Abstract
Objective
The aim of this study is to evaluate the efficacy of intra-articular injection with HYAJOINT Plus, a biofermentation-derived, high-molecular hyaluronic acid (HA), on the progression of structural changes of cartilage in patients with knee osteoarthritis (OA) by using objectively promised ultrasonography (US) evaluation.
Design
In this prospective clinical trial, 56 OA patients completed the study. One single dose of injection of HYAJOINT Plus into the knee cavity was performed. The primary efficacy outcome measure for structural change of knee joint was evaluated by US using a semiquantitative grading system. Secondary efficacy outcome measures included Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) total and subscale scores. All efficacy outcomes were measured at baseline and at first, third, and sixth month following treatment.
Results
There were significant US grade-improvement changes of cartilage between baseline and follow-up visits over medial femoral condyle and transverse overall evaluation at 3- and 6-month follow-ups, and over lateral femoral condyle, intercondylar notch, and medial longitudinal area at 6-month follow-up. The improved score change of WOMAC from baseline was significant at 1- and 3-month follow-ups in pain subscale, whereas score change from baseline was significant at 6-month follow-up in total score and all 3 subscale scores.
Conclusions
It was determined that significant improvement was found on cartilage by US after intra-articular injection with high-molecular weight, biological fermentation–derived HYAJOINT Plus. The semiquantitative grading system by US is a promising tool to identify the efficacy on cartilage band after interventions.
Introduction
Osteoarthritis (OA) is the most common musculoskeletal disease worldwide, with the knee being one of the most frequently involved sites. 1 OA is a debilitating condition associated with pain and loss of mobility that consequently undermines quality of life. 2 This highly prevalent disease and the associated disability place an unfavorable impact on both individuals and society, accounting for a substantial number of health care visits and costs in populations with access to medical care. 3 With an increase in life expectancy, OA is anticipated to become a major age-related health issue and pose a significant economic burden in coming decades.4,5
In recent years, nonsurgical and nonpharmacological interventions, including education, exercise, and weight loss, have been the most recommended interventions for lower extremity OA.6-8 Studies have focused on the biology of OA, 9 and on disease modification, still having limited certainty.8,10 Hyaluronic acid (HA) and glucocorticoids have been proposed as standard intra-articular treatments for the management of knee OA in patients who fail to respond to nonpharmacological therapy, nonsteroidal anti-inflammatory drugs (NSAIDs), or analgesics in some studies; 11 however, several clinical recommendations tend to less favor avian-derived HA than glucocorticoids for the use of intra-articular injection for knee OA, including American Academy of Orthopaedic Surgeons (AAOS) 2013, Osteoarthritis Research Society International (OARSI) 2014, National Institute for Health and Care Excellence (NICE) 2014, and American College of Rheumatology (ACR) 2012. 11 Furthermore, “the best evidence suggests that hyaluronic acid injections are equivalent to saline injections in efficacy” was pointed out in expert-voted guidelines for OA management. 7 The low clinical efficacy of avian-derived HA and even cross-linked HA might relate to limited persistence of injected HA in the treated joint as a result of physiological turnover and possibly elevated degradation of HA. In recent years, high-molecular HA have been developed, derived from biofermentation by means of using specific bacterial influence. The half-life of this new HA was found to be longer than that observed for unmodified HA (which is usually less than 1 day), while for the former is around 32 days in a rabbit model and 4 weeks in humans. 12 There are many ongoing clinical trials for this newest generation of HA; however, controversy remains concerning outcome. The existence of controversy in research and treatment consensus indicates the necessity for further well-designed studies to reveal the efficacy of intra-articular HA for OA. 11
The primary outcome measure of previously published clinical studies on HA interventions has relied solely on self-rating systems for measuring pain and joint function in symptomatic OA patients, including Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), visual analogue scale (VAS), and the Lequesne index.13-17 Nevertheless, outcome definitions that involve nonobjective measurement scales can lead to misclassification and bias in evaluation of treatment efficacy; 18 consequently, it has been proposed that a strong placebo response toward agents administered via intra-articular injection might exist. 11
High-resolution ultrasonography (US) is a widely available imaging technique for the musculoskeletal system. It has the potential advantage for repeated use on the same patient over time because of being noninvasive and inexpensive, and does not involve exposure to ionizing radiation.19,20 The US was used to evaluate patients with knee OA.21,22 A well-structured semiquantitative grading system to evaluate the status of femoral condylar cartilage has been established with good reliability and validity23,24 and this grading system has been confirmed to reflect the clinical symptoms and functions related to knee OA rather well. 25
The aim of this study is to evaluate the efficacy of intra-articular injection with HYAJOINT Plus, a biofermentation-derived, high-molecular HA, on the progression of structural changes of cartilage in patients with knee OA by using objectively promised US evaluation.
Method
Study Design, Setting, and Eligibility
This study is a prospective clinical trial under approval of the Institutional Review Board, Kaohsiung Medical University Chung-Ho Memorial Hospital (KMUHIRB-F(II)-20170062) that enrolled participants from the outpatient orthopedic and rehabilitation clinic of a medical center in Kaohsiung, Taiwan, between August 2017 and July 2020. Study staff recruited potential participants and explained the purpose of the trial and eligibility requirements for enrollment, with patients of both sexes aged above 50 years and having a diagnosis of primary knee OA being eligible to participate in this study if they fulfilled the following predefined criteria, with diagnosis of knee OA of the femorotibial compartment determined according to the clinical and radiological criteria of the American College of Rheumatology. 26 Disease severity was graded on the basis of the Kellgren and Lawrence radiographic classification system. 27 Patients with any one of the following criteria at recruitment were excluded from the study: (1) Kellgren and Lawrence grade 3 or more; (2) history or active presence of clinically significant articular and rheumatic disease other than OA, or that could be responsible for secondary OA, including a history of traumas or lesion of the knee joint and severe articular inflammation as confirmed by physical examination; (3) generalized OA; (4) presence of evidence of rapidly progressive OA prior to the trial; (5) having undergone any previous lower extremity surgery; (6) mechanical axis deviation (varus > 5°, valgus > 5°); (7) obesity defined as a body mass index (calculated as weight in kilograms divided by the square of height in meters) greater than 30; (8) history or presence of malignant disorders; (9) systemic disorders such as diabetes mellitus, hematologic diseases, severe cardiovascular diseases, infections, and immune-deficiencies; (10) systematic or intra-articular corticosteroid therapy in the previous 3 months or prior treatment with HA in the past 6 months; or (11) anticoagulants or anti-aggregate therapy or use of NSAID medications in the preceding 30 days. All recruited patients completed written informed consents.
Data Collection
At baseline recruitment, a standardized in-person interview was conducted by trained and certified study personnel according to structured questionnaires to obtain information on sociodemographic characteristics, habits of tobacco, and alcohol use. All of the OA patients underwent a complete personal medical history review and detailed physical examination.
Intervention with HA Preparations
All patients were treated with a high-molecular weight HA preparation, a single dose of injection of HYAJOINT Plus (SciVision Biotech Inc., Taiwan) 3 ml (2% microbial-fermented HA, 20 mg/ml) into the knee joint cavity by the same orthopedist. The patients were seated on a chair with knee flexed and the injection was performed from anterolateral aspect of knee after aseptic procedure with no image guidance.
Outcome Measures
All efficacy outcomes were measured at baseline and 1, 3, and 6 months following treatment. The primary efficacy outcome measure for structural change of knee joint was evaluated by US using a semiquantitative grading system. Secondary efficacy outcome measures included WOMAC total and subscale scores.
US Examination
Knee structural changes in cartilage were evaluated on serial US according to the standardized technique. All patients received US examination with commercially available US equipment (Affiniti 70; Philips, USA) using a real-time 5- to 12-MHz high-resolution linear probe. The patients were placed in supine position on an examination table. The distal weightbearing surfaces of the femoral condylar cartilage were assessed by an anterior transverse suprapatellar scan with the knee in maximal flexion over 90°, with the degree of knee flexion being established by using a standard goniometer, and then the US probe was positioned transversely above the superior margin of the patella and perpendicular to the femoral articular surface, also being dynamically tilted to facilitate better visualization of the superficial margin of the hyaline cartilage. The cartilage grading was recorded over medial and lateral femoral condyles and intercondylar notch, with the grade of transverse overall being decided by the severest one among these 3 areas. In the same manner, medial femoral condylar cartilage was assessed with the probe positioned longitudinally medial to the medial border of patella. The grading method followed the system, which classifies the cartilage from grades 0 to 6 according to parameters of sharpness, clarity, and thickness change of the cartilage band.23,25 Grade 0 refers to normal and healthy cartilage, while grade 6 refers to severely eroded cartilage. All US examinations were performed by a single experienced sonographer who was blinded to the visit time of the patient.
WOMAC Questionnaire
Symptoms of OA were assessed using the WOMAC index, which is a well-validated and widely used disease-specific questionnaire. 28 The questionnaire comprises 24 items, targeting severity of joint pain (5 items), stiffness (2 items), and physical function (17 items) reported by the patients during the preceding 48 hours. The 5-point adjectival version of the index was used. Each item was scored from 0 to 4 according to the severity (none, slight, moderate, severe, and extreme) and the total index score was expressed by the summation of the 24 constituent item scores. A higher score indicated worse symptom severity. All WOMAC evaluations were performed by one specialized well-trained clinical trial nurse.
Statistical Analysis
Data of baseline characteristics were described using means with standard deviations (SDs) for continuous variables and using frequencies (percentages) for categorical variables. The changes of clinical outcomes from baseline to the 3 follow-up visits were compared by using generalized estimating equations. Spearman correlation was used to analyze the relationship between grade changes of cartilage and WOMAC score changes. All statistical analyses were performed using SPSS software version 19 (IBM Corp., Armonk, NY). A P-value of less than 0.05 was considered as statistically significant.
Results
Fifty-eight patients were recruited. The patients were predominantly female (72.4%) and the mean age was approximately 63 years. The WOMAC scores and US findings are listed in Table 1 . Ultimately, 56 patients completed the study and were analyzed.
Demographic Characteristics and Baseline Condition of the Patients (N = 58).
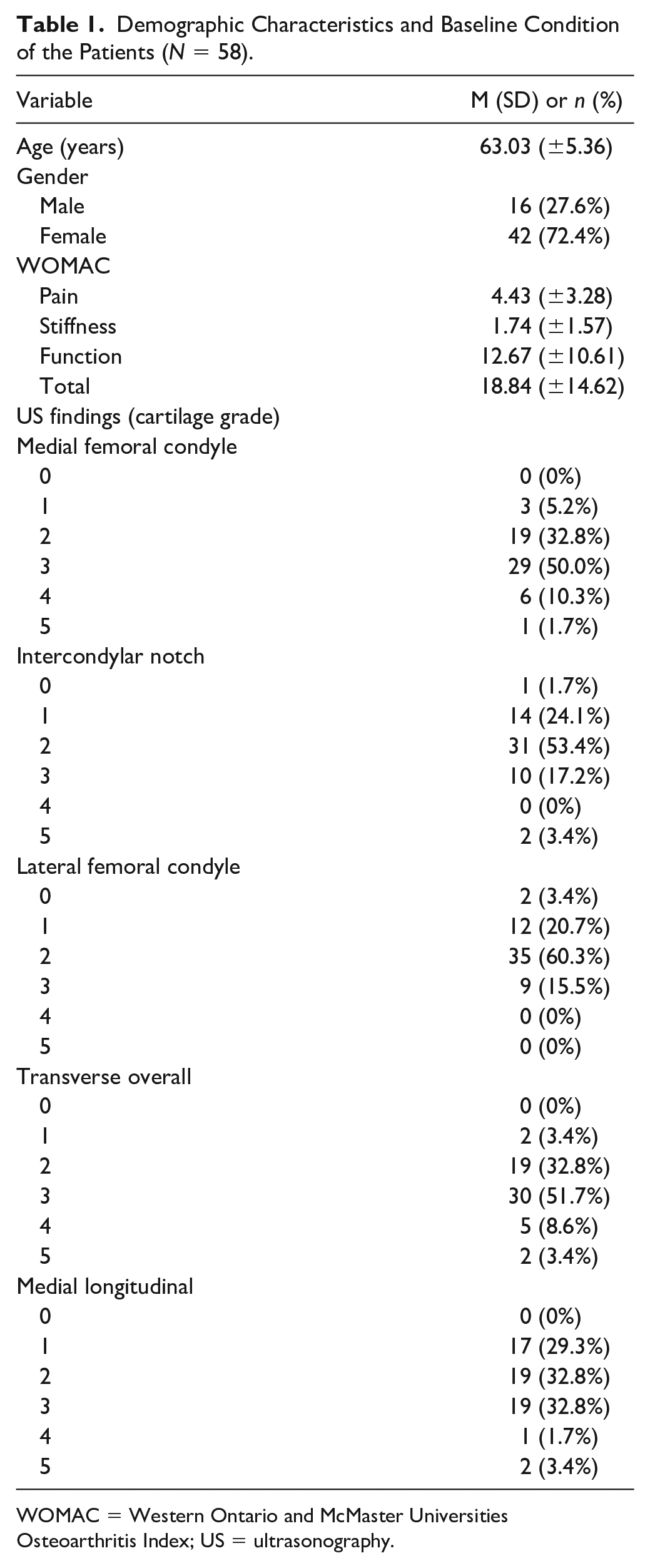
WOMAC = Western Ontario and McMaster Universities Osteoarthritis Index; US = ultrasonography.
US Findings
There were significant grade changes of cartilage compared between baseline and follow-up visits over medial femoral condyle and transverse overall evaluation at 3-and 6-month follow-ups, and over lateral femoral condyle, intercondylar notch, and medial longitudinal areas at the 6-month follow-up (
Table 2
) (
Grade Change of Cartilage Compared Between Baseline and Follow-Up Visits (N = 56).
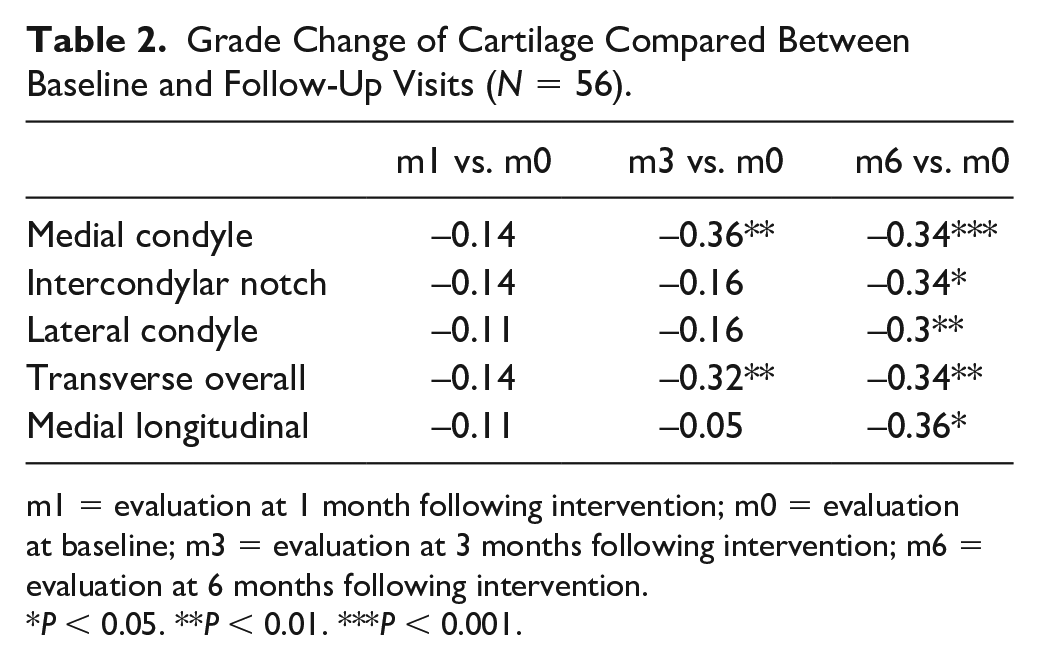
m1 = evaluation at 1 month following intervention; m0 = evaluation at baseline; m3 = evaluation at 3 months following intervention; m6 = evaluation at 6 months following intervention.
P < 0.05. **P < 0.01. ***P < 0.001.
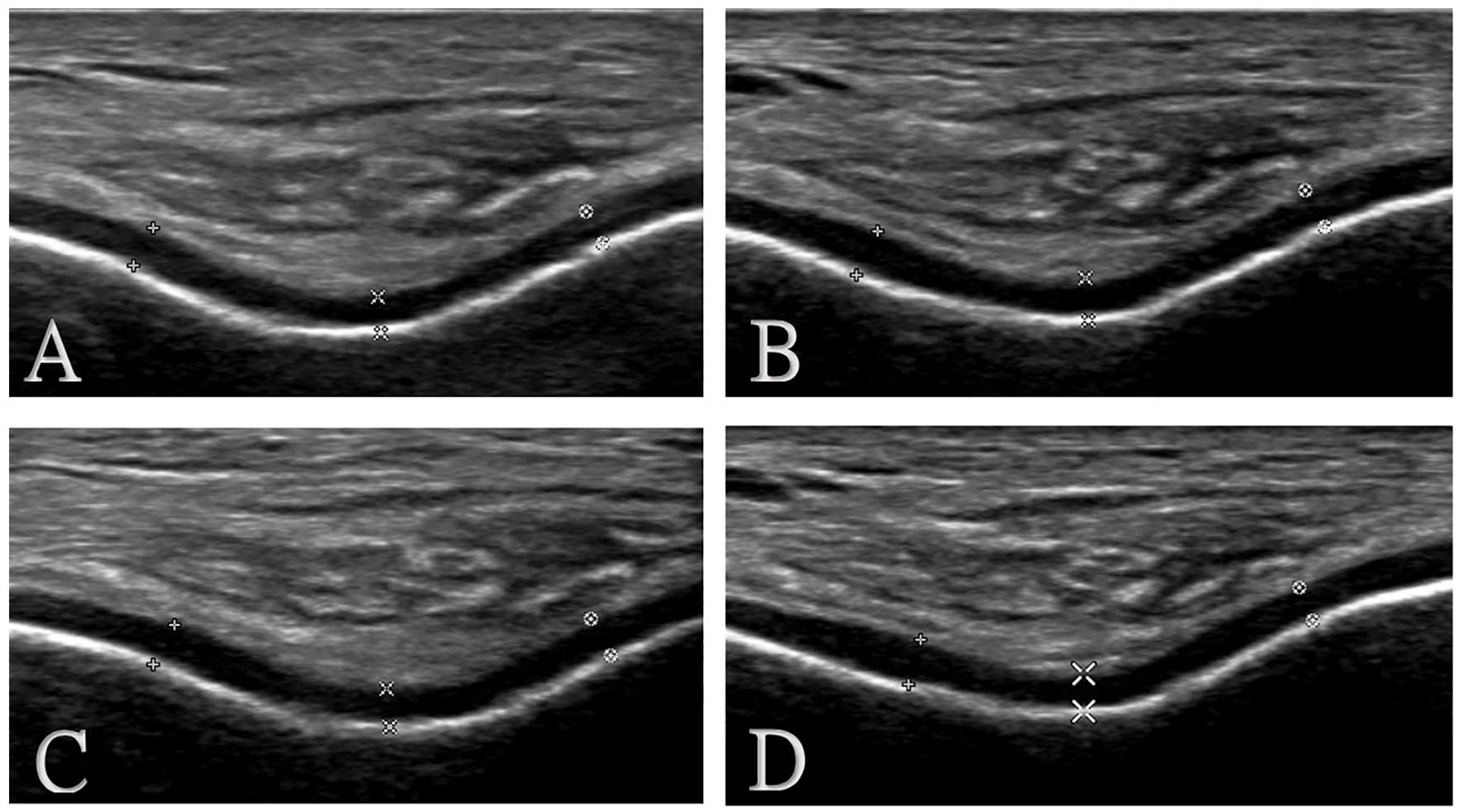
Example of US change of cartilage band in follow-up. (
WOMAC Score
There was no significant change of WOMAC score at follow-up periods, including total score and subscale scores (
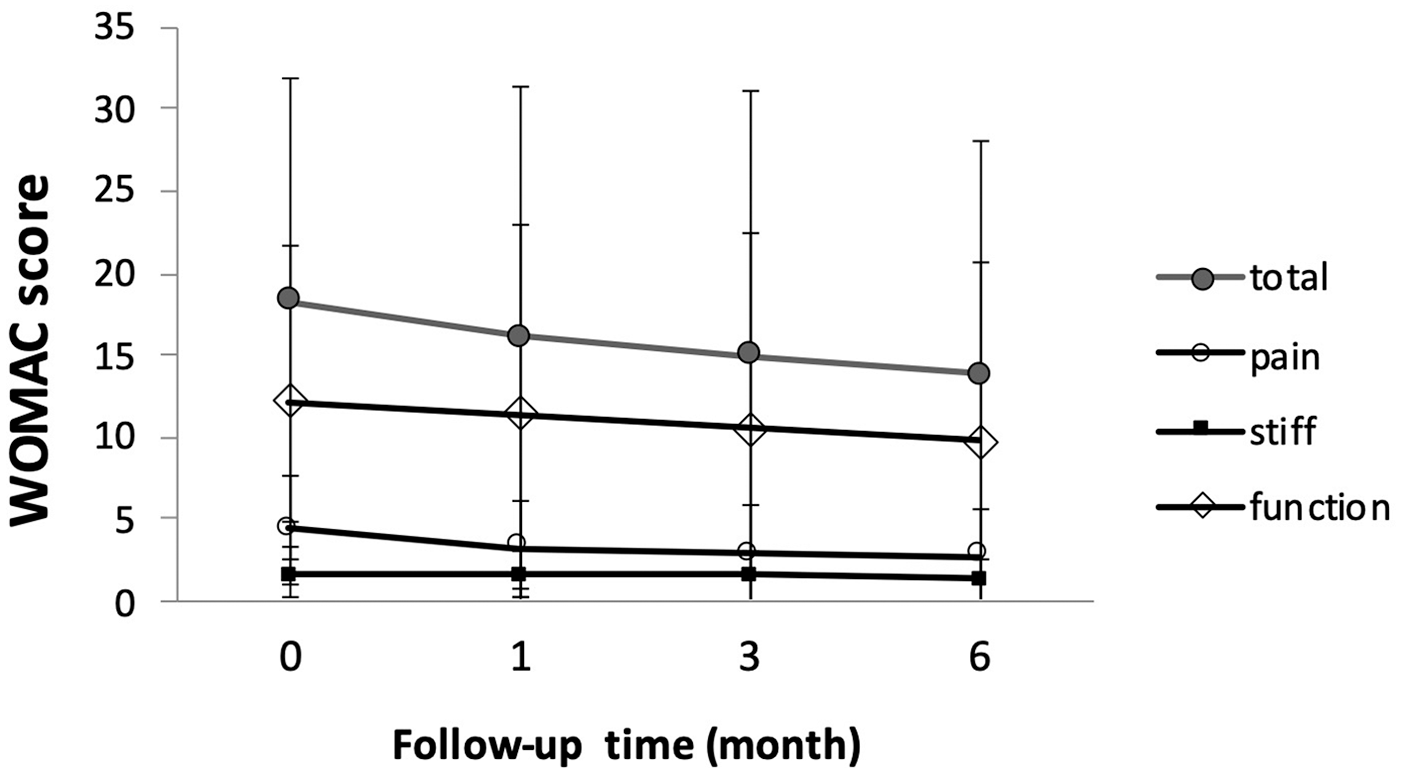
WOMAC score during follow-up (N = 56). There was no significant change of WOMAC score at follow-up periods, including total score and subscale scores. WOMAC = Western Ontario and McMaster Universities Osteoarthritis Index.
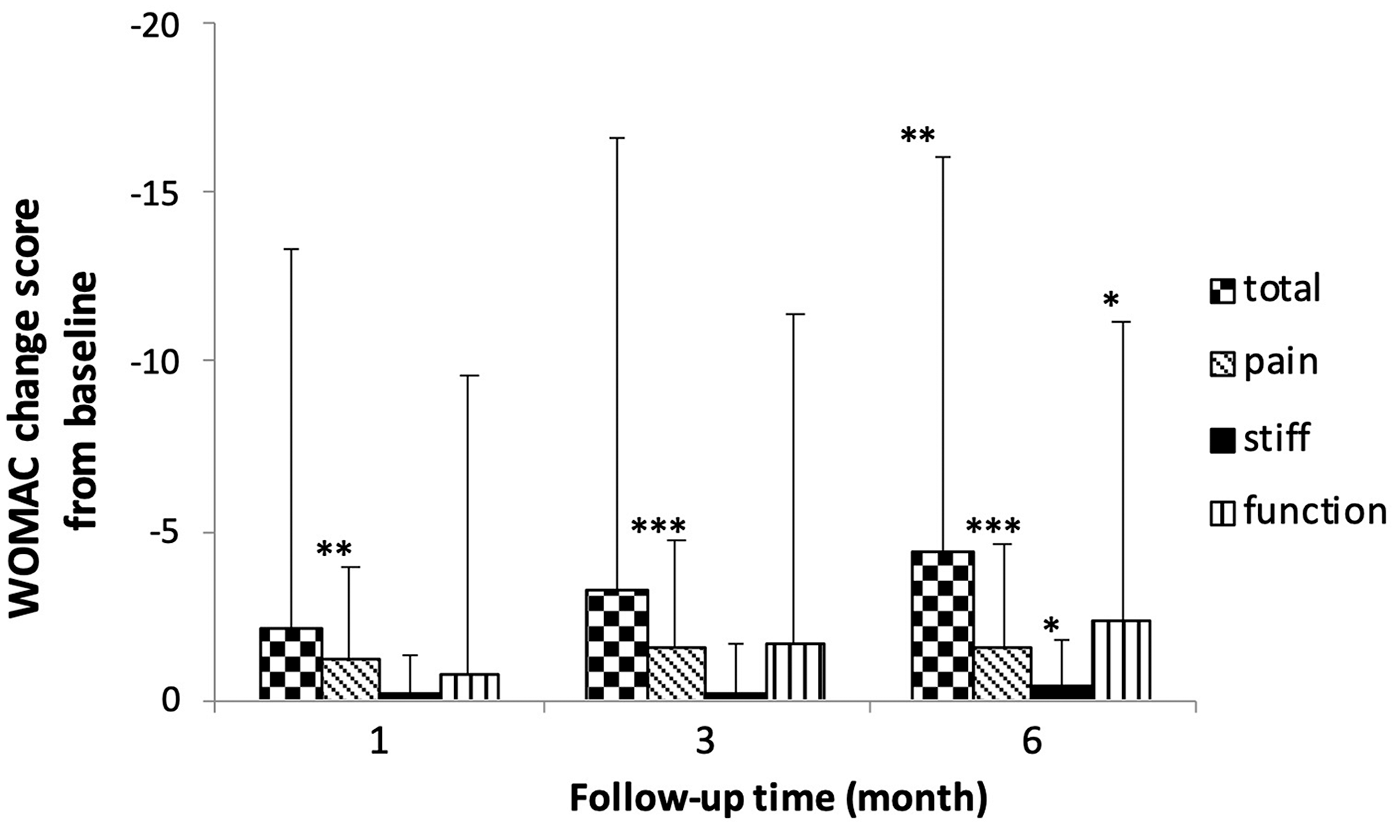
WOMAC score change from baseline (N = 56). Score change from baseline was significant at 1-month and 3-month follow-ups in pain subscale (–1.16, P = 0.002, and −1.50, P < 0.001 respectively). Score change from baseline was significant at 6-month follow-up in total score and pain/stiffness/function subscale scores (–4.36, P = 0.005; –1.59, P ≤ 0.001; –0.43, P = 0.013; –2.34, P = 0.044 respectively). WOMAC = Western Ontario and McMaster Universities Osteoarthritis Index. *P < 0.05. **P < 0.01. ***P < 0.001.
Correlation between WOMAC Score Changes and US Grade Changes at Follow-up Visits
There was no significant correlation between WOMAC score changes and US grade changes at follow-up visits ( Table 3 ).
Spearman Correlation Between WOMAC Score Changes and US Grade Changes at Follow-Up Visits (N = 56).
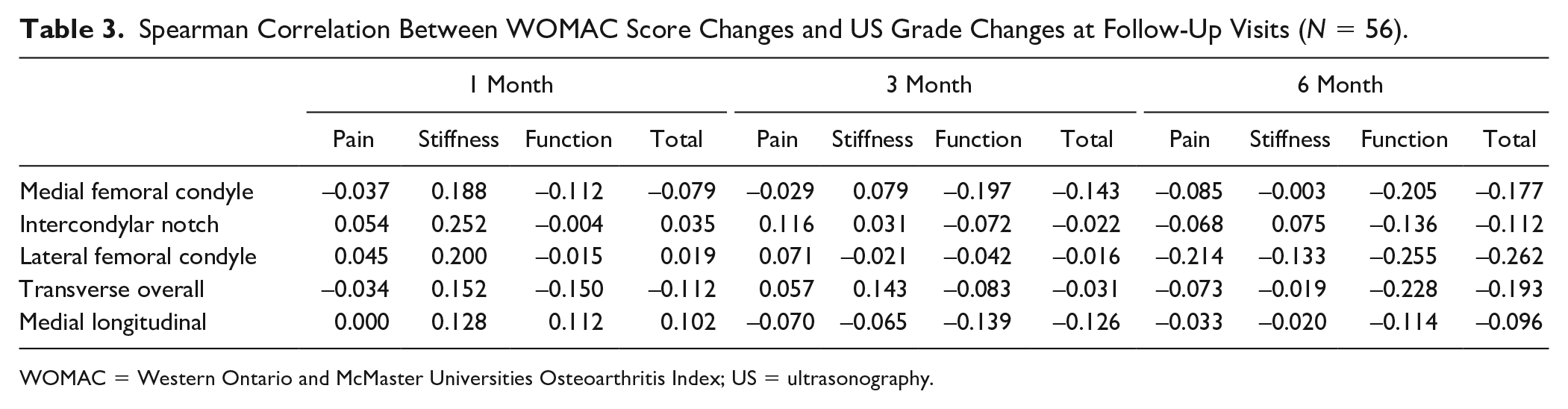
WOMAC = Western Ontario and McMaster Universities Osteoarthritis Index; US = ultrasonography.
Discussion
HA has been proposed as standard intra-articular treatment for the management of knee OA in patients who fail to respond to nonpharmacological therapy, NSAIDs, or analgesics, although few studies have used objective measurement to evaluate the efficacy. This is the first study to investigate the efficacy of HA on cartilage on structural change of cartilage by using US semiquantitative grading. We found significant grade changes of cartilage at follow-up after intra-articular injection of HYAJOINT Plus in patients with knee OA. There was also improvement in WOMAC scores at 6-month follow-up.
HA is a nonprotein, nonsulfated glycosaminoglycan (GAG). It is a major component of extracellular matrix and naturally exists in articular cartilage and synovial fluid. It consists of repeating b-1,4-
In OA cases, the concentration and molecular weight in synovial fluid both decrease. HA was proposed to treat OA almost 20 years previously and it was found that the molecular weight and source of HA products were related to efficacy and safety. High-molecular-weight products with molecular weight larger than 3000 kDa provided more favorable efficacy than those with molecular weight less than 3000 kDa. HA derived from biological fermentation, rather than avian origin, induced less flare-up response.31,32
HYAJOINT Plus, synthesized by a novel cross-linking process (i.e., 1,4-butanediol diglycidyl ether [BDDE]), has an anti-degraded feature. It is produced by microbial fermentation. With single intra-articular injection, HYAJOINT Plus was found to be beneficial in symptom evaluation, 33 functional testing, and US evaluation. 34 In this study, we found the effect not only on symptoms but on cartilage itself as shown by US evaluation. Our results showed significant improvement of cartilage grade after 3-month follow-up, although symptom improvement was noted as early as at 1-month follow-up in pain. These results are similar to those of the studies by Sun et al. 33 and Tuan et al. 34
US has provided a noninvasive tool to evaluate the status of articular cartilage band since 1984. 35 The characteristics of margin, clarity, and thickness change of cartilage band have been evaluated and a semiquantitative grading system including all the above characteristics was proposed to comprehensively evaluate the cartilage while also being well validated.23,36 Tuan et al. followed up patients after HYAJOINT Plus intervention by US evaluation; however, they only evaluated the thickness of the quadriceps and femoral condyle. In this study, we further evaluated cartilage status comprehensively by using a grading system and consequently showed significant decrease in grade of cartilage.
In conclusion, significant improvement on cartilage after intra-articular injection with high-molecular weight, biological fermentation–derived HYAJOINT Plus was found at 6 months. Further study with longer follow-up time might be necessary to investigate the duration of the effect. Semiquantitative grading system by US is a promising tool to identify the efficacy on cartilage band after interventions.
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to thank the Ministry of Science and Technology for both research and financial support (MOST 106-2314-B-037-089).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
KMUHIRB-F(II)-20170062
