Abstract
Objective
Vitrification of articular cartilage (AC) is a promising technique which may enable long-term tissue banking of AC allografts. We previously developed a 2-step, dual-temperature, multi-cryoprotectant agent (CPA) loading protocol to cryopreserve particulated AC (1 mm3 cubes). Furthermore, we also determined that the inclusion of ascorbic acid (AA) effectively mitigates CPA toxicity in cryopreserved AC. Prior to clinical translation, chondrocytes must remain viable after tissue re-warming and before transplantation. However, the effects of short-term hypothermic storage of particulated AC after vitrification and re-warming are not documented. This study evaluated the chondrocyte viability of post-vitrified particulated AC during a 7-day tissue storage period at 4 °C. We hypothesized that porcine particulated AC could be stored for up to 7 days after successful vitrification without significant loss of cell viability, and these results would be enhanced when cartilage is incubated in storage medium supplemented with clinical grade AA.
Design
Three experimental groups were examined at 5 time points: a fresh control (only incubated in medium), a vitrified − AA group, and a vitrified + AA group (N = 7).
Results
There was a mild decline in cell viability but both treatment groups maintained a viability of greater than 80% viable cells which is acceptable for clinical translation.
Conclusion
We determined that particulated AC can be stored for up to 7 days after successful vitrification without a clinically significant decline in chondrocyte viability. This information can be used to guide tissue banks regarding the implementation of AC vitrification to increase cartilage allograft availability.
Introduction
Articular cartilage (AC) serves as the smooth gliding surface covering the articulating surfaces of bones, facilitating low-friction joint motion and the transfer of forces in weight-bearing extremities. Damage to native AC is currently thought to be irreparable due to its avascular nature. This can result in osteoarthritis which is a significant burden to the health care system. 1 Focal chondral defects can be managed with a variety of surgical techniques, including cell-based resurfacing techniques that do not breach the subchondral bone, such as autologous chondrocyte transplantation (ACI) and matrix associated chondrocyte implantation (MACI). More recently, particulated AC transplantation has been developed using 1 mm3 (or less) cartilage fragments to fill the pure chondral defect. Fortunately, cartilage is an ideal transplant tissue since it is an immunoprivileged tissue and is easy to handle given its avascular and aneural state.
The mechanical mincing of AC into particulated cubes allows chondrocytes to escape and migrate from their surrounding matrix which facilitates chondrocyte proliferation and formation of a new hyaline-like cartilage matrix to repair the defect.2 -5 This technique is currently performed with fresh tissue that can only be stored up to 45 days at 4 ºC, after which it must be discarded. This often results in a waste of this precious tissue. Furthermore, approximately 14 days of that shelf life is exhausted due to requirements for infectious disease testing.6,7 Given this information, the ability to store AC long term would make it more readily available, economically feasible, and eliminate the need to throw away healthy, usable human tissue. Thus, preservation of chondrocyte viability would ensure that high-quality grafts are maintained and available for implantation at all times and would logically lead to improved clinical outcomes due to increased access at a lower cost. Long-term storage of cartilage allograft material has been a technical challenge. However, prolonged AC storage at 4 °C leads to substantial deterioration of chondrocyte viability after approximately 14 days of storage at low temperatures, despite the possibility that the integrity of the extracellular matrix is maintained.8 -10 Thus, establishing appropriate methods for long-term preservation of AC to extend tissue storage time beyond the standard 45 days is an important clinical objective. This would greatly increase the availability of donor AC tissue and make transplantation procedures more cost-effective and available to a wider population.
Although initial research using conventional cryopreservation methods can sufficiently preserve isolated chondrocytes, these techniques were unsuccessful when applied to intact cartilage tissue. This has been attributed to the formation of extracellular ice crystals which have been shown to disrupt the cartilage matrix.11 -13 Ice-free vitrification methods have been shown to be superior for post-thaw chondrocyte viability and function, thus have greater potential to allow long-term storage of intact cartilage tissue. Our group has been focused on vitrification as a means of long-term AC storage.14 -20 Vitrification uses high concentrations of cryoprotective agents (CPAs) and rapid cooling to preserve cells or tissues in a non-crystalline, amorphous solid that is in a glassy state at −196 °C.15,21 However, the use of high CPA concentrations and rapid cooling in vitrification causes various cellular stresses such as osmotic stress, chilling injury, oxidative stress, and CPA toxicity.22 -24 We previously developed an optimized vitrification protocol which successfully vitrified intact human AC with promising chondrocyte recovery and metabolic function. 15 Since then, our research has explored different ways to optimize our vitrification protocol to address current challenges in AC cryopreservation.16,18,25
We have investigated the inclusion of supplemental additive compounds as a means to reduce CPA toxicity by mitigating oxidative stress.26 -29 Various antioxidant compounds such as chondroitin sulfate or ascorbic acid (AA) have been evaluated for their ability to protect cartilage during high exposure to CPA. Notably, we have previously demonstrated that AA provides significant chondroprotection to AC during cryoprotectant exposure/removal and during short-term hypothermic storage.25 -29 We have also shown that using clinical grade AA (i.e., the pharmaceutical equivalent of this additive) provided similar chondroprotective effects to chondrocytes when compared with research grade AA, allowing our research group to translate AA supplementation into vitrification protocols intended for human cartilage. 27
More recently, our group developed a 2-step, dual-temperature, multi-cryoprotectant loading protocol that successfully vitrified AC cubes (pig and human) and showed that the cartilage maintained high cell viability and metabolic function immediately after vitrification and re-warming. This approach of vitrifying particulated AC can theoretically enable donor tissue to be stored for an indefinite amount of time. 19 However, the effects of prolonged hypothermic storage on vitrified tissue once re-warmed are unknown. From a tissue banking perspective, it is necessary to determine the amount of time for which donor AC grafts can maintain an acceptable cell viability when stored at 4 °C after vitrification and re-warming, and prior to transfer to clinical centers for transplantation.
The purpose of this study was to investigate the effects of storage duration on the chondrocyte viability of particulated AC after a vitrification and re-warming protocol. In addition, we sought to investigate whether chondrocyte viability would be further enhanced in medium supplemented with clinical grade AA. We hypothesized that porcine particulated AC can be stored for up to 7 days at 4 ºC after successful vitrification without significant loss of cell viability, and these results can be enhanced with medium supplementation of clinical grade AA during tissue storage.
Materials and Methods
Experimental Groups
A total of 3 experimental groups (N = 7 replicates for each group) were examined in this study: Group 1 (Fresh positive control): only incubated in Roswell Park Memorial Institute (RPMI) medium for 1 week; Group 2: Exposed to a CPA mixture, vitrified, re-warmed, and then incubated in RPMI medium for 1 week; Group 3: Exposed to a CPA mixture, vitrified, re-warmed, and then incubated in RPMI medium supplemented with 2,000 μM clinical grade AA (PCCA, ON, Canada) for 1 week. All experimental groups were stored at 4 °C during incubation in RPMI medium.
Sample Preparation
Porcine stifle joints from sexually mature pigs were acquired from a local meat shop (Delton, Edmonton, AB, Canada) and no animals were specifically killed for this study. Porcine joints were dissected, and the full thickness AC was shaved off the bone base of the femoral condyles. AC was mechanically minced into cubes approximately 1 mm3 in size, using a round perforated cutting block and a razor blade. The cartilage cubes were disinfected in 25 mL of phosphate buffered saline (PBS) with antibiotic-antimycotic (10,000 units/mL penicillin, 10,000 µg/mL streptomycin, and 25 µg/mL Amphotericin B [Gibco]) under a biological safety cabinet for 15 minutes. AC cubes were stored overnight at 4 °C in RPMI medium with antibiotics (240 mg/L cefoxitin sodium, 2 mL/L tobramycin, 50 mg/L vancomycin) which are routinely used by the local tissue bank (Comprehensive Tissue Center [CTC]; Edmonton, AB, Canada).
CPA Loading Protocol and Vitrification
All cryoprotectant solutions were prepared in RPMI medium. A 2-step, dual-temperature, multi-CPA loading protocol was used for this study which was developed by Wu et al. 19 Approximately 0.5 g wet weight of particulated AC (1 mm3 cubes) per experimental group were weighed on an electric balance scale (Mettler Toledo) and loaded into 50 mL conical tubes for CPA loading. For the first step of this protocol, particulated AC was incubated in a multi-CPA cocktail containing 3 Molar (M) dimethyl sulfoxide (DMSO) and 3 M ethylene glycol (EG) at 0 °C for 10 minutes. The second step involved incubation in another CPA cocktail containing 3 M DMSO, 3 M EG, and 2 M propylene glycol (PG) at −10 °C for 20 minutes. Multi-CPA solutions were prepared in RPMI medium, filtered using a sterile vacuum filter unit, and cooled in either an ice-water bath (0 °C) or a methanol cooling machine (−10 °C).
After CPA loading, particulated AC cubes were removed from the conical tubes and immediately transferred into sterile 1.8 mL cryovial tubes with a residual amount of the multi-CPA cocktail, and the cryovial tubes were plunged into liquid nitrogen (−196 °C) for vitrification for 4 days (as illustrated in Fig. 1 ). To re-warm the tissue after vitrification, the cryovial tubes were removed from liquid nitrogen and warmed in a 37 °C water bath for 30 seconds until the surrounding vitrified glass liquefied. The cartilage cubes were washed in 25 mL RPMI medium for 30 minutes at 4°C twice.
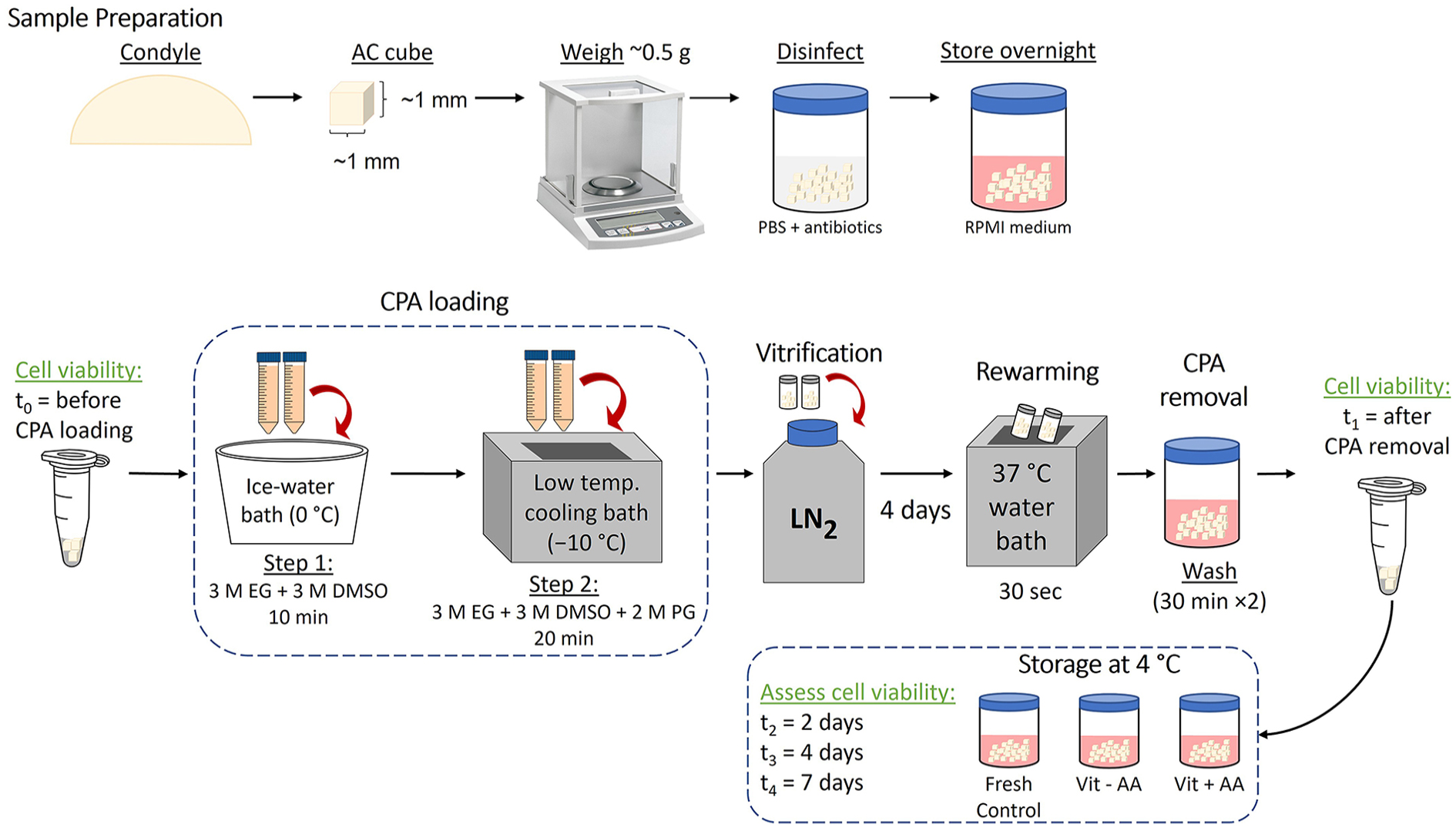
Experimental flowchart. Particulated articular cartilage (approximately 1 mm3) was obtained from the femoral condyles of porcine stifle joints. AC cubes were exposed to a multi-CPA cocktail and then samples were transferred to cryovial tubes and plunged into liquid nitrogen to achieve vitrification. To extract the cartilage, the cryovial tubes were warmed in a 37 °C water bath and then immediately washed with RPMI media to remove residual CPAs. AC cubes were immersed in a dual fluorescent stain and imaged with confocal microscopy to assess chondrocyte viability. During the 7-day tissue storage, particulated AC was incubated at 4 °C and the chondrocyte viability was measured at 5 different time points: t0 = before CPA exposure, t1 = post vitrification and CPA removal, t2 = after 2 days, t3 = after 4 days, and t4 = after 7 days storage. AC = articular cartilage; PBS = phosphate buffered saline; RPMI = Roswell Park Memorial Institute; CPA = cryoprotective agent; DMSO = dimethyl sulfoxide; EG = ethylene glycol; PG = propylene glycol; AA = ascorbic acid; LN2 = liquid nitrogen.
Assessment of Chondrocyte Viability
Chondrocyte viabilities of the cartilage cubes were assessed at 5 time points: t0 = before CPA exposure; t1 = directly after vitrification, re-warming, and CPA removal; t2 = after 2 days incubation post-vitrification/re-warming; t3 = after 4 days incubation post-vitrification/re-warming; and t4 = after 7 days incubation post-vitrification/re-warming. The sample inclusion criteria for particulated cartilage cell viability prior to vitrification were a minimum of 85% at t0; any samples that did not meet the sample criteria were excluded from the experiment. Cell viability of the cartilage cubes was assessed by using a dual fluorescent membrane integrity stain containing 6.25 µM Syto 13 (Molecular Probes, OR, USA) and 9.0 µM propidium iodide (Sigma, MO, USA) mixed in PBS. Syto 13 is a membrane-permeant nucleic acid stain which fluoresces green in viable (membrane-intact) cells. Propidium iodide is a membrane-impermeant dye that penetrates non-viable (membrane-damaged) cells and fluoresces red. At each time point, the viabilities for experimental groups were obtained by calculating the mean of the viability measurements from randomly chosen cartilage cubes. These cartilage cubes were immersed in 150 µL of the dual membrane integrity stain in a microcentrifuge tube and incubated in the dark for at least 15 minutes before imaging.
Particulated AC cubes were imaged using a Leica TCS SP5 laser confocal microscope, and all confocal images were processed using the LAS X software (Leica Microsystems). Viability 3.2, a custom-made cell-counting software (Locksley McGann, University of Alberta) was used to quantify viable and non-viable cells within the cartilage cubes.
For statistical analysis, a two-way repeated measures analysis of variance (ANOVA) with post hoc multiple comparisons was conducted using SPSS 20.0 software. Data are expressed as means ± standard deviations, and statistical significance is indicated when P < 0.05.
Results
We evaluated the chondrocyte viability of post-vitrified AC cubes that were incubated in storage medium at 4 °C for up to 7 days. We observed that the average percent cell viability of all experimental groups declined in a linear fashion over 7 days. However, it is important to note that the average chondrocyte viability for all groups was over 80% at all time periods as seen in Figure 2 . Two-way ANOVA and post hoc pairwise comparisons were performed to determine which experimental groups had a statistically significant change in cell viability and at what time point this viability change occurred.
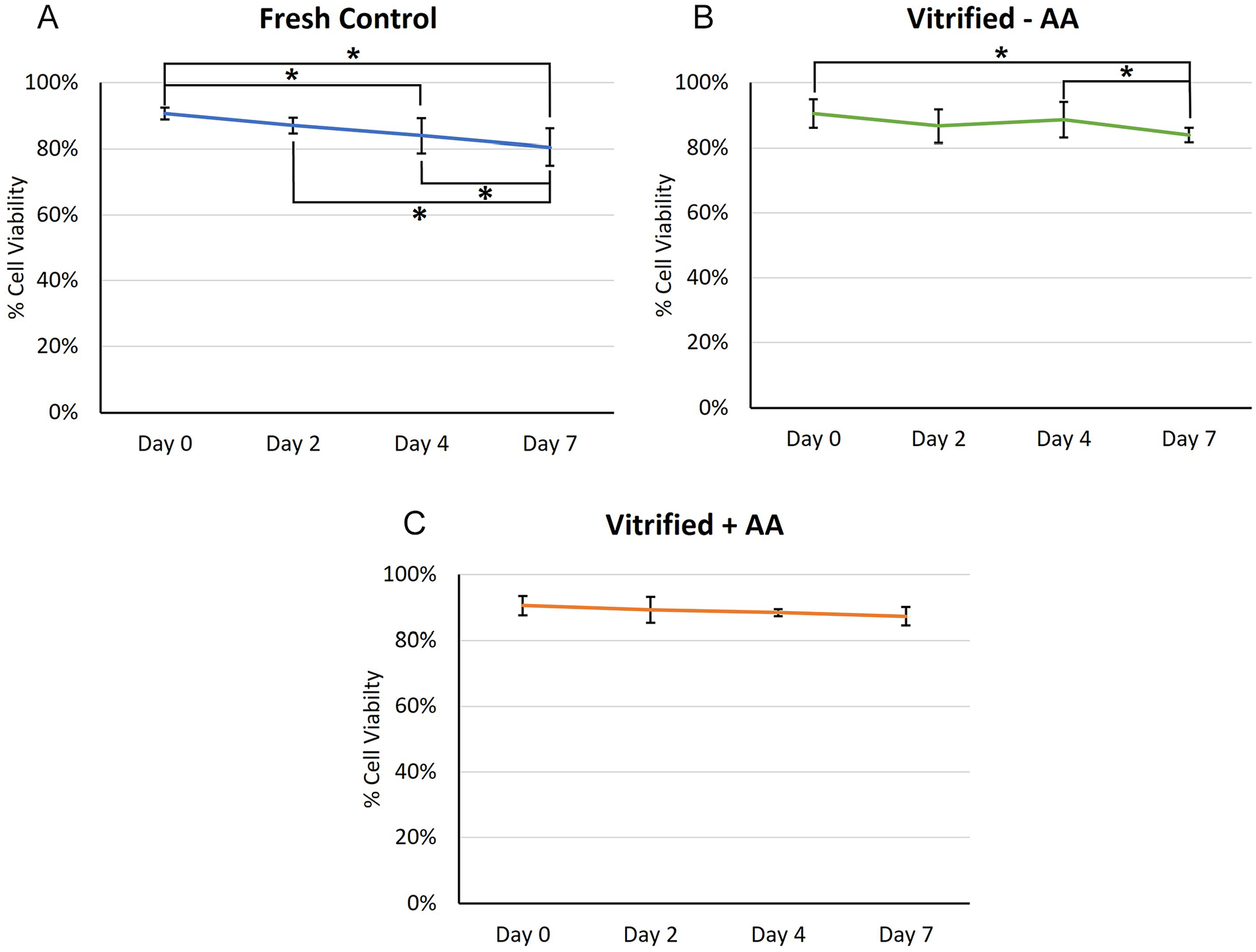
Means of percent cell viability in particulated cartilage cubes after a 7-day tissue storage period at 4 °C (N = 7) (Mean ± SD). (
Figure 2 depicts the change in chondrocyte viability over the 7 days of tissue storage. The cell viability of the fresh control declined by 10.1% over the storage period with statistically significant changes from Day 0 to Day 4, from Day 0 to Day 7, from Day 4 to Day 7, and from Day 2 to Day 7 ( Fig. 2A ). The vitrified − AA group cell viability declined by 6.6% over the storage period, and there were significant declines in cell viabilities from Day 0 to Day 7 and from Day 4 to Day 7 ( Fig. 2B ). In contrast, the percent cell viability of the vitrified + AA group decreased by only 3.3% and did not show any statistically significant differences in time point comparisons ( Fig. 2C ).
Figure 3 compares the mean chondrocyte viability of all experimental groups at each time point of viability measurements. Post hoc pairwise comparisons indicated that the cell viability of all groups is similar at Day 0, 2, and 4. However, at t4 (Day 7 of the storage period), the average cell viability of the vitrified + AA was significantly greater than that of the fresh control (P = 0.007).
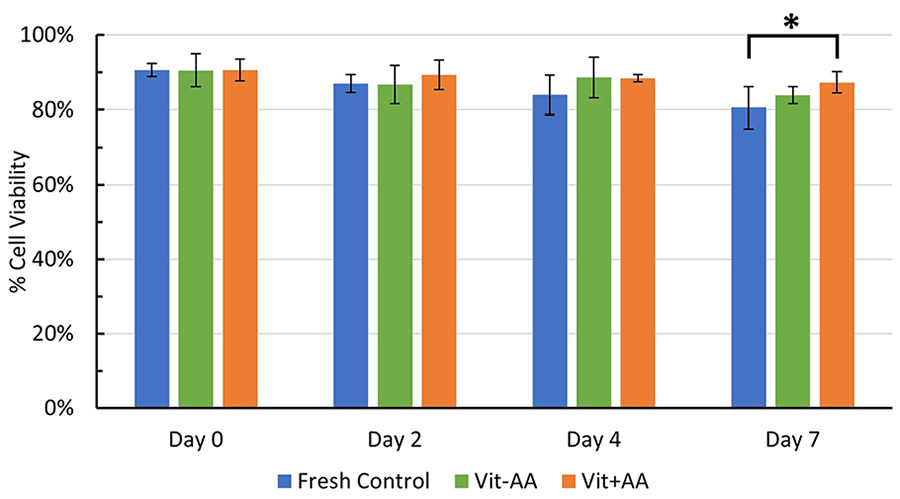
Comparison of percent cell viability means in fresh and vitrified groups at Day 0, 2, 4, and 7 (N = 7) (Mean ± SD). All groups had similar percent cell viabilities at Day 0, 2, and 4. However, at Day 7 of tissue storage, percent cell viability of the vitrified + AA group was significantly greater than in the fresh control group (P = 0.007), whereas Day 7 comparisons of cell viability with the vitrified − AA versus vitrified + AA group did not result in any statistical significance (P = 0.142). SD = standard deviation; AA = ascorbic acid. *P < 0.05.
Discussion
This study examined the effects of short-term hypothermic tissue storage on chondrocyte viability in particulated AC after it has been vitrified and warmed. The results show that the chondrocyte viability of both the fresh and vitrified AC cubes marginally declined over the 7-day storage period ( Fig. 2 ). However, the average cell viability for all experimental groups remained above 80% over the 7-day storage period which is much higher than the minimum recommended cell viability of 70% suggested for successful allograft transplantation.30,31 This finding is consistent with previous studies that show a correlation of prolonged storage time at 4 °C with a decrease in chondrocyte viability, proteoglycan loss, cellular apoptosis, and weakened matrix integrity, some of which can occur within 7 to 14 days of storage.10,32 -36
Transplantation of particulated AC allografts has been developed over the past 2 decades as a viable option for the treatment of full thickness chondral defects.7,37 Fresh allografts currently used are commercially provided as De Novo by Zimmer. 38 These grafts are typically obtained from juvenile donors and the cartilage is harvested and minced into tiny pieces which are stored for a short period of time before implantation. 2 Cartilage tissue that is not implanted by the expiry date is discarded, thus leading to the unfortunate wastage of precious tissue obtained from very young donors. Vitrification of juvenile cartilage would allow indefinite storage of cartilage tissue since vitrified cells do not age or undergo biochemical reactions.
Our research group has recently developed a technique to successfully vitrify 1 mm3 cubes of AC. 19 The current study demonstrates that this technique is reproducible when performed by different people with approximately 90% cell viability post vitrification (performed by M.C., the first author), which is similar to results obtained by Wu et al. 19 One aim of this current study was to provide guidance to tissue banks on how long they can store the re-warmed cartilage post vitrification before transferring it to the surgeon for implantation. The ability to store donor tissue temporarily at 4 °C would only be used after vitrification storage to allow sufficient time for donor tissue to be delivered to the hospital for surgery and the completion of other surgical preparations in a timely manner. This study showed that a minimum of 7 days of storage at 4 °C is acceptable post re-warming of vitrified particulated AC, and that vitrified chondrocytes survived similarly to, or better than, fresh chondrocytes over that time period.
Our results also indicated that at Day 7 of tissue storage, the mean percent cell viability of the vitrified + AA group was significantly higher than that of the fresh control; however, AA supplementation in the vitrified + AA group did not significantly improve chondrocyte viability when compared with the vitrified − AA group which contradicted our expectations ( Fig. 3 ). Although we hypothesized that the inclusion of clinical grade AA in the storage medium would enhance chondrocyte viability, the results suggest that AA supplementation was not significantly contributing to maintaining the chondrocyte viability of post-vitrified particulated AC. Based on our previous studies evaluating the effectiveness of additives on cryopreserved porcine AC, we reported the effectiveness of AA in mitigating CPA toxicity during cryoprotectant exposure, removal, and short-term hypothermic storage in culture medium. 29 It has been reported that AA supplementation effectively provides protection to chondrocytes experiencing cellular stresses through antioxidant effects which mitigates oxidative stress, as well as supporting the maintenance of cartilage matrix structures.26,39 Hence, we chose to move forward with using AA in the current study as it provided the most chondroprotection to AC subjected to CPA exposure. One limitation of our study is that we did not include AA during CPA loading steps and only investigated AA supplementation during tissue storage in the storage medium. Perhaps the chondrocyte viability in the vitrified + AA group could have been further improved if AA was supplemented throughout the entire vitrification protocol. Another interesting finding is that at Day 7, the vitrified + AA group had a cell viability that was statistically greater than that of the fresh control group. The 7 day statistically significant difference may have been due to a delay in the restart of metabolic activity post vitrification by 48 hours as noted in our previous work15,19 which may have also delayed the onset of cell metabolic stress.
Currently, hypothermic storage at 4 °C is the standard storage regimen that tissue banks utilize for AC allografts. Many studies have determined that hypothermic storage is sufficient for optimal chondrocyte survival and it has been suggested that the metabolic needs of chondrocytes are reduced at 4 °C facilitating prolonged maintenance of chondrocyte viability.35,40 However, other studies recommend that AC allografts should be stored at higher temperatures such as at room temperature or 37 °C. The rationale provided from the literature is that at physiological temperatures, the chondrocyte viability and the integrity of the AC matrix would be better preserved as it resembles in vivo conditions.41 -44 However, Mickevicius et al. 45 determined that chondrocytes stored at 4 °C have higher cell viabilities, lower apoptosis levels, and maintained glycosaminoglycan distribution in the cartilage in comparison to chondrocytes stored at 37 °C. Furthermore, tissue storage at physiological temperatures would make AC allografts more susceptible to bacterial infection. For these reasons, storing post-vitrified particulated AC at 4 °C would limit tissue wastage and provide more optimal clinical outcomes.
There is no universal standardization of AC allograft storage conditions. One major challenge in incorporating clinically relevant literature toward tissue bank practices is the wide variability in tissue sources (autopsy, transplants, etc.), tissue types (especially when comparing human and animal models), length of storage time, culture medium used, and so on.36,46 The guidelines for tissue banks in Canada and the United States require that a suitable tissue storage period is established, along with defining the preservation method, storage conditions, and packaging of the tissue product.47 -49 Our current study validates that particulated AC that has been vitrified with our protocol and re-warmed may be held in cold storage for at least 7 days while maintaining excellent chondrocyte viability, which justifies the practical implementation of vitrification as a way to cryopreserve AC allografts in tissue banks.
Conclusion
In conclusion, we determined that particulated AC can be stored for up to 7 days after successful vitrification without a clinically significant decline in cell viability. This current study can inform tissue banks of the time frame in which post-vitrified particulated AC can maintain an acceptable cell viability after tissue re-warming and before surgery, and therefore further improve access to this tissue to enhance clinical outcomes of allograft transplantation procedures. Applying practical vitrification methods to preserve intact human AC in tissue banks can increase the storage time and availability of cartilage allografts while decreasing costs associated with tissue wastage, thus allowing the current limitations of hypothermic tissue storage to be addressed.
Footnotes
Acknowledgments and Funding
J.A.W.E. holds a Canada Research Chair in Thermodynamics. K.W. was funded by the Li Ka Shing Sino-Canadian Exchange Program between University of Alberta and Shantou University. We thank Dr. Locksley McGann for the creation of the Viability 3.2 software. We also thank the Cell Imaging Centre FoMD for assistance with confocal microscopy. This research was supported by the Edmonton Orthopaedic Research Committee and a University of Alberta Department of Surgery Summer Studentship.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: N.M.J. and J.A.W.E. are coinventors of US (no. 8,758,988) and Canadian (no. 2,788,202) patents for cryopreservation of articular cartilage: N.M. Jomha et al., Cryopreservation of articular cartilage, University of Alberta. The remaining authors declare no competing interests.
Ethical Approval
The use of animal tissue in this research was approved by the Research Ethics Office at the University of Alberta.
