Abstract
Objective
In the early stages of cartilage damage, diagnostic methods focusing on the mechanism of maintaining the hydrostatic pressure of cartilage are thought to be useful. 17O-labeled water, which is a stable isotope of oxygen, has the advantage of no radiation exposure or allergic reactions and can be detected by magnetic resonance imaging (MRI). This study aimed to evaluate MRI images using 17O-labeled water in a rabbit model.
Design
Contrast MRI with 17O-labeled water and macroscopic and histological evaluations were performed 4 and 8 weeks after anterior cruciate ligament transection surgery in rabbits. A total of 18 T2-weighted images were acquired, and 17O-labeled water was manually administered on the third scan. The 17O concentration in each phase was calculated from the signal intensity at the articular cartilage. Macroscopic and histological grades were evaluated and compared with the 17O concentration.
Results
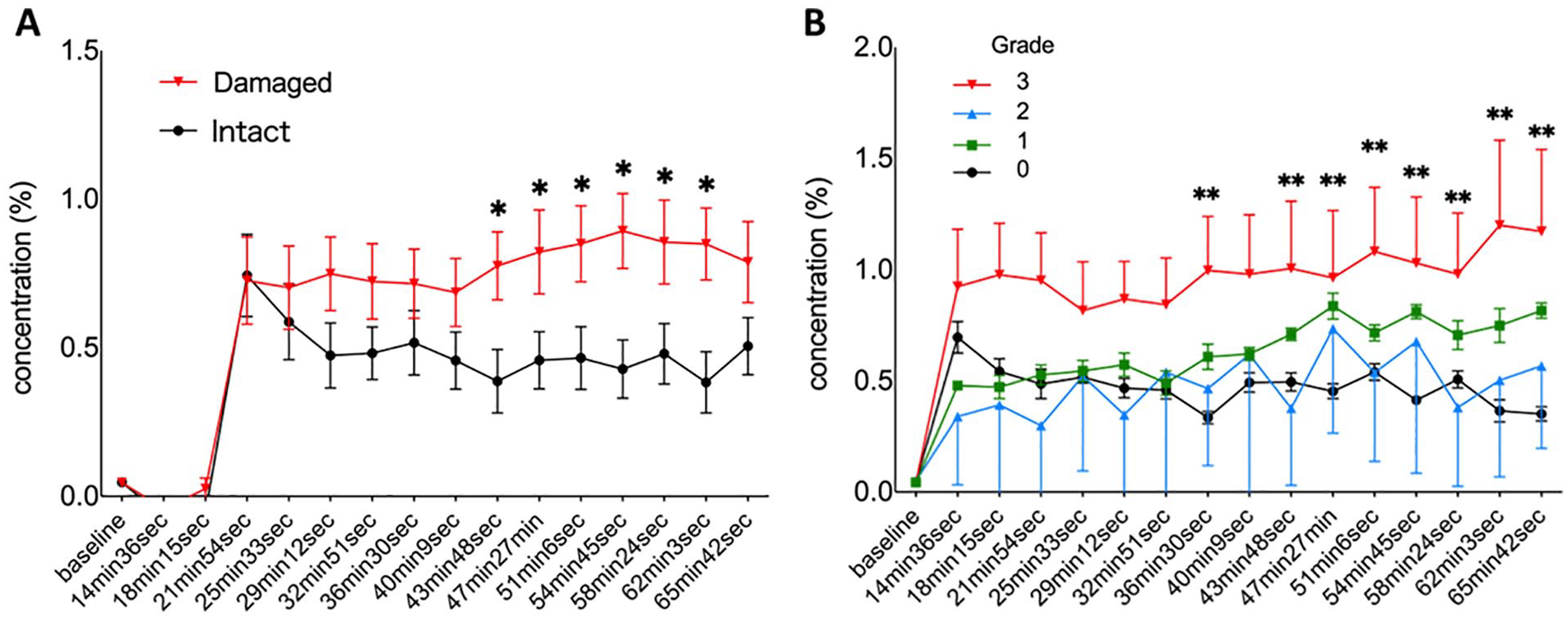
An increase in 17O concentration in the macroscopic and histologically injured areas was observed by MRI. Macroscopic evaluation showed that the 17O concentration significantly increased in the damaged site group. Histological evaluations also showed that 17O concentrations significantly increased at 36 minutes 30 seconds after initiating MRI scanning in the Osteoarthritis Research Society International (OARSI) grade 3 (0.493 in grade 0, 0.659 in grade 1, 0.4651 in grade 2, and 0.9964 in grade 3, P < 0.05).
Conclusion
17O-labeled water could visualize earlier articular cartilage damage, which is difficult to detect by conventional methods.
Introduction
Osteoarthritis (OA) is one of the most common causes of locomotor disorders characterized by cartilage degradation, bone remodeling, osteophyte formation, joint inflammation, and loss of normal joint function, which commonly affects weight-bearing joints, such as the knees, hips, spine, hands, and feet.1-3 At present, OA diagnosis relies on conventional imaging methodologies, such as radiography, magnetic resonance imaging (MRI) or ultrasound, and invasive arthroscopy. Early stage of OA has very few signals in radiography; thus, arthroscopy and MRI provide an assessment complementary. 4 Epidemiologically, OA affects a large number of patients; prevalence of radiographically confirmed knee OA in Japan has been reported to be 42.0% in males and 61.5% in females aged above 40 years. 5 However, there is still no appropriate modality for OA diagnosis, especially for early-stage OA.
Recently, there has been growing interest in developing MR techniques for the accurate diagnosing and staging of OA. For the purpose of evaluating structural alterations in articular cartilage, qualitative evaluation of cartilage has been performed by measuring the relaxation time of T1, T2, and T1ρ.6-8 Furthermore, with the recent advances in MRI technology, new MRI evaluation methods that can detect changes in the molecular structure of articular cartilage with high sensitivity are being clinically applied.9,10 However, the MRI evaluation has been inferior to arthroscopic findings in diagnosing cartilage damages. 11 While it may be possible to assess the structural properties of articular cartilage by quantifying the susceptibility of collagen fibers in articular cartilage by T1ρMRI, 8 it is theoretically impossible to evaluate the metabolic dynamics of cartilage substrates. To assess articular cartilage metabolism, there are evaluation methods such as delayed contrast-enhanced cartilage MRI (dGEMRIC), which examines the amount of glycosaminoglycans (GAGs) in the cartilage. 12 Although it is considered more sensitive than other MRI imaging methods for detecting cartilage damage, contrast-enhanced MRI involves the risk of allergic reactions.
Maintaining hydrostatic pressure in cartilage tissue affects not only mechanical properties, but also chondrocyte mechanotransduction and cell survival through apoptosis. In the early stages of cartilage damage, disruption of the hydrostatic pressure maintenance mechanism of cartilage tissue is thought to alter the mechanical properties of the cartilage matrix.13,14 An early feature of OA is the loss of GAG from cartilage tissues, and disruption of cartilage hydrostatic pressure maintenance mechanisms is thought to induce chondrocyte apoptosis. Hydrostatic pressure also acts as a modulator of chondrocyte morphology and metabolism. 15 Therefore, it is important to evaluate the ability of cartilage matrix to maintain hydrostatic pressure. On the contrary, hydrostatic pressure in cartilage tissue is maintained by the binding of water to proteoglycans within the cartilage matrix. Hence, it may be possible to estimate the hydrostatic pressure of the cartilage matrix by tracking the water molecules in the cartilage matrix. Authors slowly administered a 20% 17O-labeled water inside the joint. How much contrast was finally injected? Please, define why a single administration was not established. However, no modality has been developed that allows direct observation of water-binding capacity or tracking of water itself.
The 17O isotope, which is the only stable isotope of oxygen, has the advantage of being free from radiation exposure and allergic reactions. It can produce an MRI signal, 16 and dynamic steady-state sequences for indirect 17O imaging and absolute quantification of 17O concentration using MRI have been reported. 17 Because it can be distinguished from the host joint fluid, MRI can directly observe the dynamics of exogenous hydrogen oxide that penetrates the cartilage matrix. Therefore, it might be possible to visualize alterations in the hydrostatic pressure of the cartilage by observing the metabolic dynamics of water molecules in cartilage using 17O-labeled water by MRI.
We hypothesized that spatiotemporal alterations of cartilage detected by MRI with the 17O-labeled water would enable the visualization of the early stage of articular cartilage damage and the alteration in the hydrostatic pressure of cartilage. This study aimed to develop a novel modality of MRI using an intra-articular injection of 17O-labeled water and to evaluate its accuracy and diagnostic ability using a rabbit model.
Methods
Optimization of 17O-Labeled Water Administration
Administration of 17O-labeled water was performed using 14-week-old Japanese white rabbits. A 24G indwelling needle was inserted through the patellar tendon from the front of the knee joint, and the tip of the catheter was placed in the joint. Then, 10 mL of saline solution was administered to confirm that the joint could be injected reliably, and the joint was expanded to facilitate evaluation. A circuit that can administer the drug solution into the joint during MRI was created by connecting the indwelling needle and extension tube. During continuous MRI, 0.5 mL of 20% concentration of 17O-labeled water (Taiyo Nippon Sanso, Tokyo) was slowly administered into the joint in the examination room because large injection into the joints of small animals may shift the position of joint. The 17O concentration at the end of the experiment was approximately 1% under the influence of a pre-administered saline solution. Before performing arthrography, the effects of arthrography on living animals and amputated limbs were examined as a preliminary study. As there was no difference between the images obtained from the living animal and the amputated limb, we determined to perform arthrography by transecting the limb at the thigh to minimize the effect of body movement.
Rabbit OA Model
Japanese white rabbits (14 weeks old) were used for anterior cruciate ligament (ACL) transection surgery to induce OA. Rabbits were anesthetized with intravenous sodium pentobarbital (30 mg/kg) and ketamine (50 mg/kg). Anesthesia was maintained under oxygen isoflurane inhalation anesthesia. The right hind limb was shaved and cleansed with povidone-iodine. After local anesthesia with xylocaine (0.02 mg/kg), the skin at the medial side of the kneecap was incised about 5 cm to expose the patella and patellar tendon. After the medial joint capsule was incised, the ACL was exposed by dislocating the patella laterally with the knee flexed. The ACL was cut with a surgical scalpel, and complete resection of the ACL was confirmed by displacing the tibia anteriorly from the femur. The patella was moved back to the midline, and the fascia and skin were sutured sequentially using 3-0 polydioxanone and 3-0 nylon threads. After irrigation of the operative field with saline, the patellar dislocation was reduced, and the fascia and skin were sutured sequentially using 3-0 nylon threads. Postoperatively, euthanasia was performed if wound infection was observed or if the patient was unable to stand. Degeneration of the cartilage surface begins 4 weeks after cartilage damage and occurs in the entire cartilage layer after 8 weeks. 18 Half of the rabbits were euthanized with pentobarbital (200 mg/kg) 4 weeks postsurgery and the other half were euthanized 8 weeks postsurgery, and MRI and macroscopic and histological evaluations were performed.
Examination of MRI Conditions
MRI was conducted using a 3.0 Tesla scanner (MAGNETOM Prisma; Siemens, Tokyo) with a 4-channel small flex coil. To evaluate the most loaded part of the femur, 5-slice scan was performed centering on the most loaded part of the femur, with a coronal section tilted 45° to the bone axis. First, an MRI image of a proton density–enhanced image was taken as a reference image to identify the articular cartilage (TE, 21 ms; TR, 1600; FOV, 60 mm × 80 mm; matrix, 269 × 448; slice thickness, 2 mm; slice gap, 2.6 mm; number of excitations, 6; scan time, 3 minutes 39 seconds). Second, T2-weighted images using the 2D-fast spin echo method were performed continuously (TE, 129 ms; TR, 1600; echo train length, 12; FOV, 60 mm × 80 mm; matrix, 269 × 448; slice thickness: 2 mm; slice gap, 2.6 mm; number of excitations: 6; scan time: 3 minutes 39 seconds; number of repetitions: 18; total scan time: 65 minutes 42 seconds). The scan duration was 3 minutes 39 seconds, and the sequence was repeated 18 times (total scan time, 65 minutes 42 seconds). The voxel sizes of the 2 sequences were both 0.22 mm × 0.2 mm. The catheter was placed in the rabbit knee joint as in the optimization conditions, and 3 scans after the start of this dynamic scanning, 0.5 mL of 17O-labeled water (enriched to 20%; Taiyo Nippon Sanso Corp, Tokyo) was manually administered via the catheter. The injection rate was approximately 0.5 Â mL/s, and a 0.5 mL saline flush was performed.
MRI and Evaluation
The articular cartilage in each slice section was identified from the proton density–enhanced images, and multiple small regions of interest (ROIs) were set at 6 locations in the center, medial, and lateral sides of each condyles of articular cartilage of femur. Then, the 17O concentration in each phase of each ROI was calculated using the signal intensity of the T2-enhanced image via the following equation: 17O concentration (%) = −1/(TE×R217O) × log(S/S0) + 0.038; TE, echo time; R217O, 3.33; S, the signal intensity of ROI; S0, mean signal intensity of ROI before administration; 0.038; natural abundance of 17O. 19 The time course of 17O concentration in each ROI was evaluated, and a 17O concentration maps was created to visualize the 17O concentration change on the joint surface. The time course of 17O concentration in each ROI was also compared with the macroscopic and histological grades of cartilage damage described in the following sections.
Macroscopic Evaluation
After MRI, the rabbit knee joint was expanded, and cartilage damage in the femoral condyle was macroscopically and histologically evaluated according to the MRI slice. Osteoarthritis Research Society International (OARSI) scoring was used for macroscopic evaluation by an independent blinded observer, and score 2 or more with OARSI scoring, which stores Indian ink, was designated as macroscopic cartilage lesion. 20
Histological Evaluation
Operated limbs were fixed in 10% neutral-buffered formalin for 24 to 48 hours and decalcified in 24% formic acid and 5% formalin for 20 days. Tissue sections were prepared for 3 slices centered on the most distal part of the femoral condyle, sliced with a coronal section tilted 45° forward with respect to the bone axis to match the MRI slice. Safranin-O staining was performed, and the OARSI grade was used for histological evaluation by an independent blinded observer. 21
Statistical Analysis
Statistical analysis was performed using GraphPad Prism software (MDF Co., Ltd., Tokyo). Using the average of the signal intensity obtained from the first 3 MRI imaging as the baseline, the time course of 17O concentration in each group was evaluated using a 1-way analysis of variance. Statistical significance was set at P value <0.05.
Data Collection
The setting of the ROI was performed by 1 orthopedist, and the 17O concentration was calculated by 1 radiologist.
Research Approval
All experiments were performed according to a protocol approved by the Institutional Animal Care and Use Committee of the Graduate School of Medicine, Hokkaido University (EC approval number: 19-0140).
Results
The result of optimization is shown first. Regarding the normal knee, the ROI was set at the joint cavities shown as ROI 1 and 2 in
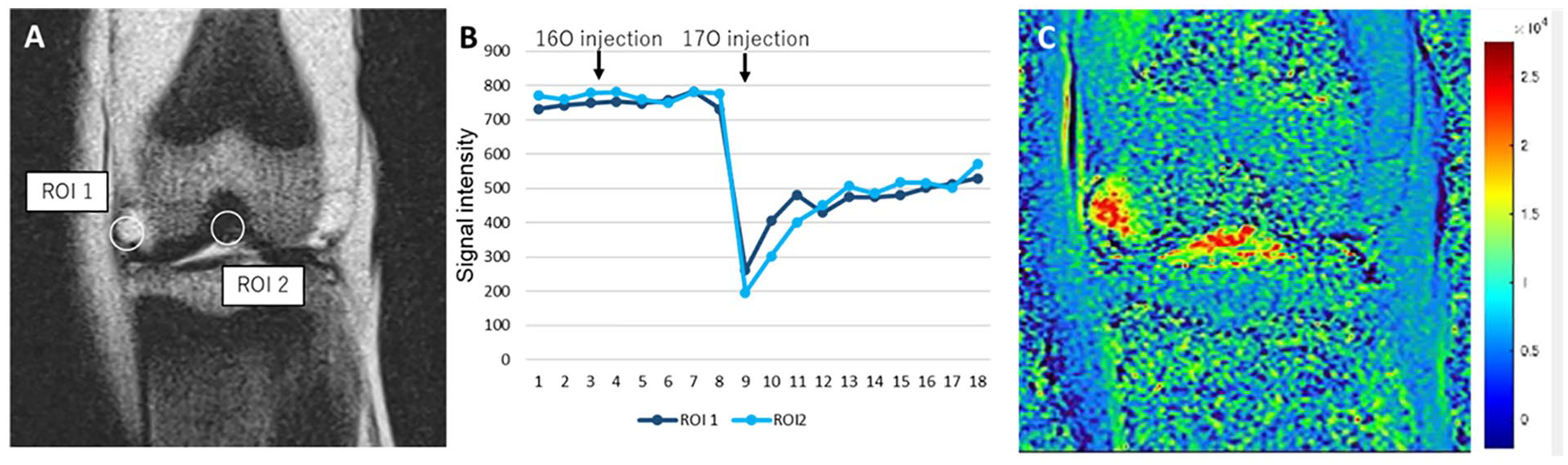
(
Macroscopic evaluation revealed 24 cartilage lesions in 10 knees. In the histological evaluation, OARSI grade 1 was 23 points, grade 2 was 3 points, and grade 3 was 6 points. ROIs were set at 27 healthy areas as controls, and the 17O concentration at each site was calculated from the MRI signal intensity.
Maps of the relative signal of 17O compared with the baseline are shown. The signal change in the dynamic scans could be visually confirmed on the 17O concentration maps of the MRI. By administering 17O local injection to the knee joint cavity at 10 minutes 57 seconds after MRI scanning was started, increased 17O concentration was observed in the knee joint cavity at 14 minutes 36 seconds. An increased 17O concentration was shown with the site that matched the macrographic cartilage lesion (
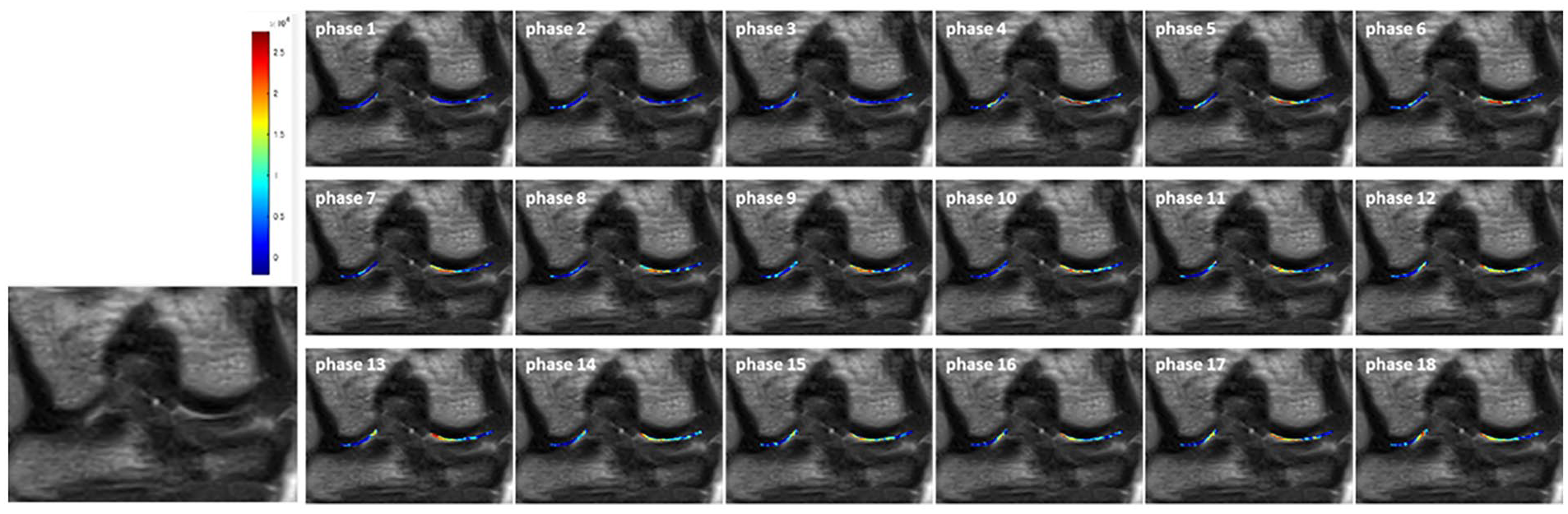
Maps of the relative signal 17O compared to baseline are shown. By administering 17O local injection to a knee joint cavity between phases 3 and 4, increased 17O concentration was shown in the knee joint cavity after phase 4. Increased 17O concentration was shown with the site that matches macrographic cartilage lesion (arrowhead).
Macrographic appearance, Safranin-O fast green staining, and 17O concentration maps of MRI of representative cases at 4 weeks (
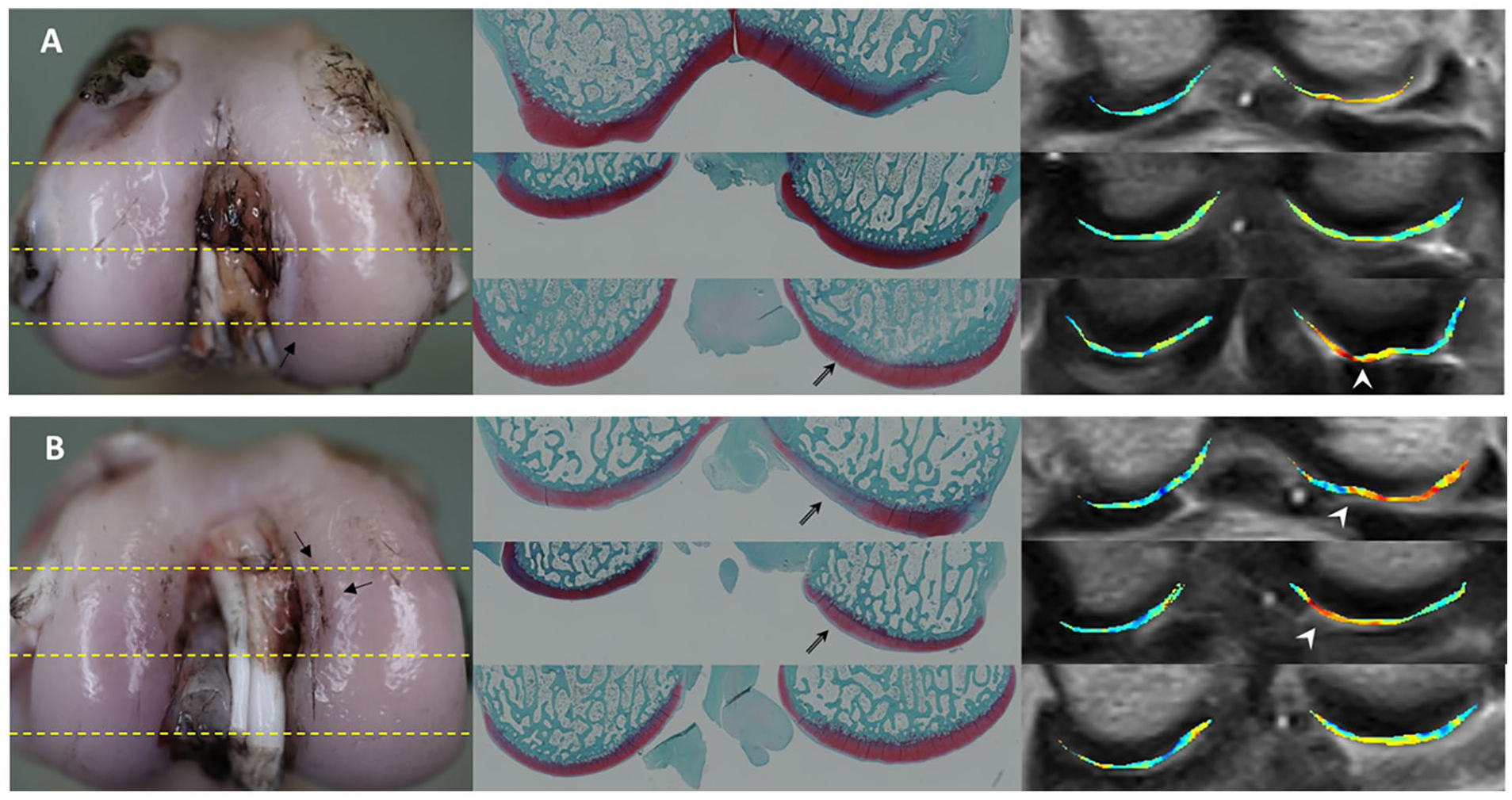
Macrographic appearance, Safranin-O fast green staining, and color-maps of magnetic resonance imaging (MRI) of 4 weeks (
The concentration-time curve between the macroscopic cartilage lesion and control site grouped by OARSI scoring is shown in
Figure 4A
. Increases in 17O concentration of cartilage lesions were apparent at 43 minutes 48 seconds after MRI scanning was started (9 phases after the injection of 17O-labeled water) compared with the control area. The concentration-time curve grouped by OARSI grading also showed that increases in density were apparent in the OARSI grade 3 group at 36 minutes 30 seconds after MRI scanning was started (7 phases after the injection of 17O-labeled water) (
(
Discussion
In this study, we established a 17O-labeled water administration procedure and MRI examination method for rabbit knees to clarify the intra-articular dynamics of water molecules in healthy and injured cartilage tissue for the first time. After intra-articular administration of 17O-labeled water, it rapidly diffused into the joint space, but there was no increase in 17O-labeled water concentration in normal cartilage. In contrast, in the OA model rabbit, the concentration of O 17 -labeled water increased significantly at the cartilage damage site after intra-articular administration. Our results revealed that the rapid influx of joint fluid containing 17O-labeled water occurred probably due to the failure of hydrostatic pressure maintenance after cartilage tissue damage.
Although advances in modality have increased the sensitivity and specificity of MRI diagnosis of cartilage damage, sensitivity to early cartilage damage is still developing.22,23 T2-weighted imaging can be used to assess the structure of the collagen fibril network. However, T2 changes in collagen fibril arrangement are affected by the magic angle effect, which may be misunderstood as degenerating normal articular cartilage. 7 T1ρMRI is specific for quantitative measurement of changes in proteoglycan content; however, the T1ρ-weighted spin-lock pulse sequence is not currently prevalent in clinical scanners. 8 Recently, MRI and computed tomography (CT) using contrast media have been used to detect early cartilage injury24,25; however, there are concerns about allergic reactions to contrast media and the risk of radiation exposure. Our results showed that 17O-labeled water could safely assess early histological cartilage injury without allergic reactions or radiation exposure.
In comparison with the macroscopic assessment, a significant increase in 17O concentration was observed, consistent with the visible cartilage injury site. Our results suggest that MRI detected macroscopic cartilage damage as 17O-labeled water flowed into the substrate from the physically damaged cartilage site. In addition, significant differences were observed 30 to 60 minutes after administration of 17O-labeled water, which is a clinically manageable timing in actual MRI. Because MRI with 17O-labeled water can detect physical damage to the cartilage surface and quantitatively evaluate the signal intensity of the ROI, it will be useful as a noninvasive and objective modality compared with semi-quantitative macroscopic evaluation.
Regarding the comparison with histological evaluation, MRI with 17O-labeled water presented a significant increase in signal intensity for grade 3 cartilage damage with an OARSI score compared with normal cartilage. This result suggested that the hydrostatic pressure maintenance mechanism of the cartilage matrix may be disrupted by damage to more than histological grade 3. Setton et al. 26 reported that damage of only 200 µm on the cartilage surface disrupted the swelling pressure mechanism. The current results of 17O-labeled water flowing into the cartilage matrix due to slight damage to the cartilage surface are consistent with this previous report. In addition, our results suggested that the hydrostatic pressure of cartilage substrate was maintained at histological grade 0 to 2, suggesting that mild histological damage, no more than histological grade 2, may have little pathological significance in OA progression. The difference between grade 1 and 2 is distinguished by a crack in the intermediate layer. For early OA, irreversible changes are considered to occur after a certain damage. Our results indicate that the hydrostatic pressure of cartilage is maintained up to the crack in the middle layer (grade 2) and may be related to the reversibility of cartilage degeneration. Wang et al. 27 reported that there was no change in the peak swelling strains in the trypsin-loaded chemical cartilage degeneration model. Our results are consistent with their report that a decrease in GAGs in the cartilage matrix is less important than superficial damage in maintaining hydrostatic pressure in the cartilage matrix. In this study, 17O-labeled water was used to directly evaluate the metabolic dynamics of water, which is fundamentally different from the conventional modality of looking at matrices such as charge and GAG. The results obtained using 17O-labeled water suggest that this modality not only helps in the early diagnosis of OA, but also provides valuable information for considering the mechanisms of OA pathogenesis.
The current study had several limitations. The first limitation is that the evaluation is based on amputated limbs and may differ from the metabolic dynamics of the living body. However, this effect should be minimal because MRI was performed within an hour of sacrificing the animal and alterations of physiological condition, charge in cartilage matrix, and chondrocyte viability were minimized in such a short time. Although we believe that in vivo evaluation is possible because living humans will be able to maintain limb position during MRI scans, experiments on living animals are required before experimenting on humans. The second limitation was that evaluations in this study were made for early-stage OA, but no evaluation was made for advanced OA. Severe cartilage damage may result in different signal intensity changes or can be undetectable because of the loss of cartilage substrates. As advanced OA can be detected by conventional examinations such as radiographic images, it is unnecessary to perform MRI with 17O-labeled water. The third limitation is that the MRI angle is tilted at 45° with respect to the bone axis, which is unusual compared with normal conditions for humans. However, considering the most loaded part of the knees in rabbits, we thought that this condition could clearly detect alterations in the cartilage matrix in this OA model. When evaluating the human knee joint, conventional coronal or sagittal image evaluations may be suitable.
Despite these limitations, this is the first study to dynamically visualize the alterations in the hydrostatic pressure of cartilage tissue in OA by administering 17O-labeled water intra-articularly and indicates the possibility of a novel modality to detect early OA.
In conclusion, we established a method using 17O-labeled water as a new contrast medium and proved its usefulness. 17O-labeled water can visualize earlier articular cartilage damage, which is difficult to detect using conventional methods. This is the first modality to evaluate the metabolic dynamics of water on cartilage without allergic reactions or radiation exposure.
Footnotes
Authors’ Note
HOKKAIDO UNIVERESITY SCHOOL OF MEDICINE DEPARTMENT OF ORTHOPAEDIC SURGERY.
Author Contributions
All authors contributed to the study design (YH, TO, KK, HK, NI), acquisition of data (YH, HK, HH), or analysis and interpretation of data (YH, TO, KH, HK, MM, JY, and NI). YH and TO drafted the article, and all authors revised it critically for intellectual content. All the authors approved the final version of the manuscript.
Acknowledgments and Funding
We would like to thank Editage (![]() ) for English language editing. Parts of this work were supported by Japan Agency for Medical Research and Development (AMED) and ZENKYOREN (National Mutual Insurance Federation of Agricultural Cooperatives). The sponsors had no role in the study design, collection, analysis, and interpretation of data; in the writing of the manuscript; and in the decision to submit the manuscript for publication.
) for English language editing. Parts of this work were supported by Japan Agency for Medical Research and Development (AMED) and ZENKYOREN (National Mutual Insurance Federation of Agricultural Cooperatives). The sponsors had no role in the study design, collection, analysis, and interpretation of data; in the writing of the manuscript; and in the decision to submit the manuscript for publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The institutional review board of Hokkaido university approved the design and publication of this study.
