Abstract
Objective
The underlying mechanisms and molecular factors influencing intervertebral disc (IVD) homeostasis and degeneration remain clinically relevant. Tenomodulin (Tnmd) and chondromodulin (Chm1) are antiangiogenic transmembrane glycoproteins, with cleavable C-terminus, expressed by IVD cells that are implicated in the onset of degenerative processes. We evaluate the organ-level biomechanical impact of knocking out Tnmd alone, and Tnmd and Chm1, simultaneously.
Design
Caudal (c5-8) and lumbar vertebrae (L1-4) of skeletally mature male and female 9-month-old wildtype (WT), Tnmd knockout (Tnmd−/−), and Tnmd/Chm1 double knockout (Tnmd−/−/Chm−/−) mice were used (n = 9-13 per group). Disc height index (DHI), histomorphological changes, and axial, torsional, creep, and failure biomechanical properties were evaluated. Differences were assessed by one-way ANOVA with post hoc Bonferroni-corrected comparisons (P < 0.05).
Results
Tnmd−/−/Chm1−/− IVDs displayed increased DHI and histomorphological scores that indicated increased IVD degeneration compared to the WT and Tnmd−/− groups. Double knockout IVDs required significantly less torque and energy to initiate torsional failure. Creep parameters were comparable between all groups, except for the slow time constant, which indicated faster outward fluid flow. Tnmd−/− IVDs lost fluid faster than the WT group, and this effect was amplified in the double knockout IVDs.
Conclusion
Knocking out Tnmd and Chm1 affects IVD fluid flow and organ-level biomechanical function and therefore may play a role in contributing to IVD degeneration. Larger effects of the Tnmd and Chm1 double knockout mice compared to the Tnmd single mutant suggest that Chm1 may play a compensatory role in the Tnmd single mutant IVDs.
Introduction
Back pain is a leading cause of disability worldwide that is strongly associated with intervertebral disc (IVD) degeneration.1-5 IVD degeneration is an aberrant, cell-mediated response to progressive structural failure 6 with degradation of extracellular matrix molecules such as glycoproteins and collagen.7,8 Nucleus pulposus (NP) dehydration and decompression can cause damaging strains on the outer lamellar annulus fibrosus (AF) resulting in loss of IVD height and structural failure.6,9 Loss of NP pressurization and AF disruption can both result in IVD height loss and cause specific alterations to axial and torsional motion segment behaviors. 9 IVDs heal poorly and can involve chronic pro-inflammatory conditions following structural disruption.10,11 The accumulation of structural IVD disruption, IVD height loss, and chronic pro-inflammatory conditions characteristic of IVD degeneration can cause disability and pain. Disabling IVD degeneration-related pathologies are an important clinical challenge that motivate the investigation of structure-function relationships to identify important biomarkers of IVD homeostasis and promising therapeutic targets.
Tenomodulin (Tnmd) and chondromodulin I (Chm1) are 2 anti-angiogenic type II transmembrane glycoproteins with cleavable C-terminal domains secreted in the extracellular matrix. They have been reported to exert functional roles in hypovascular connective tissues such as tendons, cartilage, and IVDs.12-14 Tnmd is best-known as a tendon and ligament maturation marker.12,15,16 In tendon, Tnmd regulates tenocyte proliferation at neonatal stages, collagen fibril maturation, and scar formation during early healing.17,18 The role of Tnmd in the IVD is still emerging; it is expressed by human AF cells 19 and is predominantly expressed by mouse outer AF cells. 13 Tnmd deficient (Tnmd−/−) mice demonstrated decreased AF collagen fibril diameter and nano-scale compressive stiffness, while increasing AF angiogenesis, macrophage infiltration of the outer AF, and hypertrophic-like chondrocytes in the NP. 13 These changes resulted in IVD degeneration with a decrease in disc height index (DHI). 13 Chm1, the only known homolog gene of Tnmd,13,15,16 could play compensatory roles in the maintenance of IVD structure and function. Chm1 is expressed in both human and mice IVDs with major localization in the NP, and is believed to influence NP, AF, and endplate cell proliferation.13,20 More severe IVD degeneration and angiogenic disruption was observed in Tnmd and Chm1 double knockout mice (Tnmd−/−/Chm1−/−) when compared to single knockout Tnmd−/−. 13
While Tnmd and Chm1 are implicated in structural changes to IVDs, their roles on organ-level biomechanical function remain unclear. The objective of this study was to elucidate the roles of Tnmd and Chm1 in IVD height, histomorphology, and IVD biomechanical function by knocking out Tnmd alone, or Tnmd and Chm1 together. Axial and torsional biomechanical properties, as well as creep and torsional failure properties, were evaluated which together can distinguish effects on AF tension and NP pressurization.9,21 Since Tnmd−/− IVDs showed decreased AF collagen fibril diameter and nano-scale compressive stiffness, 13 we hypothesized that Tnmd−/− and Tnmd−/−/Chm1−/− IVDs would display altered biomechanical properties due to AF collagen disruption, with more severe changes in the double knockout group.
Methods
Specimen Preparation
Skeletally mature female and male 9-month-old Tnmd−/− mice and Tnmd+/+ wildtype (WT) or Tnmd+/- heterozygous littermates, and Tnmd−/−/Chm1−/− mice were used in this study (n = 9-13 per group). WT and heterozygous IVDs were grouped together as WT samples. The generation of Tnmd−/− and Tnmd−/−/Chm1−/− mice genotype has been previously described. 17 All mice were of a C57BL/6J background. After euthanasia with CO2, caudal spines were collected and stored at −20 °C until testing. On testing day, tails were thawed in 1× phosphate-buffered saline (PBS) for 90 minutes at room temperature. Mouse husbandry, handling, and euthanasia were carried out strictly according to the guidelines of the Lower Franconia government.
Disc Height Index and Related Measurements
IVD height and vertebral body height were measured from caudal disc levels 5/6, 6/7, and 7/8 and averaged per specimen. Measurements were taken from anterior-posterior tail digital radiographs (UltraFocus, Faxitron) using established protocols. 22 DHI was calculated from IVD and adjacent vertebral body measurements, as previously described. 23
Histomorphological Scoring
Histomorphological changes were assessed in lumbar disc levels 1/2-3/4 from specimens whose caudal discs underwent biomechanical testing. Histological preparation was performed in accordance with previously established protocols. 13 In short, samples were fixed in 4% paraformaldehyde at 4 °C overnight, decalcified for 6 weeks in 10% ethylenediaminetetraacetic acid with PBS at a pH of 8.0, embedded in paraffin, sectioned at 6 µm, and stained with hematoxylin and eosin (H&E). Histomorphological scores were generated following newly developed scoring system, which characterizes structure, cellular infiltration, extent of mineralization, and evidence of clefts/fissures.13,24 The parameters evaluated were the following: NP structure (0 points, single-cell mass; 1 point, cell clusters <50%; 2 points, cell clusters >50%; 3 points, matrix-rich with little cells NP; 4 points, mineralized NP), NP clefts/fissures (0 points, none; 1 point, mild; 2 points, severe), AF/NP boundary (0 points, clear cut boundary; 1 point, round chondrocyte cells at the boundary; 2 points, loss of boundary), AF structure (0 points, concentric lamellar structure; 1 point, serpentine, widened, or rounded AF lamellae; 2 points, reversal of lamellae; 3 points, undefinable lamellar structure or penetrating the NP; 4 points, mineralized or lost AF), and AF clefts/fissures (0 points, none; 1 point, mild; 2 points, severe). For histological scoring, 6 Tnmd−/− and 6 WT animals (3 IVDs/animal and 1 tissue section/IVD) were used. For the Tnmd−/−/Chm1−/− group, 5 animals (1 IVD/animal and 1 tissue section/IVD) were used.
Biomechanical Testing and Parameter Evaluation
Following the acquisition of caudal radiographs, caudal disc level 6/7 was dissected with adjacent vertebrae intact and the excess connective tissue removed ( Fig. 1 ). Mechanical behaviors of WT, Tnmd−/−, and Tnmd−/−/Chm1−/− motion segments were assessed using custom axial (ELF3200, TA Instruments) and torsion (AR2000ex, TA Instruments) test systems following protocols as described previously. 25 WT, Tnmd−/−, and Tnmd−/−/Chm1−/− motion segments were subjected to 20 cycles of axial tension/compression at ±0.5 N at 0.1 mm/second, followed by 45 minutes of creep at −0.5 N ( Fig. 1B ). Following a 5-minute span of unloaded rehydration in a saline bath at room temperature, motion segments were subjected to torsional testing. Torsional testing began with 5-minute equilibration at 0.1 MPa pre-load, followed by 20 cycles of torsion at ±20° cycling at 0.5 Hz, and finally continuous torsion to failure at a rate of 1°/second ( Fig. 1C ).
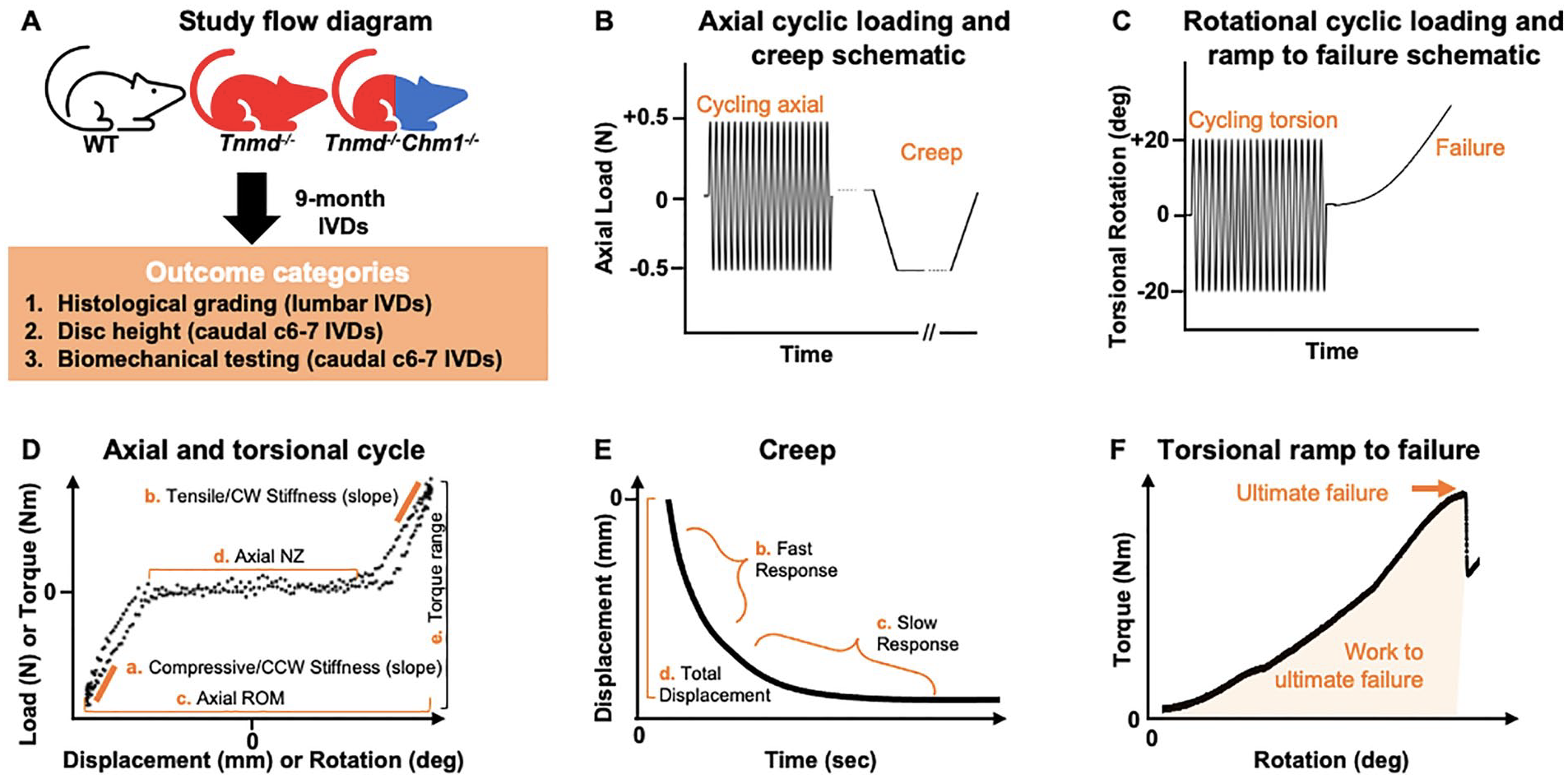
Study flow diagram with accompanying test protocols for axial tension/compression, creep, torsion, and torsional ramp to failure biomechanical parameters on representative loading curves. (
Axial data were analyzed for compressive and tensile elastic zone (EZ) stiffness, range of motion (ROM), and neutral zone (NZ) length from the 20th cycle using a custom MATLAB program 26 (MathWorks; Fig. 1D ). Compressive and tensile EZ stiffness were calculated as a linear fit to 90% to 100% of maximum load when plotted against displacement. Axial ROM was calculated as the total displacement from compression to tension. Axial NZ length was defined as the portion of the load-displacement curve where motion was produced with minimal resistance 27 and was measured from the load versus displacement curve using a custom MATLAB script, employing the double sigmoid method.25,26,28
Torsional data were analyzed for torque range and torsional stiffness from the 20th cycle ( Fig. 1D ). Torsional stiffness in the clockwise (CW) and counterclockwise (CCW) directions was averaged to obtain a single value. Torsional stiffness was calculated as a linear fit to 90% to 100% of the loading direction torque versus rotation curve and reported as an average of CW and CCW directions. Torque range was calculated as the total torque developed from fully CW to fully CCW rotations.
Creep data were analyzed for elastic response stiffness (Se), fast response stiffness (S1), slow response stiffness (S2), fast time constant (τ1), slow time constant (τ2), and total creep displacement ( Fig. 1E ), as previously described. 29
Torsional failure data were analyzed for ultimate failure torque, work to ultimate failure, and ultimate failure degree ( Fig. 1F ). These metrics were recorded at the instant where ultimate failure occurred based on rotation versus torque plots, using previously characterized definitions constituting a failure event. 30 The failure event was therefore not terminated based on visual cues, but rather permitted to complete its scheduled protocol since visual inspection was unreliable in pilot test runs. All values from these biomechanical tests were calculated using a custom MATLAB program. 26
Statistics
One-way ANOVA with post hoc Bonferroni correction for multiple group comparisons determined the role of Tnmd−/− and Tnmd−/−/Chm1−/− on DHI, histomorphological score, and biomechanical parameters with P < 0.05 denoting statistical significance. Outliers were removed using MATLAB if distributing beyond 3 scaled median absolute deviations from the median. Prism (GraphPad) and MATLAB were used to generate figures.
Results
DHI in Caudal IVDs Was Increased for Tnmd−/−/Chm1−/− Mice
No differences were found for caudal vertebral body height across all 3 groups, and no differences were found in caudal IVD height and DHI between WT and Tnmd−/− specimen. Tnmd−/−/Chm1−/− specimens resulted in significantly larger caudal IVD height and DHI compared to WT and Tnmd−/− groups ( Fig. 2 ).
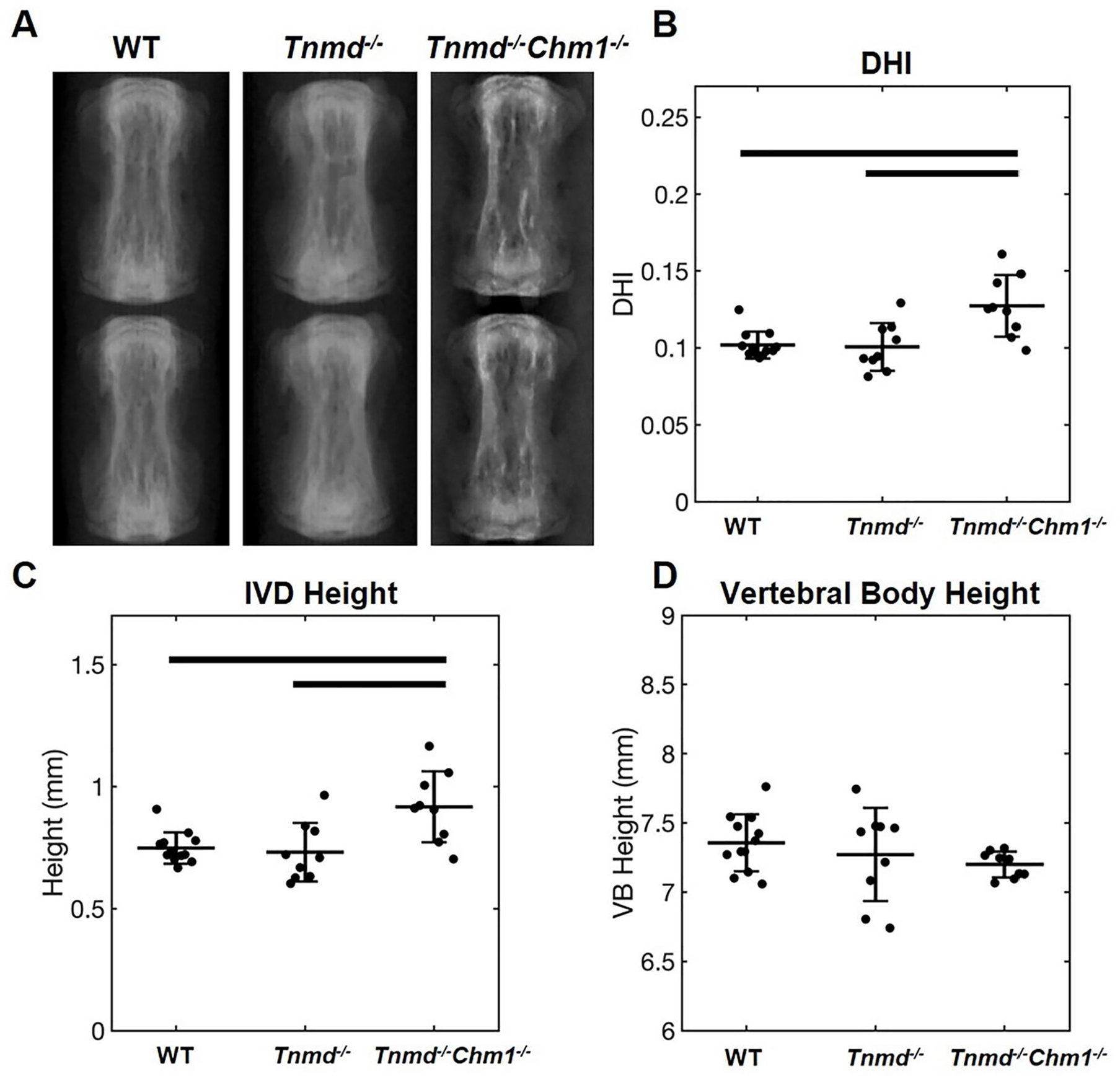
Effect of Tnmd−/− and Tnmd−/−/Chm1−/− on DHI in skeletally mature caudal IVDs. (
Histomorphological Degeneration Scores in Lumbar IVDs Were Increased for Chm1−/−/Tnmd−/− Mice
Histomorphological scores quantified NP and AF features by aggregating IVD characteristics, including cell clusters, extent of mineralization, clefts/fissures, and lamellar structure of 9-month-old lumbar IVDs. The WT group had the lowest score, with histological degeneration progressively increasing when knocking out Tnmd alone (P = 0.08) and increasing further when both Tnmd and Chm1 (P = 0.001) were deficient ( Fig. 3 ). When compared directly to the WT by t-test, the single knockout was significantly increased (P = 0.019). H&E staining revealed fewer NP cells and matrix concomitant with poor lamellar structure penetrating the NP in Tnmd−/− IVDs. In Tnmd−/−/Chm1−/− mice, more severe IVD degeneration, associated with larger NP penetration, AF fissures, and loss of AF lamellar morphology, was observed.
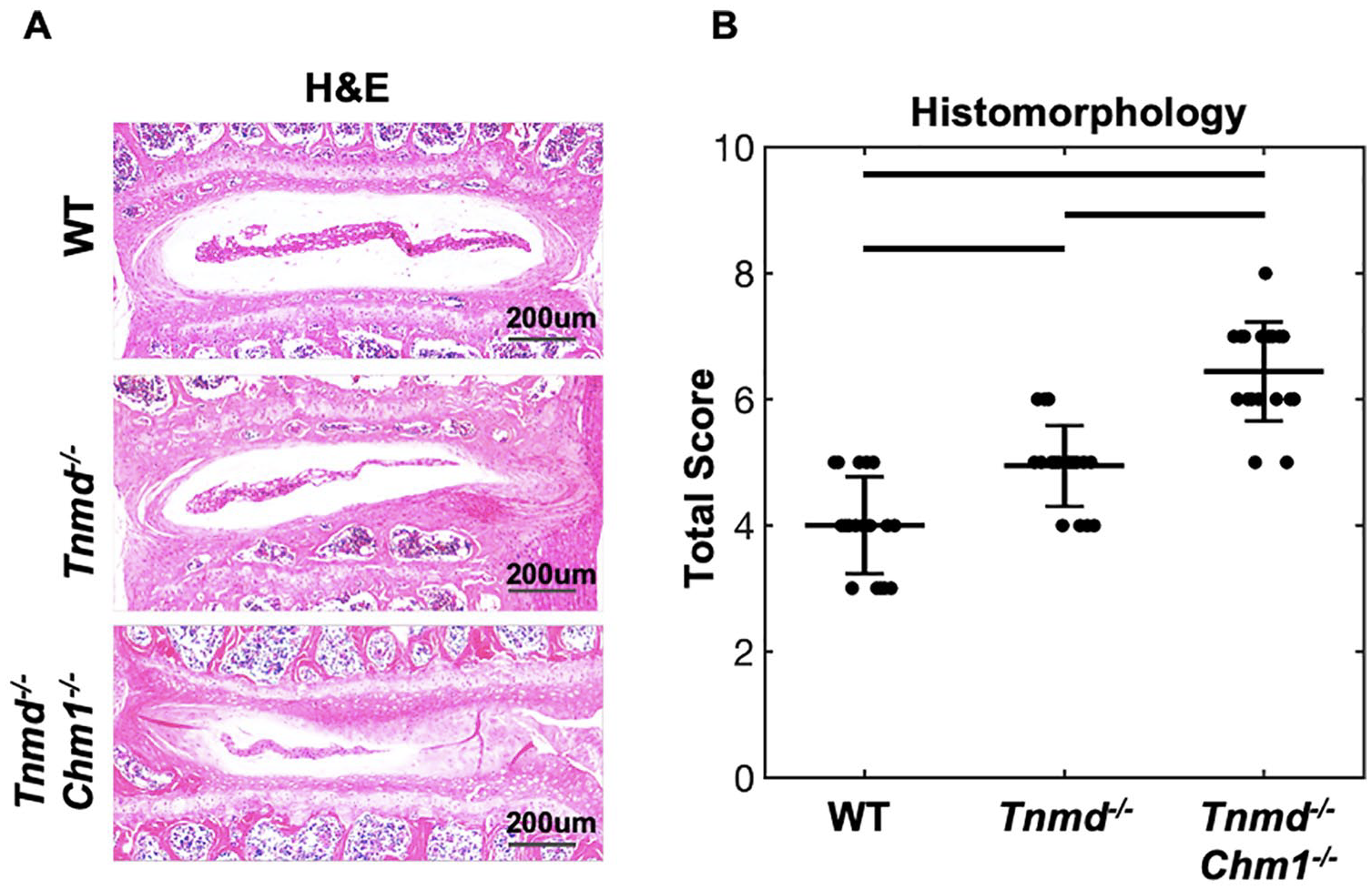
Histomorphological scoring of WT, Tnmd−/−, and Tnmd−/−/Chm1−/− lumbar IVDs. (
Axial and Torsional Biomechanical Function of Caudal IVDs Was Affected More by Tnmd−/−/Chm1−/− Than Tnmd−/− Alone
Axial compressive stiffness, but not tensile stiffness, was significantly increased for Tnmd−/−/Chm1−/− compared to WT and Tnmd−/− ( Fig. 4A and B ). Axial ROM of Tnmd−/− and Tnmd−/−/Chm1−/− were decreased relative to WT, although it did not reach statistical significance (P = 0.05) ( Fig. 4C ). Axial NZ and torsional parameters were unchanged between groups ( Fig. 4D-F ).
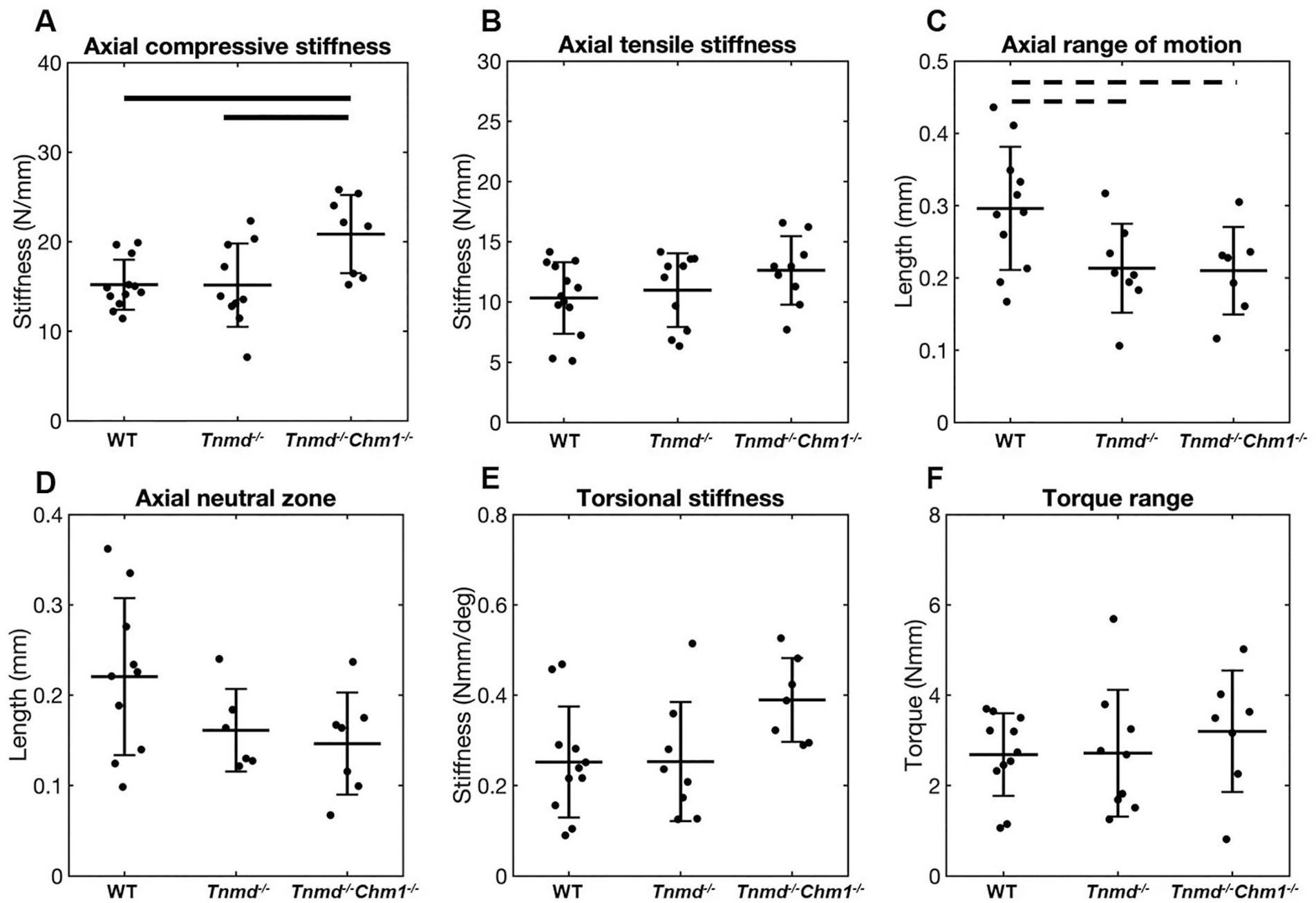
Effect of Tnmd−/− and Tnmd−/−/Chm1−/− on axial and torsional parameters. (
Creep Testing of Caudal IVDs Determined Slow Creep Time Constant Was Affected by Tnmd−/−/Chm1−/−
Creep stiffness parameters Se, S1, and S2, were unchanged between groups ( Fig. 5A-C ). Slow creep time constant (τ2) was significantly decreased in Tnmd−/−/Chm1−/− and decreased (P = 0.05) in Tnmd−/− IVDs, while the fast time constant (τ1) was unchanged between groups ( Fig. 5D and E ). Total disc creep displacement was similar between all groups (WT: 0.16 ± 0.03 mm; Tnmd−/−: 0.17 ± 0.01 mm; Tnmd−/−/Chm1−/−: 0.17 ± 0.01 mm) ( Fig. 5F ).
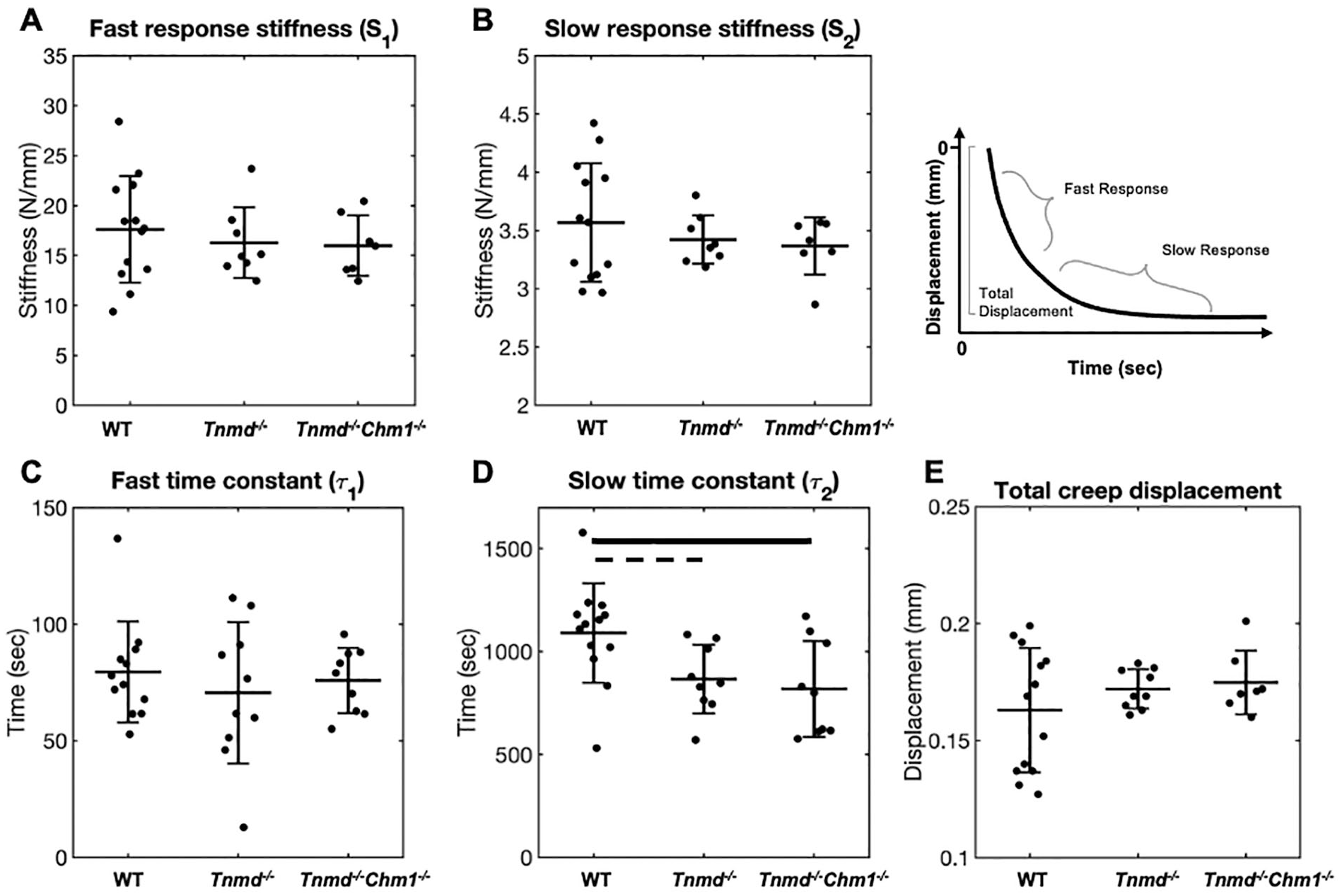
Effect of Tnmd−/− and Tnmd−/−/Chm1−/− on creep parameters. (
Torsional Failure Properties of Caudal IVDs Were Affected by Tnmd−/−/Chm1−/−
Tnmd−/−/Chm1−/− IVDs had decreased ultimate failure torque, work to failure, and degree at which failure occurred when compared to WT and Tnmd−/− groups ( Fig. 6A-C ). Tnmd−/−/Chm1−/− IVDs failed at a lesser degree of rotation, requiring significantly less torque and energy (work) to cause failure. By allowing the failure protocol to run to completion, gross inspection captured failure which occurred mostly at the endplates and at the outer AF in the mid-disc axial plane ( Fig. 6D ). The single Tnmd−/− IVDs did not demonstrate differences in torsional failure parameters compared to WT IVDs ( Fig. 6A-C ).
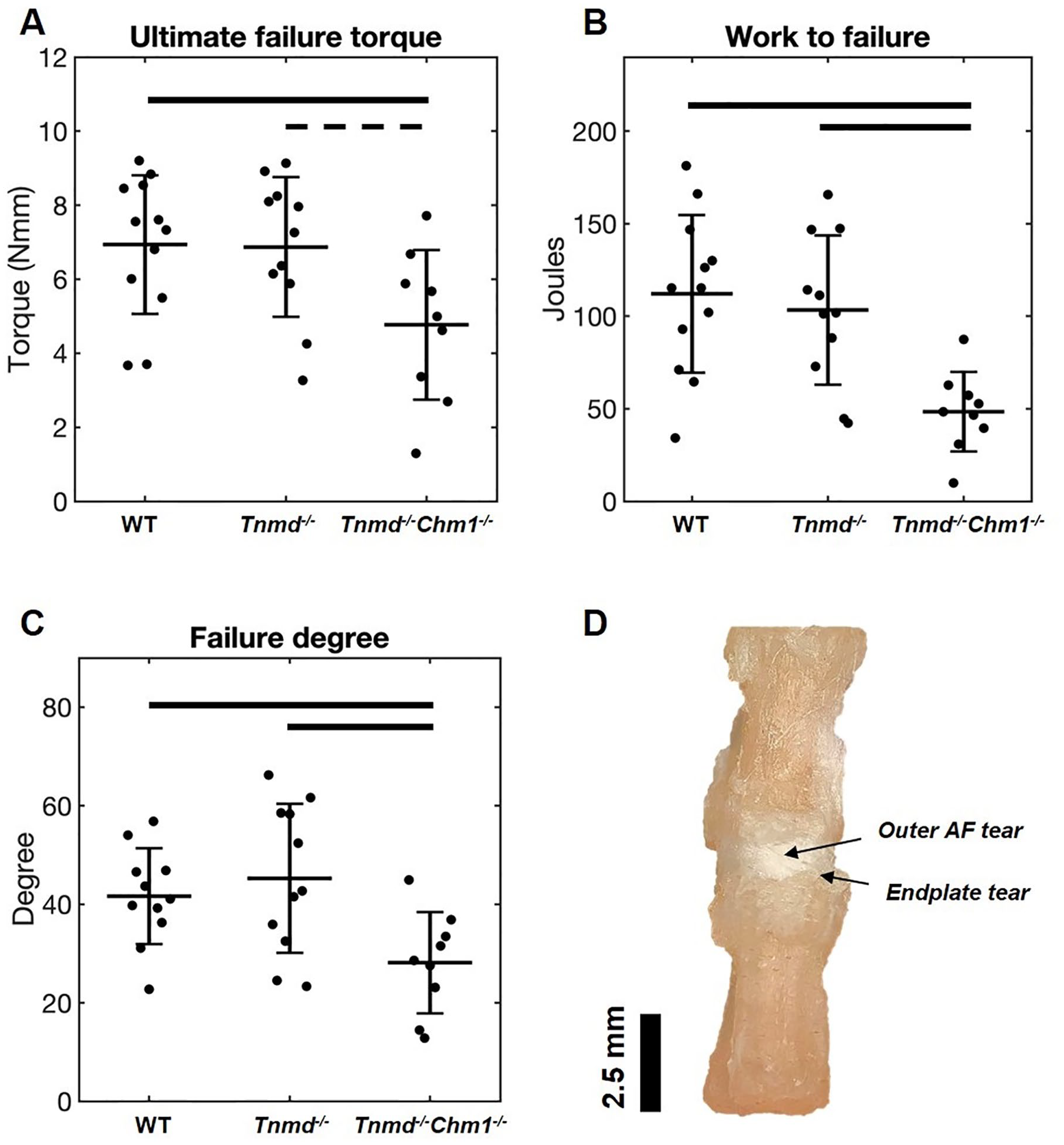
Effect of Tnmd−/− and Tnmd−/−/Chm1−/− on torsional failure parameters. (
Discussion
In order to determine the contribution of Tnmd and Chm1 on organ-level IVD motion segment function we characterized DHI (caudal IVDs), IVD histomorphology (lumbar IVDs), and biomechanical parameters (caudal IVDs) in WT, Tnmd−/−, and Tnmd−/−/Chm1−/− mice. Double knockout Tnmd−/−/Chm1−/− IVDs displayed significantly increased DHI, histomorphological degeneration scores, and axial compressive stiffness, and a decrease in creep slow time constant (τ2) and all torsional failure parameters. Single knockout Tnmd−/− IVDs were minimally impacted across these variables, suggesting Chm1 compensates for IVD organ-level biomechanical function when Tnmd is absent. The reduced torsional failure strength and decreased τ2 suggest AF collagen disruption was enhanced in the double knockout compared to the single knockout, and results suggest collagen disruption led to endplate contact which increased compressive stiffness. IVDs lacking Tnmd and Chm1 were previously shown to accelerate degenerative processes as measured by molecular markers, 13 and we now add to this knowledge by demonstrating that their deficiency increased degenerative processes with diminished inferior load carriage and fluid transport.
Viscoelastic compressive creep findings showed that the slow time constant τ2 was decreased in Tnmd−/− and Tnmd−/−/Chm1−/− mice, while other creep variables were unaffected ( Fig. 5 ). Decreased τ2 is primarily associated with increased fluid transport out of the IVD due to increased tissue permeability. 29 Creep loading results depend on preconditioning, which was satisfied by our axial cyclic loading test prior to the creep test. Furthermore, achieving intradiscal pressure equilibrium in murine specimen occurs after approximately 20 minutes, granting confidence in our slow time constant results.21,31 We propose that Tnmd and Chm1 deficiency increases fluid transport due to disrupted AF collagen structure. This inference is based on the findings that the Tnmd protein co-localizes with type I collagen, 32 where Tnmd deficiency caused decreased AF collagen fibril diameter, 13 and decreased collagen fibril diameter is associated with increased fluid transport. 33 In tendons, lack of Tnmd also decreased expression of genes related to collagen crosslinking, 32 where decreased crosslinking also correlated with decreased flow resistance. 34 Consequently, the faster fluid transport out of the IVD (as evidenced by decreased τ2) may also have resulted from disrupted AF collagen crosslinking from Tnmd deficiency. In human, fluid permeability is reported to be unaffected, increased, and decreased by IVD degeneration.29,35-37 These competing findings may be explained by the various degenerative stages, defects, and disease conditions that accumulate over decades in humans 38 and can differentially influence fluid transport and biomechanical function. 39 Taken in the context of this broader literature, we infer that even relatively minor collagen disruptions as might occur from Tnmd and Chm1 deficiency can diminish creep behaviors and alter load carriage that may accumulate in degenerative changes. We also conclude that Tnmd and Chm1 deficiency disrupted AF collagen fibril network, which resulted in more rapid fluid outflow during compressive creep.
Tnmd and Chm1 deficiency increased the histomorphological degenerative grade of lumbar IVDs and altered DHI of caudal IVDs. In humans, IVD height loss is an early sign of degeneration that can occur from alterations in AF integrity or NP pressurization.9,39 Pro-angiogenic factors increase under pathological conditions, encouraging IVD vascularity and calcification of cartilaginous tissues. 40 Tnmd and Chm1 are known anti-angiogenic proteins, and therefore increased histomorphological scores (via increased mineralization) corroborates their roles as potential affecters of early degenerative processes. The increase in score compared to the WT when Tnmd alone was knocked out was not statistically significant due to corrections from multiple pairwise analyses. However, a direct comparison between Tnmd−/− and WT parallels results previously reported in 6- and 12-month-old mice showing increased degenerative scores secondary to Tnmd deficiency. 13 In the same study, DHI measurements using histological sections showed decreased DHI of lumbar IVDs in Tnmd−/− IVDs. However, this current study shows increased DHI in Tnmd−/−/Chm1−/− IVDs. These contradictory findings are most likely related to differences in spinal levels used and specimen preparation differences. Lumbar segments were used in a prior study, 13 where histological preparation dehydrates tissues, possibly causing endplates to converge and leading to decreased DHI, especially if collagen is disrupted. The current study measured caudal IVD and vertebral dimensions in whole mouse tails via radiographs where saline-soaked tissues that maintained hydration conditions can cause swelling during specimen handling and imaging, especially if collagen is disrupted. It is also possible that collagen disruption would cause an increase in DHI in caudal levels and decrease in DHI in lumbar levels in vivo because peak forces are lower on caudal levels than on lumbar spinal levels. Since both studies and methods are well controlled, we conclude that both studies point to collagen disruption that influence DHI.
This study demonstrated that Tnmd−/−/Chm1−/− IVDs had significantly increased axial compressive stiffness, which is commonly associated with NP pressurization but can also be influenced by changes in AF collagen. 9 Loss of proteoglycans can result in NP depressurization, decreased IVD height, and altered axial biomechanical properties.9,41,42 However, Tnmd is primarily expressed in the outer AF 13 without a known influence on proteoglycans in tendons. 17 The increased compressive stiffness in this study may therefore be attributed to sudden stiffening from endplate-to-endplate contact due to AF collagen disruption. Premature endplate-to-endplate contact occurs with NP depressurization, which can happen in the setting of disrupted collagen integrity, resulting a sudden load increase when endplates contact. The axial test protocol used in this study involved displacement-control to a maximum load-limit, which can result in increased stiffness as endplate contact generates the load-limit with lower deformation. Our chosen schema provided the highest possible sensitivity to detect an effect and was deemed appropriate given that 75% of the spine biomechanics community agrees that the loading schema should be applied based on the research question. 21 We note that micro-scale modulus measured with atomic force microscopy was decreased in Tnmd−/− IVDs compared to WT IVDs and this test more directly measures the local extracellular matrix without interacting structures that comprise the motion segment. 13 We therefore attribute premature endplate-to-endplate contact as the mechanism responsible for our axial compression organ-level results.
The torsion to failure biomechanical test identified that significantly reduced torque and work were required to generate a failure when both Tnmd and Chm1 were deficient. Torsional parameters are highly sensitive and specific to AF disruption, 30 and torsional failure in this study occurred in the outer AF and at its endplate insertion. The AF contains primarily a type 1 collagen network, which is responsible for resisting motion segment torsion. 43 Tnmd is predominantly expressed in the outer AF, 13 whereas Chm1 is mostly localized to the NP but influences cell populations of the NP, AF, and endplate. 13 Torsional failure properties of this study were only affected by the double knockout, suggesting that Chm1 influences cell populations involved in AF lamellar architecture and can maintain AF structural integrity in the absence of Tnmd. It was somewhat surprising that torsional failure properties were diminished while torsional stiffness was not affected, and we attribute this to the complex lamellar architecture of the AF that permits redundancy in lamellar layers. 44 Furthermore, it highlights that Tnmd and Chm1 cause relatively minor alterations that were most detectable when forcing rotation beyond 20° (which was the maximum rotation during cyclic torsion testing).
The use of mice models enables genetic modifications, and mice and human IVDs have similar axial and torsional mechanical properties when normalized by size.45,46 Organ level biomechanical changes were sensitive enough to detect collagen changes in Tnmd−/−/Chm1−/− but not Tnmd−/− which was previously reported to have collagen disruption detectable with micro-scale mechanics measured with atomic force microscopy. 13 Motion segment tests evaluate functional biomechanical changes involve multiple interacting structures but have less sensitivity than atomic force microscopy measurements, which are highly sensitive to biomechanical changes on the micro-scale and can be localized to AF extracellular matrix. While we can conclude that Tnmd and Chm1 play interacting roles, this study did not have a single Chm1 knockout subgroup, which prevents making conclusions on the synergistic, permissive, or independent roles of Chm1 and Tnmd.
This study identified that Tnmd and Chm1 are both required to maintain IVD organ-level biomechanical integrity. Tnmd homolog Chm1 may serve a compensatory role, evidenced by our finding that only the double knockout genotype generated biomechanical phenotypic changes. Tnmd and Chm1 had direct effects on early IVD degeneration, increased fluid transport, and outer AF collagen disruption. Future studies are warranted to identify the directionality of the relationship between these proteins that contribute to the AF phenotype. These factors may also serve as biomarkers of IVD degeneration or help inform AF repair strategies.
Supplemental Material
sj-pdf-1-car-10.1177_19476035211029696 – Supplemental material for Tenomodulin and Chondromodulin-1 Are Both Required to Maintain Biomechanical Function and Prevent Intervertebral Disc Degeneration
Supplemental material, sj-pdf-1-car-10.1177_19476035211029696 for Tenomodulin and Chondromodulin-1 Are Both Required to Maintain Biomechanical Function and Prevent Intervertebral Disc Degeneration by Theodor Di Pauli von Treuheim, Olivia M. Torre, Emily D. Ferreri, Philip Nasser, Angelica Abbondandolo, Manuel Delgado Caceres, Dasheng Lin, Denitsa Docheva and James C. Iatridis in CARTILAGE
Footnotes
Author Contributions
Theodor Di Pauli von Treuheim: Planned and conceptualized the study, wrote MATLAB program, analyzed and interpreted data, wrote and edited the manuscript, and approved the final version.
Olivia M. Torre: Planned and conceptualized the study, interpreted data, led collaboration communications, and approved the final version.
Emily D. Ferreri: X-ray testing and analysis, wrote and edited the manuscript, and approved the final version manuscript writing.
Philip Nasser: Experimental set up and resource allocation.
Angelica Abbondandolo: X-ray analysis.
Manuel Delgado Caceres: IVD explantation, collaboration, communications, and reviewed, edited, and approved the manuscript.
Dasheng Lin: Histological preparation and assessment.
Denitsa Docheva: Planned and conceptualized the study, interpreted the results, and reviewed, edited, and approved the manuscript.
James C. Iatridis: Planned and conceptualized the study, interpreted the results, and reviewed, edited, and approved the manuscript.
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by R01AR057397 (JCI) from NIH/NIAMS and the EU Twinning Grant ACHILLES (H2020-WIDESPREAD-05-2017-Twinning Grant No. 810850) (DD). The investigators thank Dr. Zsuzsa Jenei-Lanzl for histological support on lumbar IVD specimens.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for this study was obtained from the Lower Franconia ethics committee.
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
