Abstract
Objective
Reliable outcome measures are essential to predict the success of cartilage repair techniques. Histology is probably the gold standard, but magnetic resonance imaging (MRI) has the potential to decrease the need for invasive histological biopsies. The 3D magnetic resonance observation of cartilage repair tissue (MOCART) score is a reliable yet elaborate tool. Moreover, literature is controversial concerning the correlation of histology and MRI.
Design
To test the applicability of the International Cartilage Regeneration and Joint Preservation Society (ICRS) II and MOCART 3D score for the evaluation of aged osteochondral regenerates in a large animal model, and to identify correlating histological and MRI parameters. Osteochondral defects in medial femoral condyles of n = 12 adult sheep were reconstructed with biodegradable bilayer implants. About 19.5 months postoperation, n = 10 joints were analyzed with MRI (3D MOCART score). Histological samples were analyzed using the ICRS II score; both pre- and post-training. The intraclass correlation coefficient, the inter-rater reliability, and the 95% confidence interval were calculated. Matching histological and MRI parameters were tested for correlation.
Results
We found a statistically significant correlation of all histological parameters. MRI parameters reflecting “overall” assessments had very strong inter-rater correlations. Statistically significant strong correlations were found for the MRI parameters defect filling, cartilage interface, bone interface, and surface. For defect overall (MRI) and overall assessment (ICRS II), we found a significant yet mild correlation.
Conclusions
The ICRS II and the 3D MOCART score are applicable to aged osteochondral regenerates. Prior training on the scoring systems is essential. Select MRI and histological parameters correlate; however, the only statistically significant correlation was found for overall assessment.
Introduction
Rigorous clinical tests are essential to prove the efficacy and safety of all cartilage repair techniques. Clinical trial endpoints should include pain and functional outcomes, imaging, and biopsies.1-4
Histology is probably the most important structural outcome measure because it allows for assessment of the morphology and quality of the regenerated tissue that cannot be depicted by imaging. Although invasive and destructive, histology is still considered the gold standard. 5 However, simple histological grading systems often have a low inter- and intra-rater reliability. 4 To address this issue, numerous histological scores have been developed previously as summarized by Hoemann et al. 4 In 2003, the Histological Endpoint Committee of the International Cartilage Regeneration and Joint Preservation Society (ICRS) published a visual assessment histological scale termed ICRS I score followed by the ICRS II score in 2010.6,7 Unlike the ICRS I score or the modified O’Driscoll score (MODS), the ICRS II score with its 14 distinct parameters uses a continuous visual analog scale (VAS) which can improve reproducibility. 7 But the ICRS II score disregards the lateral integration, which is a crucial parameter of osteochondral regeneration.4,8-11 And a second look arthroscopy together with a repair tissue biopsy exclusively provides insights at a single point of location and time in potentially actively remodeling tissue as discussed by Hoemann et al. 4 and McCarthy et al. 12
In contrast, magnetic resonance imaging (MRI) is widely available, noninvasive and therefore being extensively used for the examination of cartilage defects and its regeneration.13-15 In general, the morphology, surface, and signal intensity of the neo-cartilage should be comparable to the adjacent healthy cartilage. 16 Associated scores with low intra- and inter-rater variability are helpful for systematic and reproducible MRI evaluation. As such, various elaborated score sheets have been published previously, that is, by Roberts et al., 17 Henderson et al., 18 or Mithoefer et al. 19 The Area Measurement And DEpth Underlying Structures (AMADEUS) grading system has been shown to evaluate and grade osteochondral lesions prior to cartilage surgery.20,21 The most commonly utilized MRI score for the assessment of cartilage regenerates is probably the magnetic resonance observation of cartilage repair tissue (MOCART) that was originally designed for the evaluation after autologous chondrocyte implantation (ACI).15,22 In 2009, Welsch et al. 23 introduced the 3D MOCART score with changes and additions based on isotropic voxel MRI innovations, scientific and clinical experiences, and the needs of daily patient care. According to Trattnig et al., 24 the 3D MOCART score provides a high correlation of the MRI parameters compared with the original MOCART score. The 11 variables can be assessed by 3D isotropic MRI and/or standard 2D MR sequences and seem to be applicable to any cartilage repair technique. MRI sequences of the daily radiological routine are available and applicable as demonstrated previously.24-26 Nonetheless, the comprehensive nature of the 3D MOCART score reflects a scientific rationale rather than practicality in daily clinical routine.
However, the accuracy and value of MRI in assessing the clinical outcome remains unclear.12,15,16,22,27-29 This is also the case for clinical outcome versus histology.12,30-33 Moreover, literature on the correlation of MRI and histology is controversial,12,17,34,35 which is also due to inconsistently used clinical, histological, and MRI scores. 12 The ability to assess the outcome of a distinct cartilage repair technique in a nondestructive manner is nevertheless worth striving for.
Therefore, the objective of the current study was to test the general applicability of the histological ICRS II score and MOCART 3D score for the evaluation of aged (respectively, matured or degenerated) osteochondral regenerates and ex vivo specimens in a large animal model mean 19.5 months postoperation. Consequently, we intended to identify correlating histological and MRI parameters. To achieve this goal, the test performance before and after a training on additional data and a matched pair analysis of distinct histological criteria with radiological scores was performed.
Materials and Methods
Animal Model
We published details on the scaffold manufacturing, the animal model, and surgical procedures previously. 36 Briefly, skeletally mature sheep (n = 12, gender: female, age: 3-6 years, mean weight: 75.6 kg, range: 62-84 kg) underwent surgery under analgosedation (0.7 mg/kg ketamine, 0.5 mg/kg xylazine intramuscularly) and spinal anesthesia (1.6 mL carbostesin 0.5%). Animals received a single injection of 1 g ampicillin and 0.5 g sulbactam. Animals were positioned on their right side. After usual arthrotomy of the left knee, a sharpened, tailor-made cutter was used to punch osteochondral defects (width: 6 mm, length: 20 mm, maximum depth: 5 mm) on the weight-bearing area of the medial femoral condyles. The sample size corresponds to guidelines published previously. 37 Defects were randomly assigned either to a composite of granules of Orthoss soaked in 0.9% NaCl solution and a fitting Chondro-Gide membrane (n = 6; both Geistlich, Wollhusen/CH), or a semisynthetic tailor-made biomimetic, bilayer implant assembled of macroporous and nanofibrous poly-e-caprolactone (n = 6; PCL). After wound closure, sheep were housed in small kennels to restrict motion for at least 3 days followed by unrestricted movement. All operations were performed by the same surgeon during a time window of 4 weeks. Animals received postoperative analgesia and recovered well from surgery. After 19.5 months postoperation, animals were sacrificed. Two animals had to be euthanized at 13 months and at 16 months postoperation due to severe pneumonia (sheep #1 and #11). For these 2 animals only histologic assessment was available. After exarticulation, specimens were kept cold until immediate MRI evaluation.
Magnetic Resonance Imaging
MR experiments were conducted on a 3T MR scanner (Ingenia, v 3.2.3, Philips, Best/NL) using a dedicated microscopy coil. The 2 coil elements were positioned at the level of the collateral ligaments. Specimens were cold but not frozen and were positioned at the isocenter of the MR scanner. The sequence protocol consisted of a 2-dimensional fat saturated proton density turbo spin echo (TSE) acquisition in transversal and coronal plane, a 2D T1- and a 2D T2-weighted TSE sequence in coronal orientation and a 3-dimensional multiecho gradient echo sequence. Detailed sequence parameters have been summarized in Table 1 .
Scan Parameters of Applied MR Sequences.
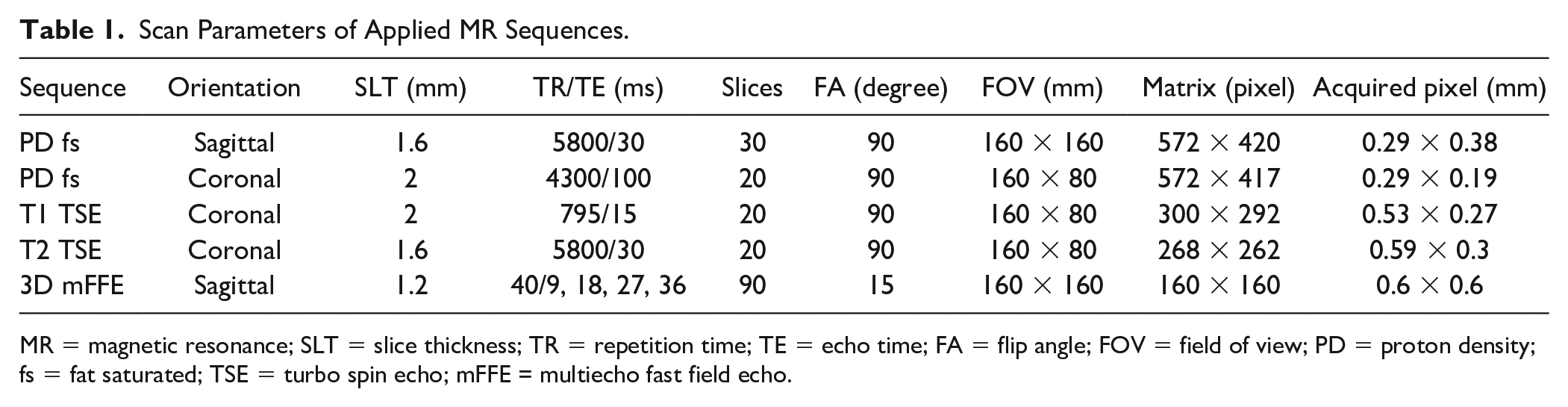
MR = magnetic resonance; SLT = slice thickness; TR = repetition time; TE = echo time; FA = flip angle; FOV = field of view; PD = proton density; fs = fat saturated; TSE = turbo spin echo; mFFE = multiecho fast field echo.
Histology
After MRI, entire joints were examined macroscopically while kept moist ( Fig. 1 ). Photographs were taken using a SONY SLT-A33 and a Tamron objective Di II 18-270 mm F/3.5-6.3. Hence, former defect sites, the articulating tibial part, the contralateral (untreated) medial femoral condyle and medial tibial plateau, and synovial tissue were collected and processed according to the ICRS recommendations. 4 Briefly, samples were fixed in buffered 4% paraformaldehyde protected from light at 5°C, and decalcified in pure ethylenediaminetetraacetic acid (EDTA) at 37°C for 13 weeks. Samples were embedded in paraffin, and serial coronal sections of 5 μm thickness were collected from 3 predetermined levels of the repair tissue (anterior, middle, posterior) to take into account obvious repair heterogeneity. Sections were stained using hematoxylin-eosin and toluidine blue standard protocols.
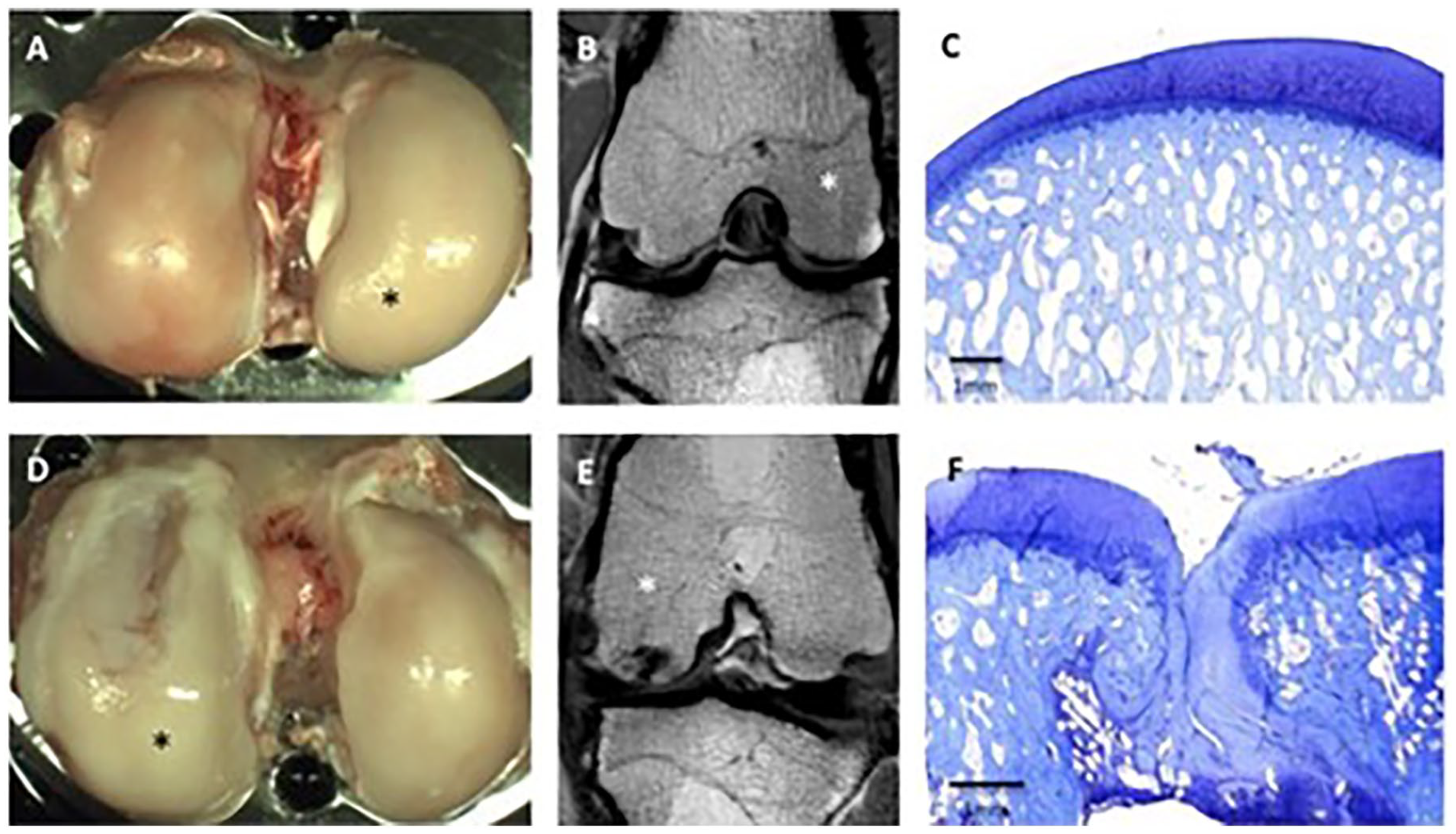
(
Histological Scoring
Three independent, blinded and experienced histologists (J.C.S., L.G., S.H. / D.V.) were asked to assess the histological slides using an Axioskop (AxioCam Mrc, KS300 3.0 software; Zeiss, Oberkochen, Germany) and using the ICRS II score. 7 After the first assessment, the readers were trained using the paper’s original pictorial material in order to depict the minimum and maximum characteristics of the different scoring parameters. 7 This training took 2 hours using slides other than derived from the study. After the preceding training, the readers assessed the specimens once again.
MRI Scoring
Three independent and experienced radiologists (F.J., I.W., and S.A.) were asked to evaluate the obtained MRI sequences in a blinded manner. The readers used a scoring sheet that comprised all MRI parameters of the 3D MOCART score. 23 An additional MRI sub-score was outlined including the parameters (1) inflammation (edema, soft tissue swelling, effusion), (2) surface/superficial assessment, (3) mid/deep zone assessment, and (4) overall assessment. These latter parameters are not included in the original 3D MOCART score and were derived from the ICRS II score using the VAS (0 worst-100 best) for better comparability. After the first assessment, the readers were trained 2 hours using representative but independent MRI images in order to depict the minimum and maximum characteristics of the different scoring parameters of the 3D MOCART. After the preceding training, the readers assessed the original MRI sequences once again.
Correlation of MRI and Histology
For the correlation of distinct histological and MRI parameters matched pairs, for example, ICRS II: surface/superficial assessment and MOCART: defect fill, or ICRS II: basal integration and MOCART: bone interface were paired to avoid accidental correlation. Also partially fitting pairs were paired, for example, ICRS II: mid/deep zone assessment and MOCART: surface or ICRS II: inflammation and MOCART: effusion. As for nonspecific parameters such as MOCART: signal intensity, those parameters were paired with related parameters, that is, ICRS II: vascularization.
Statistical Analysis
Data of overall n = 12 individuals were collected using Microsoft Excel. Data of sheep #1 and #11 were excluded in order to eliminate potential bias resulting from shortened follow-up (“na.omit” analysis). Arithmetic mean and standard deviation (SD) were calculated. The Shapiro-Wilk test was conducted for analysis of Gaussian distribution. The t test was used to determine statistical differences between independent groups. In cases of uneven distributions, the Mann-Whitney U test was run. The Shrout and Fleiss’ 38 intraclass correlation coefficient (ICC 3,1: 2-way mixed, single measures, consistency; ICC 3, k: 2-way mixed, average measures, consistency), the inter-rater (3 blinded and independent readers) reliability, and the 95% confidence interval were calculated with the correlation being weak (>0-0.49), mild (0.5-0.74), strong (0.75-0.89), and very strong (0.9-1.0; ≤0 no correlation). Level of significance was set at P < 0.05. Results were presented by scatterplots including regression lines. We obtained independent statistical consulting by Evidat (Lübeck/DE) using R3.4.3 software. Raw data will be available upon request.
Ethical Approval
The institutional review board provided approval. All experiments were approved by the German Ministerium für Landwirtschaft, Umwelt und ländliche Räume, V312-72241.122-15, 109-11/10)).
Results
Morphology
As published earlier, 36 histology revealed a significantly (P < 0.02) higher overall ICRS II score for the biologic implant compared with the synthetic implant. However, repeated-measures analysis of variance revealed no significant difference between different predetermined regions of the repair tissue (anterior, middle, posterior) or between the different treatment groups. The former defect areas were incompletely filled with fibrocartilaginous repair tissue that had a partially fragmented surface structure. Histologically, the repair tissue was well integrated, and remnants of the biodegradable implants could not be identified at all. The histological appearance was heterogeneous with fibrous or partly hyaline-like morphology. All these findings were variable and independent from implant design.
Pre-Training Histological Scoring
The pre-training histological scoring of the repair tissue revealed a strong inter-rater reliability for the histological parameters surface architecture (ICC: 0.502-0.806) and formation of a tidemark (ICC: 0.446-0.647) only. When looking at the parameters subchondral bone abnormalities/marrow fibrosis (ICC: 0.305-0.571), inflammation (ICC: 0.335-0.371), surface/superficial assessment (ICC: 0.483-0.652), mid/deep zone assessment (ICC: 0.486-0.671), and overall assessment (ICC: 0.432-0.625), we found a weak to moderate inter-rater reliability for only 2 paired raters each. Overall, we did not find a statistically significant inter-rater reliability for the majority of the histological parameters. The pre-training histological scoring of the healthy control specimens predominantly revealed a low inter-rater reliability. For only 1 pair of raters we found a strong inter-rater reliability for the parameter chondrocyte clustering (ICC: 0.759, CI: 0.2944, 0.9302). For 2 pairs of raters, we found a medium inter-rater reliability for the parameter matrix staining (ICC: 0.711, CI: 0.2584, 0.9116) and for 1 pair of raters for the parameter subchondral abnormalities/marrow fibrosis (ICC: 0.520, CI: −0.1186, 0.846). The initial high inter-rater variability led ultimately to a specific training of the readers.
Post-Training Histological Scoring
The post-training assessment of the repair tissue revealed a statistically significant correlation (P < 0.05) of all histological parameters of the ICRS II score when looking at the inter-rater reliability (ICC: −0.0009-0.9946). This was independent from pairs of raters. For select parameters and pairs of raters, we found a very strong inter-rater reliability, for example, for the parameter abnormal calcification/ossification (DV vs. LG: ICC 0.921, CI: 0.8431, 0.9607; LG vs. JSC: ICC: 0.958, CI: 0.916, 0.980) and for the parameter matrix staining (LG vs. JCS: ICC 0.979, CI: 0.957, 0.989; Fig. 2 ). In contrast, the post-training assessment of the healthy controls revealed fewer statistically significant correlations. We found correlation between 2 pairs of raters for the parameter matrix staining (LG vs. JCS: ICC 0.797, CI: 0.4202, 0.9404; LG vs. DV: ICC 0.632, CI: 0.0676, 0.887) and the parameter subchondral abnormalities/marrow fibrosis (LG vs. JCS: ICC 0.789, CI: 0.0813, 0.9488; LG vs. DV: ICC 0.774, CI: 0.1398; 0.9428). One correlating pair of raters each was identified for the parameter cell morphology (JCS vs. DV: ICC 0.580, CI: 0.0475, 0.8637), and chondrocyte clustering (JCS vs. DV: ICC 0.549, CI −0.0194, 0.8537).
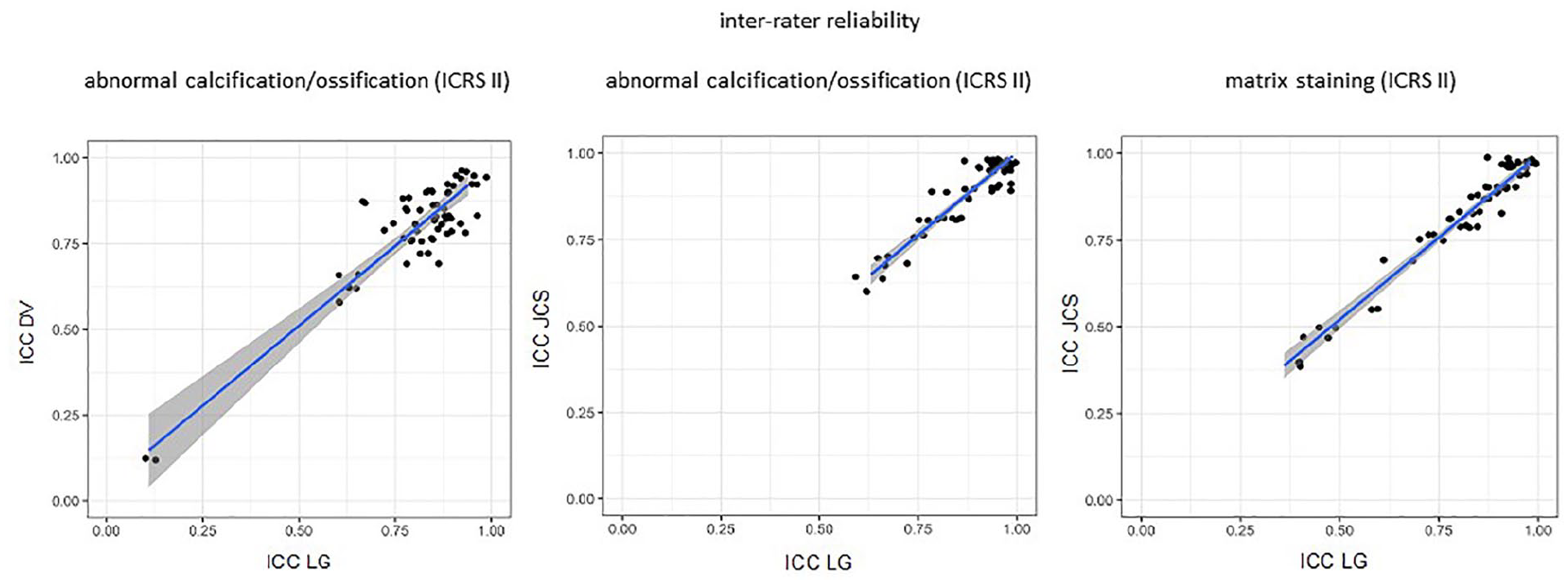
Scatterplots demonstrate a statistically significant strong inter-rater reliability (ICC) of the histological parameters abnormal calcification/ossification and matrix staining post-training. ICRS = International Cartilage Regeneration and Joint Preservation Society; ICC = intraclass correlation coefficient.
Pre-Training MRI Scoring
A significant, strong inter-rater correlation was only found for the parameter cartilage interface (ICC: 0.766, CI: 0.284, 0.937), and for the parameter bone interface (ICC: 0.8163, CI: 0.447, 0.9503). This applied for 1 pair of raters each. For all remaining MRI parameters, a weak or mild inter-rater correlation was observed inconsistently. A mild inter-rater correlation was also found when looking at the 4 parameters of the sub-score (ICC: 0.5025-0.6505). This applied for 2 pairs of raters each or less and was true for the intra-rater correlation also.
Post-Training MRI Scoring
The post-training MRI scoring revealed a strong to very strong inter-rater correlation (ICC: 0.799-0.997) of the total MOCART 3D score. In contrast to the pre-training scoring, we found for all pairs of raters a statistically significant strong to very strong correlation for the parameters defect filling (ICC: 0.741-0.971), cartilage interface (ICC: 0.688-0.951), bone interface (ICC: 0.8-1.0), and surface (ICC 0.87-1.0). There was also strong correlation for the parameter chondral osteophytes (ICC: 1.0, CI: 1.0, 1.0). For the parameter structure, we found a strong inter-rater correlation for 3 pairings (ICC: 0.8163-1.0, CI: 0.4468, 1.0). The parameters subchondral lamina, bone marrow edema, subchondral bone, and effusion had a statistically significant mild to very strong inter-rater correlation (ICC: 0.571-1.0, CI: −0.1275, 1.0), yet not for all pairs of raters. When looking at the sub-score, we found a weak to mild inter-rater correlation for the parameter inflammation for 2 pairs of raters (ICC: 0.479-0.5065, IC: -0.0767, 0.8439). The additional parameters cartilage surface overall, middle of the defect overall, and defect overall had a strong to very strong inter-rater correlation for all pairs of raters (ICC: 0.845-0.962, 0.837-0.953, and 0.78-0.93, respectively). As for the healthy (untreated) controls, we found an ICC of 1.0. This was independent from raters and/or preceding training.
Correlation of Histology and MRI
According to the study’s design, matching MRI and histological parameters were paired and correlated in order to avoid accidental correlation ( Table 2 ). For the parameters defect overall (MRI sub-score) and overall assessment (ICRS II), we found a significant yet mild correlation (ICC: 0.7302, CI: −0.1725, 0.9342; Fig. 3 ). Weak to mild correlations could be found for the remaining paired parameters (ICC: 0.0236-0.6875, IC: −1,6409, 0.9243), yet this was not statistically significant.
ICC of Paired MRI and Histological Parameters.
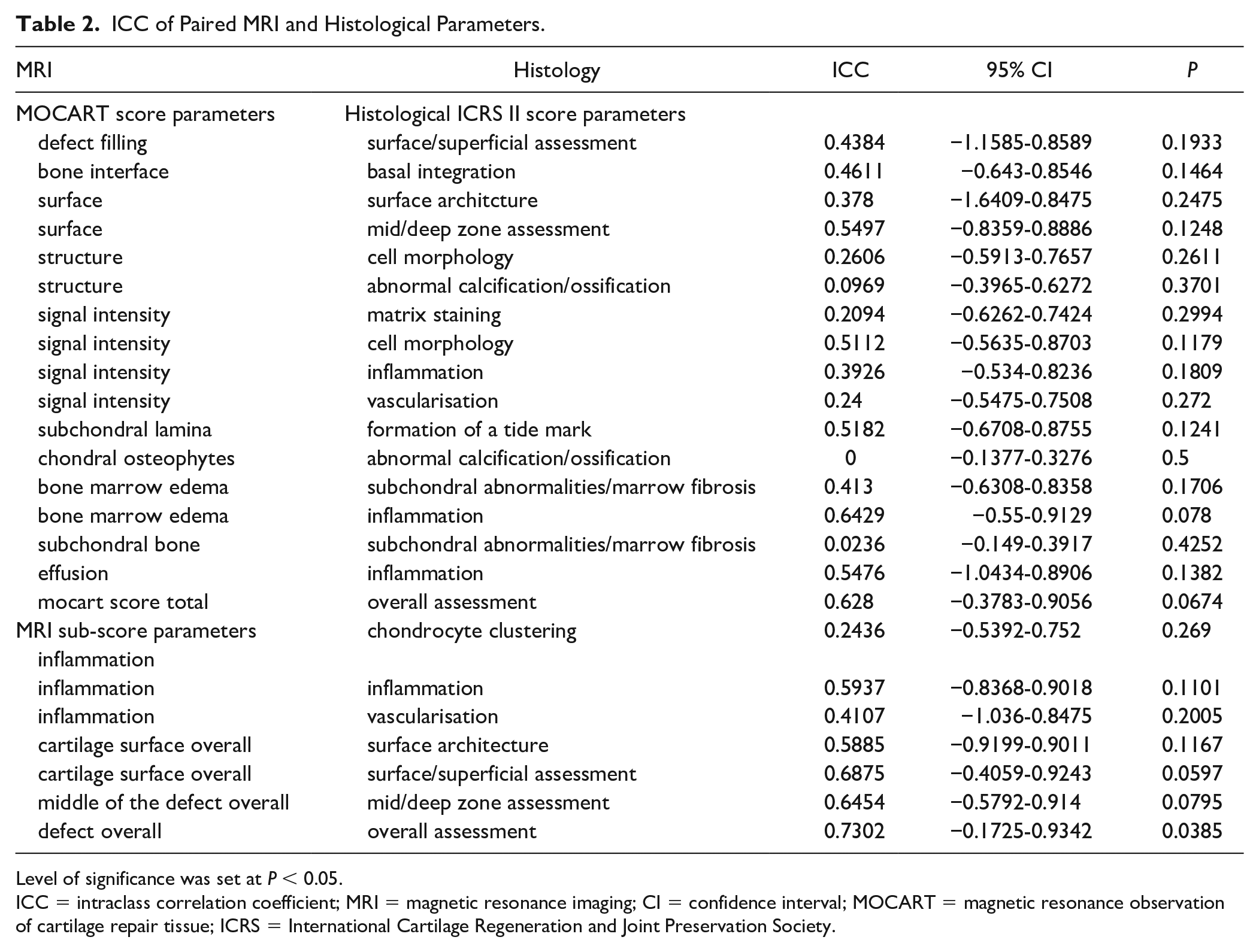
Level of significance was set at P < 0.05.
ICC = intraclass correlation coefficient; MRI = magnetic resonance imaging; CI = confidence interval; MOCART = magnetic resonance observation of cartilage repair tissue; ICRS = International Cartilage Regeneration and Joint Preservation Society.
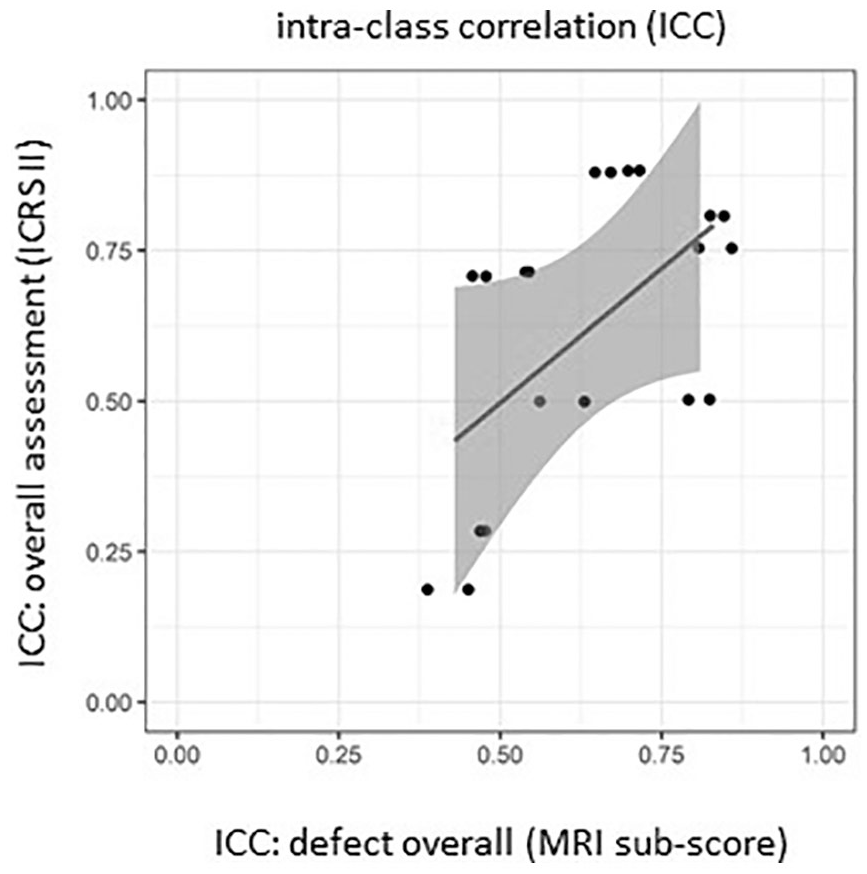
Scatterplot including regression line of the intra-class correlation of the parameters defect overall (MRI sub-score) and overall assessment (ICRS II) demonstrates a statistically significant (P = 0.0385) yet mild correlation. MRI = magnetic resonance imaging; ICRS = International Cartilage Regeneration and Joint Preservation Society.
Discussion
The first objective was to test the general applicability of the histological ICRS II score and 3D MOCART score for the evaluation of aged osteochondral regenerates and ex vivo specimens in a large animal model. In order to achieve a collective as homogenous as possible, data of the 2 animals with shortened follow-up were excluded. Our results revealed a strong inter-rater correlation of all tested histological parameters. This is consistent with the first description of the ICRS II score. 7 When looking at the investigated MRI parameters, we found strong correlations of parameters that reflect global aspects such as total/overall assessment, the interfaces, the surface and defect filling rather than subordinate specifics. In contrast, Trattnig et al. 24 demonstrated a high correlation of the 3D MOCART parameters, for example, compared with the original MOCART score. This discrepancy might be due to the fact that the 3D MOCART score was originally designed for the evaluation of humans after ACI and therefore cartilage repair tissue.15,22 Although the 3D MOCART was developed for different scientific issues, however, McCarthy et al. 12 found results consistent with correlation to clinical outcome. Taking this in account, we also conclude both the ICRS II and 3D MOCART scores to be suitable to assess cartilage regenerates in an animal model. This is consistent with other studies. Only the anatomical characteristics related to the chosen animal model (sheep) such as variable thickness of the neo-cartilage or the rigid subchondral bone are naturally waived by the 3D MOCART score.39-41
Although strictly adhering to the guidelines and recommendations for endpoints for cartilage repair studies,4,24,25 our results suggest, however, that prior training on the scoring systems is inevitable to reduce the intra- (data not shown) and inter-rater variability, which might be affected by the use of 2 different implant designs also. However, subgroup histological scoring did not show significant differences between the treatment groups. As for histology, we used the score’s original pictorial material in order to depict the minimum and maximum characteristics of the different scoring parameters, which ultimately led to a high correlation of all histological parameters. The authors of the ICRS II score noted this limitation and also the need for standardization while scoring. 7 Numerous groups of authors mention the need for training, yet there has been no detailed discussion of this issue. 42 It seems that the available scores permit a considerable range for interpretation leading to error in perception eventually.7,10,11 For example, we found a high variability of the histological parameter matrix staining, although Mainil-Varlet et al. 7 described this specific parameter to be very robust. We confirmed robustness only for the parameter overall assessment. Conceivably, a highly detailed categorization of distinct parameters is even unfavorable.7,42 For the sake of improvement, and as suggested by the authors,7,43 histological image databases are reasonable, and comparable databases for MRI images desirable. Moreover, the implementation of morphometric objective methods could help to systemize the analysis and to minimize bias. In our study, the pictorial translation of the tabular MRI score led to a significant increase in correlation of the majority of the MRI parameters. Nonetheless, our MRI results post-training were still not as consistent as for histology, for example, for the parameters bone marrow edema and inflammation maybe because minimum and maximum characteristics of these parameters are hard to depict. In contrast, Goebel et al. 44 found a strong correlation of the parameter bone marrow edema using a binary grading (yes/no). Welsch et al. 23 found a lower correlation for this parameter also. They explained this observation by using a 3-D-True-FISP sequence. 45 A similar sequence has been used in the present study (3-D-m-FFE sequence). However, a profound discussion of distinct MRI sequences particularly adapted to cartilage repair tissue is not the scope of the present study and can therefore be reviewed elsewhere.23,46
The second objective was to identify correlating histological and MRI parameters. We found a significant yet mild correlation for the parameters defect overall (MRI sub-score) and overall assessment (ICRS II). The summarized yield of the ICRS II score and the 3D MOCART score correlated as well as the remaining paired parameters. In contrast, a study published by the Oswestry group showed no significant correlation of the total MRI (MOCART and WORMS) and histological score (OsScore and ICRS II). 12 Interestingly, a negative correlation was described between the parameters subchondral bone abnormalities and formation of a tidemark (histology), and defect fill and signal intensity (MRI) as well as for the presence of osteophytes and tidemark formation. The results furthermore suggest the presence of adhesions diagnosed by MRI to positively correlate with better tidemark formation, and better tissue and cell morphology. Adhesions correlated with a higher ICRS II total yield but worse surface architecture and chondrocyte clustering. Neither the parameter structure nor the parameter signal intensity (MRI) correlated with tissue morphology (histology). The authors moreover emphasized that the presence of hyaline cartilage to any extent in the repair tissue did not correlate with a better MOCART score. Vascularization and abnormal calcifications naturally influenced the histological score yield, yet the presence of these parameters did not scale down the MOCART score yield. In contrast, Goebel et al. 44 demonstrated a correlation of the parameter defect fill (MRI) and total yield (ICRS II). It is worth mentioning, however, that the Oswestry group analyzed humans histologically 16 months after ACI, and per MRI (1.5-T) 11 or 39 months after ACI. Goebel et al. scanned sheep condyles using a 9.4-T high-field MRI. When using the Sellers score instead of the ICRS II score, the parameter subchondral bone correlated; however, a 9.4 Tesla scanner was used in this particular study. Roberts et al. 17 published a clinical follow-up after ACI earlier. The authors showed a correlation of the total yield of the Oswestry histological score and an individual MRI score. Correlation of single parameters could not be detected. Streitparth et al.39,47 described correlation of the parameters defect fill, integration, surface, signal intensity, and subchondral bone (ovine condyles, 6 months after surgery). MRI (1.5-T), however, was conducted in vivo and Gd-DTPA was administered intravenously. The use of contrast agents and quantitative MRI sequences seem nevertheless beneficial to monitor cartilage regenerates and changes within the joint cavity.46,48
The whole variety of different study designs assessing either humans or animals, using 1.5-T MRI or even experimental high-field MRI, and the highly differing endpoints is impeding the comparability. And it remains unanswered, however, which parameters are indicative for clinical success. Neither histology nor MRI are capable of depicting biomechanical properties and therefore function of the repair tissue. Supposedly, biomechanical assessment tools such as dynamic imaging or nondestructive electromechanical evaluation might be favorable to describe the properties of the repair tissue and consequently to predict the eventual clinical outcome. 36
Conclusions
Both the 3D MOCART score and the ICRS II score are suitable to assess aged osteochondral regenerates in an animal model. Prior training on scoring with both systems is essential to achieve a high inter-rater reliability. Our results suggest correlation of select MRI and histological parameters; however, the only statistically significant correlation was found for the parameter overall assessment. From our study design, the correlation of potentially matching parameters seems to depend on source of repair tissue, applied scores and sub-scores, and applied MRI sequences and scanners. A very detailed breakdown of highly specific grading parameters may even be counter-productive. It is our understanding that MRI and histology, and biomechanical assessment tools remain complementary. However, the present study was limited by small numbers, a wide range of disease severity, and degenerative changes of the aged repair tissue.
Footnotes
Acknowledgments and Funding
The authors are obliged to C. Jürgens and J. Barkhausen for giving us the opportunity to conduct the study at their facilities. The authors thank M. Klinger, R. Duschka, S. Anton, S. Hoge, D. Vogt, I. Adametz, and F. Jakob for technical assistance. Geistlich Pharma AG donated Chondro-Gide und Orthoss implants. All experiments involving animals were approved by the German Ministerium für Landwirtschaft, Umwelt und ländliche Räume (V312-72241.122-15 (109-11/10)).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The institutional review board provided approval. All experiments were approved by the German Ministerium für Landwirtschaft, Umwelt und ländliche Räume (V312-72241.122-15 (109-11/10)).
