Abstract
Objective
To investigate the anti-inflammatory effects and mechanisms of pulsed electromagnetic field (PEMF) in the treatment of osteoarthritis (OA) in the destabilization of the medial meniscus (DMM) mice.
Design
Ten-week-old male wild-type (WT), interleukin (IL)-6−/− and tumor necrosis factor (TNF)-α−/− mice undergoing DMM surgery were randomly divided into 2 groups (n = 10 each): mice with PEMF exposure and mice with sham PEMF exposure. PEMF (75 Hz, 3.8 mT, 1 h/day) or sham PEMF was applied for 4 weeks. Pain behavior of mice, histological assessment of cartilage and synovium, micro-CT (computed tomography) analysis of bone, real-time polymerase chain reaction, and immunohistochemical staining of cartilage were performed.
Results
After DMM surgery, PEMF had a beneficial effect on pain, cartilage degeneration, synovitis, and trabecular bone microarchitecture in WT mice; these protective effects were reduced in IL-6−/− and TNF-α−/− mice. In addition, PEMF downregulated IL-6 and TNF-α expression in cartilage. PEMF also ameliorated cartilage matrix, chondrocyte apoptosis, and autophagy, while deletion of IL-6 or TNF-α suppressed the effects.
Conclusions
PEMF attenuates structural and functional progression of OA through inhibition of TNF-α and IL-6 signaling. The protective effects of PEMF on chondrocyte apoptosis and autophagy are regulated by TNF-α and IL-6 signaling.
Introduction
Osteoarthritis (OA) is a multifaceted syndrome in which low-grade chronic inflammation has a central role.1,2 Proinflammatory cytokines, such as TNF-α (tumor necrosis factor-α) and interleukin-6 (IL-6), are involved in the pathogenesis of OA.3-5 A systematic review of preclinical studies suggests that TNF-α inhibition promotes cartilage repair in both in vivo and in vitro OA models. 6 There is also evidence to suggest that IL-6 inhibition 7 or knockout (KO) 8 alleviates knee OA cartilage destruction in mice. TNF-α and IL-6 have been shown to induce chondrocyte apoptosis in OA7,9 and have become a prominent treatment target for OA.
Pulsed electromagnetic field (PEMF), a noninvasive intervention, has been proposed as a potential treatment for OA. Data from a recent meta-analysis study that have assessed effects of PEMF on symptoms and quality of life suggest PEMF provides clinically significant pain relief in participants with OA. 10 Furthermore, evidence suggests that PEMF could alleviate cartilage degeneration and maintain subchondral trabecular bone microarchitecture.11-15 There was a therapeutic window of PEMF parameters; various combinations of intensity and frequency may produce different effects. Previous studies have shown that 75 Hz, 1.6 mT PEMF has a protective effect on joint cartilage and subchondral bone in knee OA guinea pigs12,14,15 and rats. 13 Furthermore, our previous studies demonstrated that 8 Hz, 3.8 mT PEMF could alleviate cartilage degeneration and subchondral bone microstructural damage. 16 In our pilot study, the findings showed that the effects of 75 Hz, 3.8 mT PEMF are better in knee OA mice compared with 8 Hz, 3.8 mT PEMF. A previous systematic review by Li et al. 17 showed that they excluded 9 randomized controlled trials with a shorter duration than 4 weeks since this time frame may be too short to assess harms and benefits based on biological plausibility. Subsequently, at least 4 weeks as the treatment duration should be chosen. Loeser et al. demonstrated that progressive histological changes of OA begin within 2 weeks after the destabilization of the medial meniscus (DMM) surgery; the early phases at 2 and 4 weeks after DMM surgery were identified with the mild cartilage lesions. 18 Our previous study showed that early PEMF treatment (begins at 4 weeks after DMM surgery) have better protective effects on cartilage degradation than delayed PEMF treatment. Time point of treatment initiation is crucial for treating OA. 13 Currently, there remains an immense need for safe and effective therapies to modify joint structure and delay OA progression, in addition to improve symptoms.
PEMF has been shown to increase extracellular matrix production19,20 and reduce the release of proinflammatory cytokines16,21 and chondrocyte apoptosis.16,22 The mechanism of PEMF in modulating chondrocyte death may be due to decreased production of proinflammatory mediators. Furthermore, chondrocyte death is a combination between apoptosis and autophagy during the pathogenesis of OA. 23 To our knowledge, there are no studies examining the role of proinflammatory cytokines in the process of PEMF modulating chondrocyte apoptosis and autophagy in OA.
In this study, we investigated the effects of PEMF on progression of OA both structurally and functionally by inhibiting TNF-α and IL-6 signaling in a murine destabilization-induced model. We also examined whether PEMF has protective effects on chondrocyte apoptosis and autophagy through inhibition of TNF-α and IL-6 signaling using TNF-α or IL-6 KO mice. For the purposes of this study, we selected the DMM model which has sufficient sensitivity to reflect human OA disease pathogenesis. 24
Methods
Animals and Experimental Design
This experimental protocol was approved by the Institutional Animal Care and Use Committee of West China Hospital, Sichuan University (IACUC permit number: 2018130A). Wild-type (WT) mice, IL-6 KO (IL-6−/−) mice, and TNF-α KO (TNF-α−/−) mice from the same litter in a C57BL/6J background were obtained from the Jackson laboratory (Bar Harbor, ME). All mice were housed in individually ventilated cages (5 mice/cage) under pathogen-free conditions in a temperature-controlled facility (22 ± 1 °C, 12-hour light/dark cycle) with ad libitum access to standard food and water. Mice were housed for 1 week for being habituated to the PEMF device. During habituation, mice were placed in the turned-off PEMF device for 1 hour over 3 consecutive days.
Sixty 10-week-old male WT, IL-6−/−, and TNF-α−/− mice in C57BL/6N background were randomly divided into 2 groups (n = 10 each): mice with PEMF exposure and mice with sham PEMF (vehicle) exposure. Random numbers were generated by Microsoft Excel. Mice were blinded to the treatment. Investigators who administered the treatment could not be blinded due to the difference between PEMF and vehicle. OA was induced by the DMM surgery on the right knee joints as described previously. 24 In brief, the right knee joint was exposed with a medial para-patellar ligament incision. Following fat pad blunt dissection, the medial meniscus (MM) and medial meniscotibial ligament (MMTL) were identified. MMTL was transected with micro-surgical scissors; DMM is complete, and medial displacement of the MM occurs. The contralateral left knee was sham operated receiving the exact same DMM surgery but left MMTL intact. Mice were excluded if there were no statistically significant differences observed in 50% paw withdrawal threshold (PWT) between baseline and 1 week after surgery. No exclusions were found in this study. Four weeks after surgery, mice were exposed to PEMF or sham PEMF for 4 weeks.
PEMF Treatment
The custom-designed PEMF exposure system comprises a waveform signal generator (DG1022U, RIGOL, Suzhou, China), an adjustable switching power supply (MS-605D, Maisheng, Dongguan, China), a power amplifier, and Helmholtz coils with a 3-coil array ( Fig. 1 ). These coils have an inner diameter of 53 cm and an outer diameter of 63 cm and were placed coaxially 22.1 cm apart from each other. The central coil and outside coil have 266 and 500 turns, respectively. The PEMF waveform used in the experiment (duty ratio, 50%; pulse width, 6.67 ms; pulse wait, 6.67 ms) repeated at 75 Hz. The peak magnetic field intensity was 3.8 mT measured using a hand-held Gaussmeter (HT201, Hengtong, Shanghai, China). The PEMF exposure was applied for 1 h/day for 4 weeks. All mice were not anesthetized when exposed to PEMF. Exposure chamber housed 5 free-roaming mice each experimental session. For the sham PEMF, mice were maintained in the same exposure area for the equal time but without turning on the PEMF system. Experiment table was randomly chosen daily, with each group treated at a different experiment table each day.
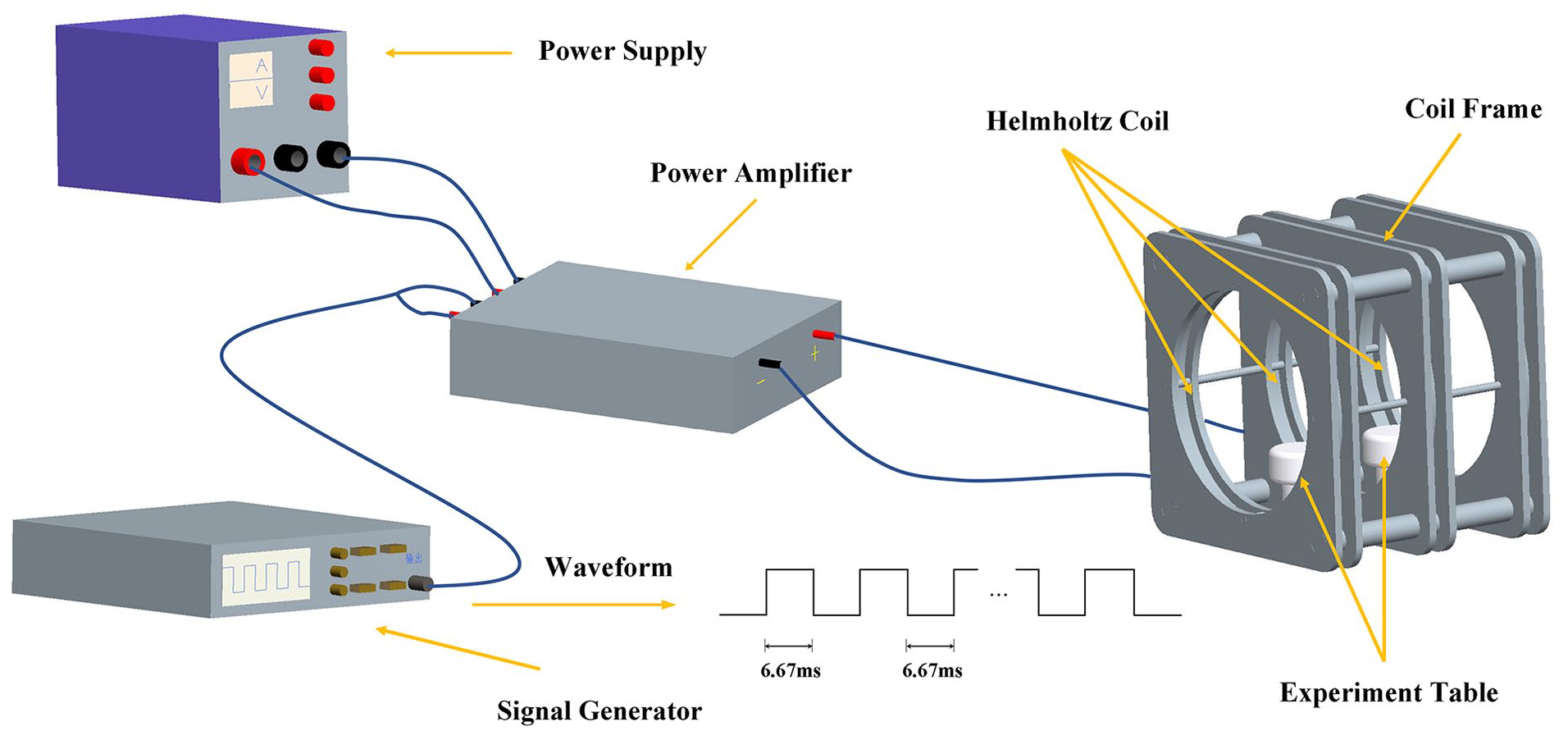
Pulsed electromagnetic field (PEMF) exposure device is composed of a power supply, a pulse signal generator, a signal amplification, and 3 Helmholtz coils. The pulse signal generator generated square wave (duty ratio, 50%; pulse width, 6.67 ms; pulse wait, 6.67 ms) with a repetitive frequency at 75 Hz. The PEMF intensity within the Helmholtz coils was 3.8 mT.
Behavioral Assessment of Pain
Pain was assessed using the Von Frey hair algesiometry (DanMic Global, San Jose, CA) with bending force (force range ≈0.07 g, 0.4 g, 0.6 g, 1 g, 2 g, 4 g, 6 g, 8 g, or 15 g). For quantifying pain behavior, hind paw withdrawal response to mechanical stimuli was assessed using the up-down method, which has been described previously.25,26 Fifty percent PWT was calculated according to the method described by Chaplan et al. 25 Measurements were made prior to DMM surgery or sham operation (week 0) and at regular intervals (week 1, 4, and 8) after DMM surgery.
Micro-Computed Tomography (CT) Analysis
After mice were sacrificed, the bilateral knee joints were harvested and fixed in 4% paraformaldehyde. Undecalcified specimens were scanned using a micro-CT system (Quantum GX, PerkinElmer, Waltham, MA) at 90 kVp and 88 µA. A hydroxyapatite phantom was used for calibration. A global threshold was used. Three-dimensional images were reconstructed to 15 µm voxel size on an Analyze 12.0 software. Regions of interest were manually selected for analysis: femoral condyle, tibial plateau, distal femur, and proximal tibia ( Fig. 3A ). Bone volume/total volume (BV/TV), bone surface/bone volume (BS/BV), trabecular thickness (Tb.Th), trabecular number (Tb.N), and trabecular separation (Tb.Sp) were calculated.
Histological Analysis
Knee joints were decalcified using 20% ethylenediaminetetraacetic acid for 7 days and then embedded in paraffin. Coronal sections (5 µm) were taken from each joint at 80 µm intervals. From each level, 3 sections were stained with Safranin O/Fast Green and toluidine blue for histologic scoring. Cartilage degeneration of femoral condyle and tibial plateau were scored according to the Osteoarthritis Research Society International (OARSI) scoring system. 27 Synovitis was measured by examining the enlargement of the synovial lining cell layer and density of the cells using a previously published scoring system.28,29 The maximum OARSI and synovitis scores were separately recorded.
Immunohistochemical (IHC) Staining
For IHC staining, knee joint sections were deparaffinized with xylene and rehydrated through ethanol series, followed by treatment with 3% H2O2 and citrate buffer. Sections were then blocked with 10% normal goat serum for 30 minutes and incubated overnight at 4 °C with primary antibody, including MMP-13 (1:200; ab39012, Abcam, Cambridge, MA), Aggrecan (Acan; 1:200; ab3778, Abcam), Bax (1:200; ab32503, Abcam), mTOR (1:200; ab2732, Abcam), Beclin-1 (1:500; ab62557, Abcam), and Cleaved Caspase-3 (1:200; Asp175, Cell Signaling Technology, Danvers, MA), followed by the biotinylated secondary antibody and DAB peroxidase substrate.
Real-Time Polymer Chain Reaction (RT-PCR) Analysis
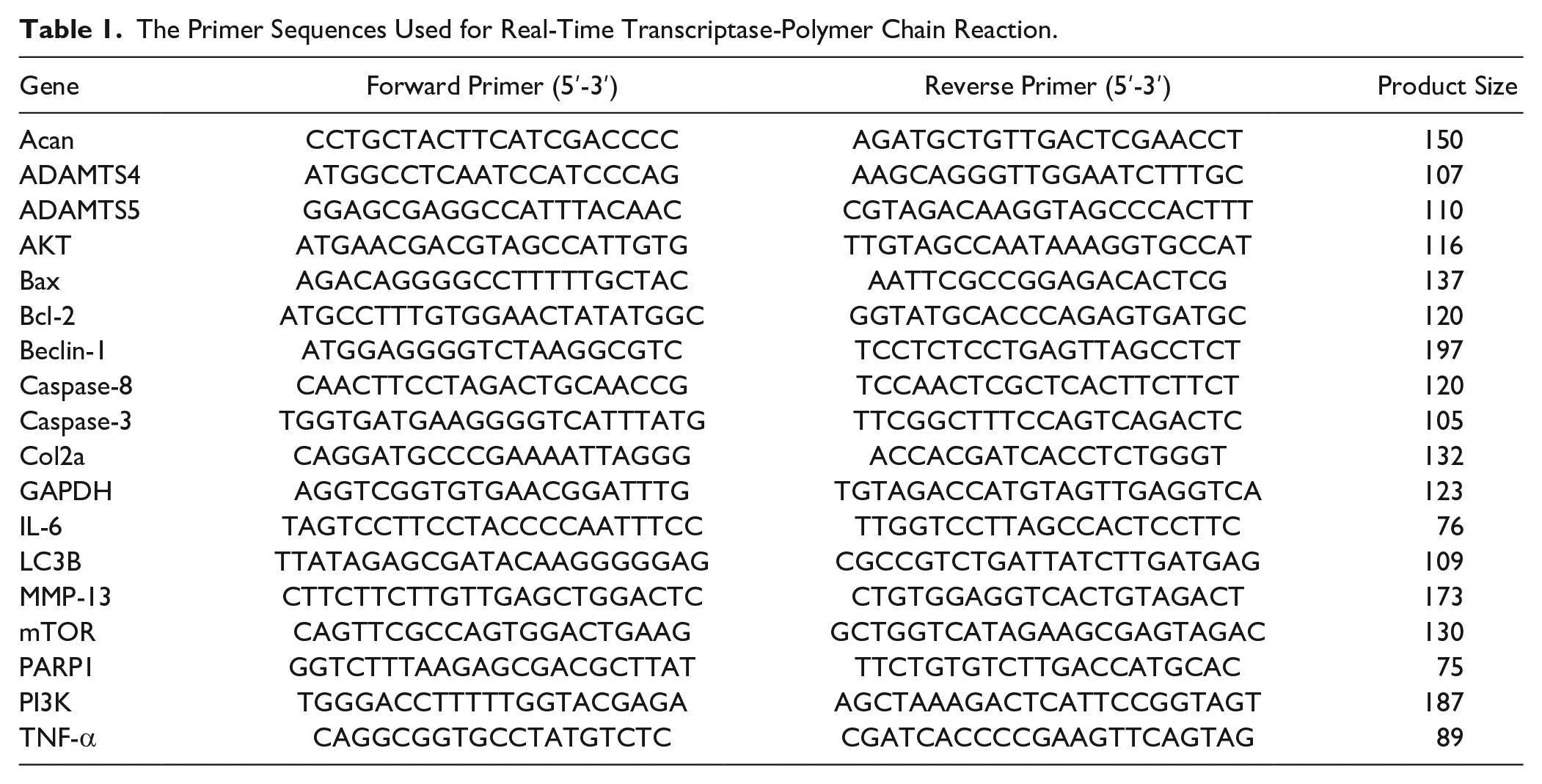
Cartilage samples from tibial plateau and femoral condyle were dissected with a scalpel under a microscope. Total RNA was isolated using Trizol reagents (Invitrogen Life Technologies, Carlsbad, CA) and subsequently reverse transcribed into cDNA using the PrimeScript RT reagent kit (Takara Bio, Japan). mRNA levels were detected using a CFX RT-PCR system (Bio-Rad, Hercules, CA) with the SYBR Premix Ex Taq II kit (Takara Bio, Japan). The primers were designed (using the Primer-BLAST tool available from the NCBI website) and synthesized by Sangton Biotech (Shanghai, China). The primers used in this study are shown in Table 1 . The mRNA levels of each gene were normalized to the GAPDH mRNA level. Relative mRNA levels were calculated using the 2−ΔΔCt method.
The Primer Sequences Used for Real-Time Transcriptase-Polymer Chain Reaction.
Statistical Analysis
G Power was used for sample size calculation. The following were chosen: α = 0.05 and β = 0.2 were chosen. The effects of PEMF on pain were evaluated in OA mice for the first time. Then, we acquired effect size according to our pilot data. In this study, data were presented as means and 95% confidence interval. Statistical analyses were performed using STATA (version 14, StataCorp, College Station, TX). To examine the differences between groups, we used one-way ANOVA followed by a Bonferroni multiple comparison test when variances were homogeneous, and Welch’s ANOVA followed by a Games-Howell post hoc test when variances were heterogeneous. Assumptions were checked using the Shapiro-Wilk test for normality and Bartlett’s test for homogeneity of variance. P values less than 0.05 were considered statistically significant.
Results
PEMF Attenuates OA Pain through Inhibition of TNF-α and IL-6 in the DMM Mice
The decreased 50% PWT was observed in WT mice at 1 week after DMM surgery and persisted through 8 weeks; this reduction was attenuated in the PEMF-treated WT mice at 8 weeks after DMM surgery ( Fig. 2A ). In contrast, PEMF had no significant effect on increasing 50% PWT in IL-6−/− and TNF-α−/− mice, though IL-6−/− and TNF-α−/− mice displayed the higher 50% PWT when compared with WT mice after DMM surgery ( Fig. 2B and C ).
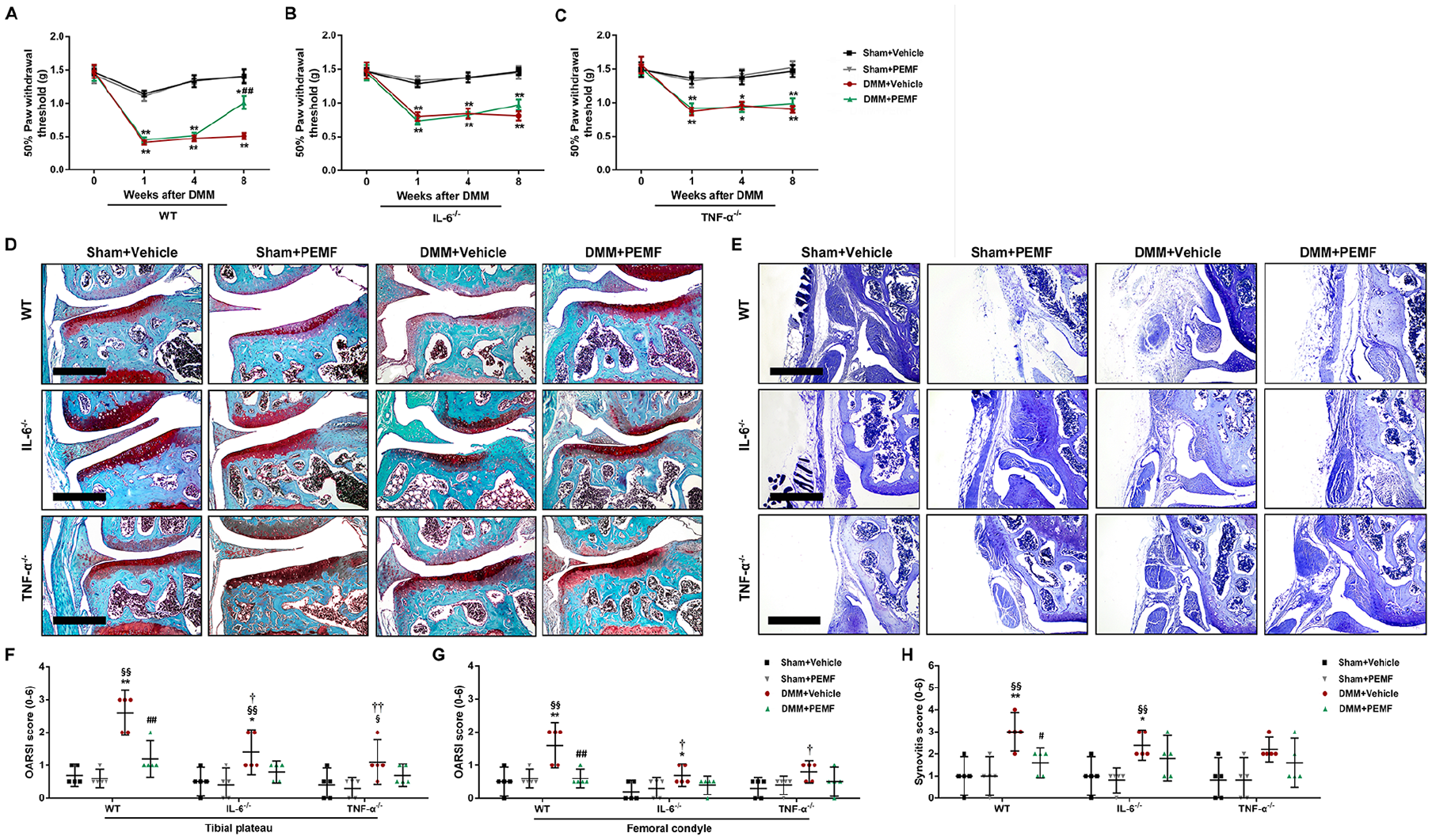
Pulsed electromagnetic field (PEMF) attenuates osteoarthritis through inhibition of TNF-α and IL-6 in the destabilization of the medial meniscus (DMM) mice. At 4 weeks after DMM surgery or sham operation, wild-type (WT) mice, IL-6 knockout (IL-6−/−) mice, and TNF-α knockout (TNF-α−/−) mice were treated with PEMF or vehicle (sham PEMF) for 4 weeks. (
PEMF Attenuates Cartilage Degradation through Inhibition of TNF-α and IL-6 in the DMM Mice
After DMM surgery, cartilage lesions and proteoglycan loss were observed in WT mice by Safranin O/Fast Green staining; IL-6−/− and TNF-α−/− mice displayed the milder cartilage degradation. PEMF, compared to vehicle, showed less severe cartilage lesions in WT mice after DMM surgery, which was in contrast to the findings from PEMF-treated IL-6−/− and TNF-α−/− mice ( Fig. 2D ). We also observed that the OARSI scores of the tibia and femur were significantly reduced in the PEMF-treated WT mice compared to vehicle after DMM surgery, while no statistically significant differences were observed between PEMF and vehicle groups in IL-6−/− and TNF-α−/− mice ( Fig. 2F and G ).
For mRNA levels, PEMF, compared to vehicle, decreased the expression of proinflammatory cytokines TNF-α and IL-6 ( Fig. 4A and B ) and catabolic factors MMP-13, Adamts4, and Adamts5 (Fig. 4C-E), and upregulated the expression of anabolic factor Acan ( Fig. 4F ) in articular cartilage in WT mice after DMM surgery. There were no statistically significant differences in the expression of MMP-13, Adamts4, Adamts5, Col2a, and Acan between PEMF and vehicle groups in IL-6−/− and TNF-α−/− mice after DMM surgery (Fig. 4C-G). For protein levels, IHC results consistently showed that the increased expression of MMP13 in articular cartilage after DMM surgery was reduced in the PEMF-treated WT mice, and the decreased expression of Acan was upregulated. The IL-6−/− and TNF-α−/− mice had significantly lower MMP-13 than the WT mice after DMM surgery, whereas similar staining was observed after PEMF treatment in WT, IL-6−/− and TNF-α−/− mice ( Fig. 5 and Suppl. Fig. S1).
PEMF Alleviates Synovitis through Inhibition of TNF-α and IL-6 in the DMM Mice
Synovitis was increased in WT mice after DMM surgery; the increased synovitis was attenuated in IL-6−/− and TNF-α−/− mice ( Fig. 2E ). For synovitis score, after DMM surgery, there was a statistically significant decrease in PEMF-treated WT mice when compared with vehicle-treated WT mice, while no statistically significant differences were observed between PEMF and vehicle groups in IL-6−/− and TNF-α−/− mice ( Fig. 2H ).
PEMF Maintains Trabecular Bone Microarchitecture through Inhibition of TNF-α and IL-6 in the DMM Mice
Micro-CT analysis of subchondral bone in tibial plateau showed that PEMF increased the integrity of subchondral bone in WT mice after DMM surgery, but not in IL-6−/− and TNF-α−/− mice ( Fig. 3B ). Furthermore, in 3D analysis of subchondral bone, BS/BV of tibial plateau was reduced, and Tb.Th of tibal plateau and BV/TV of femoral condyle were increased after PEMF treatment when compared with vehicle in WT mice after DMM surgery. There were, however, no statistically significant differences observed in BV/TV, BS/BV, Tb.Th, Tb.Sp, and Tb.N between PEMF and vehicle groups in IL-6−/− and TNF-α−/− mice after DMM surgery (Fig. 3C-L).
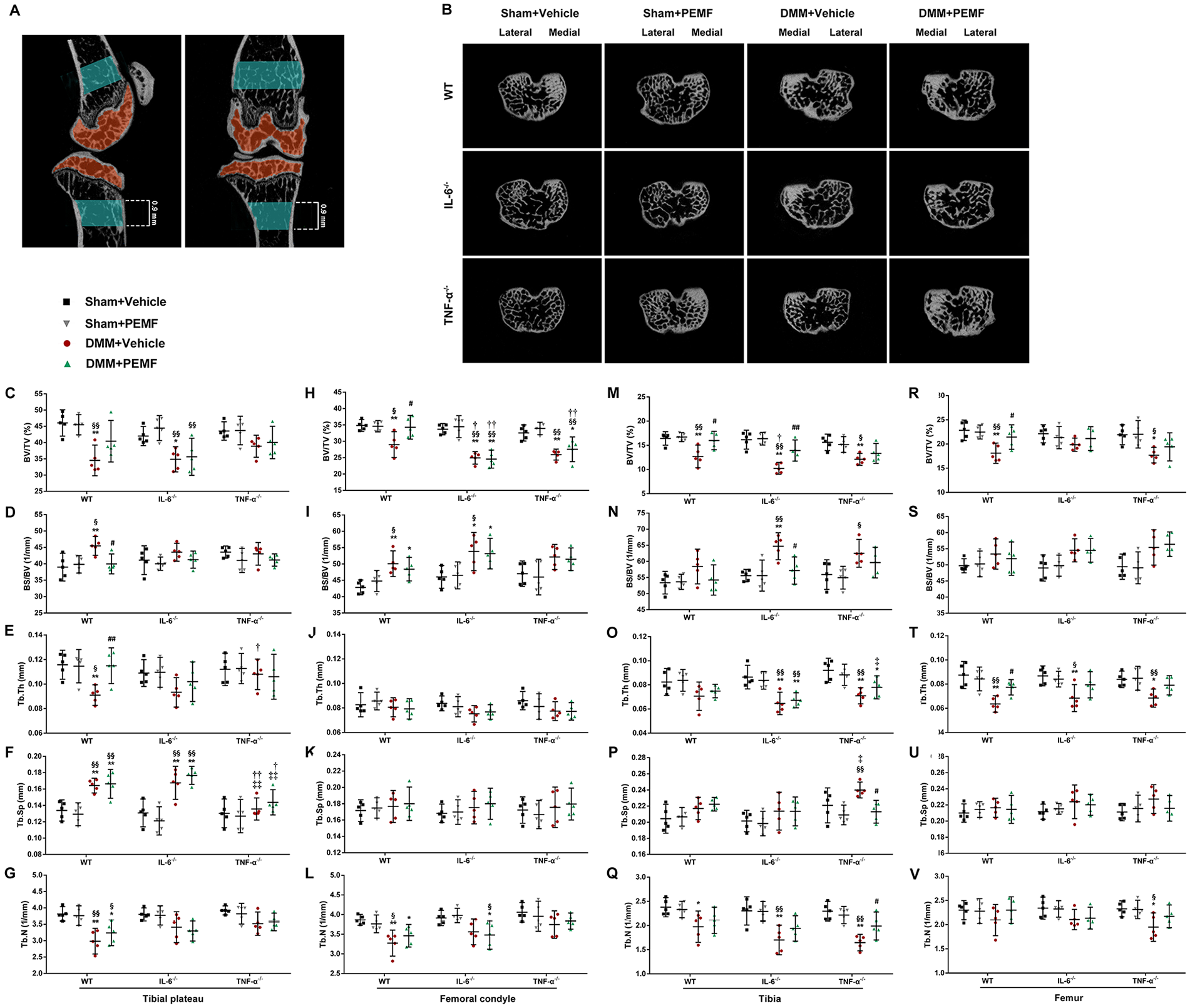
Pulsed electromagnetic field (PEMF) maintains trabecular bone microarchitecture through inhibition of TNF-α and IL-6 in the destabilization of the medial meniscus (DMM) mice. (
In 3D analysis of bone, the decreased BV/TV of tibia and femur and Tb.Th of femur were attenuated in the PEMF-treated WT mice after DMM surgery. There were no statistically significant differences in all parameters of tibia and femur between IL-6−/− and WT mice after PEMF treatment, though PEMF-treated IL-6−/− mice had significantly higher BV/TV and lower BS/BV in tibia than vehicle-treated IL-6−/− mice after DMM surgery. Similarly, PEMF had no significant effect on the tibial and femoral bone in TNF-α−/− mice (Fig. 3M-V).
PEMF Inhibits Chondrocyte Apoptosis via IL-6 and TNF-α In Vivo
The gene expression of apoptosis markers, including Bax, Caspase-8, Caspase-3, and PARP1, was significantly increased in articular cartilage after DMM surgery in WT mice, while the expression of Bcl-2 was reduced. We further observed that this effect was inhibited by PEMF in WT mice. Similarly, PEMF significantly reduced the expression of Bax, Caspase-8, and Caspase-3 in IL-6−/− mice after DMM surgery; this inhibition was, however, not observed in TNF-α−/− mice (Fig. 4H-L). Last, IHC results confirmed that the increased expression of Cleaved Caspase-3 in articular cartilage after DMM surgery was reduced in the PEMF-treated WT mice; in contrast, no significant differences were observed between PEMF and vehicle groups in IL-6−/− and TNF-α−/− mice after DMM surgery ( Fig. 5 and Suppl. Fig. S1).
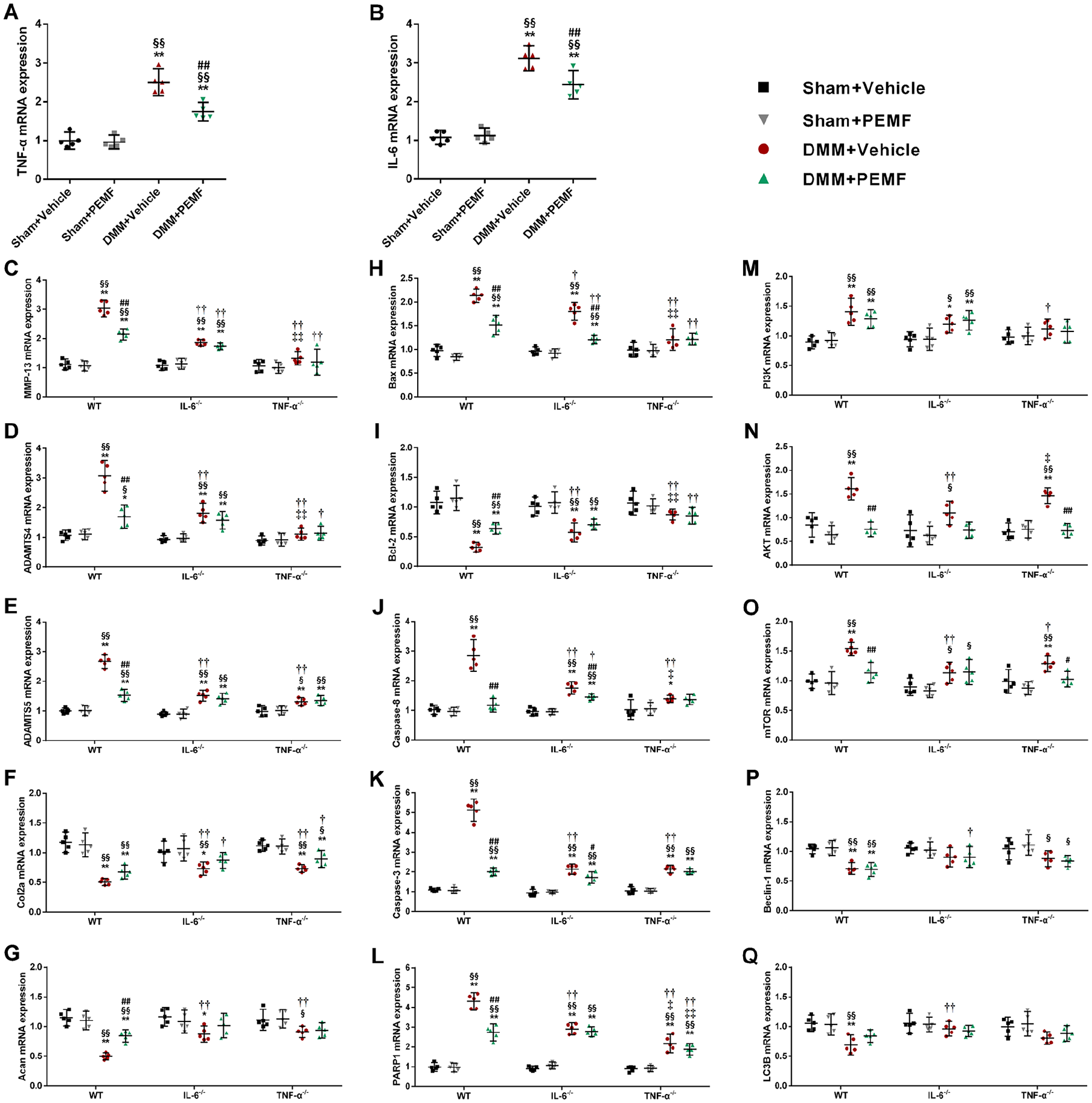
PEMF regulates gene expression of joint cartilage from tibial plateau and femoral condyle via IL-6 and TNF-α in the destabilization of the medial meniscus (DMM) mice. (
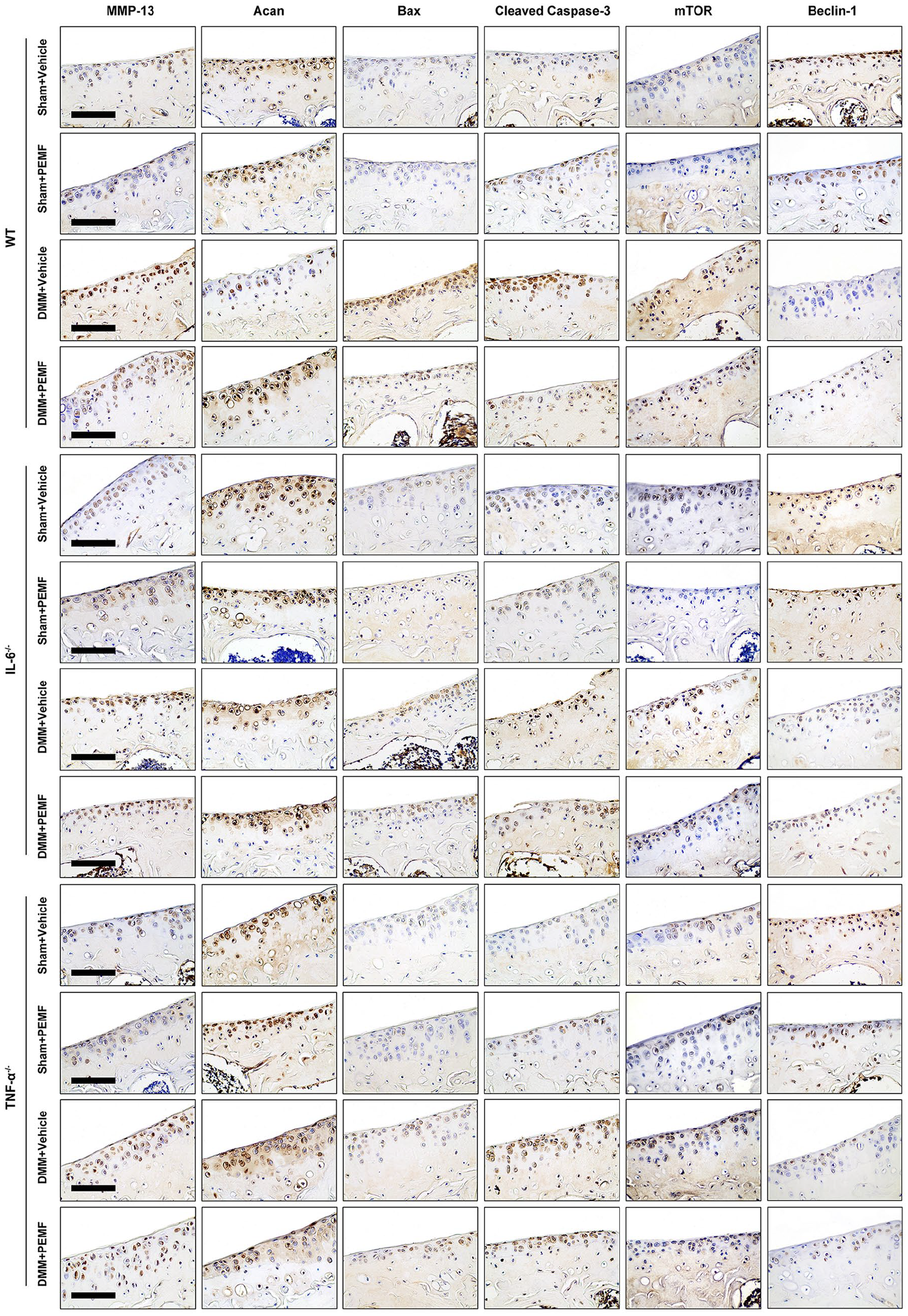
PEMF regulates protein expression of joint cartilage from tibial plateau via IL-6 and TNF-α in the destabilization of the medial meniscus (DMM) mice. Representative immunohistochemistry staining of MMP-13, Acan, Bax, Cleaved Caspase-3, mTOR, and Beclin-1 (n = 3). Magnification: 40×. Scale bar: 100 mm.
PEMF Promotes Chondrocyte Autophagy via IL-6 and TNF-α In Vivo
We further observed a significant downregulation in the mRNA expression levels of AKT and mTOR with no significant differences in the expression of PI3K, Beclin-1, and LC3B after PEMF treatment when compared with vehicle in WT and TNF-α−/− mice after DMM surgery. After DMM surgery, TNF-α−/− mice had lower expression of AKT and mTOR genes than WT mice; this change was reduced after PEMF treatment. We also observed that there were no statistically significant differences in the expression of all autophagy mediators above after PEMF treatment compared with vehicle in IL-6−/− mice after DMM surgery (Fig. 4M-Q). Last, as shown by IHC staining, PEMF resulted in decreased expression of mTOR protein in WT mice after DMM surgery, while this downregulation was not observed in the PEMF-treated IL-6−/− and TNF-α−/− mice ( Fig. 5 and Suppl. Fig. S1).
Discussion
Our study suggests that PEMF, after joint injury, attenuates OA progression through inhibition of IL-6 and TNF-α signaling; data were derived from pain, the degree of cartilage degradation and synovitis, and trabecular bone characteristics in mice. Furthermore, PEMF protects chondrocytes from apoptosis and autophagy, at least partly, via IL-6 and TNF-α signaling.
In this study, protection from PEMF treatment was observed in pain, cartilage in tibial plateau and femoral condyle, synovium, and trabecular bone in WT mice after DMM surgery, indicating that PEMF had a beneficial effect on symptom and structural changes in OA. However, the effects of PEMF were reduced in IL-6−/− and TNF-α−/− mice. Taken together, this would suggest that IL-6 and TNF-α may be critical factors in the effects of PEMF on functional and structural progression of OA. Our previous study reported that PEMF can reduce serum TNF-α levels in OA rabbit. 16 Similarly, Ongaro et al. observed that PEMF decreased proinflammatory cytokine IL-6 in human osteoarthritic synovial fibroblasts. 21 Here, they only found that PEMF had protective effects against proinflammatory cytokines in OA. We extended these studies to describe the importance of proinflammatory cytokines in the effects of PEMF on OA progression. The pathway regulated by these cytokines was showed in Supplementary Figure S2.
Our data showed a beneficial effect of PEMF on cartilage matrix metabolism, chondrocyte apoptosis, and autophagy, while deletion of IL-6 or TNF-α suppressed the effects, indicating the potential value of IL-6 and TNF-α in chondroprotective effects of PEMF. This finding is consistent with our previous studies in which the expression of Bax and Caspase-3 were reduced in the articular cartilage of OA rabbits and rats after PEMF treatment.16,22 Furthermore, our results suggest that TNF-α and IL-6 signaling might act synergistically with apoptosis (Bax/Caspase-3) and autophagy (AKT/mTOR) mediators to promote the degeneration of cartilage. Proinflammatory cytokines have been shown to be associated with chondrocyte death. Increased apoptosis and decreased autophagy in cartilage would result in matrix degradation. 30 Although the mechanism by which PEMF modulates chondrocyte apoptosis and autophagy may be associated with proinflammatory mediators, the detailed mechanisms have not been fully investigated. Further investigation is required to confirm the role of cell death mediators (e.g., mTOR) in the PEMF treatment.
There were several strengths to our study. First, our primary aim was to explore the effects of PEMF on attenuating OA progression by inhibiting TNF-α and IL-6 signaling. To our knowledge, we are the first to use TNF-α or IL-6 KO mice for examining the effects of PEMF. We provide strong evidence that TNF-α and IL-6 signaling could be important mediators during the PEMF treatment of OA. Furthermore, TNF-α and IL-6 signaling might, in part, contribute to the effects of PEMF on chondrocyte apoptosis and autophagy. Subsequently, we analyzed the trabecular bone microarchitecture of proximal tibia and distal femur, which would provide full spectrum of bone characteristics in knee OA mice.
There are several potential limitations in our study. First, the TNF-α or IL-6 KO mice used in our study were TNF-α or IL-6 global KO mice, but not cartilage-specific KO mice. TNF-α or IL-6 global knockout may impact on key innate immune cell function. Second, the contralateral limbs were used as sham-operated. Although previous studies selected the same method,29,31,32 this approach may subsequently affect behavior assessment of pain. In addition, we selected regions of interest manually slice by slice for analyzing subchondral bone characteristics. While ideally regions of interest would be selected across all available slices automatically. Our methods are in agreement with previous methods used to measure relative values. 33
In conclusion, we observed that PEMF attenuates structural and functional progression of OA through inhibiting the expression of TNF-α and IL-6. Our data suggest that TNF-α and IL-6 signaling may be critical factors in the protective effects of PEMF on chondrocyte apoptosis and autophagy. This study may have clinical relevance for combined application of PEMF and anti-TNF-α or anti-IL-6 monoclonal antibody in the treatment of OA. Further studies are required to confirm whether combined effects of PEMF and anti-TNF-α or anti-IL-6 monoclonal antibody are suppressed.
Supplemental Material
sj-jpg-1-car-10.1177_19476035211049561 – Supplemental material for Pulsed Electromagnetic Field Attenuates Osteoarthritis Progression in a Murine Destabilization-Induced Model through Inhibition of TNF-α and IL-6 Signaling
Supplemental material, sj-jpg-1-car-10.1177_19476035211049561 for Pulsed Electromagnetic Field Attenuates Osteoarthritis Progression in a Murine Destabilization-Induced Model through Inhibition of TNF-α and IL-6 Signaling by Xiaotian Yang, Hua Guo, Wenwen Ye, Lin Yang and Chengqi He in CARTILAGE
Supplemental Material
sj-tif-1-car-10.1177_19476035211049561 – Supplemental material for Pulsed Electromagnetic Field Attenuates Osteoarthritis Progression in a Murine Destabilization-Induced Model through Inhibition of TNF-α and IL-6 Signaling
Supplemental material, sj-tif-1-car-10.1177_19476035211049561 for Pulsed Electromagnetic Field Attenuates Osteoarthritis Progression in a Murine Destabilization-Induced Model through Inhibition of TNF-α and IL-6 Signaling by Xiaotian Yang, Hua Guo, Wenwen Ye, Lin Yang and Chengqi He in CARTILAGE
Footnotes
Author Contributions
XY, LY, and CH contributed to the conception and design of the work. XY, HG, and WY contributed to the acquisition and interpretation of data. All authors contributed to drafting the work and revising the content critically. All authors have approved the final version.
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the National Natural Science Foundation of China (No. 81972146 and No. 8210090170).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for this study was obtained from the Institutional Animal Care and Use Committee of West China Hospital, Sichuan University (No. 2018130A).
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
