Abstract
Objective
Kartogenin (KGN) has proven as a both chondrogenic and chondroprotective drug for osteoarthritis (OA) therapy. However, being a small hydrophobic molecule, KGN suffers from rapid joint clearance and inability to penetrate cartilage to reach chondrocytes following intra-articular administration. As such multiple high doses are needed that can lead to off-target effects including stimulation and tissue outgrowth. Here we design charge-based cartilage targeting formulation of KGN by using a multi-arm cationic nano-construct of Avidin (mAv) that can rapidly penetrate into cartilage in high concentrations owing to weak-reversible electrostatic binding interactions with negatively charged aggrecan-glycosaminoglycans (GAGs) and form an extended-release drug depot such that its therapeutic benefit can be reaped in just a single dose.
Design
We synthesized 2 novel formulations, one with a releasable ester linker (mAv-OH-KGN, release half-life ~58 h) that enables sustained KGN release over 2 weeks and another with a non-releasable amide linker (mAv-NH-KGN) that relies on mAv’s ability to be uptaken and endocytosed by chondrocytes for drug delivery. Their effectiveness in suppressing cytokine-induced catabolism was evaluated in vitro using cartilage explant culture model.
Results
A single 100 μM dose of cartilage homing mAv-KGN was significantly more effective in suppressing cytokine-induced GAG loss, cell death, inflammatory response and in rescuing cell metabolism than a single dose of free KGN; multiple doses of free KGN were needed to match this therapeutic response.
Conclusion
mAv mediated delivery of KGN is promising and can facilitate clinical translation of KGN for OA treatment with only a single dose.
Introduction
Osteoarthritis (OA) is characterized by chronic inflammation and cartilage degradation and is one of the most common joint disorders affecting millions of people worldwide.1,2 There are no approved disease-modifying treatments that can delay or prevent OA progression.3,4 Current treatment options like non-steroidal anti-inflammatory drugs (NSAIDs), intra-articular (IA) injection of hyaluronic acid (HA)-based products or triamcinolone encapsulating poly(lactic-co-glycolic acid) (PLGA) microparticles can only help manage OA pain.4-7 Thus, there is an urgent need to develop effective therapeutic interventions that can not only inhibit OA-associated catabolic activity but also upregulate anabolic processes to enable repair at cell and tissue level. 8 It is also important that these therapies are delivered to their cell and matrix target sites inside cartilage in a sustained manner for an effective treatment.9-12 Following IA administration, most macromolecules clear out rapidly from the joint space via lymphatics or blood vessels resulting in short intra-joint residence times.5,10,13,14 In addition to this, their transport and penetration into cartilage is hindered due to its dense negatively charged matrix comprising high density of aggrecan-glycosaminoglycans (GAGs) and collagen type II meshwork giving it a nanometer range pore size.5,10,12,14
Kartogenin (KGN) is a small molecule that promotes the selective differentiation of mesenchymal stem cells (MSCs) into chondrocytes through interactions with actin-binding protein filamin A (FLNA), core binding factor β (CBFβ), and transcription factor RUNX1. 15 KGN functions by breaking the balance between CBFβ and FLNA in the cytoplasm and translocating CBFβ into the nucleus, where CBFβ interacts with RUNX1 to form the CBFβ-RUNX1 complex. 15 This complex activates the transcription of chondrogenesis-related proteins and enhances synthesis of cartilage matrix proteins like collagen type II and aggrecan. 15 In addition, KGN exhibits chondroprotective effects by stabilizing the interaction of CD44 receptors to hyaluronan while blocking the inflammatory cytokine-induced expression of aggrecanase. 16 However, being a small hydrophobic molecule (MW ~ 317.3 Da), 17 KGN suffers from rapid joint clearance following its IA administration.18,19 As such, multiple high doses are needed that can potentially lead to off-target effects such as stimulation and outgrowth of normal tissues.19,20 For example, a recent rat study evaluating multiple doses of KGN for wound healing in the tendon-bone junctions (TBJs) reported that KGN spread out from the intended location and resulted in excessive growth of patellar and surrounding cartilage like tissues, which completely hid the patellar tendon. 20
KGN has gained significant attention lately as a disease-modifying OA drug.15,16,19,21-25 For example, 125 μM KGN was shown to be effective in suppressing anterior cruciate ligament transection (ACLT)-induced subchondral bone and cartilage degradation but weekly IA injections were required until 12 weeks. 25 Similarly, weekly IA administered KGN combined with microfracture was found to increase production of hyaline like cartilage tissue and improve the cartilage repair assessment (ICRS) scores compared with microfracture alone in a rabbit cartilage defect model. 26 Recently, a more potent and stable analogue of KGN, KA34 was shown to be effective in cartilage repair and pain modulation in injury models of rats and dogs with weekly or biweekly IA injections. 22 These preclinical studies have led to a Phase I clinical trial (NCT03133676) to evaluate the safety and tolerability of single and multiple IA doses of KA34 in OA patients. These are promising developments further highlighting the importance of developing sustained release formulations to reduce the need for multiple injections, which is also associated with local injection site pain, inflammation and discomfort. Particle and gel-based systems are under consideration for enhancing intra-joint residence time of small molecule drugs like KGN but none of these delivery methods enable cartilage targeting or penetration.18,24,27-29
Our group has shown that electrostatic interactions are effective in targeting cartilage, a tissue with a very high negative fixed charge density.10,30-33 We showed previously that Avidin, a glycoprotein, due to its optimal size (< 10 nm which is the estimated cartilage matrix pore size) 34 and net charge (between +6 and +20), is an effective carrier for intra-cartilage drug depot delivery.34-37 We synthesized a multi-arm Avidin nano-construct (mAv) by conjugating four 8-arm branched polyethylene glycols (PEGs) to its biotin binding sites38,39 and showed that it can penetrate through the full thickness of cartilage in high concentrations (112x higher than the equilibration bath) resulting in a long intra-cartilage residence time (>7 days) in both healthy and arthritic cartilage owing to weak-reversible binding with negatively charged aggrecans.13,31,39
Here, our goal is to design cartilage targeting formulations of KGN by using mAv that can penetrate rapidly into the tissue in high concentrations and form an extended-release drug depot such that its therapeutic benefit can be reaped in just 1 dose. We synthesize 2 novel formulations, one with a releasable ester linker that enables sustained KGN release over 2 weeks and another with a non-releasable amide linker that relies on mAv’s ability to be uptaken and endocytosed by chondrocytes for drug delivery (
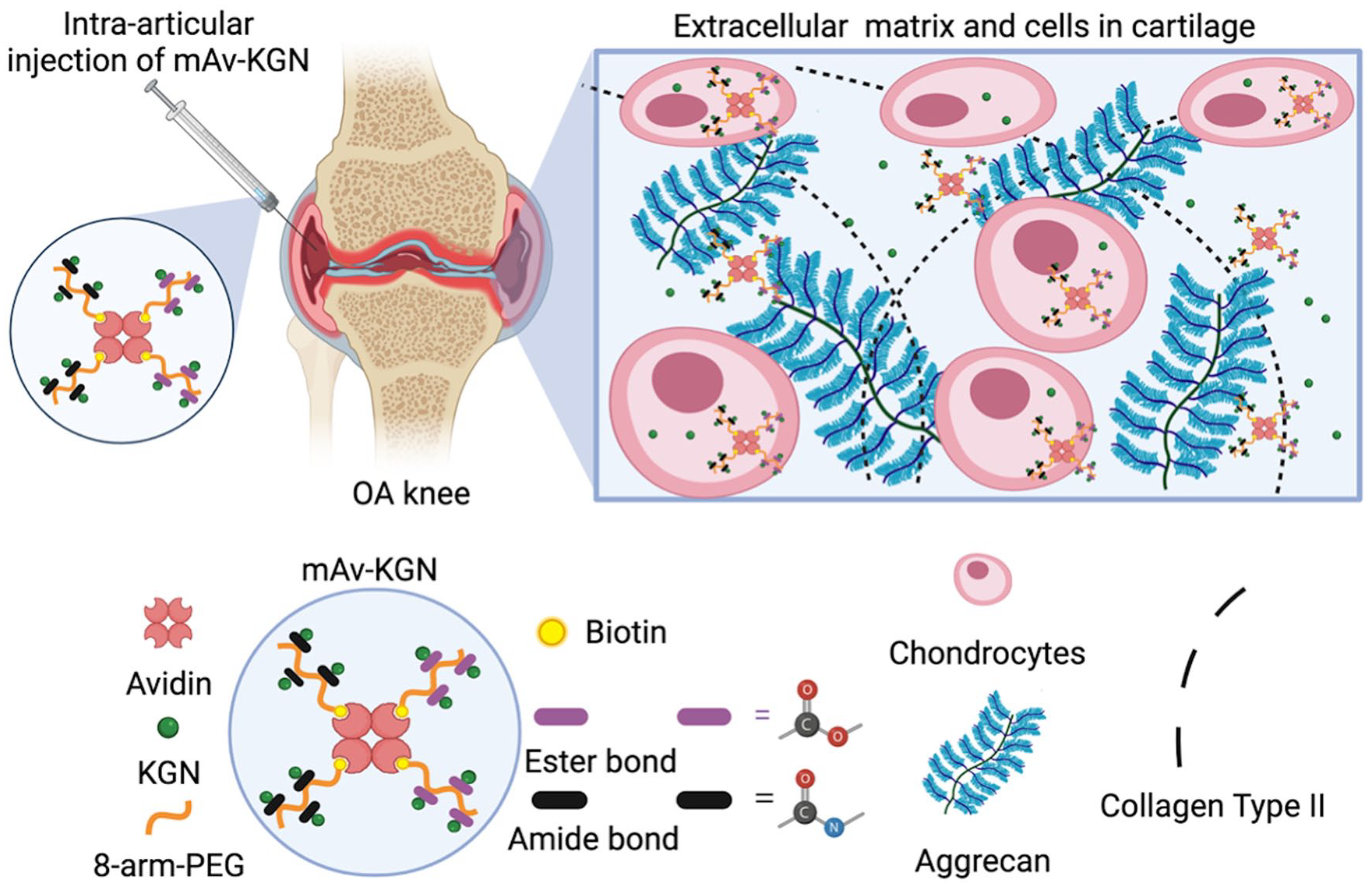
Schematic of charge-based intra-cartilage delivery of KGN conjugated to multi-arm Avidin nanocarrier (mAv-KGN) by using either a releasable ester linker or a non-releasable amide linker. Following its intra-articular administration, mAv-KGN can rapidly penetrate through the full thickness of negatively charged cartilage in high concentrations due to electrostatic interactions thereby creating an intra-cartilage drug depot. The optimal net positive charge of mAv enables its rapid and high intra-cartilage uptake and long-term retention via weak-reversible binding with negatively charged aggrecan-GAGs. Therapeutic doses of KGN are either released via hydrolysis of ester linker or in case of amide linker, mAv-KGN is uptaken by chondrocytes. KGN = Kartogenin; mAv = multi-arm Avidin nano-construct; GAGs = glycosaminoglycans; OA = osteoarthritis; PEG = polyethylene glycol.
Methods
Materials
The 10 kDa 8-arm PEG amine hydrochloride salt was purchased from Advanced Biochemicals (Lawrenceville, GA). N-Hydroxysuccinimido (NHS)-biotin, NHS, 1-ethyl-3-(3 dimethylaminopropyl) carbodiimide hydrochloride (EDC), N-hydroxysulfosuccinimide (NHSS), Avidin (Av), 3.5 kDa molecular weight cut-off (MWCO) and 7 kDa MWCO SnakeSkin dialysis tubing was purchased from Thermo Fisher Scientific (Waltham, MA). Kartogenin (KGN) was purchased from Tocris bioscience; Proteinase-K was purchased from Roche Diagnostics (Risch-Rotkreuz, Switzerland). Dulbecco’s Modification of Eagle’s Medium (DMEM) was from Cellgro (Manassas, VA). HEPES, non-essential amino acids (NEAA), penicillin-streptomycin Antibiotic-Antimycotic (PSA), and trypsin-EDTA phenol red were purchased from Gibco (Carlsbad, CA). Ascorbic acid and L-proline were from Fisher Bioreagents (Pittsburgh, PA). Propidium iodide (PI) was obtained from Thermofisher Acros Organics (Geel, Belgium). Human recombinant interleukin (IL)-1α and Oncostatin-M (OSM) were from PeproTech (Rocky Hill, NJ). Antibodies for type II collagen immunohistochemistry was acquired from the Developmental Studies Hybridoma Bank (University of Iowa), while the Vectastain Elite ABC kit was from Vector Laboratories (Burlingame, CA). 3-hydroxpropionic acid, dimethyl sulfoxide-d6 (DMSO-d6) containing 0.03% (v/v) tetramethylsilane, fluorescein diacetate (FDA), fluorescein isothiocyanate isomer I (FITC), dimethylaminopyridine (DMAP), resazurin sodium salt, Griess reagent, histology reagents, and other salts were purchased from Sigma-Aldrich (St. Louis, MO).
Biotinylation of 8-Arm PEG
Eight-arm PEG-amine was biotinylated by reacting with NHS-biotin to get 1 biotin per PEG as previous described.38,39 Briefly, 8-arm PEG-amine (10 mg, 0.001 mmol, 1 equiv.) was reacted with NHS-biotin (1.7 mg, 0.005 mmol, 5 equiv.) for 2 hours under gentle rotation at room temperature (RT). Excess NHS-biotin was removed from the PEG-biotin conjugate solution using dialysis tube (7.0 kDa MWCO) for 24 hours against phosphate buffer saline (PBS) and following another 24 hours against water at room temperature. Eight-arm PEG-Biotin-amine (PEG-Biotin) was then lyophilized, weighed, and stored at −20°C for future use.
Synthesis of PEG-OH-KGN and PEG-NH-KGN
Briefly, 3-hydroxypropinoic acid (31 μl of 30% solution in water, 0.1 mmol, 100 equiv.) was firstly activated with EDC (14 mg, 0.067 mmol, 67 equiv.), and NHSS (16mg, 0.073 mmol, 73 equiv.) in PBS, then reacted with 8-arm PEG-Biotin (10 mg, 0.001 mmol, 1.0 equiv.) to form PEG-OH (as shown in
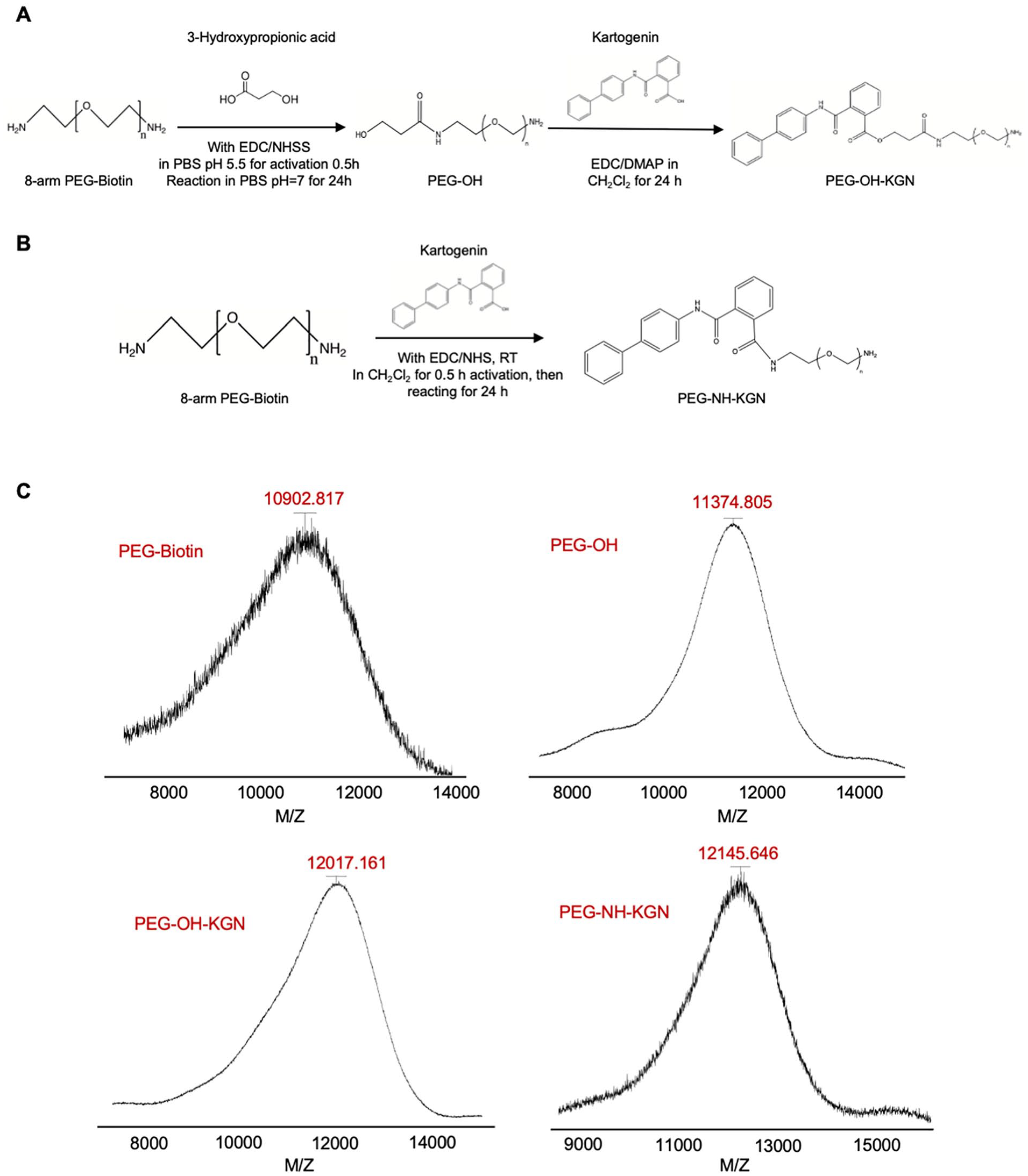
Schematic of KGN conjugation with 8-arm PEG-amine by forming (
To form an amide linker between the 8-arm PEG-Biotin-amine and KGN, carboxyl group in KGN (25mg, 0.08 mmol, 80 equiv.) was firstly activated with EDC (28mg, 0.134 mmol,134 equiv.) and NHS (14mg, 0.121 mmol, 121 equiv.) in anhydrous CH2Cl2 for 0.5 hours (as shown
Characterization of PEG-OH, PEG-OH-KGN, and PEG-NH-KGN
Matrix-assisted laser-desorption ionization time-of-flight mass spectrometry (MALDI-TOF MS) was used to confirm the molecular weight of PEG-Biotin, PEG-OH, PEG-OH-KGN, and PEG-NH-KGN. 2 µL of PEG-OH, PEG-OH-KGN, or PEG-NH-KGN (1 mg/ml) was mixed with equal amount of sinapic acid matrix (10 mg/ml), and the change in molecular weight between unmodified and modified molecules and the drug loading content were confirmed using MALDI-TOF spectrometry (Bruker Microflex II). The data were analyzed using flexAnalysis software from Bruker.
Conjugation with KGN was further confirmed with HPLC. 1 mg/ml of each purified compound was injected into HPLC (Agilent Technologies 1260 infinity II) equipped with a Variable Wavelength Detector using a Poroshell 120 EC-C18 column (4.6 x 150 mm). A gradient of solvent A (0.1% trifluoroacetic acid [TFA] in water) and solvent B (0.1% TFA in acetonitrile) was used. The concentration of solvent B was increased linearly from 5% to 65% over 15 minutes. Column temperature of 30 °C and a flow rate of 1.0 ml/minute were used. KGN was detected at UV = 280 nm.
The chemical shift values for KGN, PEG-OH, PEG-OH-KGN and PEG-NH-KGN were analyzed using 500 MHz 1H NMR (Varian Inova, Agilent Technologies). One to two milligrams of each of the 4 purified compounds were dissolved in 700 μl DMSO-d6 at RT. The obtained NMR data were analyzed using MestRe Nova software. The 4 compounds KGN, PEG, PEG-OH-KGN and PEG-NH-KGN were characterized by Fourier transform infrared spectroscopy (FTIR) (Billerica MA, Bruker). 0.5 mg of each sample was dissolved in 1 ml dichloromethane (DCM) and read at 4000-650 cm−1 range using 30 scans. The spectrum was analyzed used Octopus software (Billerica MA. Bruker).
In Vitro Drug Release Study
The lyophilized products, PEG-OH-KGN (10 mg) and PEG-NH-KGN (10 mg), were dissolved in 1 ml PBS in a dialysis tube (3.5 kDa cutoff) and then put in a 50 ml tube containing 25 ml release media (pH = 7.4 PBS). The setup was put on slow shaking inside the incubator at 37 °C. At different time points, 300 μl of release media was collected and analyzed using HPLC to measure the amount of KGN released as described in section above. An equal amount of fresh media was replaced.
Synthesis of mAv-OH-KGN and mAv-NH-KGN
Purified biotin containing PEG-OH-KGN and PEG-NH-KGN were reacted with Avidin in DI water in 5:1 molar ratio for 30 minutes under gentle shaking at room temperature to form releasable mAv-OH-KGN and non-releasable mAv-NH-KGN using Avidin-biotin reaction. The conjugation was confirmed using 4’-hydroxyazobenzene-2-carboxylic acid (HABA) dye assay and gel electrophoresis as described previously.38,39 Both formulations were synthesized right before their use in biological experiments described below.
Bioactivity of Single-Dose mAv-KGN in Cytokine Challenged Cartilage Explant Culture OA Model
Cartilage explants harvested from 1- to 2–week-old calf knee joints (procured from Research 87, Boylston, MA) were equilibrated separately in culture media for 2 days prior to treatment as described previously.40-42 Cartilage explants were then treated with or without a combination of cytokines, Oncostatin M (OSM) (3 ng/ml) + IL-1α (2 ng/ml), for 8 days in combination with (1) a single dose of 100 μM free KGN, (2) a continuous dose of 100 μM free KGN, (3) a single dose of mAv-OH-KGN (100 μM KGN), or (4) a single dose of mAv-NH-KGN (100 μM KGN). Media was changed every 2 days and the cytokine combination (OSM + IL-1α) was replenished at each medium change. Single-dose treated explants were subjected to the drug or drug carrier for only the first 2 days and in the following media changes, the media did not contain any therapeutic, thereby simulating a single intra-articular injection in vivo.39,40 KGN was replenished throughout the culture duration for the continuous dosing condition. Combination of OSM + IL-1α was chosen based on prior published literature demonstrating that this combination cytokine can trigger a more aggressive GAG loss by degrading both aggrecan and hyaluronan43-45 and break the interactions with CD44 12 that can be rescued by KGN. 16 Previous work has shown that treatment with 10 to 100 μM KGN can be effective in suppressing cytokine-induced catabolism in cartilage explant culture model.15,16,21,23 We, therefore, chose single dose of mAv-KGN with effective KGN concentration of 100 μM for intra-cartilage drug depot delivery and compared its efficacy with a single or continuous dosing of equivalent concentration of free (unmodified) KGN (the dosing is described in more detail in the “Results” section).
After 8 days of culture, cartilage explants were weighed and digested in proteinase K as previously described.39,40,42 The cumulative GAGs released to the media and residual GAGs in the digested explants were measured using the 1,9-dimethylmethylene blue (DMMB) assay. 46 Nitrite content was measured using the Griess assay as an indicator of nitric oxide (NO) release from tissues.40,47 Sodium nitrite was used as the standard.
Cartilage explant cell viability was analyzed by staining cartilage explants with fluorescent cocktails of FDA (4.0 mg/ml) and PI (10.0 mg/ml) for live (green) and dead (red) cells, respectively. Slices were washed with PBS and imaged at 4x magnification (Nikon Eclipse Ts2R). The live and dead images were overlaid using ImageJ. At the end of the culture, tissue explants were incubated with alamarBlue assay for 3 hours in dark at 37°C and 5% CO2. Cell metabolic activity was then estimated by measuring fluorescence at 530 nm excitation and 590 nm emission wavelengths.
Statistical Analysis
Data are presented as Mean ± Standard Deviation. For all studies, n = 6-8 explants per condition and experiments were repeated using explants from at least 3 animals. A general linear mixed effects model was used with animal as a random variable. For comparisons between different treatment conditions, Tukey’s Honestly Significant Difference test was used. P < 0.05 was considered statistically significant.
Results
Synthesis and Characterization of PEG-OH-KGN and PEG-NH-KGN
Based on change in molecular weight measured using MALDI-TOF, we estimated 6.55 OH groups per PEG-OH, 2.15 KGN per PEG-OH-KGN, and 4.15 KGN per PEG-NH-KGN (
The 1H NMR spectra of KGN showed prominent resonance peaks “1-13” at δ = 7.0-8.0 ppm, which correspond to the protons in the aromatic ring of KGN, consistent with previous reports (
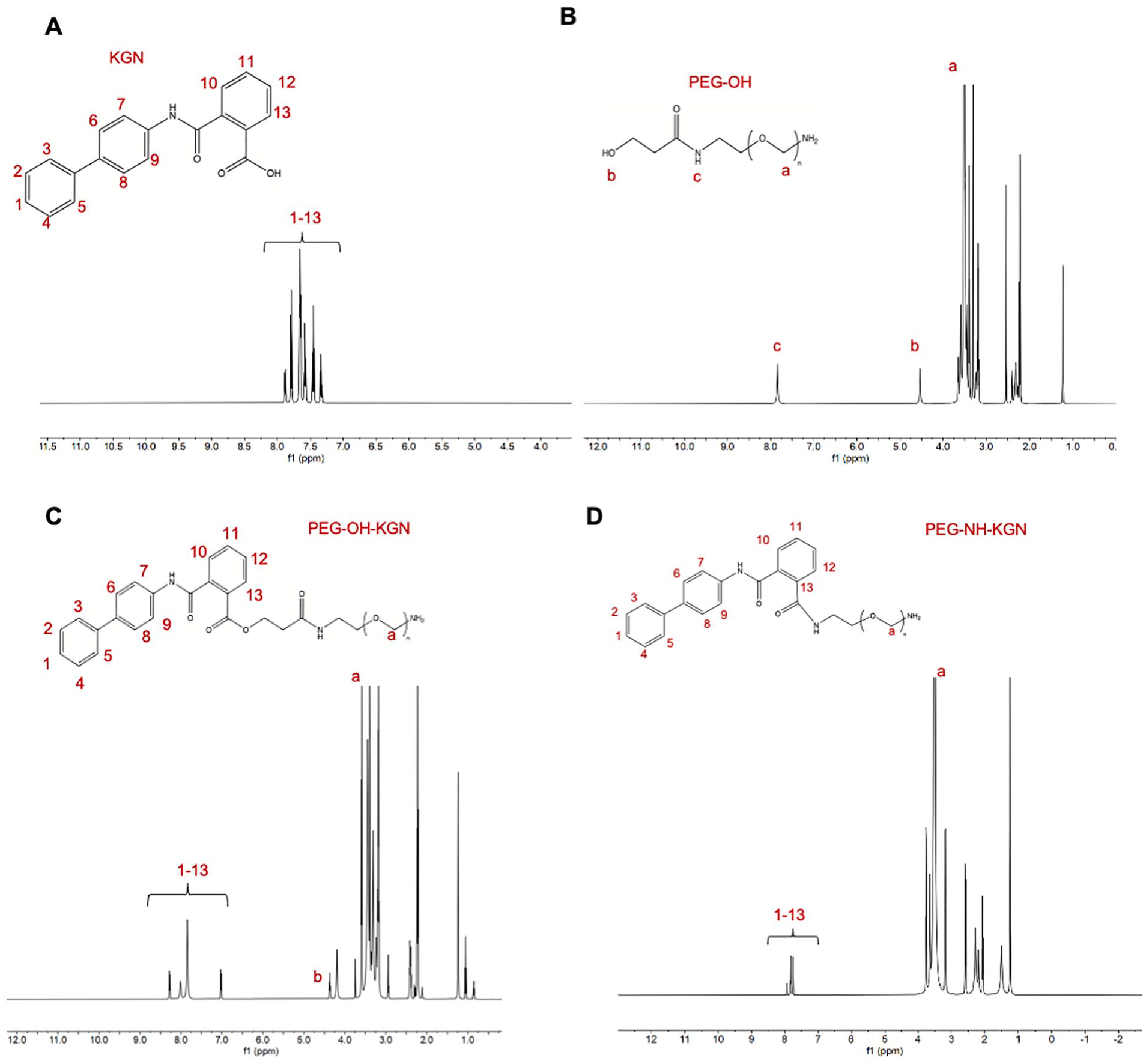
1H NMR spectra of (
The FTIR spectrum of KGN shows C=C bend in the aromatic ring of KGN (peaks at 1537 cm−1 and 1596 cm−1) (
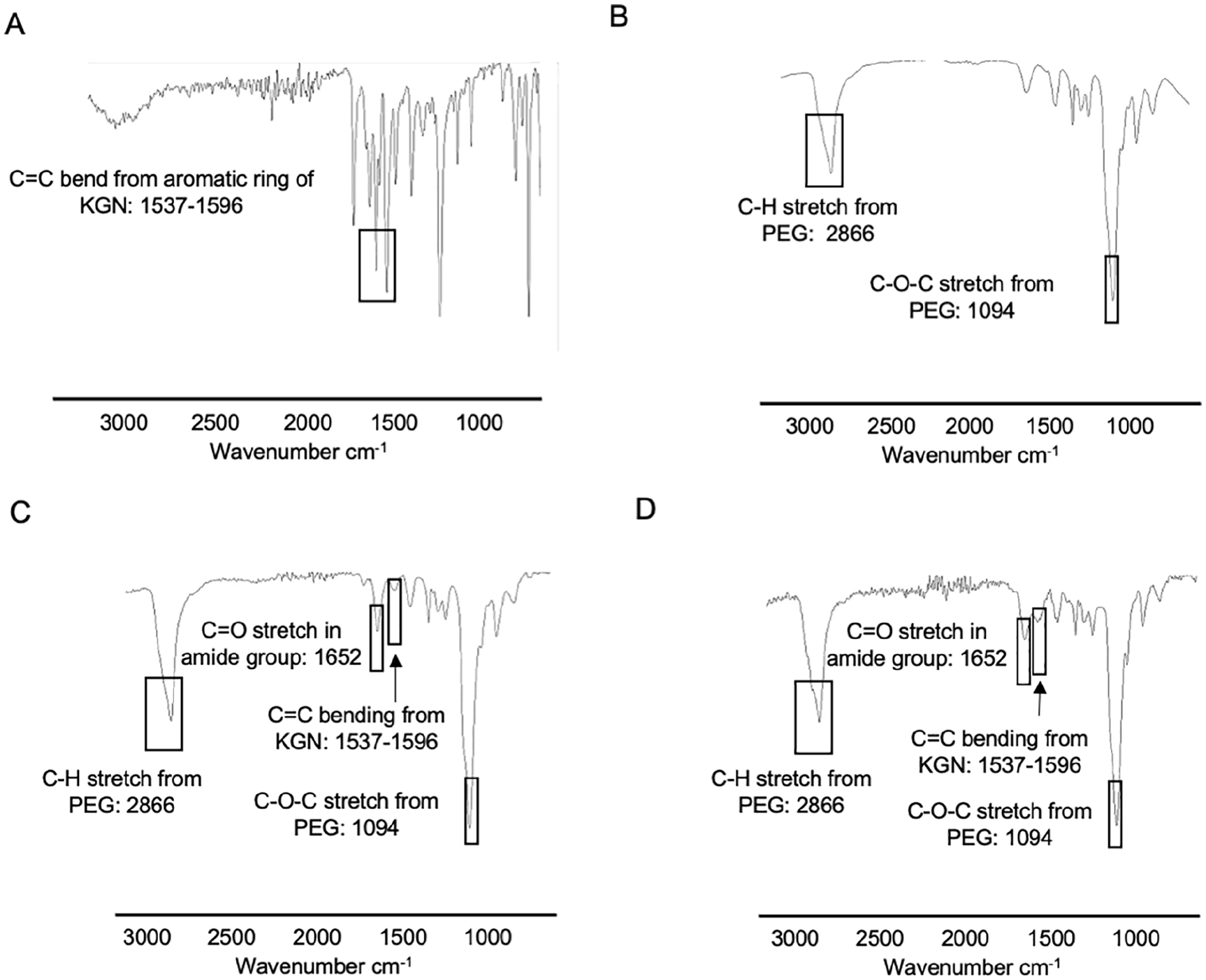
FTIR spectra of (
In Vitro Drug Release and Cytotoxicity
Figure 5A
shows that KGN was released in a sustained manner from the releasable formulation, PEG-OH-KGN (black curve), containing the hydrolysable ester linker resulting in a half-life of about 58 hours. The non-releasable formulation containing amide linker, PEG-NH-KGN (red curve) remained stable releasing only about 5% of total KGN loaded over a test period of 300 hours (12.5 days). Treatment with either conjugate at 100 μM KGN dose did not show any change in cell viability compared with untreated control (
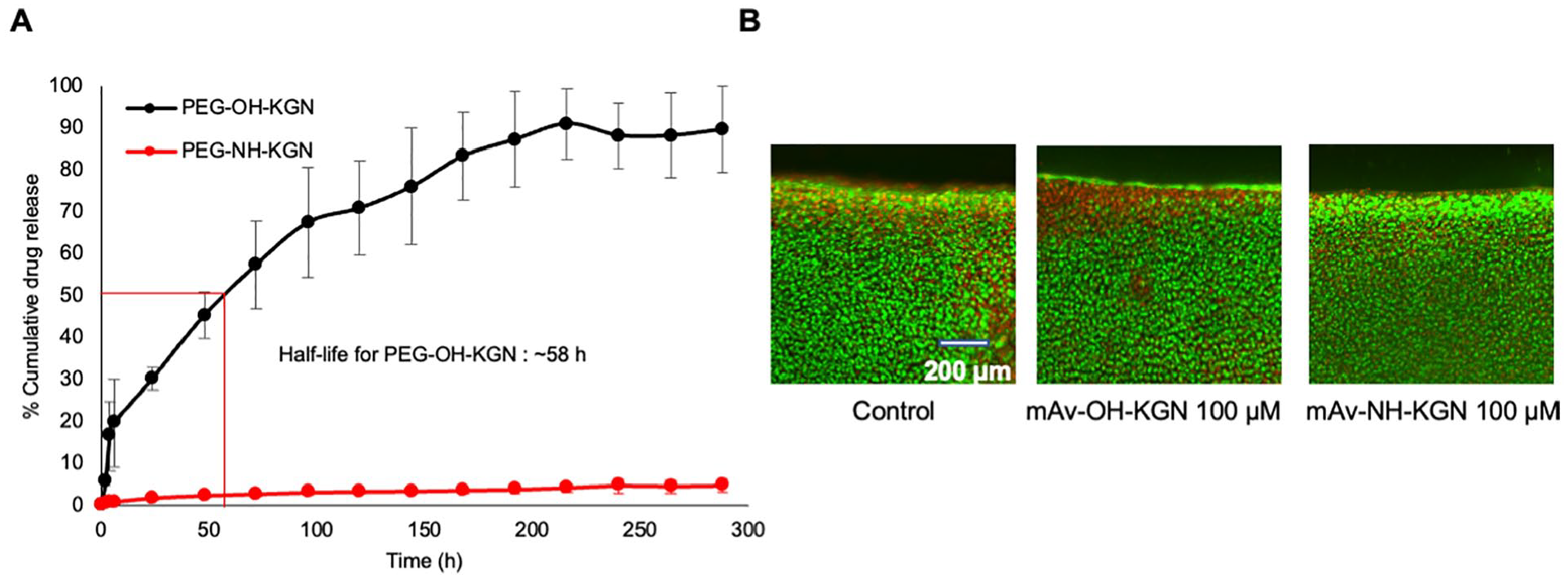
(
Bioactivity of Single-Dose mAv-OH-KGN or mAv-NH-KGN in ΟSM + IL-1α Challenged Cartilage Explant Culture OA Model
We first compared the effectiveness of single versus continuous doses of 100 μM free KGN in suppressing OSM + IL-1α–induced catabolic activity in cartilage. Treatment with OSM + IL-1α significantly increased GAG loss over the 8-day culture period compared with control (P < 0.0001) (red curve,
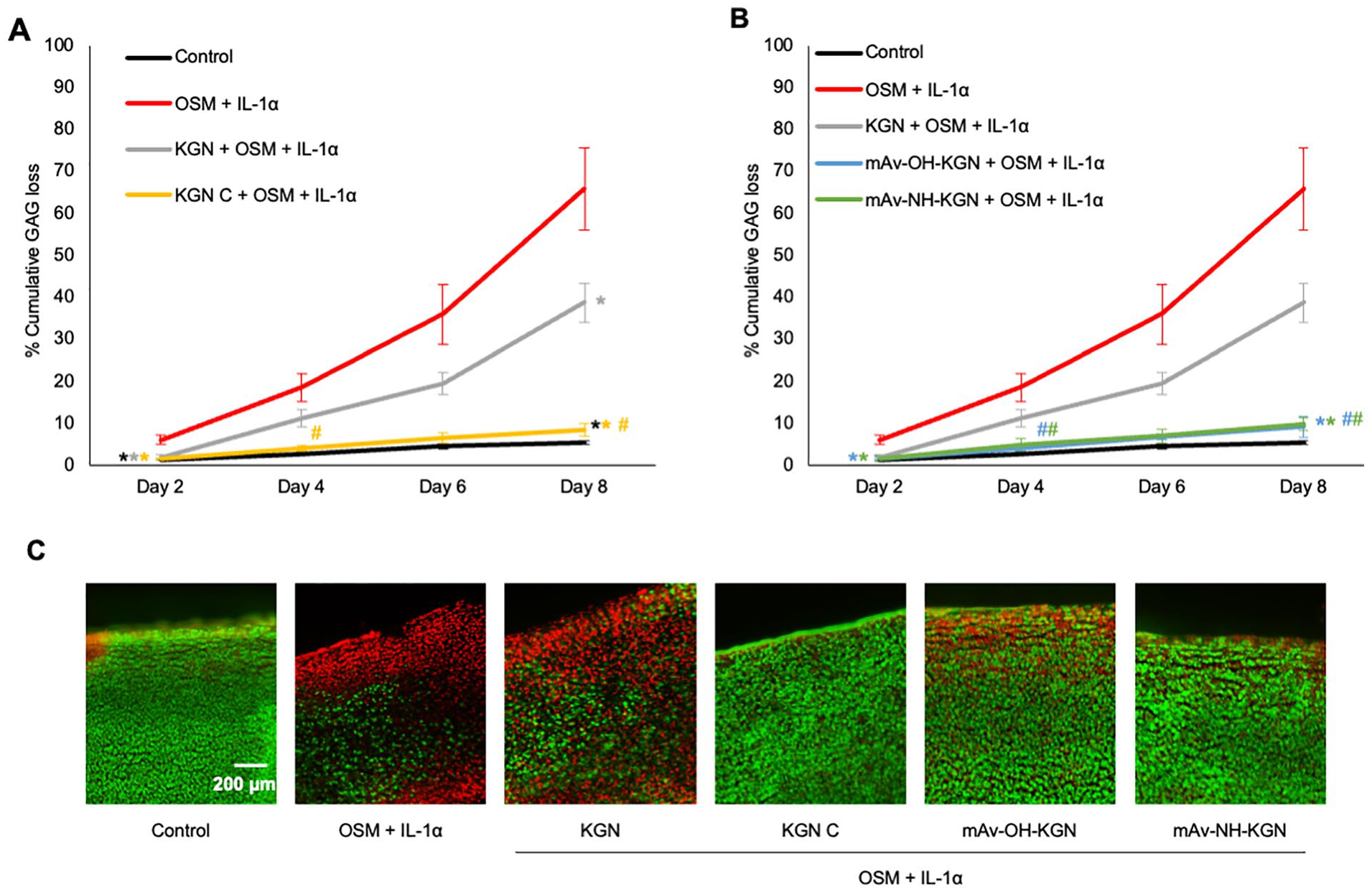
Effectiveness of single-dose mAv-KGN treatment in suppressing OSM + IL-1α–induced GAG loss and chondrocyte death. % cumulative GAG loss over 8 days: OSM + IL-1α–treated cartilage explants treated with (
To evaluate the bioactivity of single dose of mAv-OH-KGN and mAv-NH-KGN, equivalent amounts containing 100 μM KGN were tested and compared with free KGN. Since both these formulations can form intra-cartilage depots, a single dose containing 100 μM KGN was chosen as it is sufficient to provide therapeutic KGN doses over the entire culture duration. The choice of dosing is further explained here. We have shown that a sustained (continuous) dosing of 100 μM KGN throughout the culture duration is effective in suppressing cytokine-induced catabolism (
Both single dose of mAv-OH-KGN and mAv-NH-KGN suppressed OSM + IL-1α–induced GAG loss throughout the culture duration significantly greater than single dose of free KGN (P < 0.05 starting at Day 4), bringing levels down close to that of the untreated control as expected (
Nitrites are a reactive oxygen species (ROS) triggered by an upregulation in inflammatory activity such as the presence of cytokines in an OA environment.40,47 As expected, OSM + IL-1α treated explants produced 10x and 20x higher amounts of nitrite compared with control at 2 and 8 days of culture (P < 0.0001), respectively (
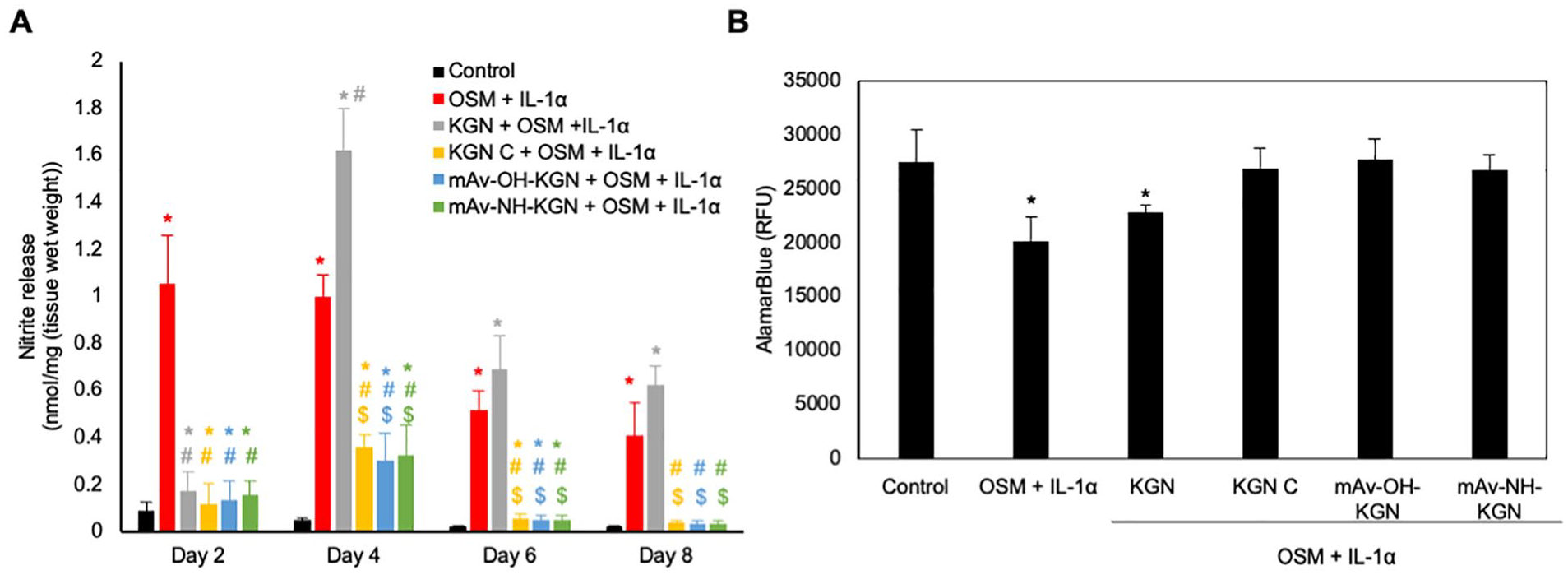
Effect of mAv-KGN on nitrite release and cell metabolism in OSM + IL-1α treated cartilage. (
Discussion
It is necessary that drugs are delivered in a sustained manner inside cartilage to reach chondrocytes in therapeutic doses over several days for effective treatment of OA. Cartilage, being a dense, avascular and one of the most negatively charged tissues in our bodies, hinders transport of IA administered macromolecules.10,30 Targeted delivery to cartilage has remained an outstanding challenge in the field, which has limited effectiveness of OA therapeutics and their clinical success. 48 Here, we demonstrate that a branched PEGylated cationic protein, Avidin-based nanocarrier (multi-arm Avidin, mAv) can penetrate into the tissue to form an intra-cartilage depot owing to electrostatic binding interactions and provide sustained release of pro-anabolic small molecule OA drug, KGN16,21 to chondrocytes over several days. This is timely work as IA administered KGN analogue is currently being evaluated in a clinical trial for OA therapy. 22
We designed 2 mAv-KGN formulations with high drug loading content. The releasable mAv-OH-KGN formulation with a hydrolysable ester linker comprised 8.5:1 molar ratio of KGN to mAv and the non-releasable mAv-NH-KGN with an amide linker had 16.6:1 KGN to mAv by mole. As a result, for a single shot intra-cartilage delivery of 100 μM KGN, less than 10 µM of Avidin was needed—a dose that has been shown to be well within safe limits; our extensive studies evaluating the dose dependent response of Avidin and mAv showed no change in GAG loss, chondrocyte viability, or biosynthesis rates of protein and GAGs with doses within 100 µM range.35,37,39 A low 10 µM concentration of Avidin was shown to be non-immunogenic when administered IA in rabbits.
37
A 30-minute intravenous infusion of 100 mg Avidin was found to be well tolerated and safe in a prospective Phase III substudy evaluating Avidin’s effectiveness in clearing a systemically injected biotinylated drug in patients with deep vein thrombosis.
49
In the preceding Phase 1 dose studies, single or repeated 25 to 100 mg doses of intravenous administered Avidin showed a low incidence of asymptomatic antibody response and no cases of hypersensitivity,
50
further strengthening the clinical prospects of using Avidin as a drug delivery carrier. Moreover, our current design of mAv incorporates high molecular weight 8-arm PEGs (
KGN has gained significant attention as a chondrogenic factor since its first discovery and use for cartilage repair in 2012. 15 Recently, the anabolic effect of KGN on cytokine challenged chondrocytes was confirmed to be mediated by SMAD-1 phosphorylation 16 —these SMAD transcriptional modulators are exhibited by the aggrecan, CD44 and hyaluronan synthase-2 genes.55,56 KGN also reduced IL-1 β stimulated release of aggrecan G1 domain fragments from human cartilage explants by inhibiting both ADMASTS5 mediated aggrecanase activity as well as by blocking the proteolytic cleavage of the hyaluronan receptor, CD44. 16 KGN ameliorated OA-induced cartilage degradation by attenuating oxidative stress by suppressing miR-146a, which is known to upregulate the protein levels of NRF2 (a key transcription regulator of antioxidant defense system inhibiting apoptosis and mitochondrial dysfunction) and down-regulate the expression of matrix degradation enzymes thereby preserving the expressions of collagen type II and aggrecans in IL-1β-treated chondrocytes. 21 OSM in combination with IL-1 can induce greater G1 and hyaluronic acid (HA) release from cartilage than either OSM or IL-1 alone. 43 We, therefore, used a combination cytokine treatment of OSM + IL-1α in our in vitro bovine cartilage OA model to validate the chondroprotective and chondrogenic effect of one dose mAv-KGN and continuous dose of free KGN, which may be induced by protecting G1 and stabilizing HA-CD44 interaction or by attenuating oxidative stress by suppressing miR-146a. Similar to our results, sustained dose of KGN has been shown to suppress OSM + tumor necrosis factor (TNF)-α–induced nitrite release from chondrocytes and GAG loss from cartilage explants. 15 Since mAv can be uptaken by other negatively charged soft tissues inside the joint including fat pad, synovium, ligaments, menisci and synovial fluid, 35 mAv-KGN is expected to target these as well and elicit a disease-modifying OA effect in the entire joint. In addition, mAv-KGN can facilitate differentiation of synovial fluid residing MSCs to chondrocytes, which are known to be overexpressed in early stages of OA.57,58
Current methods for developing sustained release formulations of KGN utilize materials like chitosan, 24 thermogel, 18 PAMAM-PEG, 17 amphiphilic polymers polyurethanes, 59 or HA PEG hydrogels. 29 These delivery systems, however, cannot penetrate cartilage to reach chondrocytes residing in tissue deep zones either owing to their large size,17,18,24,59 negatively charged surfaces, 29 or aggregating nature.18,29 The hydrodynamic diameter of mAv even after KGN conjugation is within the 10 nm pore size limit of cartilage matrix. mAv-KGN is thus not sterically hindered, and its intra-cartilage transport and binding is significantly enhanced by electrical charge interactions. Use of high cationic charges is associated with cytotoxicity concerns but that can be minimized by incorporating structure modifications like PEGylation, spatial charge distribution, use of reducible linkers for easy breakdown and by controlling the dose32,60—several of these features were incorporated in mAv-KGN design. PEGylation also increased its drug loading content thereby requiring smaller doses of Avidin for KGN delivery. mAv-KGN, therefore, has high translational potential due to its simple Avidin-biotin based chemistry, 38 no cytotoxicity and its ability to reduce the amount of drug dose and the frequency of administration.
A limitation of this study is that synovial fluid was not used. Synovial fluid has negatively charged hyaluronic acid that could potentially hinder transport of cationic carriers into cartilage. However, our in vivo work evaluating intra-articular kinetics and biodistribution in rats and rabbits has shown that Avidin can rapidly penetrate through the full thickness of cartilage in high concentrations35-37 potentially owing to 20x higher negative fixed charge density of cartilage than the joint fluid. 30 Future work will evaluate the efficacy of mAv-KGN in vivo using post-traumatic OA models. As OA has a range of causal factors, and one class of OA drugs may not be effective for all clinical cases, a broad applicability of drug delivery system is critical. 10 mAv offers itself as a broad charge-based platform that can enable sustained delivery of a variety of small molecules and their combinations to cartilage as well as to scaffolds for chondral repair.39,61
Conclusion
In summary, we designed cartilage targeting and homing formulations for delivery of a pro-anabolic OA drug, KGN by using a branched cationic protein-based carrier, multi-arm Avidin. By using the weak-reversible binding nature of charge interactions, we showed that mAv-KGN can create a concentrated drug depot inside the negatively charged cartilage and effectively suppress cytokine-induced catabolism in cartilage with only a single dose. The hydrolysable ester linkers enabled sustained KGN release in therapeutic doses over several days while the formulation with non-releasable amide linker elicited chondroprotective response following direct uptake by the chondrocytes. A single dose of free KGN, on the other hand, was not effective. This charge-based cartilage targeting delivery system, therefore, can reduce the required number of injections and drug dosage. It paves way for facilitating clinical translation of various other OA drugs, which is currently limited due to a lack of tissue targeting methods and/or concerns around drug toxicity.
Supplemental Material
sj-tiff-1-car-10.1177_19476035221093072 – Supplemental material for Single-Dose Intra-Cartilage Delivery of Kartogenin Using a Cationic Multi-Arm Avidin Nanocarrier Suppresses Cytokine-Induced Osteoarthritis-Related Catabolism
Supplemental material, sj-tiff-1-car-10.1177_19476035221093072 for Single-Dose Intra-Cartilage Delivery of Kartogenin Using a Cationic Multi-Arm Avidin Nanocarrier Suppresses Cytokine-Induced Osteoarthritis-Related Catabolism by Tengfei He, Irfhan Shaw, Armin Vedadghavami and Ambika G. Bajpayee in CARTILAGE
Supplemental Material
sj-tiff-2-car-10.1177_19476035221093072 – Supplemental material for Single-Dose Intra-Cartilage Delivery of Kartogenin Using a Cationic Multi-Arm Avidin Nanocarrier Suppresses Cytokine-Induced Osteoarthritis-Related Catabolism
Supplemental material, sj-tiff-2-car-10.1177_19476035221093072 for Single-Dose Intra-Cartilage Delivery of Kartogenin Using a Cationic Multi-Arm Avidin Nanocarrier Suppresses Cytokine-Induced Osteoarthritis-Related Catabolism by Tengfei He, Irfhan Shaw, Armin Vedadghavami and Ambika G. Bajpayee in CARTILAGE
Supplemental Material
sj-tiff-3-car-10.1177_19476035221093072 – Supplemental material for Single-Dose Intra-Cartilage Delivery of Kartogenin Using a Cationic Multi-Arm Avidin Nanocarrier Suppresses Cytokine-Induced Osteoarthritis-Related Catabolism
Supplemental material, sj-tiff-3-car-10.1177_19476035221093072 for Single-Dose Intra-Cartilage Delivery of Kartogenin Using a Cationic Multi-Arm Avidin Nanocarrier Suppresses Cytokine-Induced Osteoarthritis-Related Catabolism by Tengfei He, Irfhan Shaw, Armin Vedadghavami and Ambika G. Bajpayee in CARTILAGE
Footnotes
Acknowledgments and Funding
This work was funded by the National Institutes of Arthritis Musculoskeletal and Skin Diseases (AR075121). Funding sources had no involvement in study design, collection, analysis, interpretation of data or in the writing of this manuscript. We are grateful to Dr. Jason Guo, Director of the NMR facility for providing access and Mengqi Reng, PhD student in Chemical engineering Department at Northeastern University for providing suggestion on NMR spectrum. We would like to acknowledge to Dr. Ke Zhang from Northeastern University for graciously allowing us to use their MALDI-TOF facility and CILS core facility at Northeastern University for providing access to use 2-photon microscopy. We would also like to thank Yang Wenhui for assistance in artwork and the rest of the Bajpayee Lab for helpful discussions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was not sought for the present study because this research did not require Ethics Board approval because it does not involve human or animal subjects.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
