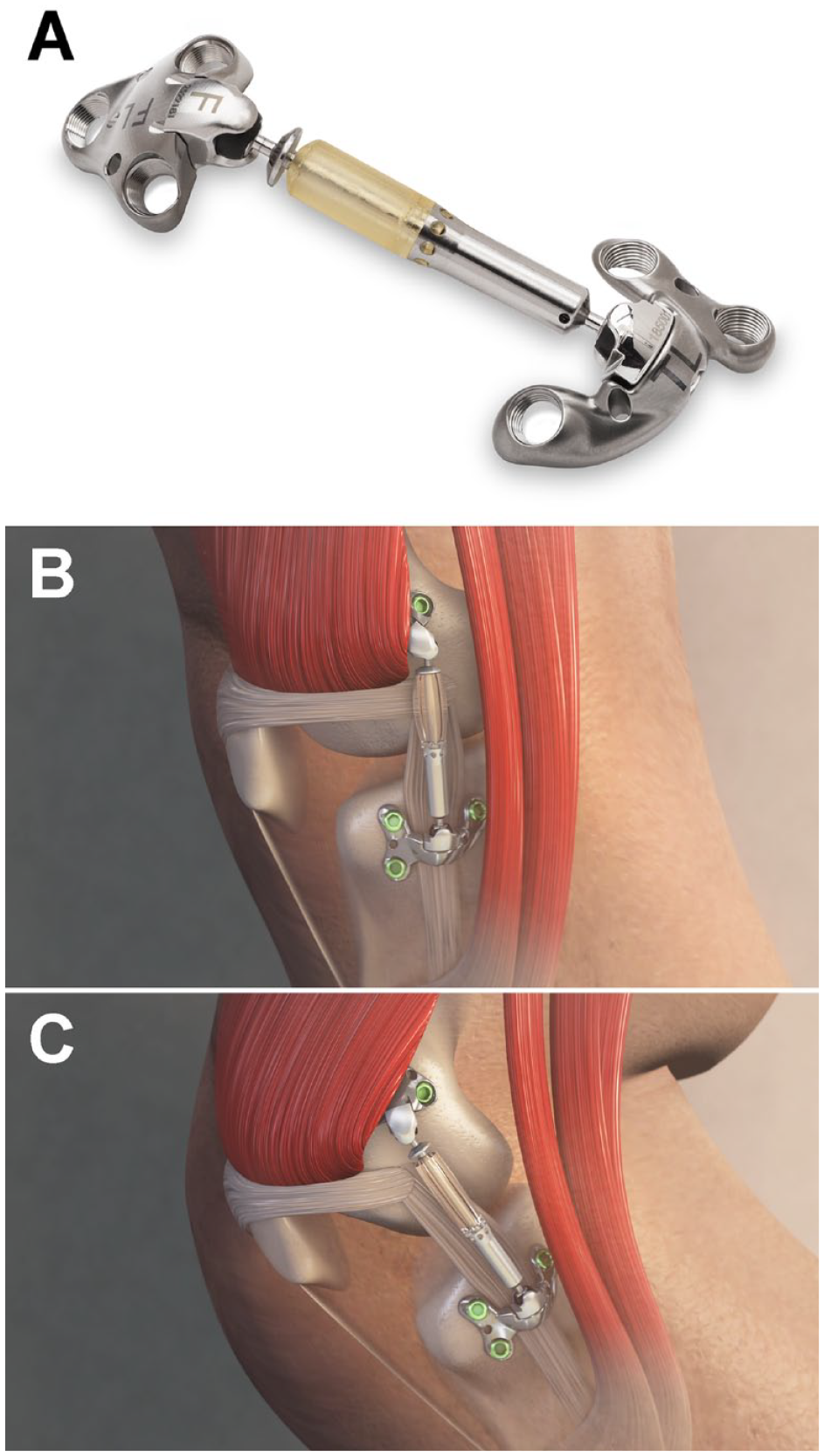
Implantable shock absorber. (
A recently validated SIFK severity scoring system, 14 based on radiologic findings such as Kellgren-Lawrence (K-L) grade 15 and meniscus tearing/extrusion, was able to accurately stratify patients into groups at low, medium, and high risk of progressing to arthroplasty through 4 years. 14 Patients classified as high risk based on SIFK score had a 39 times higher risk of arthroplasty, and 95% converted within 2 years of SIFK diagnosis. SIFK patients represent a clearly defined population at risk of imminent arthroplasty; thus, a treatment option that can reduce this arthroplasty risk serves important clinical utility.
The purpose of the current study was 2-fold: (1) to compare 2-year freedom-from-arthroplasty rates in subjects with medial-compartment knee OA and SIFK who received an ISA compared with a matched cohort of patients treated with non-surgical therapy (Control), and (2) to assess the ability of SIFK scores to predict 2-year rates of arthroplasty in these 2 patient populations. Subjects in the ISA group were derived from an ongoing multi-site interventional trial, while the Control arm subjects were matched (1:1 ratio) by a combination of SIFK score, age, and body mass index (BMI) from a database of non-surgically managed patients with a diagnosis of SIFK on MRI. Experimental groups were compared as a whole, and after segregation according to SIFK severity.
Method
Study Oversight
This study was performed under the Mayo Clinic Institutional Review Board (IRB ID#15-000601). The study was designed and performed in compliance with the principles of Good Clinical Practice guidelines and the International Organization for Standardization (ISO) 14155, and in accordance with the tenets of the Declaration of Helsinki. All subjects in the ISA group provided written informed consent and no additional imaging was obtained specifically for this analysis; the institutional review board (IRB) waived informed consent requirements for Control group subjects because of the retrospective study design using anonymized patient data.
Study Participants and Matching
All study subjects had SIFK present at study entry, confirmed by MRI. Baseline and final radiographs, in addition to MRIs, were evaluated independently by 2 reviewers to confirm the diagnosis and location of SIFK, and meniscus and cartilage characteristics in both Controls and ISA subjects. Subjects from the ISA group were selected from enrollees in an ongoing medial knee OA clinical trial (ClinicalTrials.gov: NCT03671213 and NCT03838978), which is a prospective, multicenter study that enrolled subjects at 10 investigational sites in the United States and Europe. Inclusion criteria for the ISA trial included persons (1) age 25 to 65 years, (2) BMI <35 kg/m2, (3) body mass <300 lbs, and (4) diagnosed with unilateral OA pain in the medial knee compartment that continued after ≥6 months of non-operative treatment (physical therapy, intra-articular corticosteroid injections, bracing). Subjects with K-L Grade 4 were allowed, except those with large marginal osteophytes in the medial compartment or bony erosion.
Control subjects were identified using a retrospective search of our institutional radiology database from 2005 to 2017, using previously defined search criteria. 14 Radiographs and MRIs were reviewed independently by two reviewers, and patients with any identifiable SIFK were included. Patients with secondary osteonecrosis due to medical treatment (e.g., radiation, steroid treatment) and medical conditions (e.g., tumors, infection), and patients with evidence of prior surgery were excluded.
Control subjects with radiologic findings of SIFK were matched 1:1 to ISA subjects with SIFK according to SIFK score risk group, 14 age (±5 years), and BMI (±5 kg/m2). Subject matching by these criteria was performed using MedCalc v.19.2.6 software (MedCalc Software Ltd, Ostend, Belgium). Twenty-one subjects were enrolled in each study group.
Implantable Shock Absorber
The ISA is placed subcutaneously, but outside the joint capsule and superficial to the MCL, during an outpatient procedure using surgical techniques familiar to orthopedic surgeons. No weightbearing or range of motion restrictions are required. The ISA provides load absorption at low knee flexion angles, reducing peak cartilage-to-cartilage compressive forces by 31% and peak cartilage-to-meniscus compressive forces by 32%. 16 This unloading is equivalent to that of HTO, as demonstrated in a cadaver model. 17
SIFK Scoring
The SIFK score is a recently validated system that uses defined radiographic findings of patients with SIFK to predict the likelihood of progression to arthroplasty. 14 The SIFK score is easily calculated based on 5 clinical parameters: (1) lateral meniscus extrusion, (2) lateral meniscus root tear, (3) K-L Grade 4, (4) SIFK on the MFC, and (5) medial meniscus extrusion, with maximum scores for these individual parameters being 4, 2, 2, 2, and 1, respectively. 14 Thus, the maximum possible SIFK score (highest risk) is 11 points. The SIFK scoring system classifies patients as low (score 0-2), medium (score 3-4), or high risk (score ≥5) of undergoing arthroplasty within 4 years. Low-, medium-, and high-risk subjects had respective 4-year conversion rates of approximately 9%, 40%, and 80%. 14
Statistical Analysis
Data were extracted and standardized to arithmetic means and standard deviations as a measure of variance, taking sample size into account. Continuous variables are reported as mean ± standard deviation with the mean weighted for sample size where applicable. Differences between continuous variables were evaluated using a 2-sample, 2-tailed, Wilcoxon rank-sum test. One-way analysis of variance (ANOVA) was used to evaluate differences in Control subject age and BMI according to SIFK severity grouping. Categorical variables were reported as frequencies with percentages. Differences between categorical variables were evaluated using a χ2 analysis or Fisher exact test to account for small sample bias. Kaplan-Meier curves were used to evaluate survival. Analysis software included JMP v.12.0.1 (SAS Institute Inc., Cary, NC), Prism v.5.03 (GraphPad Software Inc., San Diego, CA), and Excel (Microsoft, Redmond, WA).
Power Analysis
An a priori power analysis was performed for sample-size calculation. 18 We conducted a hypothetical superiority analysis using the following parameters: 1:1 matching study with a conservative Group A (Control or matched group) arthroplasty-free proportion of 0.6, Group B (ISA) arthroplasty-free proportion of 0.8, and a superiority margin of 0.1. The analysis revealed that the current study needed a total of 20 patients in Group B (ISA). The group proportions were samples from previously established arthroplasty rates of medium-risk patients in a validated study. 14
An additional a priori power analysis was conducted for a 2-sample log-rank test to determine the sample size at an alpha level of 0.05, a power of 0.8, hazard ratio of 4, and a sample proportion of 1:1 in the cohorts. 18 This revealed a total of 16 patients would be needed per group.
Results
Subjects and Demographics
Our Control subject pool for matching comprised 249 patients, 68% female, with a mean age of 64.6 ± 10.5 years, and a median follow-up of 3.4 years. 14 Ultimately, 21 Control subjects were well-matched to an ISA counterpart and were enrolled and analyzed.
Of the 81 ISA subjects with medial knee OA who were initially screened, 39 were excluded because of no pre-operative MRI-confirmed SIFK. Of the remaining ISA subjects with confirmed SIFK at baseline (42 of 81, 52%), we enrolled 21 ISA individuals for whom we were able to identify 21 Controls who were reciprocally and precisely matched for SIFK score, age, and BMI. Thus, our study was comprised of 21 ISA subjects and 21 matched Control subjects.
Subject demographics, matching parameters, and SIFK scoring details are provided in Table 1. The whole-cohort mean age was 52.3 ± 8.7 years, mean BMI 29.5 ± 3.9 kg/m2, 40% female, and 29% of all subjects had K-L Grade 4. Control and ISA subjects were well-matched by age, BMI, and SIFK score. In both groups, SIFK scores categorized 19% of subjects (n = 4/group) as being low risk, 52% (n = 11/group) medium risk, and 29% (n = 6/group) at high risk of progression to arthroplasty. Of the five radiologic parameters comprising the SIFK score, SIFK on MFC, medial meniscus extrusion, and K-L grade were similarly common occurrences in both groups, whereas lateral meniscus aberrations were relatively rare.
Patient Characteristics of ISA and Control Cohorts Demonstrating Matching by SIFK Score, Age, and BMI.
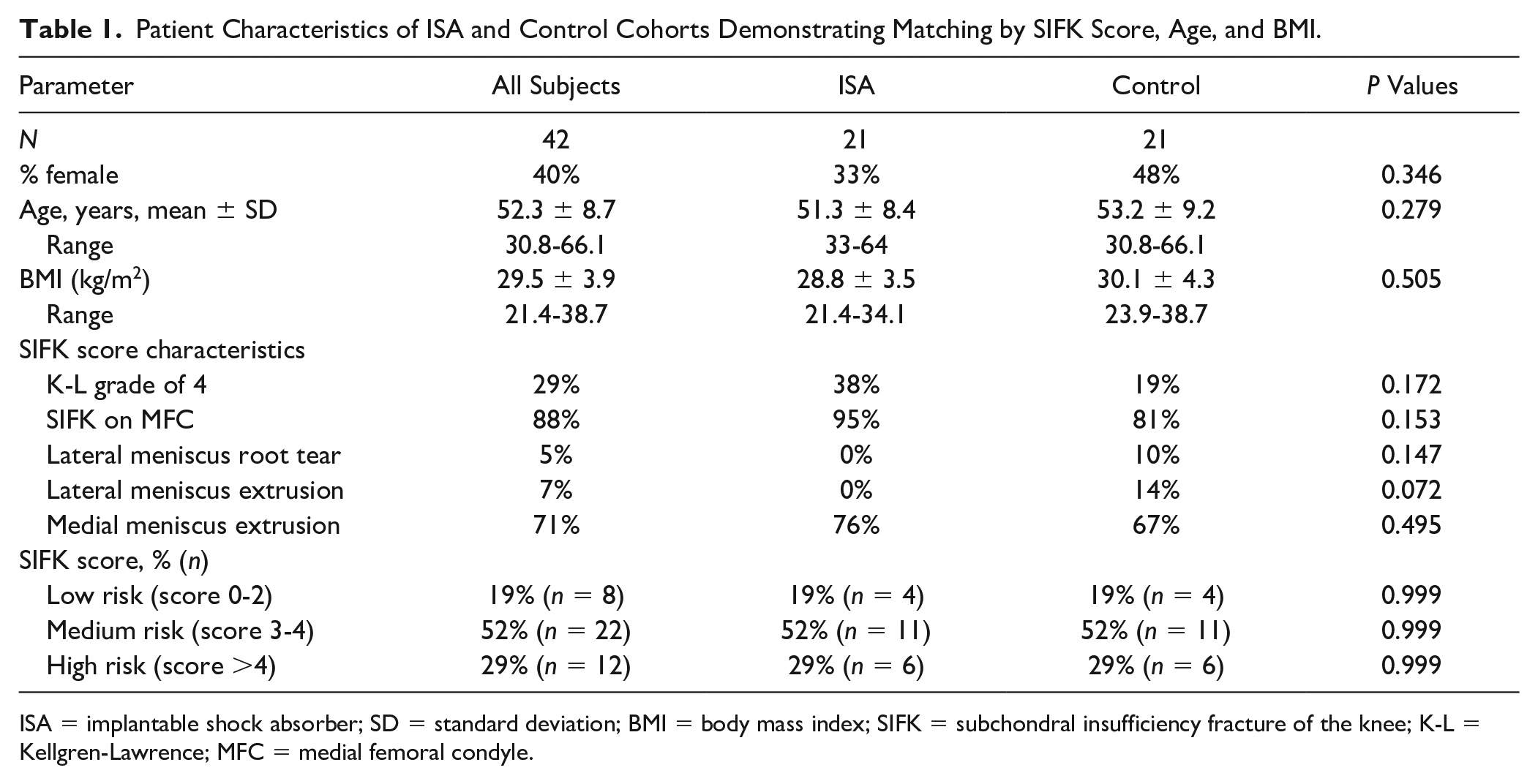
ISA = implantable shock absorber; SD = standard deviation; BMI = body mass index; SIFK = subchondral insufficiency fracture of the knee; K-L = Kellgren-Lawrence; MFC = medial femoral condyle.
Of ISA subjects, 62% (13/21) had a mean 1.6 previous procedures before undergoing ISA implantation. Procedures included meniscectomy (43%), microfracture or drilling (24%), debridement (19%), removal of chondral flaps or fragments (14%), meniscus repair (5%), osteochondral allograft of the medical femoral condyle (5%), and previous anterior cruciate ligament (ACL) reconstruction (5%). No ISA subject had osteotomies or meniscus transplants prior to ISA implantation. No Control subject had undergone prior knee surgery per inclusion criteria.
Follow-Up Duration
The mean total follow-up duration for all study subjects was 3.0 years. The mean follow-up durations for the Control group and the ISA group were 4.1 ± 2.8 years and 1.9 ± 0.2 years, respectively (P = 0.005). All ISA subjects completed their 2-year study follow-up visit, and the follow-ups occurred during a pre-specified window to facilitate subject scheduling.
Overall Progression to Arthroplasty
Overall, 19% of all study subjects progressed to unicompartmental or total knee arthroplasty (UKA or TKA) during the 2-year follow-up, with 100% of these cases occurring in the Control group versus 0% of ISA subjects (P = 0.001). The overall rate of arthroplasty in the Control group was 38% (n = 8/21 subjects). The mean time-to-arthroplasty in these Control subjects was 0.9 ± 0.7 years (range = 0.1-1.8 years).
Survival by Treatment and SIFK Severity
Of the 21 Control subjects, the median 1-year and 2-year arthroplasty-free survival probabilities were 76% and 55%. The 1- and 2-year rates for the ISA cohort were both 100%, significantly higher than the Control group (P = 0.001; Fig. 2 ).
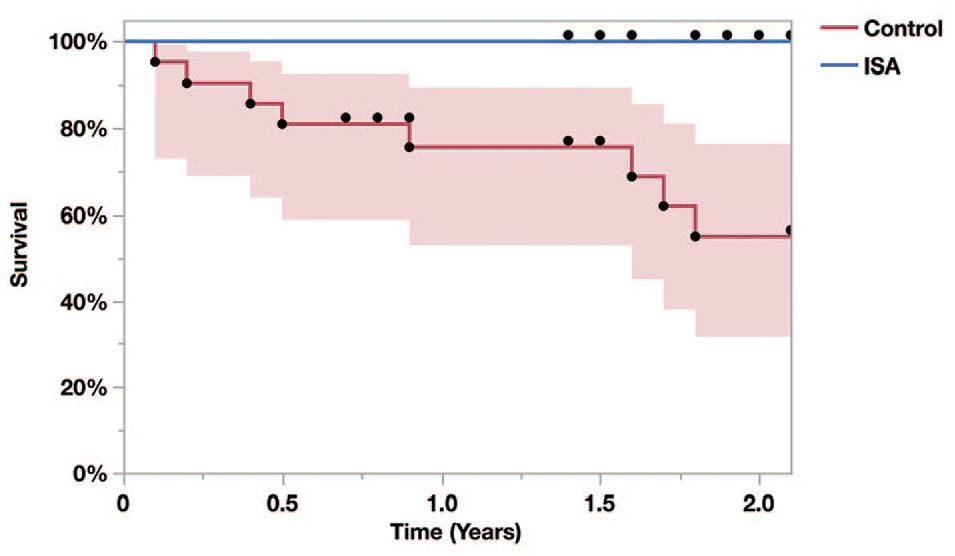
Whole-cohort 2-year freedom-from-arthroplasty: 100% (21/21) of the ISA subjects remained free-from-arthroplasty during the 2-year observation window. The median probabilities of Control subjects remaining free-from-arthroplasty at 1 and 2 years were 76% and 55% (P < 0.001 vs. ISA at both time points). Overall, 8/21 (38%) Control subjects required arthroplasty during the study period. Shading indicates 95% confidence intervals. ISA = implantable shock absorber.
No patients at low-risk, as stratified by SIFK severity scores, had progressed to arthroplasty in either the Control group or the ISA group through final follow-up ( Fig. 3A ). A sub-analysis was conducted between Control and ISA group with respect to subjects with medium- and high-risk SIFK scores. Medium-risk subjects in the ISA group had median 1-year and 2-year arthroplasty-free survival probabilities (100%, 100%) that trended higher than the Control group (90%, 68%, P = 0.07; Fig. 3B ). Among all subjects with high-risk SIFK scores, subjects who received an ISA also had significantly higher freedom-from-arthroplasty probabilities at 1 and 2 years, enjoying 100% survival at both time points, compared with high-risk patients in the Control group (33%, 0%, P = 0.002; Fig. 3C ) who had all progressed to arthroplasty before 2 years, with more than half undergoing surgery within 1 year.
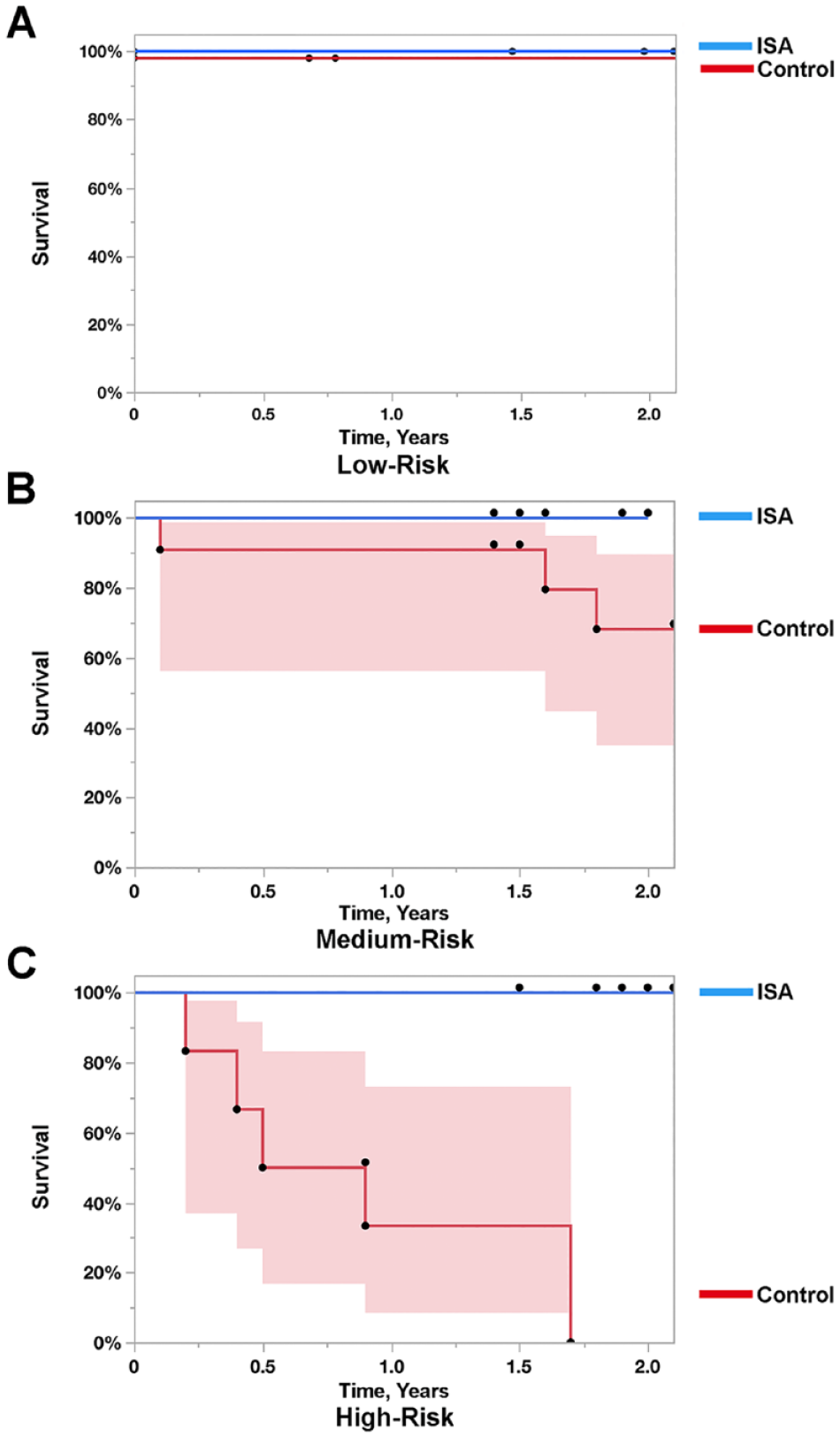
Freedom-from-arthroplasty through 2 years according to SIFK severity. None of the 21 subjects who received an ISA progressed to arthroplasty, regardless of their SIFK severity score. Non-surgically treated Control subjects underwent arthroplasty during this timeframe at a rate dependent on SIFK severity. (
Discussion
The primary findings of this study, which investigated progression to arthroplasty in subjects with knee OA, comparing subjects treated with an ISA with a matched Control group with non-surgical treatment are 2-fold: (1) Treatment with the ISA is associated with a reduced rate of arthroplasty through at least 2 years in subjects with medial-compartment knee OA and SIFK, and this protection appears especially strong in patients with high SIFK scores; and, (2) SIFK severity scores are predictive of progression-to-arthroplasty within 2 years in unoperated patients with similar knee OA degenerative symptoms who are only treated with non-surgical therapy. Patients with medium- and high-risk SIFK scores and OA represent a clearly defined population at increased risk of imminent arthroplasty, and intervention with the ISA appears to positively affect this risk.
Repetitive stress-induced SIFK is increasingly recognized as a collection of interlinked pathological phenomena with important implications in driving knee degeneration,3 -7 and SIFK may be a more appropriate designation than SONK for describing the tissue-level mechanisms of pathogenesis.4,5 Recently, degenerative meniscus tears and posterior root tears have been associated with development of SIFK. 4 Because the meniscus is the natural shock absorber of the knee, an insufficient meniscus increases contact pressures in the joint and facilitates an overloaded mechanical environment. It follows that reducing overload with an ISA may positively affect this mechanical environment, allowing the joint to achieve a state of relative mechanical stability.
Prognostic forecasts suggesting need for surgery cause stress that may influence postoperative functional outcomes and affect quality-of-life. 19 The SIFK scoring is easily performed during routine clinical evaluation, using standard imaging techniques. High-risk individuals with elevated SIFK scores could be better informed about potential benefits and risks of different treatment paths, thus making educated treatment decisions with realistic expectations for various outcomes. Incorporation of SIFK scoring may aid the surgeon in gauging the likelihood of disease progression-to-arthroplasty and facilitate dialogue and shared decision making with the patient regarding the suitability of alternative interventions.
During the SIFK scoring system development, it was initially tested for predicting 4-year conversion-to-arthroplasty rates in previously unoperated subjects with SIFK. 14 Of the validation cohort, 27% (66/249 subjects) underwent arthroplasty within 4 years; in our study, 14% of Control subjects advanced to arthroplasty within 1 year. The 4-year risk of progression-to-arthroplasty in low-, medium-, and high-risk SIFK groups of the earlier study was 8.8%, 40.4%, and 78.9%, respectively. 14 This resembles the respective 0%, 32%, and 100% conversion rates through 2 years that we observed in low-, medium-, and high-risk Control subjects.
The validation of the SIFK score determined BMI was not a significant factor in the model. In the general population, overweight and obese patients require TKA at a younger age than individuals of normal weight.20,21 However, average BMI appeared unrelated to SIFK score or 2- to 4-year arthroplasty rates in both the prior SIFK-prognostic study 14 and the current study, albeit with a maximum allowable BMI of 35. In a different OA-to-arthroplasty predictive model based on anthropomorphic and clinical parameters, BMI was closely correlated with arthroplasty risk, but this relationship disappeared when radiographic findings were additionally incorporated into the model. 22 In our model, inclusion of radiographic SIFK criteria similarly supersedes BMI in relative predictive importance.
Implantation of the ISA was associated with 100% freedom-from-arthroplasty through 2 years in the current matched pairs study. The average patient age in the ISA group was 52 years, with some subjects as young as 33 years old. Arthroplasty rates are steadily rising in younger patients <55 years old. 23 However, younger patients are more prone to experience early arthroplasty failure, with 1-year revision surgery rates estimated to be up to 5-fold higher than that in individuals older than 65 years, 24 and long-term implant survivorship may be unacceptably low in these patients if revision surgery is required.25,26 Therefore, the potential of the ISA to delay premature arthroplasty in a medium- or high-risk SIFK score patient is clinically compelling.
The ISA was developed as a reversible joint-sparing option for medial knee OA to overcome the efficacy shortcomings of conservative therapy while avoiding the highly invasive nature of arthroplasty that often involves bone cutting and removal. 27 ISA subjects in this current analysis were initially recruited to a study for medial knee OA, and the relatively common finding (52% of subjects) of MRI-observed SIFK on baseline screening indicates both the potential prevalence and clinical impact of risk stratification and appropriate intervention.
In a clinical trial that followed 25 persons aged ≤65 years for 2 years after receiving an ISA for mild-to-moderate severity medial knee OA, 100% of subjects reported Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) pain score improvement ≥20%, with almost all (96%) indicating a clinically relevant improvement in ability to perform daily activities. 13 No subject was reported to have progressed to arthroplasty during this timeframe, a result that mirrors the 100% avoidance of arthroplasty achieved through 2 years in participants of the current study, which included a similar rate of subjects with high-risk SIFK severity scores.
Primary limitations of our study were its relatively small sample size, and exclusion of persons >65 years old, which was due to the inclusion criteria of the primary ISA study from which subjects in this study were recruited. Our a priori power analysis was intended to identify survival difference between Control and ISA groups, so statistical power was lower when comparing survival across different SIFK severity subgroups. The ISA and Control groups were followed for different mean time points due to study design; namely, the matched pairs from the Control arm were selected from a historical database of previously identified patients. This limitation is mitigated, however, because all conversions in the Control arm occurred before 2 years, the minimum follow-up required for the ISA subjects. In addition, Kaplan-Meier analysis accounts for this differential follow-up, though does not address this discrepancy completely.
Although we only report 2-year ISA results here, these early results are encouraging. In addition, longer-term outcomes will be collected from the ongoing ISA trials, with analyses and reporting extended out to at least 5 years. Overall, the authors believe an implant like the ISA may be most useful for young patients with medial-compartment OA who wish to pursue active lifestyles and prefer joint replacement alternatives.
Conclusion
This study demonstrates the excellent effectiveness of the ISA in reducing progression-to-arthroplasty for at least 2 years after implantation, even in patients with very high-risk for progression. This study also highlights the utility of the SIFK scoring algorithm for predicting 2-year risk of conversion to arthroplasty. The ISA offers a useful load-reduction approach that may effectively delay or supersede the need for arthroplasty in certain patients.
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Moximed Inc., the manufacturer of the ISA device.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Institutional Review Board at Mayo Clinic, Rochester, MN, USA.
