Abstract
Objective
Biologics are increasingly used for cartilage repair and osteoarthritis (OA) treatment. This study aimed to provide an overview of the clinical trials conducted on this subject.
Design
Two-word combinations of two sets of key words “cartilage”; “joint”; “osteoarthritis” and “biologics”; “stem cells”; “cell implantation” were used to search the database of ClinicalTrials.gov and supplemented with searches of PubMed and EMbase. The registered trials were analyzed for clinical conditions, completion status, phases, and investigated biologics. Recently completed trials with posted/published results were summarized.
Results
From 2000 to 2022, a total of 365 clinical trials were registered at ClinicalTrials.gov to use biologics for cartilage repair and OA treatment. Since 2006, the number of registered trials accelerated at an annual rate of 16.4%. Of the 265 trials designated with a phase, 72% were early Phase 1, Phase 1, and Phase 2. Chondrocytes and platelet-rich plasma (PRP) were studied in nearly equal number of early- and late-stage trials. Mesenchymal stem/stromal cells (MSCs) were the most commonly investigated biologics (38%) and mostly derived from bone marrow and adipose tissue (70%). In last 5 years, 32 of the 72 completed trials posted/published results, among which seven Phase 3 trials investigated chondrocytes, PRP, bone marrow aspirate concentrate, hyaluronic acid, collagen membrane, and albumin.
Conclusions
There was a rapid increase in the number of registered clinical trials in recent years, using a variety of biologics for cartilage repair and OA treatment. Majority of the biologics still require late-stage trials to validate their clinical effectiveness.
Introduction
Therapeutic biologics are components or products of living organisms for medical applications. Under this category, the U.S. Food and Drug Administration (FDA) lists “a wide range of products such as vaccines, blood and blood components, allergenics, somatic cells, gene therapy, tissues, and recombinant therapeutic proteins” (www.fda.gov). Biologic products are increasingly used in cartilage repair and osteoarthritis (OA) treatment. 1 To many of the products, however, their clinical and cost effectiveness, protocol optimization and indication specification are still to be proven or validated by clinical trials.2,3 The clinical applications of specific biologics or relevant therapies have been reviewed or analyzed in recent literature.4-6 What is lacking is an overview of the clinical trials used a diverse array of biologics for the treatment of cartilage defect and OA, whether the trials are just initiated, ongoing, or completed, and with or without published results. Such an overview would not only sum the accomplished clinical studies but also demonstrate trends of biologic product development and areas of clinical concerns.
ClinicalTrials.gov maintained by the National Library of Medicine at the National Institutes of Health is the largest clinical trial registry in the world. The registry publishes details of study design, intervention, outcome measures, eligibility criteria, investigator and sponsor information, and results of the trials in a standardized format. In fact, more than half of the clinical trial results are only available at ClinicalTrials.gov. 7
The goal of this study was to provide an overview of the clinical trials used biologics for the treatment of cartilage defect and OA. This study systematically examined Clinicaltrials.gov, with supplementary searches of PubMed/Medline and EMbase databases, for relevant trials. Rather than limited to one or one category of biologics, this study included a variety of biologics for cartilage repair and OA treatment. The clinical trial data were analyzed by status, phases, and categories of biologics. The results of the trials completed in the last 5 years were presented in a summary.
Methods
1. ClinicalTrials.gov: A search of clinical trials was performed at www.ClinicalTrials.gov on January 16, 2022, using two-word combinations of two sets of key words: (1) cartilage, joint, osteoarthritis; (2) biologics, stem cells, cell implantation. No limitations and filters were applied. The returns of nine inquiries were downloaded in csv file format and assembled into one document, after removed duplicates, of 1514 trials.
Irrelevant trials were deleted, based on the following exclusion criteria: (1) trials about non-biologic implants, hemostasis, physical therapies; (2) trials about supplements/drugs via oral or intravenous administration; (3) trials collecting disease samples or about biochemical analysis; (4) biologics for imaging and diagnostic applications; and (5) trials on other medical conditions, such as rheumatoid arthritis, inflammatory arthritis, and joint infection. A total of 356 trials remained for further analysis.
2. PubMed/Medline: To implement ClinicalTrials.gov, a PubMed search was conducted on January 16, 2022, using the same nine sets of key words, with filters of “Clinical Trial” and “ClinicalTrials.gov Identifier.” After removing duplicates and irrelevant papers, using the same exclusion criteria applied to the data of ClinicalTrials.gov, 16 publications were identified by nine inquiries. Of them, three had a ClinicalTrials.gov identifier unique to the ClinicalTrials.gov dataset and were manually added into the trial list of ClinicalTrials.gov.
3. EMbase: A search on EMbase was conducted on January 16, 2022, using the same nine sets of key words as for ClinicalTrials.gov and PubMed/Medline. An EMbase term “ClinicalTrials.gov” was used in each inquiry. After removed duplicates and irrelevant papers, using the same exclusion criteria applied to the data of ClinicalTrials.gov, 11 studies were identified by nine inquiries. Of them, six had a unique ClinicalTrials.gov identifier and were manually added in the ClinicalTrials.gov trial list.
As the results of searching 3 databases, 365 clinical trials used biologics for cartilage repair and OA treatment and registered at ClinicalTrials.gov were included in this study.
4. Data sorting and analysis: In ClinicalTrials.gov, the trial phases are listed as Early Phase 1, Phase 1, Phase 2, Phase 3 and Phase 4. Based on clinical significance, this study introduced Early Phase to include Early Phase 1, Phase 1 and Phase 2, and Advanced Phase to include Phases 3 and 4. When two trial phases were indicated for a trial, the trial was counted by the lower phase.
The trial status in ClinicalTrials.gov registry is designated as active, not recruiting, by invitation, recruiting, suspended, terminated, withdrawn, completed, and unknown. For analysis, the status of the trials in this study was re-grouped as incomplete (= active + not recruiting + recruiting + by invitation), terminated (= suspended + terminated + withdrawn + no longer available), complete and unknown.
The trials were sorted by the year scheduled to start, status and phase of the trials, type of biologics, disease conditions and individual joints. Particularly, the trials completed between 2016 and 2021 and having results posted in ClinicalTrials.gov database or published in journals were summarized.
Statistical analysis: Average annual increase rate was calculated with
Results
1. Trend of the trials: The 365 trials included in this study were started or scheduled to start between March 2000 and April 2022. Between year 2000 and 2005, there was only one or none trial registered per year. By January 16, there was 12 trials scheduled to start in the year of 2022. For a full calendar year between 2006 and 2021, the number of registered trials fluctuated but trended to increase, with an average annual increase rate of 16.4% (
2. Phases of the trials: There were 21 trials that did not provide information about trial phases and 79 trials entered “not applicable” for trial phase clarification. Among the remaining 265 trials, 10 trials were classified as Early Phase 1, 51 trials as Phase 1, 56 trials as Phase 1/Phase 2, 64 trials as Phase 2, 11 trials as Phase 2/Phase 3, 50 trials as Phase 3 and 23 trials as Phase 4 (
3. Status of the trials: Among the 365 trials, the status of 60 trials were unknown and 145 had been completed. There were 13 trials withdrawn from the registry, 2 trials suspended, and 14 trials terminated. Among the 131 incomplete trials, 71 were recruiting, 2 were recruiting by invitation, 22 active but not recruiting, and 36 not yet recruiting (
4. Trial conditions and joints: Of the 365 trials, several trials involved in multiple or unspecific joints and treated for both OA and cartilage defects. There were 322 trials (88%) for the medical condition of OA only or included OA as a sub-group (
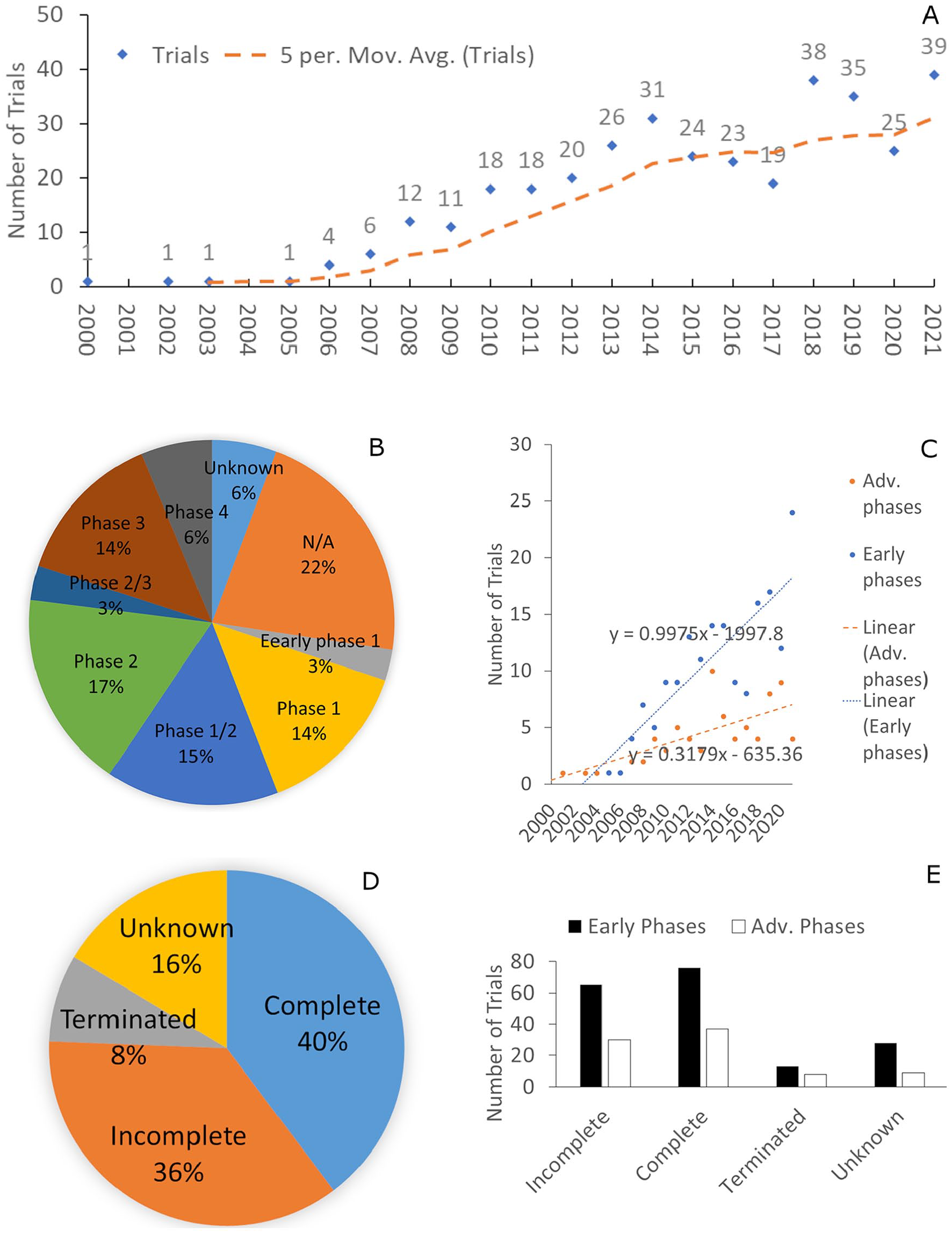
Analysis of 365 clinical trials on biologics for cartilage repair and osteoarthritis treatment. (
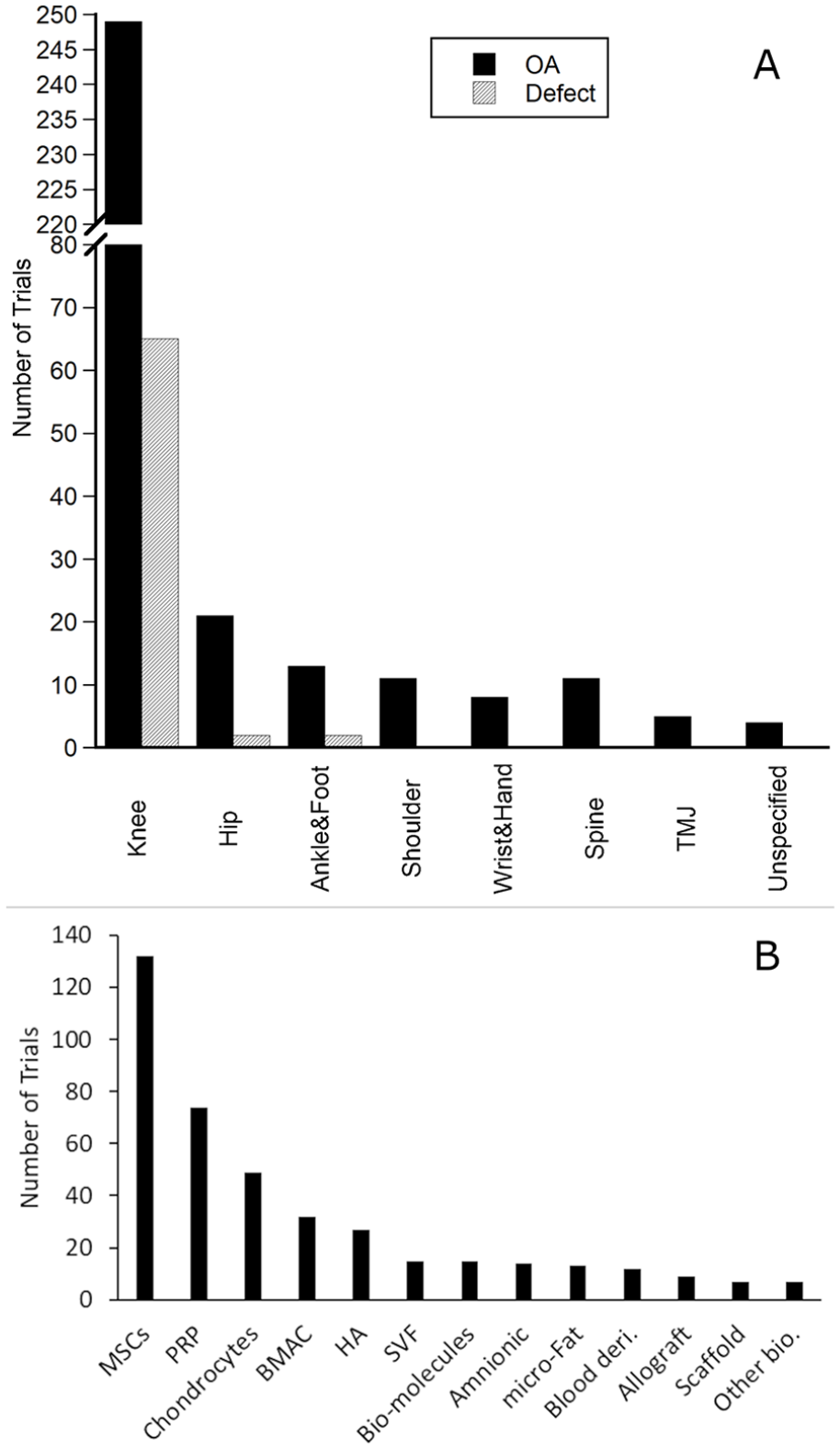
(
There were 69 trials (19%) that targeted articular cartilage defects, mostly in the knee. A few trials involved in hip and ankle cartilage lesions.
5. Biologics in the trials: A wide range of biologic products were used for the treatment of cartilage defects and OA in the 365 trials (
5.1. MSCs: Of 132 trials, most involved in one type of MSCs but a few trials applied MSCs of different tissue origins. The common tissue origins of MSCs were bone marrow and adipose tissue ( Table 1 ). MSCs used in the trials were also derived from umbilical cord blood or the cord, amnionic tissues, peripheral blood, and dental pulp. The majority of the MSC trials targeted at the medical condition of OA (96%) in the knee (91%). The estimated enrollment for these trials varied from 1 to 480 (median 30 and average 60 participants per trial). Among the trials, 68% were in Early Phase and 9% in Advanced Phase. About 37% of the trials were active or recruiting, 36% complete, and 6% terminated or withdrawn from the registry.
5.2. PRP: For the 74 trials about PRP, all were designed for OA treatment and enrollments were estimated from 10 to 4,000 (median 70). The trials were performed mostly on the knee (75%). Other joints in this group of trials included hip (n = 4), shoulder (n = 3), wrist and hand (n = 2), ankle (n = 2), and spine (n =2), TMJ and unspecified joints. Among the 74 trials, 37% were in Early Phase and 32% in Advanced Phase. Of 74 trials, 31 focused on the therapeutic effectiveness, preparation protocols, and application regimen of PRP for OA. In other trials, PRP was either compared with other biologics or as a conjunct reagent of a biologic therapy. PRP therapy was compared with HA in 16 trials for OA treatment. In 20 trials, intraarticular injection of PRP was compared with other cell/tissue biologics, such as MSCs, BMAC, SVF, micro-Fat, and amniotic tissue products, for OA treatment. Four trials compared intraarticular injection of PRP with corticosteroids and three trials compared PRP with visco-supplementation, for OA treatment. Intraarticular injection of PRP was also compared with hydrogel (NCT04519047), acetaminophen (NCT01782885), and physical exercise (NCT04697667) in OA treatment.
5.3. Chondrocytes: There were 49 cell therapy trials using chondrocytes, with estimated enrollments from 6-510 (median 46.5; average 87). Chondrocyte therapies were mostly applied for cartilage defects (71%). There were 19 chondrocyte trials in Early Phase and 20 in Advanced Phase.
Summary of Clinical Trials on MSCs, PRP and Chondrocytes for the Treatment of Cartilage Defect and OA.
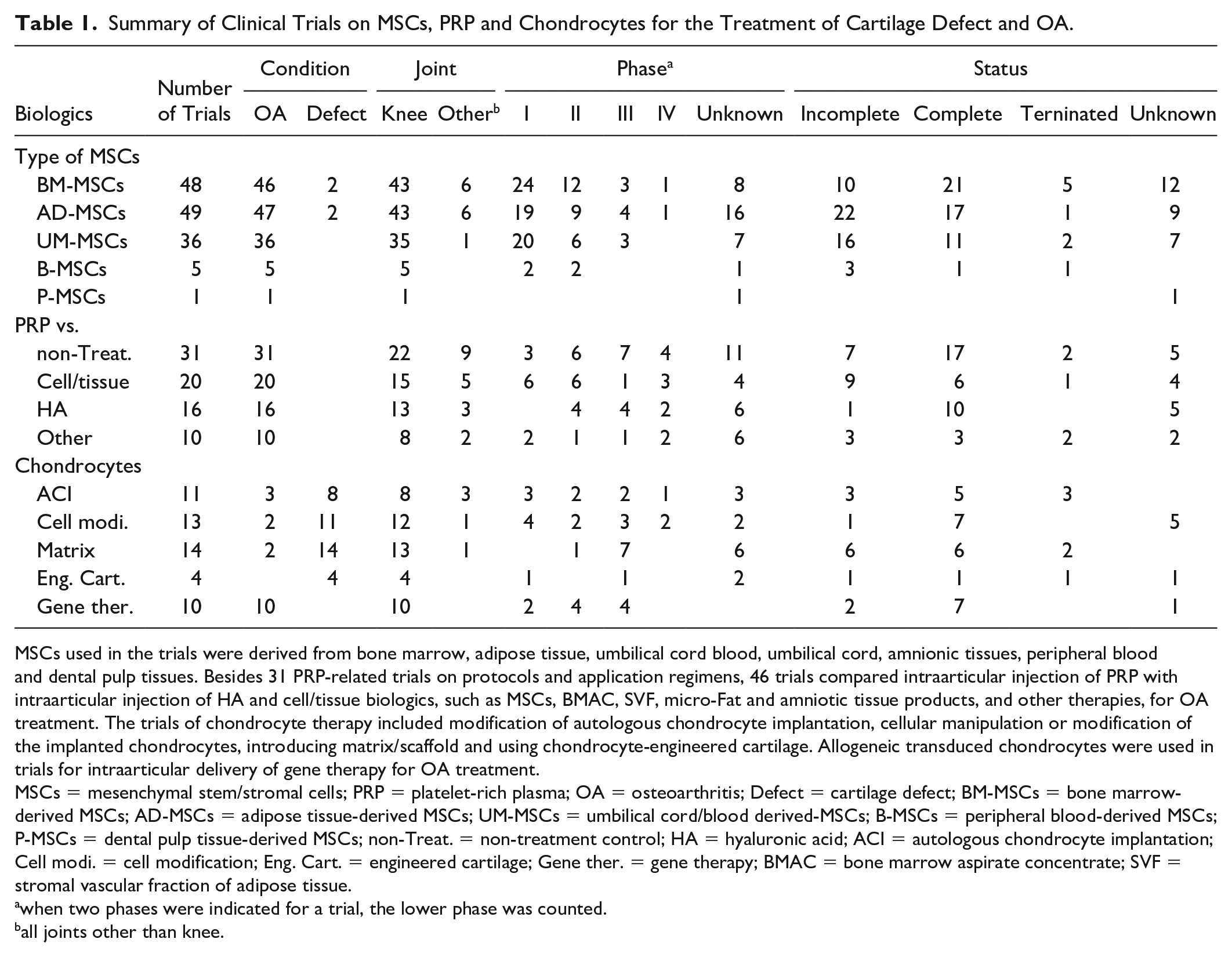
MSCs used in the trials were derived from bone marrow, adipose tissue, umbilical cord blood, umbilical cord, amnionic tissues, peripheral blood and dental pulp tissues. Besides 31 PRP-related trials on protocols and application regimens, 46 trials compared intraarticular injection of PRP with intraarticular injection of HA and cell/tissue biologics, such as MSCs, BMAC, SVF, micro-Fat and amniotic tissue products, and other therapies, for OA treatment. The trials of chondrocyte therapy included modification of autologous chondrocyte implantation, cellular manipulation or modification of the implanted chondrocytes, introducing matrix/scaffold and using chondrocyte-engineered cartilage. Allogeneic transduced chondrocytes were used in trials for intraarticular delivery of gene therapy for OA treatment.
MSCs = mesenchymal stem/stromal cells; PRP = platelet-rich plasma; OA = osteoarthritis; Defect = cartilage defect; BM-MSCs = bone marrow-derived MSCs; AD-MSCs = adipose tissue-derived MSCs; UM-MSCs = umbilical cord/blood derived-MSCs; B-MSCs = peripheral blood-derived MSCs; P-MSCs = dental pulp tissue-derived MSCs; non-Treat. = non-treatment control; HA = hyaluronic acid; ACI = autologous chondrocyte implantation; Cell modi. = cell modification; Eng. Cart. = engineered cartilage; Gene ther. = gene therapy; BMAC = bone marrow aspirate concentrate; SVF = stromal vascular fraction of adipose tissue.
when two phases were indicated for a trial, the lower phase was counted.
all joints other than knee.
There were 11 trials used autologous chondrocyte implantation (ACI) with technical modification or in comparison with other cartilage repair procedures. They were mostly applied for repairing cartilage lesions (8/11) and in the knee (8/11). There were 13 trials featured manipulations or cellular modifications of the implanted chondrocytes. For example, there were trials supplemented autologous chondrocytes/chondrons implantation with allogeneic MSCs (NCT03672825, NCT04236739) or performed chondrogenic selection of the cultured chondrocytes before implantation (NCT00414700). Chondrocytes were implanted in the forms of cell sheet (NCT01694823), spheres (NCT01694823), or encapsulated in beads (NCT01050816). Fourteen trials incorporated extracellular matrix, synthetic scaffold, and hydrogel to ACI (NCT719576, NCT1947374, NCT560664). A series of trials (n = 10) on intraarticular delivery of allogeneic chondrocytes with augmented transforming growth factor- β1 (TGF-β1) expression were conducted in OA joints. Four trials were conducted on implantation of chondrocyte-engineered cartilage for repairing cartilage defects.
5.4. BMAC: In 32 trials, BMAC was used mostly for OA treatment in the knee ( Table 2 ). The estimated enrollment of the BMAC trials was from 6 to 4,000 (mean = 439; median = 37). In most of the trials (n = 27), BMAC was delivered by intraarticular injection. BMAC was also applied on scaffold (n = 3) or incorporated in osteochondral graft (n = 2) for implantation. In eight trials, BMAC was investigated in comparison with other biologics or drugs, such as HA. 34% of the trials were in Early Phase and 19% in Advanced Phase.
5.5. HA: There were 27 trials about HA, all for OA or degenerative joints (23 in knee, 2 in hip, 1 in shoulder and 1 in TMJ). The estimated enrollment for the 27 HA-related trials was from 14 to 270 (median = 70; mean = 91). Among them, three trials investigated HA alone and 24 trials compared HA with other biologics/therapies for OA treatment ( Table 3 ). 33% of the HA-related trials were in Early Phase and 41% in Advanced Phase.
5.6. SVF and micro-fat: Fifteen trials applied SVF for the treatment of cartilage defect and OA. Except of one trial (NCT05120700), in which SVF and synovial stromal cells were engineered for repairing cartilage defect, all other trials injected autologous SVF intraarticularly for the treatment of OA. For similar clinical indications, autologous micro-fragmented fat tissue was also used in the trials (n = 13). In this SVF and micro-Fat group, 76% of the trials were Early Phase and 24% Advanced Phase.
5.7. Allograft: There were nine trials used various allogeneic tissues for OA treatment or cartilage repair. For example, meniscal allograft was used to reconstruct the osteoarthritic metatarsophalangeal (MTP) joint of the big toe and the basal joint of the thumb (NCT01673347, NCT01542515). There were several trials on neonatal cartilage (NCT00791245), osteochondral allograft or the entire joint incorporated with autologous BMAC (NCT03719417), prepared as a biologic joint, for transplantation. Decellularized osteochondral allograft was prepared and transplanted for osteochondral defect treatment (NCT02430558).
5.8. Biologic products made from amniotic tissue/fluid, placental tissue and umbilical cord/blood were investigated in 14 trials, for treatment of OA in the knee (n = 10), ankle (n = 2), shoulder (n = 1), and unspecified joints.
5.9. Bio-molecules: Several antibodies/antagonists against key molecules of OA pathology, such as Interleukin-1 receptor (IL-1R) and Tumor Necrosis Factor-α (TNF-α), were developed for joint injection (n = 15). This category included a series of trials (n = 5) that intraarticularly delivered DNA plasmid with a variant Interleukin-10 (IL-10) transgene for the treatment of OA in the knee and lumbar spine (NCT04124042, NCT04841512). Recombinant human growth factors (osteogenic protein-1, NCT00456157) and platelet-derived growth factor-18 (NCT01033994) were used in OA joints in two trials.
5.10. Blood-derived biologics: Blood derivatives were injected intraarticularly in 12 trials for OA treatment. A series of trials (n = 9) were focused on a product of low molecular weight fraction of 5% human serum albumin. Autologous protein solution (APS) prepared from the cellular components of the blood was used in three trials for OA.
5.11. Scaffold: Seven trials used a variety of acellular scaffold for cartilage regeneration. The biologic scaffold included products made of extracellular matrix (NCT05082831), bi-layer matrix made from collagen and glycosaminoglycan (NCT03808623), bi-layer scaffold featuring varied pore sizes and molecular compositions (NCT01791062).
5.12. Other biologics: There were eight trials of OA treatment used intraarticular injection of Botulinum Toxin type A (NCT01518257), hydrogel (NCT05086068), HA-based visco-supplementation products (NCT01625013) and chitosan (NCT02323451). One trial (NCT02696876) used a novel synovium harvesting device to implement microfracture procedure with implantation of autologous synovium to facilitate biological repair of cartilage.
6. Results of clinical trials in last 5 years.
Summary of Biologics Used in Clinical Trials for Treatment of Cartilage Defect and OA.
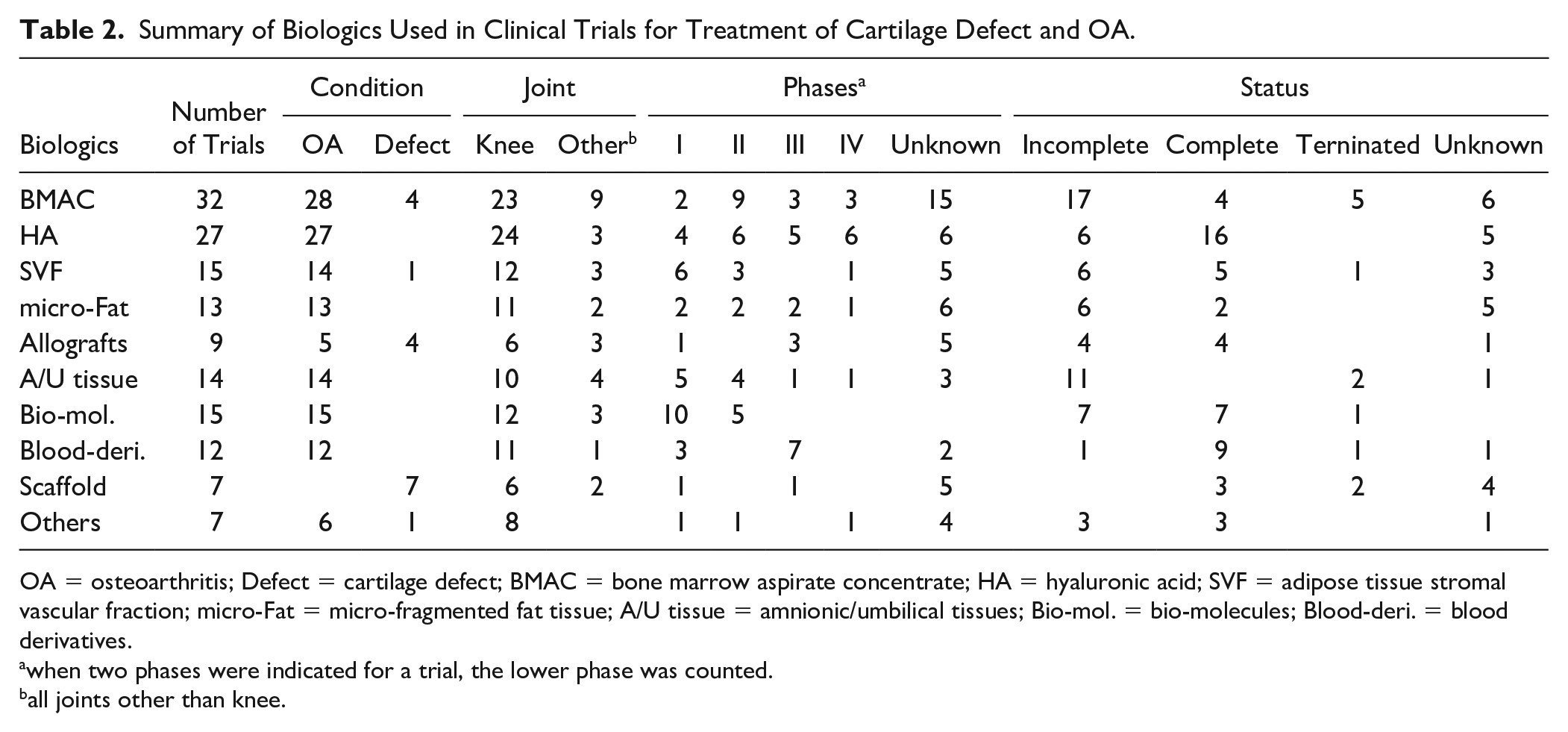
OA = osteoarthritis; Defect = cartilage defect; BMAC = bone marrow aspirate concentrate; HA = hyaluronic acid; SVF = adipose tissue stromal vascular fraction; micro-Fat = micro-fragmented fat tissue; A/U tissue = amnionic/umbilical tissues; Bio-mol. = bio-molecules; Blood-deri. = blood derivatives.
when two phases were indicated for a trial, the lower phase was counted.
all joints other than knee.
HA, in Comparison With Other Biologics, Used in Clinical Trials for OA Treatment (n = 24).
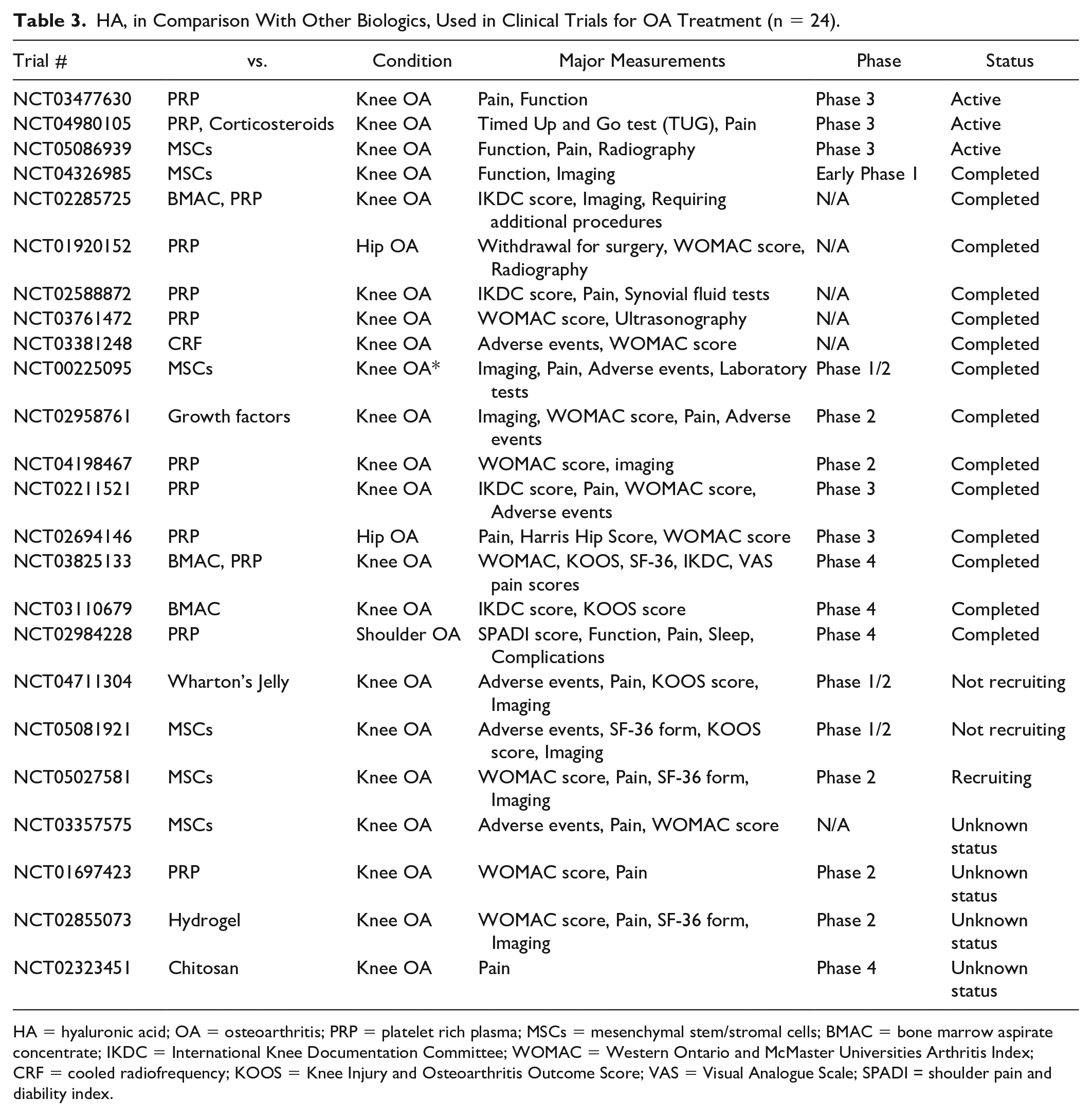
HA = hyaluronic acid; OA = osteoarthritis; PRP = platelet rich plasma; MSCs = mesenchymal stem/stromal cells; BMAC = bone marrow aspirate concentrate; IKDC = International Knee Documentation Committee; WOMAC = Western Ontario and McMaster Universities Arthritis Index; CRF = cooled radiofrequency; KOOS = Knee Injury and Osteoarthritis Outcome Score; VAS = Visual Analogue Scale; SPADI = shoulder pain and diability index.
Among the 365 trials included in this study, 72 trials were completed between 2016 and 2021, and 13 trials posted their results on ClinicalTrials.gov. Additionally, 19 trials published results in the journals indexed by PubMed and EMbase ( Table 4 ). Seven trials with results were phase 3 trials:
Clinical Trials on Biologics for the Treatment of OA and Cartilage Defect Completed Between 2016 and 2021 and Having Posted/Published Results (n = 32).
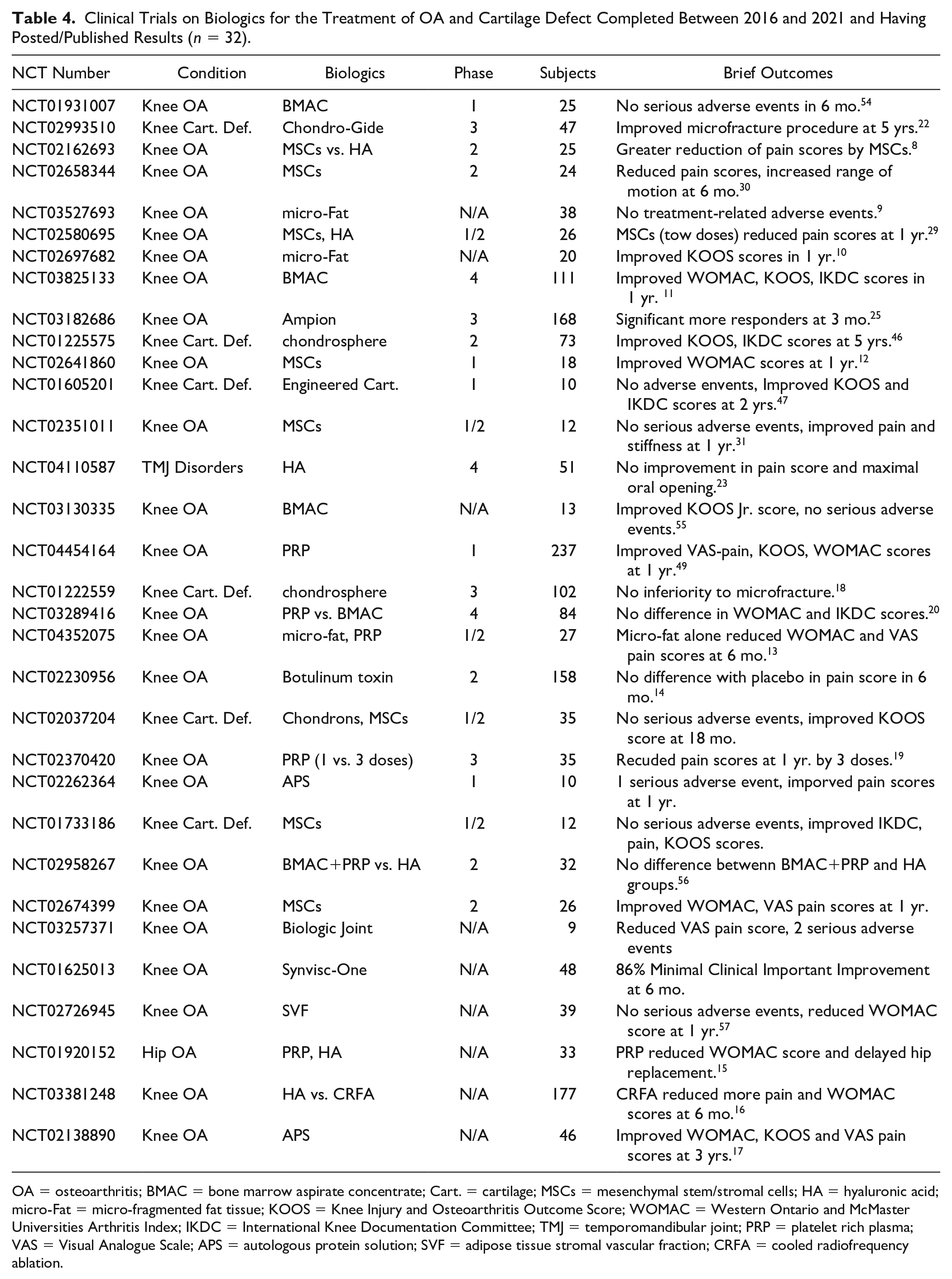
OA = osteoarthritis; BMAC = bone marrow aspirate concentrate; Cart. = cartilage; MSCs = mesenchymal stem/stromal cells; HA = hyaluronic acid; micro-Fat = micro-fragmented fat tissue; KOOS = Knee Injury and Osteoarthritis Outcome Score; WOMAC = Western Ontario and McMaster Universities Arthritis Index; IKDC = International Knee Documentation Committee; TMJ = temporomandibular joint; PRP = platelet rich plasma; VAS = Visual Analogue Scale; APS = autologous protein solution; SVF = adipose tissue stromal vascular fraction; CRFA = cooled radiofrequency ablation.
In a non-inferiority trial of ACI (NCT1222559), autologous chondrocytes were cultured in spheroids and implanted in cartilage defect in the knee (n = 34; defect size 2.7 ± 0.8 cm2).
18
Microfracture was performed in the controls (n = 32; defect size 2.4 ± 0.8 cm2). At 2-year follow up, the improvement of Knee Injury and Osteoarthritis Outcome Score (KOOS;
Single and triple intraarticular injections of PRP for knee OA (mostly Kellgren-Lawrence grade 2) were compared (NCT02370420). At 12-month follow-up, both single and 3 biweekly intraarticular injections of PRP significantly reduced joint pain and improved function.
19
But the reduction of visual analogue scale (VAS) pain score from baseline was 86% in the triple injection group (n = 17), whereas it was 37% in the single injection group (n = 18; P = 0.02). Similarly, there was greater reduction of Western Ontario and McMaster Universities Arthritis Index (WOMAC;
Intraarticular injections of PRP and BMAC were compared for knee OA in 84 participants (NCT03289416). The PRP and BMAC were prepared with similar devices and protocols.
20
At 1-year follow up, both groups showed similar improvements in WOMAC and International Knee Documentation Committee (IKDC;
Intraarticular delivery of BMAC via superolateral, anteromedial, and anterolateral portals of knee arthroscopy were compared in 111 knee OA patients (NCT03825133). 21 The improvements of KOOS, WOMAC, and VAS pain scores were not statistically different among the three portals of BAMC delivery at days 3, 7, 14, and 21 post-operatively.
To treat cartilage defect, a collagen membrane was either glued or sutured to cover the defect after microfracture (NCT02993510). The group of microfracture with a membrane coverage over the defect (n = 30) sustained improvement (>80) of Modified Cincinnati Score (
HA was injected into the TMJ after arthroscopy for TMJ disorder in 26 patients and 25 patients had arthroscopy alone (NCT04110587). No differences were observed between the two groups in VAS pain score, maximum mouth opening, and disk position by MRI at months 3, 6, 9, and 12. 23
The ultra-filtrate (<5 kDa) of 5% human serum albumin was injected into OA knees (n = 144; Kellgren-Lawrence grade 4; NCT3182686). The control group injected saline. Assessed with OMERACT-OARSI responder criteria, 24 71% (95% confidence interval: 63%–78%) of the albumin group were responders at 3 months, which exceeded the 30% threshold for clinical benefit. 25
Discussion
This study analyzed 365 trials that used biologics for the treatment of cartilage defect and OA, and registered at ClinicalTrials.gov. In contrast to only four trials registered between 2000 and 2005, trial registration accelerated after 2006. The increased clinical trials on this subject may attribute to a growing clinical interest, increased availability of commercial biologics, and regulatory and public demand. 26
Of the 365 trials, more than 70% were Early Phase trials (from early phase 1 to phase 2), which investigates the safety and effectiveness of a biologic product in small groups of patients. The Early Phase trials registered at a faster pace than the Advanced Phase (phases 3 and 4) trials during the same period. Although they are essential for moving any biologics toward clinic applications, Early Phase trials have much less immediate clinical impacts.
Majority of the 365 trials targeted OA in the knee, which is in line with a high prevalence of knee OA. 27 Among a group of diverse biologics used in the trials, MSCs accounted for more than a third of the trials (132/365). It is evident of aggressive clinical translation of recent progresses in stem cell and cartilage research. 28 There were, however, far fewer Advance Phase trials than Early Phase trials (12 vs. 94). Recently completed Early Phase trials showed no serious adverse events after intraarticular injections of umbilical cord-derived MSCs, 29 adipose tissue-derived MSCs 30 or bone marrow-derived MSCs. 31 Still, regulations and standardized protocols of processing, storage, transplantation and administration are required to guard the safe application of MSCs. 32 In small cohorts, the injected MSCs showed to improve pain and functions of the OA joints. In a phase 3 trial (NCT01041001, NCT01626677), implantation of umbilical cord-derived MSCs in knee cartilage defects exceeded the minimal clinically important difference (MCID) for pain and function improvements at 5-year follow up. 33 A meta-analysis of 13 randomized controlled clinical trials, including a total of 434 participants, however, concluded that intraarticular injection of MSCs has no therapeutic effect on OA. 34 Randomized controlled phase 3 trials, with sufficient sample sizes, are needed to validate the effectiveness of MSC therapies for cartilage repair and OA treatment.
Unlike MSCs, chondrocytes were used more in Advanced Phase trials than Early Phase trials. Developed from the original ACI technique, 35 recent trials addressed the biological properties of the chondrocytes, such as the chondrogenic potential of the implanted chondrocytes (NCT719576, NCT414700).36,37 Autologous chondrocytes were isolated as chondrons (NCT04236739, NCT02037204), which retain the pericellular matrix of the isolated chondrocytes and are more robust in matrix production and viability than chondrocytes.38,39 The concept of chondron was also introduced to mix autologous chondrocytes with fibrin before implantation (NCT1050816). Autologous chondrons and allogeneic MSCs were combined for implantation for enhanced chondrogenesis (NCT02037204). 40 The matrix/scaffold used in ACI trials not only facilitates the handling of chondrocytes during surgery but also provides the implanted chondrocytes 3-dimensional anchorage,37,41-44 which is essential to stabilize chondrocyte phenotype, maintain high viability and increase matrix production. 45 In the same line of strategy, there were trials to grow autologous chondrocytes in the forms of cell sheet or spheres for implantation (NCT01225575, NCT01222559, NCT01694823).18,46 A recent trial used nasal cartilage as a source of autologous chondrocytes and cartilage engineering (NCT01605201). 47 Allogeneic transduced chondrocytes were used for intraarticular delivery of gene therapies for OA (NCT2072070). 48
PRP was another biologic product more commonly used in the trials for OA treatment. Trials (NCT02370420, NCT04454164) demonstrated that multiple intraarticular injections increased the efficacy of PRP and extended the duration of its therapeutic effects for knee OA.19,49 A big topic of the PRP trials was to gauge its efficacy, in comparison with other biologics, such as BMAC (NCT03289416) 20 and HA (NCT01670578, NCT02211521), for OA.50,51 Varied PRP preparation protocols used in the trials were a source of controversy of how PRP should be defined or standardized. 52
Although it is controversial to characterize BMAC and SVF as minimally manipulated MSCs, 53 they were used as such in a number of trials (NCT01931007, NCT03825133, NCT03130335, NCT03289416, NCT02958267, NCT02726945) for OA treatment. The available results of these trials indicated both BMAC and SVF were safe and therapeutic for OA applications.20,21,54-57
Tissue biologics used in the trials for cartilage repair and OA treatment included micro-fragmented fat, particulated neonatal cartilage, and micronized particulate amnion and umbilical cord. Blood derivatives, antibodies, antagonists, growth factors and gene therapies used in the clinical trials targeted more specific OA pathologies. This class of biologics presented great diversity of biologic products and were used mostly in the Early Phase trials, with a few posted or published outcomes.
Because of considerable variables a biologic product inherited by its biological origin and built in during processing, several clinical trials may be needed to reach a consensus on its clinical effectiveness. 58 ClinicalTrials.gov provides a portal to access relevant clinical trials and monitor their progress. Detailed trial protocols published by the registry are not often available in research articles. In this study, 7 of the 32 recently completed trials posted their results only at ClinicalTrials.gov. Additionally, the trials ended by suspension, termination and withdrawn may have valuable information but are rarely published in journals.
In summary, the 365 clinical trials registered at ClinicalTrials.gov demonstrated a rapid increase of using biologics for the treatment of cartilage defect and OA. A large portion of the trials used MSCs for the treatment of OA but they were mostly Early Phase trials. This study showed that ClinicalTrials.gov is a useful source of information about biologics for cartilage repair and other applications.
Supplemental Material
sj-docx-1-dst-10.1177_19476035221093065 – Supplemental material for The Current Status of Clinical Trials on Biologics for Cartilage Repair and Osteoarthritis Treatment: An Analysis of ClinicalTrials.gov Data
Supplemental material, sj-docx-1-dst-10.1177_19476035221093065 for The Current Status of Clinical Trials on Biologics for Cartilage Repair and Osteoarthritis Treatment: An Analysis of ClinicalTrials.gov Data by Zijun Zhang and Lew Schon in CARTILAGE
Footnotes
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
