Abstract
Objective
A major obstacle in the clinical translation of engineered auricular scaffolds is the significant contraction and loss of topography that occur during maturation of the soft collagen-chondrocyte matrix into elastic cartilage. We hypothesized that 3-dimensional-printed, biocompatible scaffolds would “protect” maturing hydrogel constructs from contraction and loss of topography.
Design
External disc-shaped and “ridged” scaffolds were designed and 3D-printed using polylactic acid (PLA). Acellular type I collagen constructs were cultured in vitro for up to 3 months. Collagen constructs seeded with bovine auricular chondrocytes (BAuCs) were prepared in 3 groups and implanted subcutaneously in vivo for 3 months: preformed discs with (“Scaffolded/S”) or without (“Naked/N”) an external scaffold and discs that were formed within an external scaffold via injection molding (“Injection Molded/SInj”).
Results
The presence of an external scaffold or use of injection molding methodology did not affect the acellular construct volume or base area loss. In vivo, the presence of an external scaffold significantly improved preservation of volume and base area at 3 months compared to the naked group (P < 0.05). Construct contraction was mitigated even further in the injection molded group, and topography of the ridged constructs was maintained with greater fidelity (P < 0.05). Histology verified the development of mature auricular cartilage in the constructs within external scaffolds after 3 months.
Conclusion
Custom-designed, 3D-printed, biocompatible external scaffolds significantly mitigate BAuC-seeded construct contraction and maintain complex topography. Further refinement and scaling of this approach in conjunction with construct fabrication utilizing injection molding may aid in the development of full-scale auricular scaffolds.
Keywords
Introduction
Microtia, the congenital absence of part or all of the external ear, is a socially and psychologically significant deformity. The current gold standard for surgical correction, which involves multistage operative techniques popularized by Brent 1 and Nagata, 2 utilizes autologous costal cartilage to correct microtia. In expert hands these approaches can lead to reasonable facsimiles of the external ear. However, they are technically challenging and, in the majority of cases, provide a suboptimal aesthetic result. 3 Because it is sculpted from rib fibrocartilage, the resultant auricle is unnaturally stiff compared to the elastic native ear. 4 Furthermore, the obligatory chest wall donor site is a source of morbidity and pain, especially in the pediatric population.5,6 As with so many aspects of reconstructive surgery, this clinical challenge enhances the opportunity for tissue engineering to improve upon current practice by introducing a solution that obviates the need for a technically challenging 2-stage procedure that creates significant donor morbidity.
Prosthetic ears or a combination of prostheses and autologous tissues have been described for external ear reconstruction and have been made easier to fabricate with the aid of 3-dimensional (3D) printing techniques.7,8 Although porous polyethylene has been used with great success by a limited number of surgeons to produce a full-scale scaffold with no or limited donor site morbidity, 9 most other practitioners have found this material to have a high rate of complications, including extrusion and infection.10-12 Furthermore, like autologous costal cartilage, this material is inherently stiff.
Over the past decade, 3D-printed bioengineered and cell-seeded ear scaffolds have also been studied as alternative autologous cartilage implants for auricular reconstruction and assessed in terms of cell survival, neocartilage formation, and biomechanical properties that imperfectly resemble the native human ear.8,13-15 It is clear from much of the published literature that the major remaining obstacle to successful clinical translation of auricular engineering is the fabrication of constructs that are able to withstand the intrinsic contractile and external compressive forces experienced in vivo. These forces remain a significant cause of volume loss and blunting of the complex auricular topography, producing a less than ideal aesthetic outcome.16,17
In our previous work, anatomically faithful, full-scale human chondrocyte laden hydrogel constructs implanted in vivo produced healthy, viable elastic cartilage throughout the entirety of the scaffold. 18 However, the constructs contracted to approximately 50% of their original length and width, and also demonstrated flattening and loss of topographical detail. We hypothesized that this was largely due to a combination of extrinsic compressive forces exerted by the overlying soft tissue and intrinsic contractile forces created by the chondrocytes as the initially soft, low modulus scaffold matures. Mitigation of these forces will be a crucial step in allowing for the translation of this approach to auricular tissue engineering. In the current work, we explored whether 3D-printed external scaffolds composed of biocompatible polylactic acid (PLA), to protect the soft chondrocyte-collagen hydrogel constructs as the cartilage matures, would decrease volume loss and preserve topography.
Material and Methods
External Scaffold Design and Fabrication
Two different scaffolds were created in the 3D-design software Sketchup (Trimble, Sunnyvale, CA), optimized for 3D-printing with a Makerbot Replicator 5th Generation and printed using PLA (MakerBot Filament, MP05776) for in vitro and in vivo study: (1) an 8-mm disc-shaped scaffold (8 mm diameter × 2 mm height) designed as 2 printable parts which could be assembled by snapping together (
Fig. 1A
,
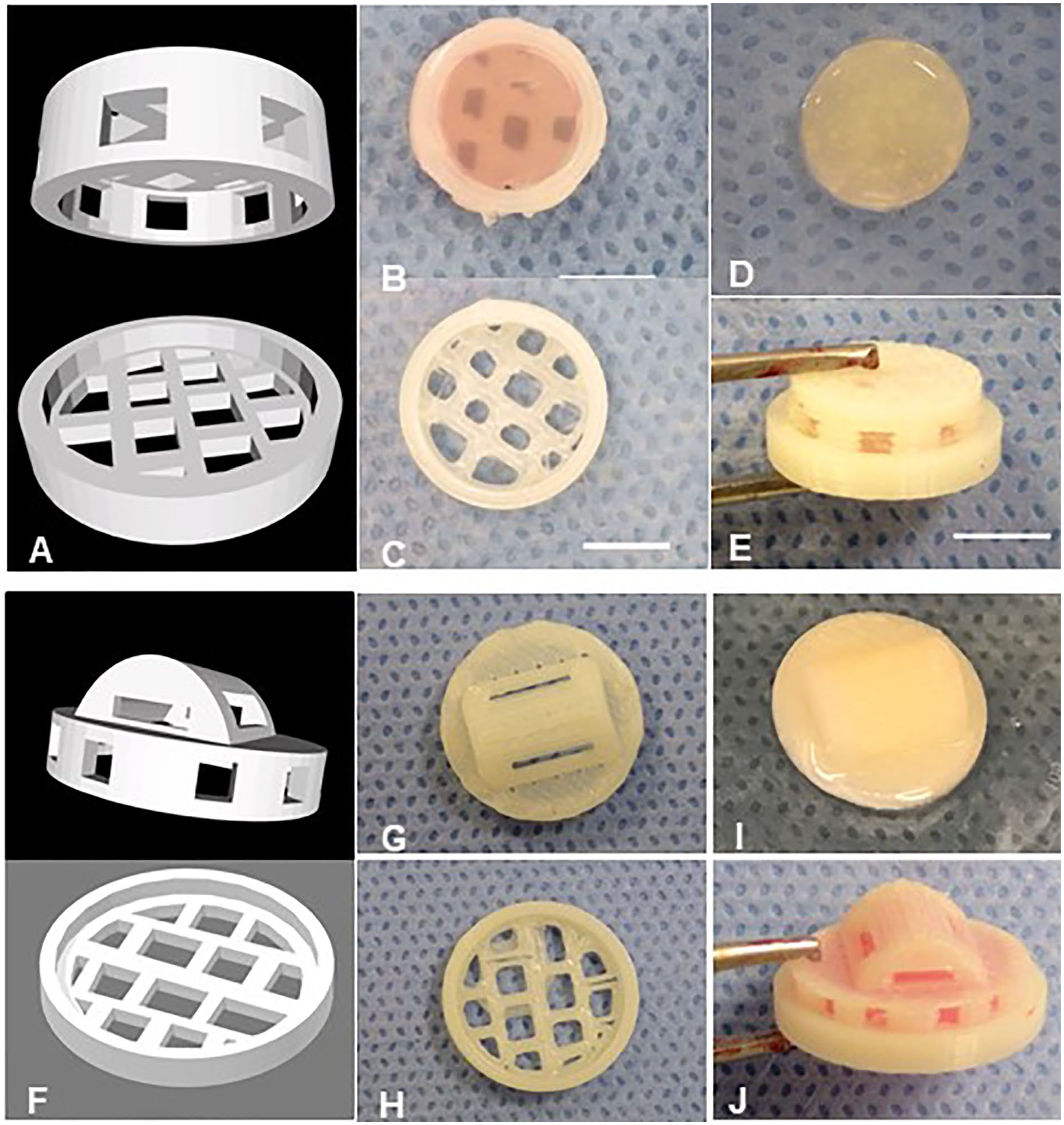
Three-dimensional-printed PLA external scaffolds: 8 mm disc-shaped scaffold (
Isolation of Bovine Auricular Chondrocytes
Bovine auricular chondrocytes (BAuCs) were isolated as previously described. 18 Ears were obtained from freshly slaughtered 1- to 3-day-old calves (Gold Medal Packing, Oriskany, NY, and a local abattoir in Brooklyn, NY). Auricular cartilage was dissected from the skin and perichondrium, minced into 1 mm3 pieces and digested overnight in 0.2% collagenase II (Worthington Biochemicals Corp., Lakewood, NJ), 100 μg/mL penicillin and 100 μg/mL streptomycin in Dulbecco’s modified Eagle’s medium (DMEM, 1×). Chondrocytes were filtered, washed, counted, and then placed in culture.
In Vitro: Acellular Collagen Constructs
This study was designed to investigate whether the presence of an external scaffold would affect construct volume in the absence of cells within the hydrogel. The 2 techniques utilized involved (1) a preformed construct that is secondarily placed within the external scaffold (S) versus (2) an injection molding technique where the construct is formed in situ within the external scaffold (SInj) to increase interfacial bonding between the collagen and the external scaffold. Type I collagen stock was extracted from rat tails, lyophilized, and reconstituted as previously described.18-20 One percent neutralized collagen was homogeneously mixed and allowed to undergo thermal gelation at 37 °C for 1 hour within a negative polydimethylsiloxane (PDMS) mold (matching the interior dimensions of the 8-mm disc-shaped scaffold). Half of the constructs were then removed from the PDMS mold and used as a “naked/N” (non-scaffolded) control group ( Fig. 1D ). The other half of the constructs were placed into 8-mm disc-shaped external scaffolds as a “Scaffolded/S” experimental group ( Fig. 1B ). For the third group, “Injection molded/SInj” constructs were prepared by injecting an acellular collagen suspension into external scaffolds housed within a negative PDMS mold in a slow and steady rate to avoid creating air bubbles, intended to maximize the surface contact between the scaffold and the hydrogel during gelation ( Fig. 1E ). All of the constructs were cultured within 6-well plates in complete media (DMEM, 1× + 10% FBS + 100 μg/mL penicillin and 100 μg/mL streptomycin) for 1 and 3 months, at which point construct volume was assessed. At least 10 samples per time point were studied for each group.
In Vivo: BAuC-Seeded Collagen Constructs
All animal care and experimental procedures were in compliance with the Guide for the Care and Use of Laboratory Animals, 21 and were approved by the Weill Cornell Medical College Institutional Animal Care and Use Committee (protocol # 2011-0036).
The next experimental phase involved the addition of chondrocytes to the collagen hydrogels. Similarly, BAuC-seeded collagen constructs were prepared in both 8-mm disc-shaped and 12-mm ridged scaffolds for 3 groups: naked (N), scaffolded (S), and scaffolded/injection molded (SInj). One percent collagen was homogeneously mixed with 25 million/mL BAuCs at passages 2 to 3 and gelled at 37 °C for 1 hour with or without external scaffolds in PDMS molds. 18 Cell-loaded constructs were cultured in DMEM media overnight before implantation. The next day, 6 constructs were implanted subcutaneously on the bilateral dorsa of 10-week-old nude rats (Charles River, Wilmington, MA). After 3 months, the animals were sacrificed and the constructs were explanted and scanned with microCT. Each explant was then fixed in 10% formalin, paraffin-embedded, and stained with Safranin O/Fast green to assess proteoglycan distribution, Picrosirius red to assess collagen organization, and Verhoeff’s/Van Gieson to assess the presence of elastin fibers. Images were taken using a Nikon Eclipse TE2000-S microscope (Nikon Instruments, Melville, NY). A total of 6 samples per time point were studied for each group.
Volume, Base, and Angle Measurement
Constructs were imaged via Inveon Pre-clinical MicroPET/CT/SPECT scanning (CTI/Siemens, Knoxville, TN) and digitally reconstructed with Inveon Research software to assess volumetric and base area changes over time. Regions of interest (ROIs) were created in digitally reconstructed images by selecting voxels using thresholds for pixel intensity and a combined 3D ROI created from the coronal, axial, and sagittal views was generated to acquire a precise volume and base area measurement of all the explanted constructs. Preservation of the “helical rim” ridge feature was evaluated objectively by measuring the angle between the rim and the base of the constructs on both sides with Imaris (Bitplane, Belfast, UK; Fig. 4A).
Statistical Analysis
Data analyses were performed using an unpaired t-test to compare gross measurements and angle preservation of the constructs. At least 4 constructs per group were analyzed. A value of P < 0.05 was used as a threshold for statistical significance. All data were expressed as means ± 1 SD.
Results
In Vitro Studies
Acellular collagen constructs showed a consistently high base area preservation at the 1- and 3-month time points in all groups ( Table 1 ). There was no statistically significant difference in these metrics regardless of the presence or absence of a protective external scaffold at either time point ( Fig. 2B ). At 1 month, mean volume preservation among the naked (N) was 71.9 ± 6.2% compared to 70.8 ± 6.5% in the scaffolded (S) group (P > 0.05). This same trend was observed at 3 months, when mean volume preservation was found to be 59.7 ± 21.2% in the naked (N) group compared to 59.1 ± 23.3% in the scaffolded (S) group (P > 0.05). When the injection molding technique was utilized to form the discs, mean volume preservation was somewhat improved after 1 and 3 months in culture (80.3 ± 12.6% at 1 month and 73.5 ± 19.9% at 3 months), although this did not reach statistical significance (P > 0.05; Fig. 2A ).
Volume and Base Area Preservation of 8-mm Disc-Shaped Acellular Collagen Constructs After 1 Month and 3 Months of In Vitro Culture a .
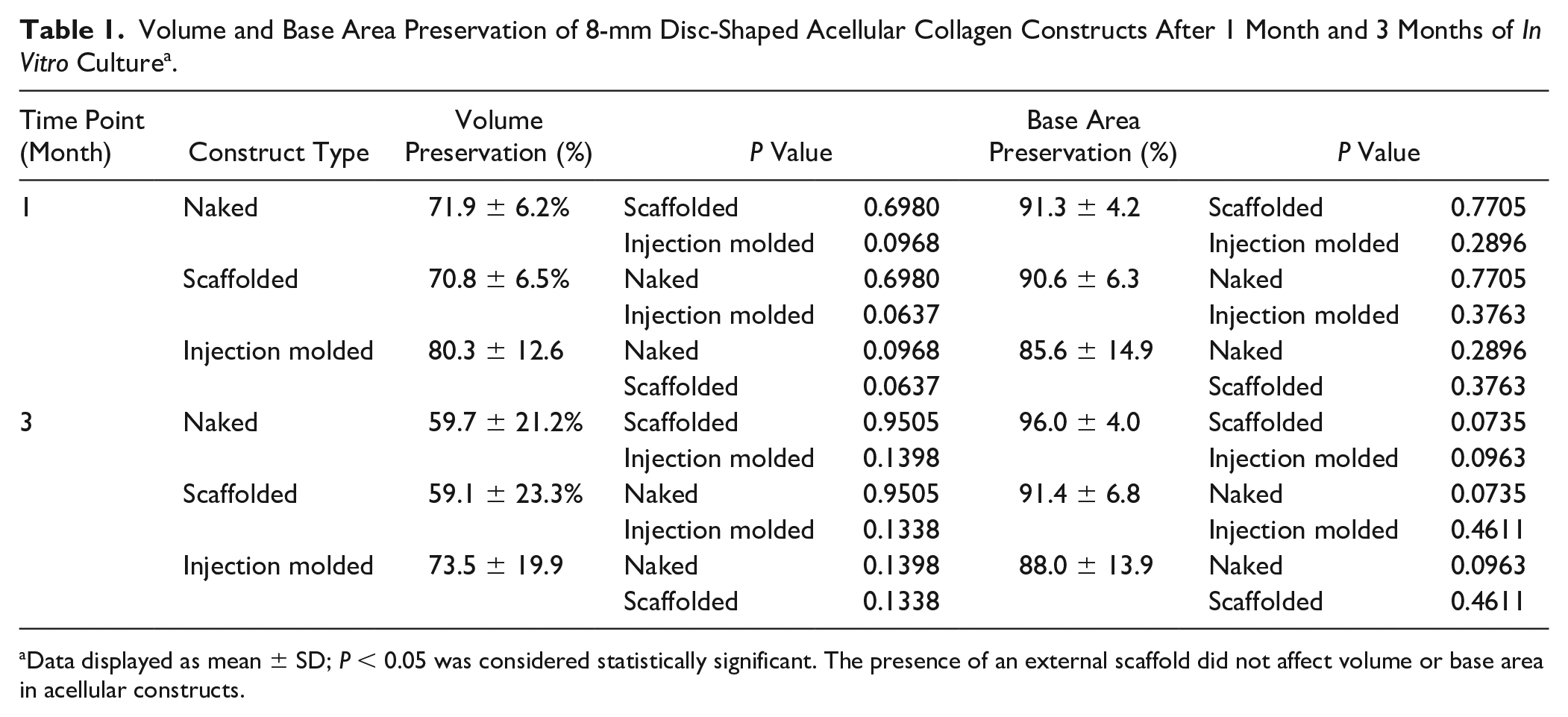
Data displayed as mean ± SD; P < 0.05 was considered statistically significant. The presence of an external scaffold did not affect volume or base area in acellular constructs.
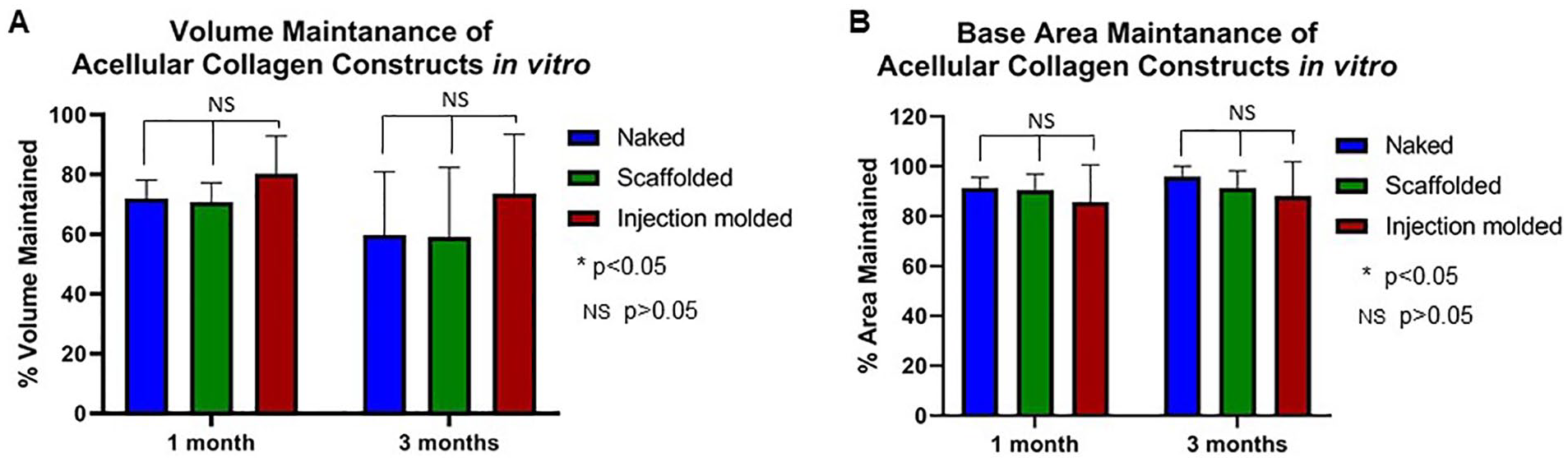
Volume and base area maintenance of 8-mm disc-shaped acellular collagen constructs after 1 month and 3 months in vitro culture. (
In Vivo Studies: Effect of Scaffolding and Construct Fabrication Methodology on Disc Dimensions
In vivo, the presence of an external scaffold significantly decreased volume and base area loss after 3 months compared to the naked (N) groups ( Table 2 ). Specifically, the improvement in volume and base area preservation was significant in the 8-mm BAuC-seeded scaffolded (S) constructs compared to the naked (N) group (90.5 ± 13.8% [S] vs. 64.0 ± 0.6% [N], P < 0.05); constructs formed in situ using injection molding demonstrated complete volume preservation after 3 months in vivo (108.0 ± 2.1% [SInj], P < 0.05; Fig. 3A ). The volume of tissue in this latter group was marginally greater than the starting volume due to formation of through the pores of the scaffolds (a result of the injection molding technique). The injection molded (SInj) constructs were also noted to have significantly greater base area preservation after 3 months compared to the naked (N) group (91.2 ± 2.4% [SInj] vs. 46.0 ± 3.0% [N] for the 8-mm discs and 86.9 ± 10.3% [SInj] vs. 39.6 ± 4.2% [N] for the 12-mm ridged discs respectively, P < 0.05). The same finding was observed when base area preservation was compared between the injection molded (SInj) and scaffolded (S) group for the 8 mm discs (91.2 ± 2.4% [SInj] vs. 70.3 ± 11.5% [S], P < 0.05; Fig. 3B ).
Volume and Base Area Preservation of 8-mm Disc-Shaped and 12-mm Ridged BAuC-Seeded Collagen Constructs after 3 Months In Vivo a .
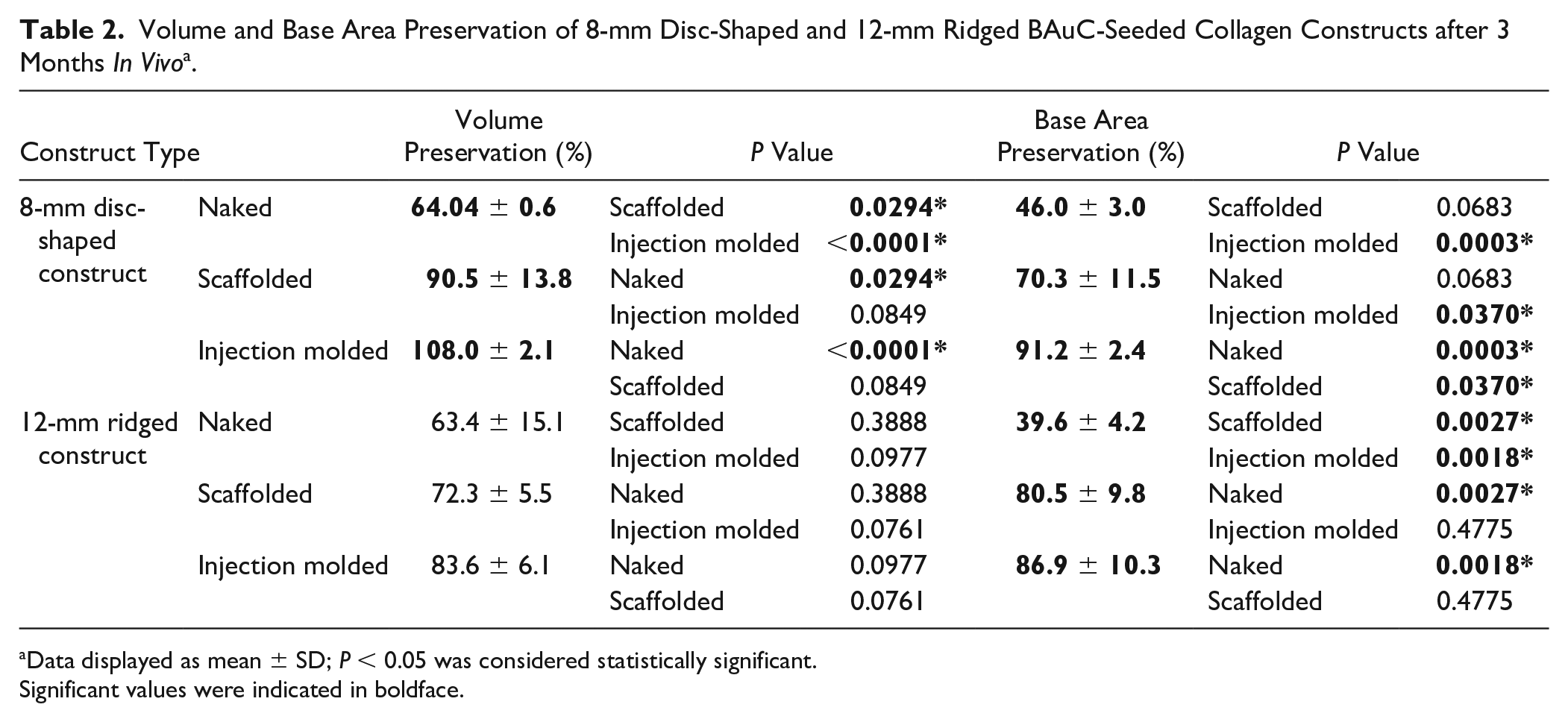
Data displayed as mean ± SD; P < 0.05 was considered statistically significant.
Significant values were indicated in boldface.
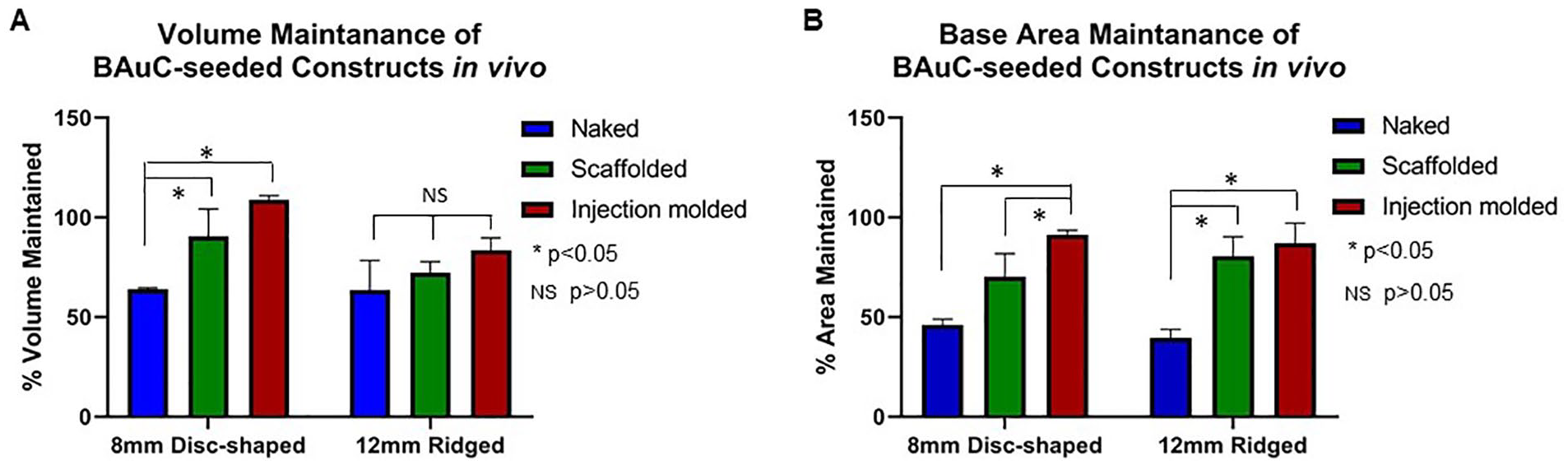
Volume and base area maintenance of 8-mm disc-shaped and 12-mm ridged BAuC-seeded collagen constructs after 3 months in vivo. (
External Scaffolds Preserve Construct Topography In Vivo
At the time of 12-mm ridged construct fabrication, the angle formed between the ridge feature and the top of the disc measured 110° (
Fig. 4A
). After 3 months in vivo, the naked (N) constructs lost their ridge features entirely resulting in flattened discs. In contrast, the injection molding methodology demonstrated significant improvement in preservation of the construct angle between the ridge feature and the top of the disc (133.9 ± 12.2° [SInj]) compared to both the scaffolded (147.9 ± 9.2°) (S) and naked groups (178.0 ± 11.4°) (N) (P < 0.05;
Fig. 4B
and
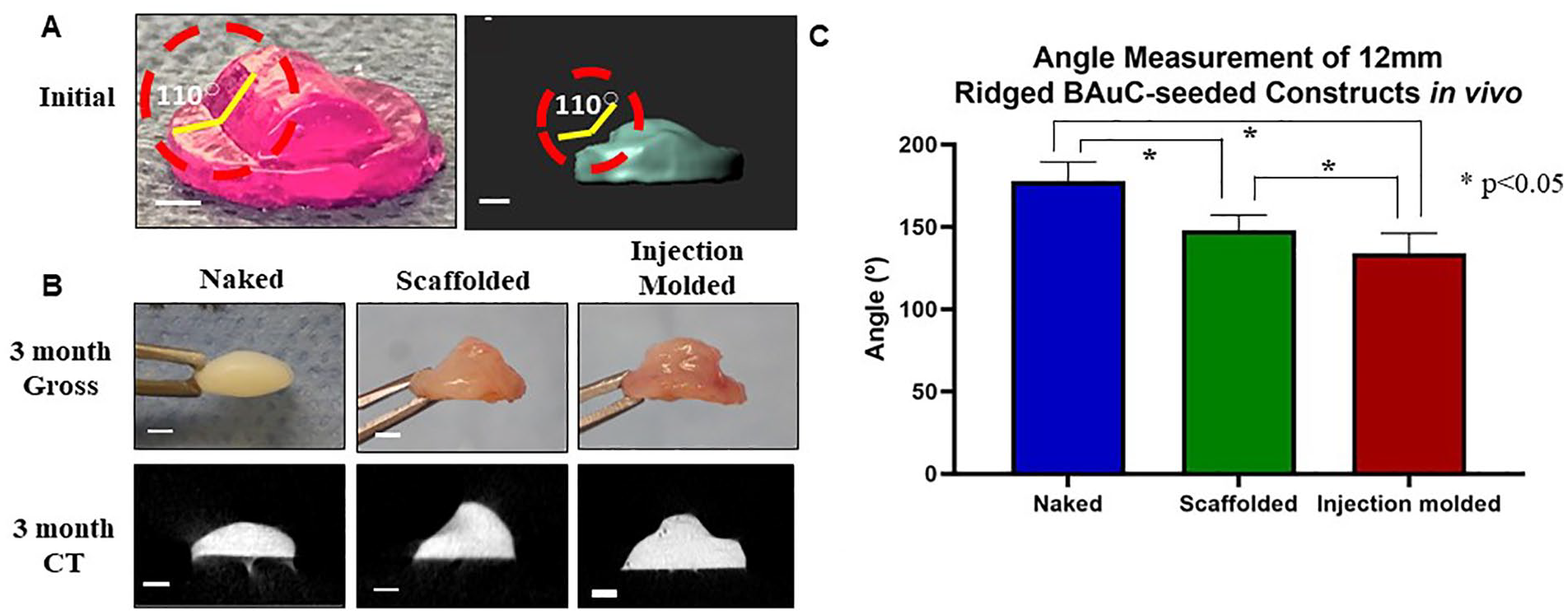
External scaffolds preserve construct topography in vivo. (
External Scaffolds Do Not Impair Healthy Cartilage Formation
Upon gross examination at the time of explantation, there was a fibrous connective tissue capsule with ingrowth through the pores of scaffolds; there was no evidence of degradation of PLA-printed scaffolds noted at 1 and 3 months. All discs had a glossy white cartilaginous appearance, along with a texture and elastic flexibility similar to that of auricular cartilage. Engineered 12-mm ridged scaffolded constructs displayed the histological features of auricular cartilage after 3 months in vivo ( Fig. 5 ), and similar results were observed in 8-mm scaffolded constructs (data not shown). Picrosirius red staining showed the formation of bundled collagen fibers along the surface, resembling the perichondrial layer characteristic of native ear cartilage. Safranin-O staining demonstrated the formation of a rich proteoglycan matrix within the interior of the constructs after 3 months in vivo. In addition, all constructs displayed development of cellular lacunae within proteoglycan-dense tissue and Verhoeff’s staining confirmed the development of a nascent elastin fiber network. Taken together, these data demonstrate that the external scaffold does not impede sufficient flow of nutrients in vivo, which is necessary for chondrocyte survival, function, and neocartilage formation.
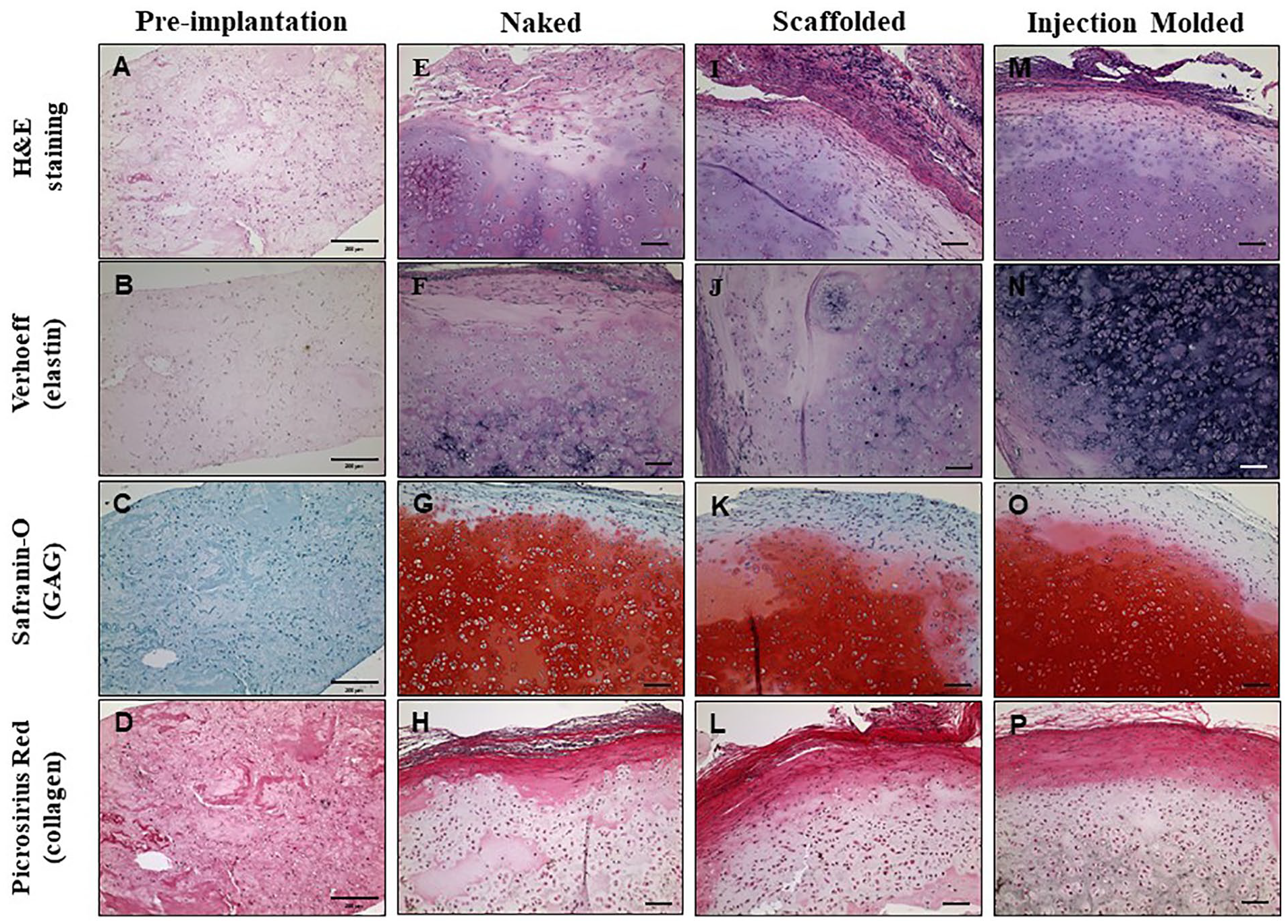
Histologic analysis of 12-mm ridged BAuC-seeded naked (N), scaffolded (S), and injection molded (SInj) constructs pre-implantation and after 3 months in vivo. Representative images of H&E staining (
Discussion
Despite considerable progress in the field of auricular tissue engineering, perhaps the preeminent obstacle to the creation of full-scale engineered constructs remains the significant contraction and inevitable loss of the intricate patient-specific morphological details that define the unique structure of the human external auricle as cartilage matures in vivo.18,22 In a clinical scenario, when a construct sculpted from costal cartilage is placed in its new, “proper” anatomic location, compressive forces generated by the scalp overlying the scaffold (which normally sits on an unyielding bony layer underneath), do not deform the construct because of the stiff nature of the rib-derived fibrocartilage from which it is fabricated.23-25 Although this stiffness is somewhat advantageous in this surgical scenario, in the long term the firmness of the reconstructed auricle contributes to its artificial feel and suboptimal aesthetic appearance. In contrast, in our prior auricular engineering work, the loose skin of rats likely did not exert a significant compressive force on the constructs. Thus, we can attribute the loss of volume and topographical detail observed primarily to the effect of intrinsic contractile forces within the forming cartilage. 26
Intrinsic contractile forces generated by individual cells on a microscopic level within a fibrillar matrix can produce a significant aggregate force which can result in macroscopic changes in the shape and size of that matrix. The contractile forces are directly transmitted from cells to the surrounding extracellular matrix (ECM) in the process of cell migration, fiber remodeling and focal adhesion formation, resulting in ECM deformation and contraction.27,28 Various ECMs that have been widely used in tissue engineering, such as collagen and fibronectin, largely encourage cell adhesion. 29 Cells adhere to the matrix and other cells with the help of proteins, such as integrins, cadherins, and selectins. These serve as anchors, binding to specific ECM proteins to assist cells in creating forward motion and, ultimately, matrix contraction.28,30,31
The use of an internal scaffold (i.e., wire) has previously been documented in the literature as a means of preventing contraction of collagen scaffolds, but this approach does not mitigate loss of external topography. In addition, an internal wire scaffold is obligatorily permanent within the construct, which can cause inflammation and negatively affect neocartilage formation.16,17 An external scaffold may be a superior option to an internal support in auricular engineering16,17 given that it can either be removed after cartilage formation or made from a material that biodegrades as the mature cartilage forms.8,32,33 In contrast to an internal wire, an external support can serve as protection from extrinsic compression forces without affecting neocartilage formation within the maturing matrix. Furthermore, an external scaffold may be particularly suited for auricular engineering since it can be designed and 3D-printed to match specific patient anatomy.
Visscher et al. demonstrated that after 28 days in vitro an external porous scaffold made of polycaprolactone (PCL) completely eliminated contraction of articular chondrocyte/MSC-seeded collagen matrices placed within them while still allowing for formation of a cartilaginous matrix. 14 While this study showed impressive results without any loss of construct volume, we found that collagen discs seeded with bovine auricular chondrocytes protected by PLA porous scaffolds demonstrated significantly less volume loss than naked, non-scaffolded constructs after 3 months in vivo. Given the relatively short timescale studied by Visscher et al., it is unclear whether the lack of contraction observed after only 28 days would hold true after 90 days (as in the current study) or longer. Furthermore, the high cell density within the constructs (25 millions/mL collagen) in the current work versus 1 million cells/50 µL hydrogel used by Visscher’s group also may have contributed to the increased degree of contracture seen in our constructs. Although Visscher et al. asserted evidence of early cartilage formation, their supportive data, including immunohistochemistry staining for collagen II and second harmonic imaging of collagen, as well as the paucity of chondrocytes and associated cartilaginous matrix seen, did not convincingly support their claims. In contrast, after 3 months in vivo we observed copious deposition of GAG and elastin surrounding numerous healthy chondrocytes within our scaffold matrices, demonstrating that the original collagen type I hydrogel had matured into elastic cartilage.
Although mitigation of volume loss of the engineered construct is important, for the purpose of auricular engineering, faithfully mimicking the complex topography of the patient-specific external ear is far more crucial to preservation of the aesthetic appearance of the engineered tissue. As contraction of the maturing construct can result in significant blunting and distortion of normal anatomy, we designed slightly larger (12-mm wide) porous PLA discs that featured a ridge on the surface of the disc to simulate the shape of the helical rim. After 3 months in vivo, the helical rim feature remained better preserved in the injection molded (SInj) and scaffolded (S) groups compared to the naked (N) group, where the rim feature was essentially lost resulting in a flat disc of cartilage. Importantly, in the S and SInj groups the presence of an external PLA scaffold did not interfere with the formation of healthy cartilage within the constructs.
Although the 8 mm × 2 mm (height × width) discs fit snugly within the scaffold, on a micro-scale there was still a fair amount of “space” between the construct and the surrounding PLA. In order to test the hypothesis that an increased degree of interfacial bonding between the hydrogel and the surrounding scaffold might further decrease construct volume loss, we fabricated constructs in situ within the external scaffolds (as opposed to making a sheet and punching constructs out) by injection molding the chondrocyte/collagen hydrogel directly into the scaffold. This resulted in the hydrogel maximally attaching to all exposed PLA surfaces prior to implantation, including within the pores. Compared with conventional methods, injection molding allows for the creation of a more fully interconnected porous network 34 and has been shown to be particularly well suited for the rapid fabrication of homogenous volumes with high accuracy. 35 Our results are consistent with previous work demonstrating that mechanically anchoring a cell-seeded collagen construct to an external structure helps to maintain shape fidelity over extended culture in vitro. 36 In the current study, the presence of the hydrogel within the pores of the scaffolds, combined with minimal contracture, likely resulted in a final average construct volume marginally greater than the starting volume (since 100% volume was defined as the space within the walls of the scaffold).
Injection molding has been shown to effectively compensate for material shrinkage during the production process because bubbles nucleated from the sudden decrease in pressure during fabrication continue to grow inside the mold cavity. 34 This has proven true in multiple prior studies wherein injection molding has been used to produce living tissues with high shape accuracy and very little radial shrinkage. 19 Accordingly, in the current study, the injection molded (SInj) BAuC-seeded constructs showed significantly greater volume and base preservation compared to the naked (N) group, and significantly greater base area preservation compared to the scaffolded (S) 8-mm disc-shaped constructs after 3 months in vivo.
We chose PLA to fabricate the scaffolds given that it is biodegradable, biocompatible, and can be easily and economically 3D-printed in intricate shapes (such as a human auricle). Furthermore, PLA is Food and Drug Administration–approved and is frequently used as a scaffold in tissue engineering, mainly due to its cell compatibility, strength, and low cost.14,37 Furthermore, PLA has a high enough elastic modulus to protect the collagen scaffold containing BAuCs from compressive forces that would be encountered if implanted under the tight scalp skin (unlike the minimal compression exerted by loose rodent skin). 38 The slow degradation of PLA in vivo is advantageous in that it retains its integrity throughout the time required for the collagenous matrix to transform into elastic cartilage. As we have previously shown that after 3 months in vivo, our cartilage constructs demonstrate mechanical properties that are similar to that of native cartilage, and these are maintained after 6 months in vivo, it is critical that the PLA can maintain its stability throughout this time period.18,39 Full-scale ear constructs will likely require a longer period of maturation in vivo in order for the much larger volume of collagenous matrix to form cartilage. Determination of the utility of the external PLA scaffold in preventing loss of volume and anatomic fidelity in full-scale human ear constructs is currently underway.
This work is not without limitations. The utilization of bovine cells demands ultimate progression to human cells in order to produce a truly clinically translatable result. The use of a rodent model is limiting due to the loose nature of rodent skin which does not accurately mimic the compressive forces that are experienced by scaffolds placed under human auricular/scalp skin. 40 Future directions of this work include transitioning to the use of human cells, repeating this study with a full-scale ear model, and utilizing a larger animal model that could more accurately mimic human skin and its biomechanical properties.
Conclusion
Custom designed, contour-matched, 3D-printed biocompatible/biodegradable external scaffolds do not impede formation of mature elastic cartilage from collagen-based hydrogels. Importantly, scaffolds fabricated using an injection molding methodology protect auricular cartilage constructs from external compression and intrinsic contractile forces leading to significant mitigation of contraction and maintenance of the complex topography. Our unique technique can be used to create custom scaffolds that contour to any form, enabling the fabrication of engineered autologous cartilage tailored to the individual patient without the compression, contraction, and loss of topography that has thus far impeded translation to the clinic.
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this research was provided by NIH Grant 5T35EB006732, NYSTAR, 3D BioCorp, and the Empire State Stem Cell Fund through New York State Department of Health Contract #C30293GG. Opinions expressed here are solely those of the authors and do not necessarily reflect those of the Empire State Stem Cell Board, the New York State Department of Health, or the State of New York.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All animal care and experimental procedures were approved by the Weill Cornell Medical College Institutional Animal Care and Use Committee (Protocol # 2011-0036).
Animal Welfare
All animal care and experimental procedures were in compliance with the Guide for the Care and Use of Laboratory Animals.
