Abstract
Objective
The aim of this study was to evaluate levels of consensus in rehabilitation practices following MACI (autologous cultured chondrocytes on porcine collagen membrane) treatment based on the experience of an expert panel of U.S. orthopedic surgeons.
Design
A list of 24 questions was devised based on the current MACI rehabilitation protocol, literature review, and discussion with orthopedic surgeons. Known areas of variability were used to establish 4 consensus domains, stratified on lesion location (tibiofemoral [TF] or patellofemoral [PF]), including weightbearing (WB), range of motion (ROM), return to work/daily activities of living, and return to sports. A 3-step Delphi technique was used to establish consensus.
Results
Consensus (>75% agreement) was achieved on all 4 consensus domains. Time to full WB was agreed as immediate (with bracing) for PF patients (dependent on concomitant procedures), and 7 to 9 weeks in TF patients. A progression for ROM was agreed that allowed patients to reach 90° by week 4, with subsequent progression as tolerated. The panel estimated that the time to full ROM would be 7 to 9 weeks on average. A range of time was established for release to activities of daily living, work, and sports, dependent on lesion and patient characteristics.
Conclusions
Good consensus was established among a panel of U.S. surgeons for rehabilitation practices following MACI treatment of knee cartilage lesions. The consensus of experts can aid surgeons and patients in the expectations and rehabilitation process as MACI surgery becomes more prevalent in the United States.
Introduction
MACI (autologous cultured chondrocytes on porcine collagen membrane), a third-generation autologous chondrocyte implantation (ACI) product, was the first Food and Drug Administration (FDA)–approved product that applies the process of tissue engineering to grow chondrocytes on scaffolds using healthy cartilage tissue from the patient’s own knee. 1 ACI, a restorative cellular treatment for symptomatic cartilage defects of the knee, was first described by Brittberg et al. 2 in 1994. First-generation ACI was administered surgically with direct injection of expanded autologous chondrocytes into the cartilage defect secured using a harvested autologous periosteal flap that required extensive suturing and was associated with an increased need for subsequent surgical procedures. MACI improves the delivery of chondrocytes by seeding the chondrocytes onto a stable scaffold trimmed to the size and shape of the cartilage defect that is secured in place using fibrin sealant, thereby eliminating the need for the periosteal flap.
The rehabilitation program post MACI treatment is critical to optimizing good surgical outcomes through protection of the implant while healing. Ability for personalization of rehabilitation strategies is important due to the variability in lesion location, lesion size, need for concomitant surgeries, and patient characteristics such as age and body mass index (BMI). The postoperative ACI/MACI rehabilitation protocol 3 was developed to support the progression of return to full weightbearing (FWB) and range of motion (ROM), as well as muscle strengthening and conditioning, following treatment with ACI/MACI.
The specific timing and steps of the rehabilitation protocol used with first-generation ACI treatment have been adjusted over time with the changeover to third-generation ACI (MACI), largely driven by the extensive work of Ebert and Edwards in Australia.4-10 The rehabilitation procedure following first-generation ACI was lengthy (up to 18 months) due to the requirement of delayed WB to protect the repair site.11,12 The potential for acceleration of the MACI rehabilitation program may be possible due to the advantages of MACI compared with previous generations of ACI in that MACI is the application of a stable scaffold with uniform distribution of cells using simpler technique with smaller incisions. In addition, the more streamlined procedure allowing a reduction of time spent in surgery may potentially have less impact on quadriceps; quadriceps strength deficits have been frequently observed following knee surgery.13-15 Quadriceps weakness may result in decreased tolerance to standing, walking, or running and therefore difficulty returning to previous levels of activity. 16
With the discontinuation of Carticel (first-generation ACI) from the market in 2017 and the expanding use of MACI in the United States since product launch in early 2017, the aim of this study was to evaluate levels of consensus in rehabilitation practices following MACI treatment based on the experience of U.S. orthopedic surgeons. Furthermore, the goal of the study was, with consensus of experts, to address pervasive patient and surgeon questions regarding the rehabilitation process that are common in the realm of articular cartilage treatment and concerns regarding the rehabilitation process following MACI.
Methods
To evaluate current practices of rehabilitation following MACI treatment, we used the modified Delphi technique, a method for structuring group communication among experts to promote the development of consensus-based guidelines. 17 The Delphi survey method is used for collecting opinions from experts by using a number of questionnaire rounds, feedback of responses, and the opportunity for participants to modify their responses. 18 Our approach for the full evaluation required various methods, including review of the literature, interviews with experts, and iterative consensus of an expert panel. There are no defined rules for the size of an expert panel. Linstone et al. 19 suggests a minimum of 7 experts. Orthopedic surgeons with experience performing MACI procedures and knowledge of rehabilitation practices were invited to participate in the 2 consensus surveys and face-to-face meeting.
A list of questions was devised based on the current MACI rehabilitation protocol, a literature review, and input from a smaller panel among the authors. These questions were drafted with the aim of addressing areas of current variability within MACI rehabilitation practices (see Supplemental Appendix 1 for the complete list of questions). Four known areas of variability (domains) related to the stages of rehabilitation following MACI treatment were identified prior to the study based on previous rehabilitation practices following cartilage repair and on studies published in the literature.5,6,8,9 The 4 domains included WB, ROM, return to work, and activities of daily living (ADL), and return to recreational activities and sports ( Table 1 ). Within each of the domains are goal/milestones that may be dependent on lesion and patient characteristics. To assess the impact of patient and lesion characteristics on goals/milestones within each domain, 4 general patient profiles were developed based on common patient complexity scenarios and challenges with the intent of establishing boundaries for the timing of rehabilitation steps for those patients that might be regarded as having a less challenging rehabilitation (i.e., shorter duration to achieve milestone) compared with those with more challenging rehabilitation (i.e., longer duration) due to age, number of lesions, and/or level of activity. The patient profiles were stratified by lesion location (patellofemoral [PF] and tibiofemoral [TF]), and by “less challenging” and “more challenging” patient characteristics of age (24 vs. 50 years) and job-level activity (low vs. high), number of defects (1 and multiple) and lesion size (<3 cm2 and ≥5 cm2) as shown in Table 2 . These scenarios were designed to ascertain the impact of the cartilage lesions only on rehabilitation; therefore, assumptions were made that patients within each profile had normal alignment (i.e., no need for osteotomy), and normal ligament status and meniscus status. The 4 patient profiles were provided to the surgeons along with the survey questions to aid in the consensus process.
Consensus Domains (Milestones).
Patient Profiles (Stratified by Defect Location).

Patients 1 and 3 are considered to have a “less challenging” rehabilitation due to treatment of a single defect, smaller lesion size, younger age, and lower job-level physical activity.
Patients 2 and 4 are considered to have a “more challenging” rehabilitation due to treatment of multiple defects, larger lesion size, older age, and higher job-level physical activity.
A 24-item survey was constructed and disseminated for rounds 1 and 2 of the consensus process by Trinity Life Sciences, Cambridge, Massachusetts (Supplemental Appendix 1). The Delphi scoring method was modified between rounds 1 and 2 by using the same survey questions in each round but redefining the scoring approach (i.e., direct choice of specific times or goals in round 1, to ranking of aggregate scores from round 1 in round 2). Round 3 was used to further narrow areas of agreement and identify continued disagreement where possible.
In round 1, the panel of surgeons received the questions via email and provided responses by direct choice of specific times or goals for each domain. In round 2, conducted by phone interview, aggregate findings from round 1 were shared with each individual panel member who was then asked to rate the level of appropriateness of aggregate responses on a scale from 1 (not at all appropriate) to 9 (extremely appropriate). In round 3, areas of continued disagreement were targeted for an in-person consensus panel. All surgeons participating in rounds 1 and 2 were invited to participate in round 3. A face-to-face consensus conference was held with the expert panel on July 10, 2019.
Following completion of each round, data were exported and analyzed by Trinity Life Sciences, Cambridge, Massachusetts. Consensus was defined as a minimum of 75% agreement by the surgeons. For consensus in round 3, consensus was obtained by the moderator by verbal acceptance/lack of acceptance (yes or no) at the end of the discussion of each subpart of each domain. The moderator ensured that at least 6 of the 8 panel members (75%) were in agreement with the recommendations before moving to discussion of the next domain.
Characteristics of Expert Panel
Vericel has a record of the number of patients treated by each orthopedic surgeon per year due to the autologous nature of the product. The surgeons invited to participate had treated at least 10 patients per year with MACI. Twelve surgeons agreed to participate in rounds 1 and 2 of the Delphi Survey. Of the 12 surgeons, 25% (3/12) had <10 years in practice, 58% (7/12) had between 10 and 20 years, and 17% (2/12) had ≥30 years in practice. All but one surgeon had sports medicine fellowship training. The majority of surgeons, 75% (9/12), practiced in an academic setting and 25% (3/12) practiced in private or community health settings. The surgeons learned the MACI surgical technique through a range of training types, including their residency, peers, fellowship, and company-sponsored training. The self-reported percentage of patients treated with MACI among each surgeon’s patients with cartilage injuries requiring surgery ranged between 10% and 75% per year, with an average of 40 patients (range 10-70) treated with MACI per year. Eight of the 12 surgeons were available to participate in the face-to-face (round 3) consensus meeting.
Characteristics of MACI Patients Treated by Surgeon Participants
The majority of MACI patients treated by the surgeon participants are typically 25 to 55 years old with moderate to high activity level. The most common defect characteristics treated by surgeons were single defect lesions size 3 to 5 cm2 and the patella was the most common location.
Results
Overview of Rounds (All Domains)
In all domains, the variability of the approach to rehabilitation following MACI treatment was reflected in the survey results of rounds 1 and 2, where the distributions of responses were clustered around specific timepoints but did not demonstrate a clear consensus for any of the timepoint variables in any of the domains. Therefore, for the face-to-face meeting (round 3) lead by a trained moderator from Trinity Life Sciences, the moderator focused on the areas where the responses were clustered to obtain agreement with the panel of surgeons. The following sections outline the discussion that occurred during round 3 by domain, followed by a tabular presentation of the results for each domain variable. The tables presented for each domain (
Tables 3
Consensus on Weightbearing: Summary Following Round 3.
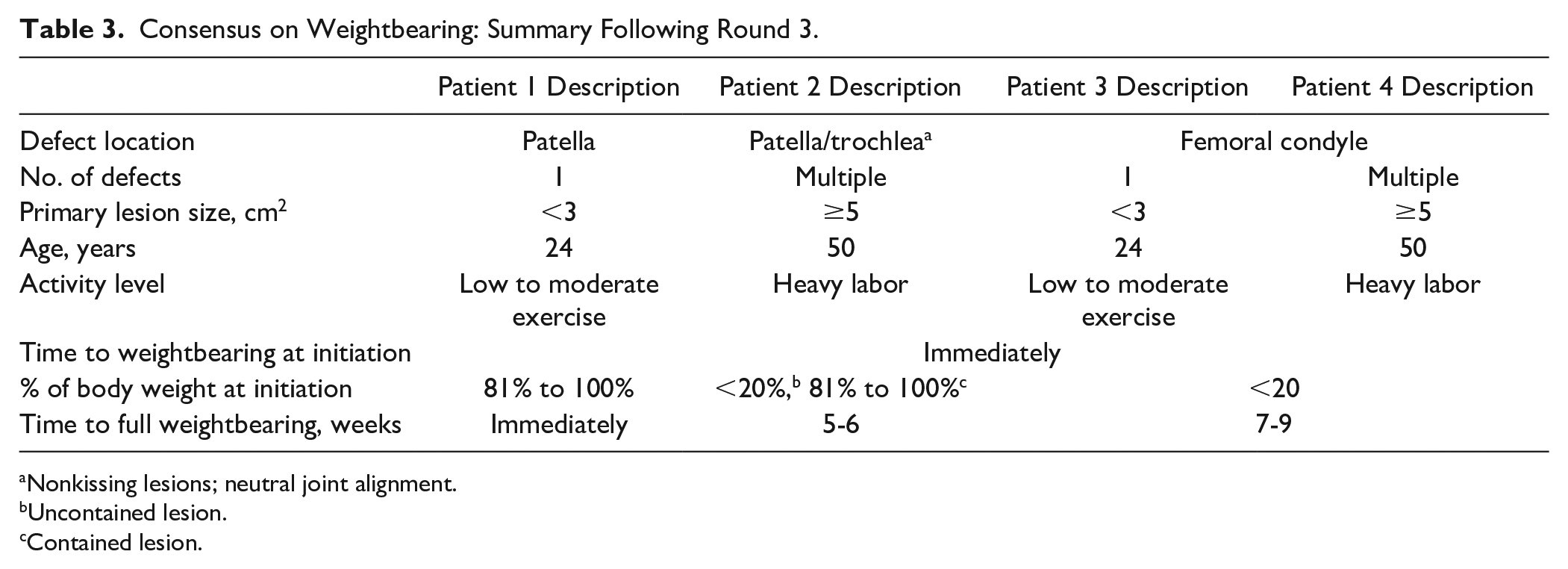
Nonkissing lesions; neutral joint alignment.
Uncontained lesion.
Contained lesion.
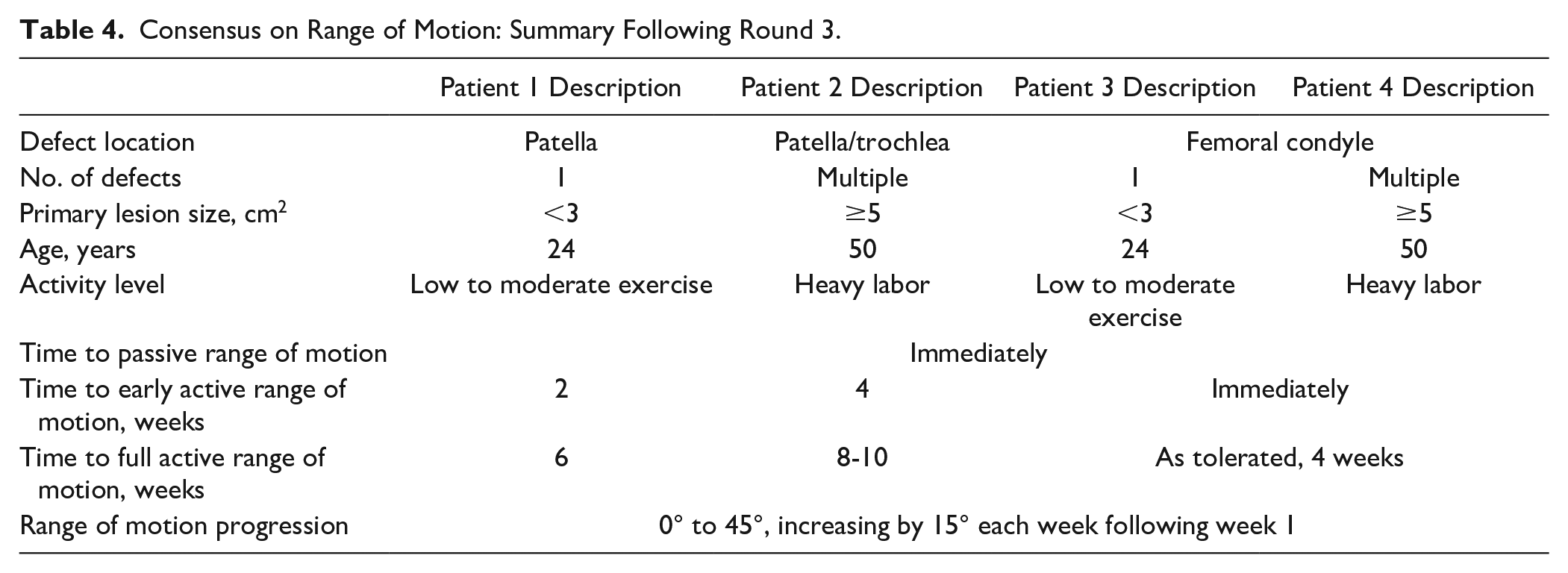
Consensus on Range of Motion: Summary Following Round 3.
Consensus on Work and Activities of Daily Living (ADL): Summary Following Round 3.
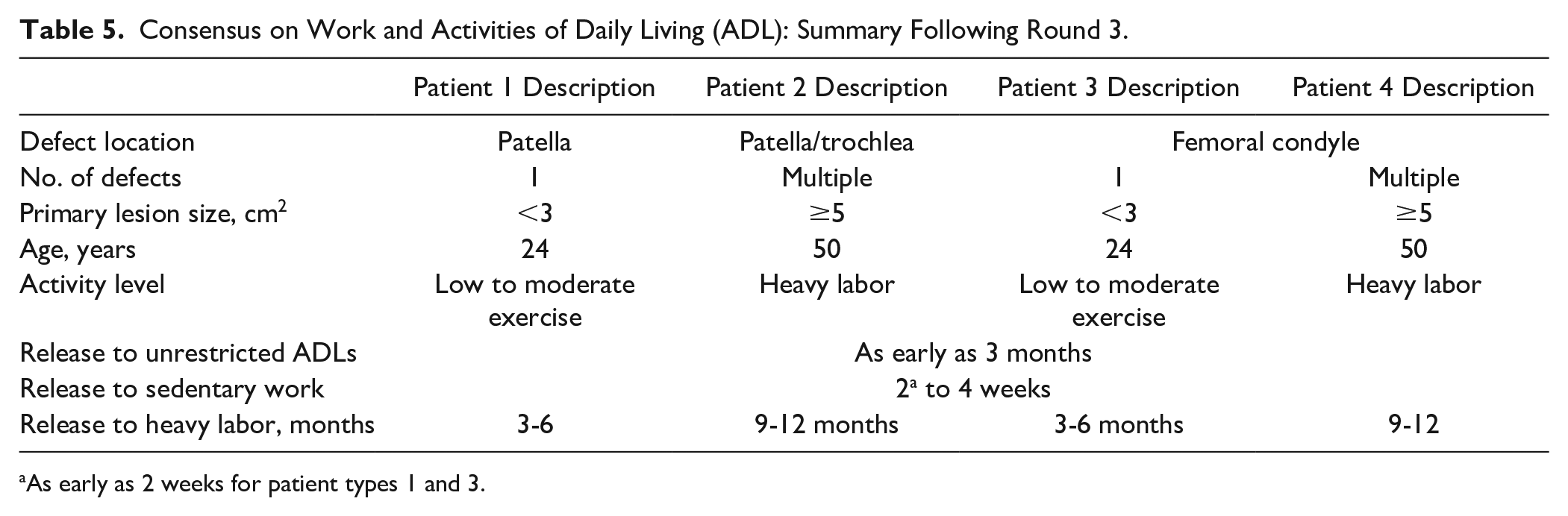
As early as 2 weeks for patient types 1 and 3.
Consensus on Return to Recreational Activities and Sports: Summary Following Round 3.
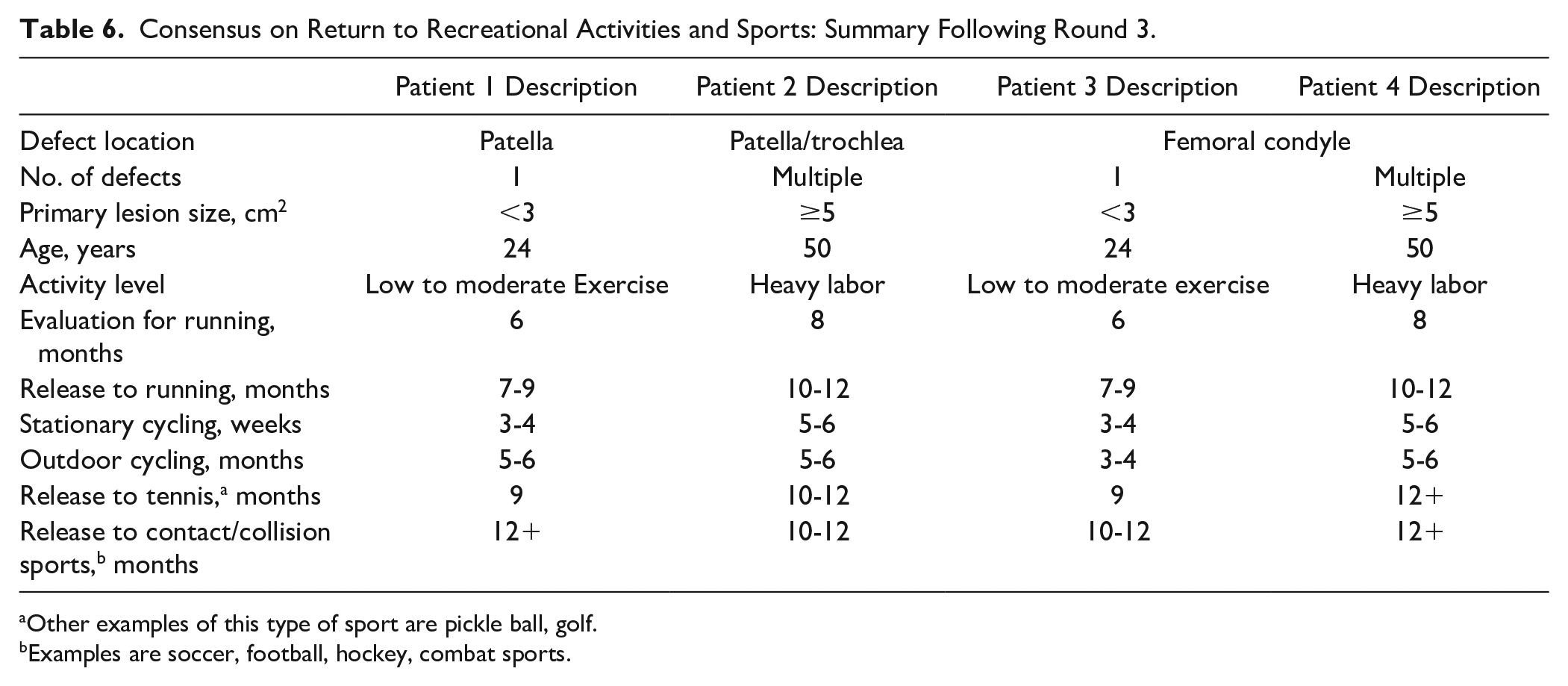
Other examples of this type of sport are pickle ball, golf.
Examples are soccer, football, hockey, combat sports.
Weightbearing Consensus
Consensus was achieved for the timing of initiation of WB, percentage of body weight at initiation, and for the time to FWB ( Table 3 ).
Round 3 Discussion
Following the face-to-face meeting, consensus (≥75% agreement) was obtained on all variables within the WB domain as shown in Table 3 . During the meeting, it was agreed that time to the initiation of WB could begin immediately for all patients as long as there was adjustment to the percentage of body weight at initiation, dependent on patient and lesion factors. Factors to take into consideration across patient types included
Well-contained patella/femoral trochlea lesions could be initiated at 81% to 100%.
Lesions poorly contained would be reduced to initiate WB using flat foot up to 20% body weight. In addition, if any of the lesions were poorly contained, the overall timing of WB may have more extended timelines.
Lesion location and concomitant surgeries were seen as the most important patient factors determining rehabilitation decision-making when it comes to WB. All surgeons indicated that patient 2 would often receive an osteotomy, which would extend the timing of many of the rehabilitation steps. Recommendations by the expert panel did not factor in the extended rehabilitation timelines in case of additional osteotomy procedures such as high tibial or distal femoral osteotomies.
Milestones of gaining normal gait pattern and confidence walking without crutches were seen as the most important milestones to move from initial to full WB. It is important to note that “improved pain” was not included in the list of important milestones since it was felt that this variable was obvious in terms of high importance, but surgeons indicated that they would have included it in the list of variables.
Range of Motion Consensus
Consensus was achieved on all variables within the ROM domain as shown in Table 4 .
Round 3 Discussion
During the face-to-face meeting, the panel discussed the definition of passive versus active ROM. Early active ROM was defined by the surgeons as short arc (0° to 30°). Passive ROM should be initiated across patient types immediately following surgery with more intense (active) ROM initiated over time. Passive ROM was defined as continuous passive motion (CPM) machine or manual passive motion performed by a rehabilitation specialist. Specific recommendations for bracing (locked and unlocked) were discussed; however, there was variability in opinion between the surgeons. Active ROM will vary across patient types and concomitant procedures associated with the lesion. All active ROM milestones and timeframes can progress so long as patients are progressing without any red flags such as swelling, catching, locking, or pain. The presence of any of these red flags requires slower progression to full ROM. There should be a potential to define specific criteria as part of rehabilitation guidelines that will allow patients to further progress once the criteria are met. However, for simplicity it was agreed by the panel that across all patient types (even with concomitant osteotomies), a stepwise progression from 45°, increasing by 15° each week would make sense. A standardized progression of ROM allows guidelines to be straightforward and physicians to follow them easily, while still being adjustable depending on the patient. At a patient’s initial (e.g., 2 weeks postoperative) visit, physicians can evaluate patient’s progress and make adjustments to ROM progression as necessary. Progression allows patient to reach 90° by week 4, at which point patients can continue progression as tolerated. The panel estimated that the time to full range of motion would be 7 to 9 weeks on average.
Work and Activities of Daily Living Consensus
Consensus was achieved on all variables within the Work and ADL domain as shown in Table 5 .
One of the goals of the Delphi consensus survey was to obtain more specific information on the return to work and the return to ADL for inclusion in the MACI Rehabilitation Manual. 3
Round 3 Discussion
During the face-to-face meeting, discussion revolved around level of activity that is performed in the patient’s specific job responsibilities; the ranges of time (in months) would be dependent on whether a patient had a fully sedentary job, a job that required some standing or extended standing, or one that required more twisting/turning and exertion. In addition, transportation issues were discussed as well as progression of the patient’s overall rehabilitation program; however, no recommendations were made regarding transportation issues in the discussion. Some patients (i.e., patient types 1 and 3) may be able to return to work at the early end of the range (i.e., 2 weeks), then increase time spent at work and return to their full job duties by the end of the 1-month range. Returning to a normal daily routine and ADLs were seen as the most important milestones, with number of concomitant surgeries and lesion size seen as the most important patient factor in making decisions on returning to work and ADLs.
Return to Recreational Activities and Sports
Another goal of the Delphi consensus survey was to obtain more specific information on the return to recreational activities and sports. Consensus was achieved on all variables of return to recreational activities and sports and is shown in Table 6 .
Round 3 Discussion
During the face-to-face meeting, surgeons noted that in order for time frames to be relevant for individual patients, patients must reach required goals/milestones during the evaluation phase. Most likely, the physician for patient 4 would recommend not to return to running if the patient had multiple lesions, but they might be able to achieve a modified running program (e.g., fewer miles) within 10 to 12 months on meeting criteria. For recreational activities (e.g., local gym), the panel felt that this question was too broad given the wide range of activities performed at a gym and therefore could not reach consensus for this milestone (data not shown). For stationary cycling (low intensity; not a workout), surgeons felt some patient types (patients 1 and 3) may be able to return in 3 to 4 weeks but most patients would take 5 to 6 weeks to return to stationary cycling. Return to recreational exercise activities and return to preinjury activities were seen as the most important milestones for returning to sports, and lesion size and number of concomitant surgeries were the most important factors determining return to recreational activities and sports.
Discussion
The rehabilitation program following MACI treatment is critical to optimizing surgical outcomes through protection of the implant and physiological healing. The change from first to third-generation ACI (MACI) has allowed for more accelerated rehabilitation, supported by studies described in the literature.4-10 To determine levels of consensus in rehabilitation practices based on the experience of U.S. orthopedic surgeons with MACI, we used a modified Delphi technique for the development of consensus-based guidelines. The goal of this study was to refine the timing for mechanical/functional milestones, including WB and ROM, and to provide information regarding personal milestones such as timing for return to work and sports. The most important finding of this study was that relatively good consensus among U.S. surgeons of rehabilitation practices was established for MACI treatment of knee cartilage lesions.
The treatment and rehabilitation of patients with cartilage lesions is complex and needs to be carefully planned and often individualized based on patient and lesion characteristics. Interestingly, while executing the Delphi study, we observed that surgeon responses from the expert panel reflected evaluations reported in the literature in that responses tended to cluster around a range of timepoints depending on whether they took a conservative or more aggressive approach to the timing associated with rehabilitation. This observation is similar to one regarding a survey of post ACL rehabilitation practices in Australia by Ebert et al., 20 where responses revealed a range of differences within the group. In our study, these differences may be potentially due to each surgeon’s experience with MACI (e.g., number and types of patients) during the short time since approval in the United States, and/or biases from the previous more conservative approach to rehabilitation practices following treatment with ACI. Therefore, the modified Delphi approach worked well in this study to allow consolidation of the timing of milestones between rounds, and then gaining agreement (≥75%) in the face-to-face meeting in round 3.
For mechanical/functional milestones of WB and ROM, in general, current rehabilitation practices following treatment of a PF lesion allows a faster progression of WB in the extension position but requires a slower progression of ROM compared to TF repairs. 21 Similar to Ebert et al., 20 in patients with TF lesions there was consensus for immediate WB using a stepwise WB approach. In randomized controlled trials, this has been shown to be safe and effective, and allows for earlier return of normal gait pattern and therefore muscular function and control.8,10,22 Interestingly, there was also consensus in PF lesions for immediate full WB in locked brace initially. This is a significant difference from historical reports in the literature of first generation ACI where the challenge of delayed WB was noted for both PF and TF lesions.11,12 For time to full WB consensus was obtained for both TF (7-9 weeks) and PF lesions (immediate dependent on containment and concomitant procedures). Consensus by the panel on milestones for ROM were also obtained with time to early active ROM immediate for TF patients (full active 6-10 weeks) and 2 to 4 weeks for PF patients (full active as tolerated at 4 weeks). For TF patients this allows early increases in joint motion with slower interval increases in WB, and for PF patients this approach allows for immediate WB with the knee in full extension or restricted ROM. These rehabilitation steps provide protection dependent on location of lesions (TF vs PF) but allow for mobilization of the knee following surgery since even short-term immobilization may have detrimental effects and may induce articular cartilage atrophy.23-25 In addition, early mobilization enhances metabolic and nutritional activity of the cartilage and prevents joint stiffness.26,27
Return to normal activities and work are pivotal steps in a patient’s recovery. There are few studies of the timing of return to ADL and/or work following ACI/MACI published in the literature, and there is limited information for the type and demand of the work. 28 Therefore, obtaining consensus on these milestones was important so that realistic expectations for patients and surgeon guidance can be established for MACI. One case series study by Pestka et al. 29 found the mean duration of return to work following treatment with second-generation ACI was 13.6 weeks (range 2-53 weeks). Differences were found depending on work intensity (no physical labor vs labor with heavy physical strain). The consensus reached by our expert panel is in alignment with this study in that release to heavy labor may be as early as 3 to 6 months for less challenging patients (patients with single lesions in PF or TF), and 9 to 12 months for patients with larger lesion size and multiple lesions. Release to sedentary work can be as early as 2 to 4 weeks for all patient types depending on rehabilitation and transportation (e.g., driving or getting on/off public transport) issues. Release to unrestricted ADL was agreed by the panel to be as early as 3 months for all patient types.
Similar to the milestones of ADL or work, there is very little information in the literature regarding timing of return to recreational activities or sports following ACI, and none in the literature regarding MACI. For our Delphi study, the expert panel felt that questions regarding return to recreational activities (e.g., return to local gym) were too broad given the wide range of activities performed at a gym and therefore they could not reach consensus on the questions within this milestone. As would be expected, release to stationary cycling had the shortest timing of the sports activities with release at 3 to 4 weeks for less challenged patients (both PF and TF) and 5 to 6 weeks for more challenged patients. Release to outdoor cycling could be as early as 3 to 4 months for less challenged patients with a single PF lesion, and 5 to 6 months for the 3 other categories of patients. Evaluation for return to running, that is, assessment to ensure the patient has sufficient quadriceps strength and neuromuscular control to start a running progression, could begin at 6 months (less challenged) or 8 months (more challenged patient), with release to running at 7 to 9 months or 10 to 12 months, respectively. Release to tennis followed a similar pattern. Notably, the consensus for the timing for return to running for the less challenged patient groups in our Delphi study is similar to the timing recommended for release to running for osteochondral allografts with no concomitant procedures. 30 Release to soccer and football had the longest timing for return (10-12 months) since these are high-impact, twisting, and pivoting sports. Other examples of high-impact sports having both contact and collisions, additionally challenging the cartilage, that may require longer rehabilitation time include ice hockey and combat sports.
Limitations
Limitations of the modified Delphi method using consensus from a panel of experts, includes potential bias in the selection of experts. In our study, surgeons were selected without specific knowledge of their approach to rehabilitation practices (i.e., more conservative or more aggressive approaches). However, the panel’s answers to the survey questions reflected the range of approaches and timing for rehabilitation following MACI treatment that are reported in the literature. While the modified Delphi method allowed us some flexibility to narrow the timing for milestones between rounds 1 and 2, the face-to-face consensus meeting became important in establishing consensus. This could have introduced bias in that participants of the meeting may have felt compelled to conform to the group view, a criticism of consensus methodology. 31 To prevent this, a white paper that detailed all results from the meeting was sent to all participants within a few months of the meeting to provide an opportunity for comment.
Additional limitations are in the scope of the patient characteristics included in the patient profiles. While we tried to set up boundaries for each type of lesion (TF or PF) by describing more and less challenging patient and lesion types, the assumption was that patients had normal alignment and normal meniscus and ligament status. Future studies could assess the impact of osteotomies and other complexities in the timing of milestones and goals.
Conclusions
The aim of this study was to evaluate levels of consensus in rehabilitation practices following MACI treatment based on the experience of U.S. orthopedic surgeons. While patient care is, and should be, individualized due to the variability of patient and lesion characteristics, this study can provide guidelines and consensus opinion on standard practice to optimize surgical outcomes in rehabilitation programs post MACI treatment. The most important finding of this study was that relatively good consensus of rehabilitation practices was established for MACI treatments of knee cartilage lesions among U.S. surgeons. The consensus of experts can aid surgeons and patients in the expectations and rehabilitation process as MACI surgery becomes more prevalent in the United States.
Supplemental Material
Appendix_1 – Supplemental material for Consensus on Rehabilitation Guidelines among Orthopedic Surgeons in the United States following Use of Third-Generation Articular Cartilage Repair (MACI) for Treatment of Knee Cartilage Lesions
Supplemental material, Appendix_1 for Consensus on Rehabilitation Guidelines among Orthopedic Surgeons in the United States following Use of Third-Generation Articular Cartilage Repair (MACI) for Treatment of Knee Cartilage Lesions by David C. Flanigan, Seth L. Sherman, Brian Chilelli, Wayne Gersoff, Deryk Jones, Cassandra A. Lee, Alison Toth, Caryn Cramer, Victor Zaporojan and James Carey in CARTILAGE
Footnotes
Acknowledgments and Funding
The authors thank Andrew Kocher, MPT, CMPT for facilitation of survey question development, and Trinity Life Sciences, Cambridge, Massachusetts for facilitation of survey development and consensus procedures. The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: David Flanigan has received income from Smith & Nephew for services other than consulting; consulting fees from Ceterix Orthopaedics Inc., Linvatec, Smith & Nephew, DePuy Synthes, Zimmer Biomet, and Medical Device Services Inc.; honoraria from Vericel; and hospitality payments from Linvatec. Seth Sherman received funding from Smith & Nephew and Arthrex for services other than consulting; consulting fees from Joint Restoration Foundation Inc., Vericel, Flexion Therapeutics, Olympus America, Arthrex, and Linvatec; speaking fees from Linvatec; education payments from Evolution Surgical; hospitality payments from Joint Restoration Foundation Inc., Vericel, Flexion Therapeutics, Smith & Nephew, Zimmer Biomet, Aesculap Biologics, Linvatec, and Arthrex; honoraria from Flexion Therapeutics; Vericel, and Joint Restoration Foundation Inc.; and travel and lodging payments from Smith & Nephew, Arthrex, Vericel, Linvatec, and Joint Restoration Foundation Inc. Wayne Gersoff has receive consulting fees, honoraria, food and beverage payments, and travel and lodging payments from Vericel. Deryk Jones has received consulting fees from Medical Device Services Inc., and Amniox Medical Inc.; food and beverage payments from Medical Device Services Inc., Vericel, Musculoskeletal Transplant Foundation Inc., Smith & Nephew, Flexion Therapeutics, and Acumed; honoraria from Musculoskeletal Transplant Foundation, Flexion Therapeutics, and Vericel; and travel and lodging payments from Linvatec, Flexion Therapeutics, TissueTech, Medical Device Business Services, Vericel, Acumed, Musculoskeletal Transplant Foundation Inc., and Amniox Medical. Cassandra Lee has received consulting fees from KCI USA, education payments from Arthrex and Sequoia Surgical, food and beverage payments from Arthrex, Vericel, Sequoia Surgical, and Smith & Nephew; honoraria from Vericel; travel and lodging payments from Vericel, Smith & Nephew, Arthrex, and Sequoia Surgical. Alison Toth has received honoraria, travel and lodging payments, and food and beverage payments from Vericel. James Carey has received consulting fees, travel and lodging payments, food and beverage payments, and honoraria from Vericel. Caryn Cramer and Victor Zaporojan are employed by Vericel.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
