Abstract
Objective:
To assess intraindividual biological variability of serum cartilage specific biomarker Coll2-1 and define the best standardized conditions for blood sampling.
Design:
Blood samples were taken from 116 subjects with knee osteoarthritis (OA) at a single time point (PRODIGE study) and from 15 healthy subjects under various conditions, including fasting condition, sampling time and season, blood treatment, and type of blood collection tube (COVAR study). Type II collagen-specific biomarker Coll2-1 was directly measured in serum using an immunoassay.
Results:
There was no significant difference on Coll2-1 values between samples collected at any of the 5 sampling times or at any of the sampling days measured. None of the sampling parameters tested had a significant impact on Coll2-1 value (clotting time, clotting temperature and temperature of blood centrifugation, type of tube). On the contrary, differences were found in between subjects and between subjects with knee OA and healthy subjects.
Conclusion:
Coll2-1 measurement is not affected by sampling specific conditions, circadian rhythm or seasons but was found elevated in subject with knee OA indicating that Coll2-1 serum variation is not linked to the study environment, but to cartilage degradation in OA. Coll2-1 assay is sufficiently robust for use in OA clinical trials.
Clinical Significance
Coll2-1 is a specific biomarker of cartilage degradation measured by a robust test that could be helpful for the monitoring of intervention efficacy in patients with knee osteoarthritis.
Introduction
Osteoarthritis (OA) management requires the identification of effective biomarkers for the monitoring of treatment efficacy. 1 In the future, biomarkers might help at identifying OA patients most likely to receive clinically important benefits from an intervention and individuals at earlier stages of OA in order to institute treatment at a time more amenable to disease modification. In addition, biomarkers might help select progressors and by this allow reduction of both sample size and duration of clinical trials investigating structural effects. Many efforts have been carried out in that direction for the past decade and it is now recommended to collect biospecimens in all OA clinical trials, when resources permit.2,3
Biomarkers validation is a long process that involves analytical and clinical validations. Biomarker analytical validation ensures that the biomarker value reflects the biological system and not the methodological variability. It is characterized by the analysis of test accuracy, precision, repeatability, reproducibility and evaluation of sensitivity and specificity for the intended use, as well as the identification of limits of quantification, detection and of blank.2,4,5 Moreover, the investigation of robustness and stability is essential to ensure that the test is not affected by method parameters. 4 Numerous analytical and preanalytical factors may affect the performance of a test. Preanalytical factors include but are not restricted to physiology of the participant and biospecimen collection and handling practices. On the opposite side, analytical factors cover the use of validated assays, the monitoring of controls, reference samples and analysis of test reproducibility within the testing session, the randomization of samples, the recording of freeze-thaw cycles, and the standardization of technical staff training in assay performance. 5
We have developed a competitive and specific immunoassay (enzyme-linked immunosorbent assay [ELISA]) that recognizes the linear form of Coll2-1, a 9 amino acid sequence (HRGYPGLDG) specific of type II collagen which is released during cartilage degradation. 6 Coll2-1 can be defined based on the BIPED classification system proposed by Bauer et al. 7 for OA biomarkers, as a biomarker useful for studying the burden of disease, 8 the prognostic 9 and, the diagnosis. 9 In terms of efficacy of intervention, it was demonstrated that Coll2-1 serum concentration decreased after hyaluronic acid (HA)10,11 or autologous plasma-rich platelet (PRP) injections. 12 A decrease of Coll2-1 has also been observed in OA patients after 14 and 84 days of treatment with bio-optimized curcumin. 13
This study aimed to investigate the influence of environmental factors on the level of Coll2-1 measured in the serum by this immunoassay and to provide recommendations to standardize its use in clinical trials. More precisely, we have investigated not only fasting, resting state, circadian rhythm, and seasonality but also sampling-specific factors, such as type of clotting tubes, clotting patterns, and centrifugation temperature. Finally, we have provided recommendations for the use of Coll2-1 in clinical trials.
Materials and Methods
Biological samples were collected with written informed consent from individuals in accordance with International Conference on Harmonisation Good Laboratory Practice (ICH GLP).
Heathy subjects were recruited from the COVAR study, a monocentric study conducted by ARTIALIS SA in Belgium and registered as NCT02348944. Eligible healthy subjects were men or women, aged older than 18 years who do not present with severe liver or renal insufficiency or infectious diseases (HIV, hepatitis B or C). Fifteen subjects were selected based on their history and absence of OA risk factors. A key criterion was the absence of joint trauma, pain, and discomfort. No analgesic, anti-inflammatory drug, or food supplement were used by these subjects before and during the time of the study. From these subjects, blood was collected at specific time points during a 68-week testing phase. The protocol has been submitted to the central Ethics Committee (EC) from the University Hospital of Liege on July 2, 2013 and approved by July 16, 2013 (B707201317939). Participant flow diagram of COVAR study is illustrated in Figure 1 .
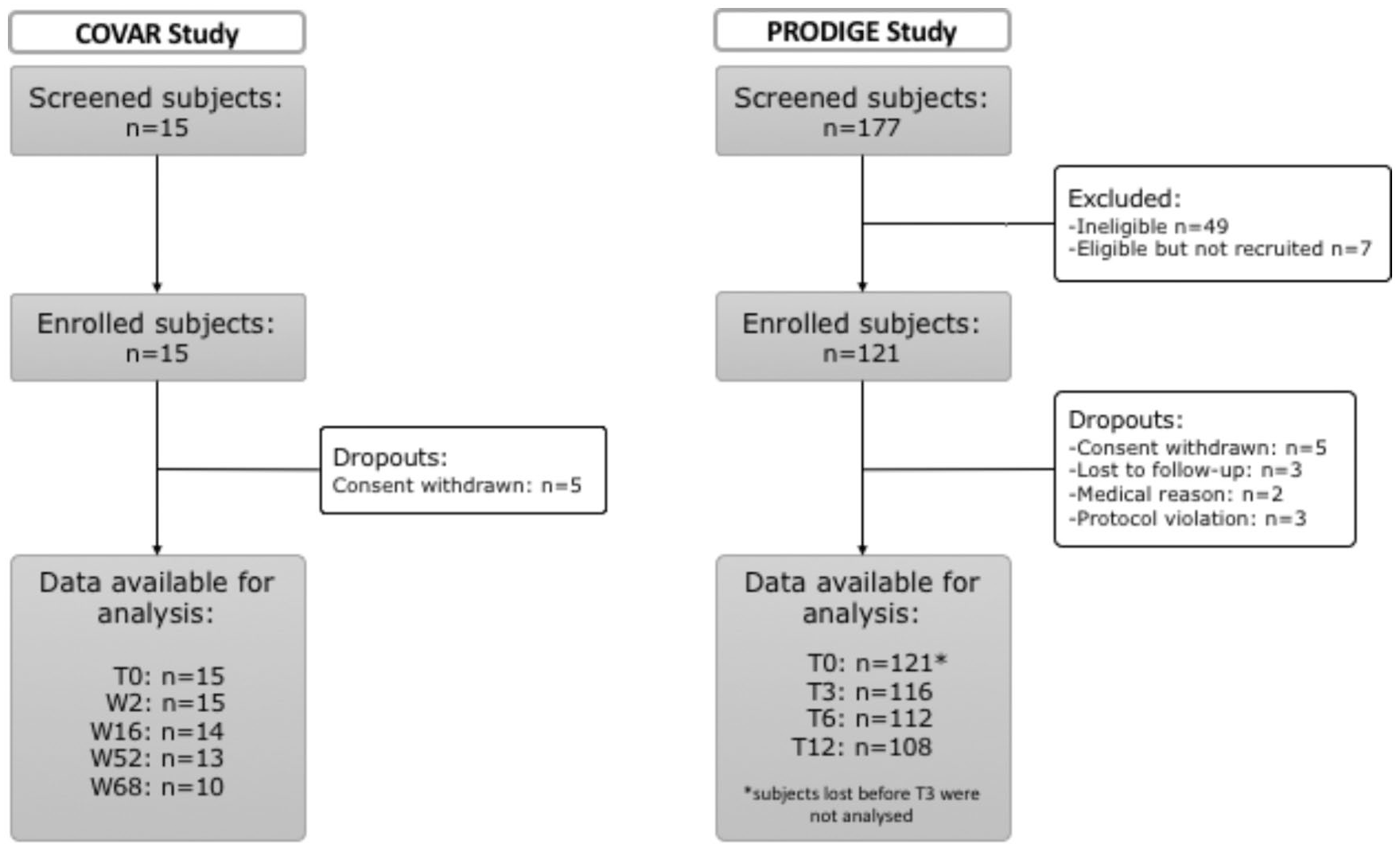
Participant flow diagrams of COVAR study (left) and PRODIGE study (right).
Patients with knee OA were recruited from the PRODIGE study, a multicenter study conducted by ARTIALIS SA in Belgium and France registered as NCT02070224. A total of 121 subjects suffering from knee OA were followed during 1 year. Knee OA was based on the American College of Radiology (ACR) criteria and confirmed by X-ray report and magnetic resonance imaging (MRI) assessed centrally. The protocol has been submitted to the Belgian central Ethics Committee (EC) from the University Hospital of Liege on October 22, 2013, to the French Ethics Committee (Comité de Protection des Personnes EST III [CPP III]) on October 31, 2013, and to the French National Agency for Medicines and Health Products Safety (ANSM) on October 31, 2013. The protocol has been approved by EC on January 13, 2014, by CPP on April 10, 2014 and by ANSM on January 8, 2014. The national number was B707201318719 for Belgium and ID RCB 2013-A01368-37 for France. Patient flow diagram of PRODIGE study is shown in Figure 1 .
Blood Sampling
Peripheral blood samples were collected by antecubital venepuncture with the subject in a sitting position. Samples were allowed to clot before being centrifuged at 1000 × g for 10 minutes. The sera were aliquoted and stored at −20°C until their analysis.
As detailed in Table 1 , blood was collected at inclusion (baseline, T0), and after respectively 2 (W2), 16 (W16), 52 (W52), and 68 (W68) weeks. Blood was also drawn at different times of the day: 8 am, 9 am, 12 pm, 2 pm, and 5 pm. Samplings at 8 am and 12 pm were performed before meals while samplings at 9 am and 2 pm were collected after meals. Meals were taken within the hospital facility and food intake was recorded. Sampling at W2 was done on a Monday after a free weekend, while sampling at baseline was conducted on a Thursday during a working week to investigate the influence of daily activities and stress on biomarker levels. Subjects agreed not to participate in any physical activity or otherwise stressful events the day before visit W2, that visit was considered being after a resting state.
Drawing Procedure for Healthy Subjects (COVAR Study).
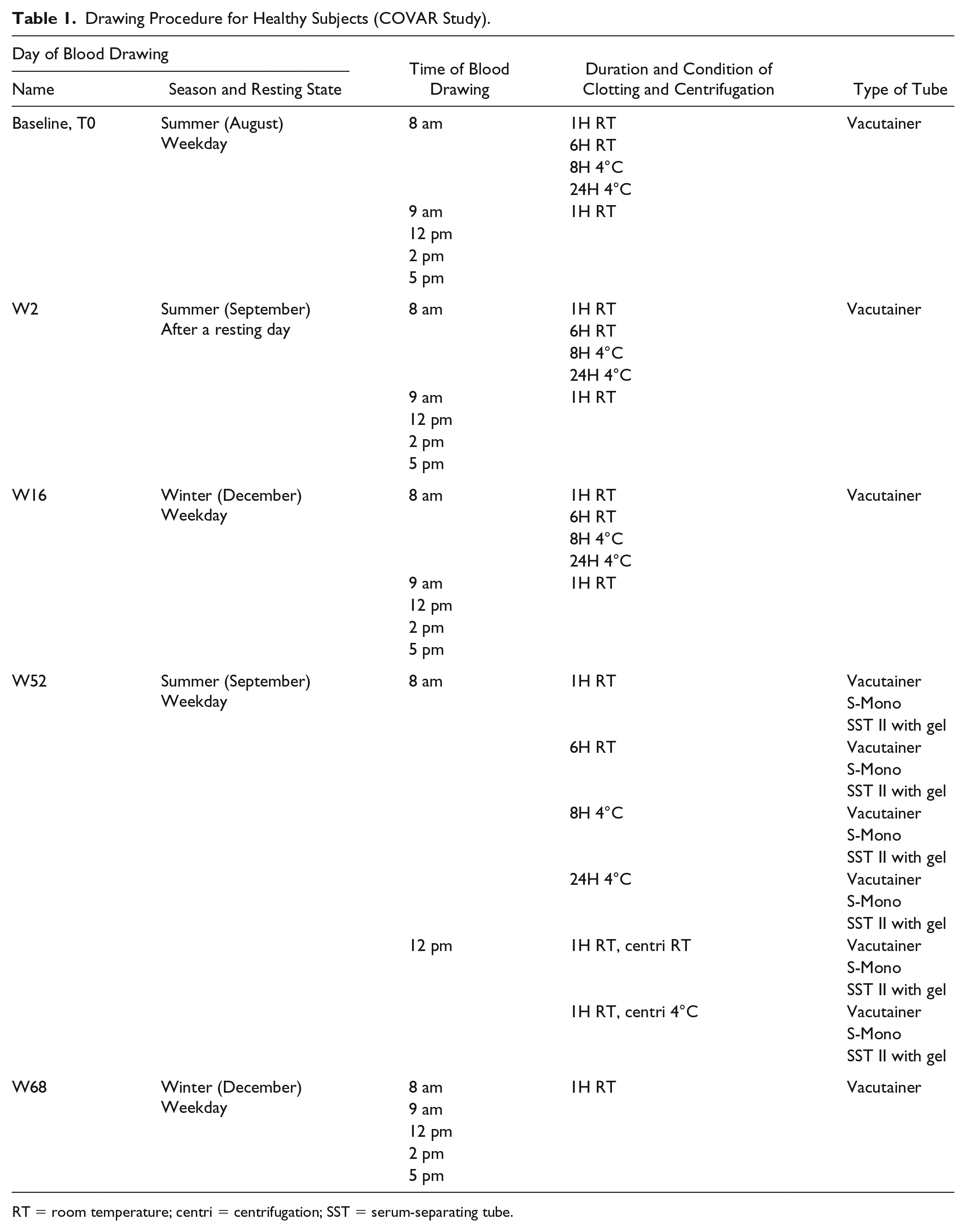
RT = room temperature; centri = centrifugation; SST = serum-separating tube.
Baseline, W2 and W52 were taken during the summer, while W16 and W68 samples were collected during the winter to investigate the association between seasons and serum Coll2-1 levels.
At baseline, W2 and W16, 4 tubes were collected at 8 am and allowed to clot either during 1 or 6 hours at room temperature (RT), or during 8 or 24 hours at 4°C to study the effect of clotting protocol, on serum Coll2-1 levels. At W52, blood was collected at 8 am and 12 pm in 3 different types of blood collection tubes (vacutainer, S-Mono, and serum-separating tubes [SST] II advanced tubes) to investigate the impact of clotting tubes on biomarker levels. Finally, samples taken at 12 pm at W52 were either centrifuged at room temperature or at 4°C for an hour to study the effects of centrifugation temperature on serum Coll2-1 values.
Testing
Testing was operated by the centralized biomarker platform (ARTIALIS SA, Liège, Belgium) with regard to the Good Clinical Laboratory Practice guidelines (GCLP/WHO). Coll2-1 concentration was measured by competitive immunoassay (ARTIALIS SA, Liège Belgium). One testing session was performed for each cohort (OA and healthy). Samples were allocated randomly to the different wells/plates according to a pre-established randomization list. In addition to the 1000nM control used in the 2 sessions, 2 serum samples, respectively, from OA or healthy patients, were added to monitor interplate coefficient of variation (CV%) within each testing session. Nonconforming values (CV of replicates > 20%; threshold fixed by ARTIALIS SA quality system to comply with ISO 13485) were retested 3 times.
Analysis
Analyses were performed using Microsoft Excel 2013 and the Statistical Analysis Software Statistica (Statsoft, France). Comparisons of the Coll2-1 level means in various conditions were performed by the analysis of variance (ANOVA). Because F tests are one-sided, effect size (η2) were computed with 90% confidence interval (CI). General linear model evaluating all conditions together was performed as well. Multiple regression analysis was used to evaluate the associations with age, body mass index (BMI), and OA status. For sex influence analysis, Kolmogorov-Smirnov test was used. A P value <0.05 was considered significant. Confidence intervals containing zero were considered nonsignificant. No correction for multiple testing was made. No power calculation was performed. All collected data were analyzed.
Results
Subjects Demographics
Mean age of healthy subjects was 31.6 ± 9.7 years and BMI was 23.6 ± 4 kg/m2. Ten (66.7%) subjects were female and 5 (33.3%) were male. Among OA subjects, 59% were female and 41% were male. Mean age of OA population was 62.7 ± 8.5 years and mean BMI was 29.8 ± 5.1 kg/m2. Kellgren and Lawrence (K&L) grade 14 was assessed for the OA population: 46% of the 121 subjects had a K&L grade of 2 and 39% had a K&L grade of 3. Subjects with unknown K&L grade were included based on MRI demonstrating knee osteoarthritis. Mean OA duration was 8 ± 7 years and mean pain (during the 24 hours preceding the visit) evaluated on a visual analogue scale (VAS) was 38 ± 28 mm. Subjects’ demographics and OA history can be found in Table 2 .
Demographics of Healthy and OA Subjects and OA History.

SD = standard deviation; OA = osteoarthritis; KL = Kellgren and Lawrence; NA = not applicable.
Analytical Performances
In total, 635 samples were collected from healthy subjects, of which 2 (0.3%) could not be analyzed because volume was too low, 71 (11.3%) had to be retested to get conclusive data (CVs of replicates <20%) and 2 presented too much variability (CVs of replicates >20%) and were excluded from this study. From OA subjects, 116 samples were collected at baseline, of which 20 (17.2%) were retested. One sample presented too much variability (CV of replicates >20%) and was excluded from the study.
In order to evaluate the precision, internal control with known concentration of Coll2-1 were used. CVs of all controls were below 15%, which is in accordance with OARSI (Osteoarthritis Research Society International) recommendations. 2 Calibration curves were generated following OARSI guidelines: 2 standards (internal controls), 1 blank, and 7 nonzero samples covering the expected range. As suggested by OARSI guidelines, reproducibility was assessed using 2 healthy and 2 OA reference serum samples for healthy population testing and OA population testing, respectively. Mean intra-assay coefficients of variations were below 20% for internal controls and reference serum samples. Mean inter-assays coefficients of variations were below 20% for healthy serum samples (n = 21 plates) and OA serum samples (n = 26 plates). Finally, mean inter-assays coefficient of variations between the plates from the 2 testing sessions (n = 47 assays) was 8% for kit internal control samples (700-1300 nM). Moreover, bilateral Grubb’s test showed there was no outlier among any control or reference serum samples on the 47 plates. The detection and quantification limit was 67 nM.
Circadian Rhythm and Seasonal Variation
Circadian rhythm and seasonal variation were assessed on samples collected from healthy subjects. There was no intraday (diurnal) Coll2-1 variation analyzed at baseline, week 2, and week 16 (T0, n = 12, P = 0.53, η2 = 0.054 with 90% CI = [0, 0.112]; W2, n = 15, P = 0.53, η2 = 0.043 with 90% CI = [0, 0.092]; W16, n = 14, P = 0.89, η2 = 0.018 with 90% CI = [0, 0.03]; pooled data baseline, W2, W16, n = 41, P = 0.90, η2 = 0.039 with 90% CI = [0, 0.012]), meaning that Coll2-1 levels was not significantly associated with time of sampling and fasting condition ( Fig. 2A ). Analysis of seasonal Coll2-1 variation in subjects at baseline (August, summer), W2 (September, summer, collection after a resting day), W16 (December, winter), W52 (September, summer), and W68 (December, winter) at 8 am and 12 pm revealed no significant difference (P = 0.70, η2 = 0.054 with 90% CI = [0, 0.109], at 8 am, n subjects = 9; P = 0.93, η2 = 0.027 with 90% CI = [0, 0.041], at 12 pm, n subjects = 9, pooled data [8 am and 12pm]: P = 0.83, η2 = 0.043 with 90% CI = [0, 0.006], n = 18) ( Fig. 2B ). These results indicated that neither resting state nor season caused changes in Coll2-1 levels.
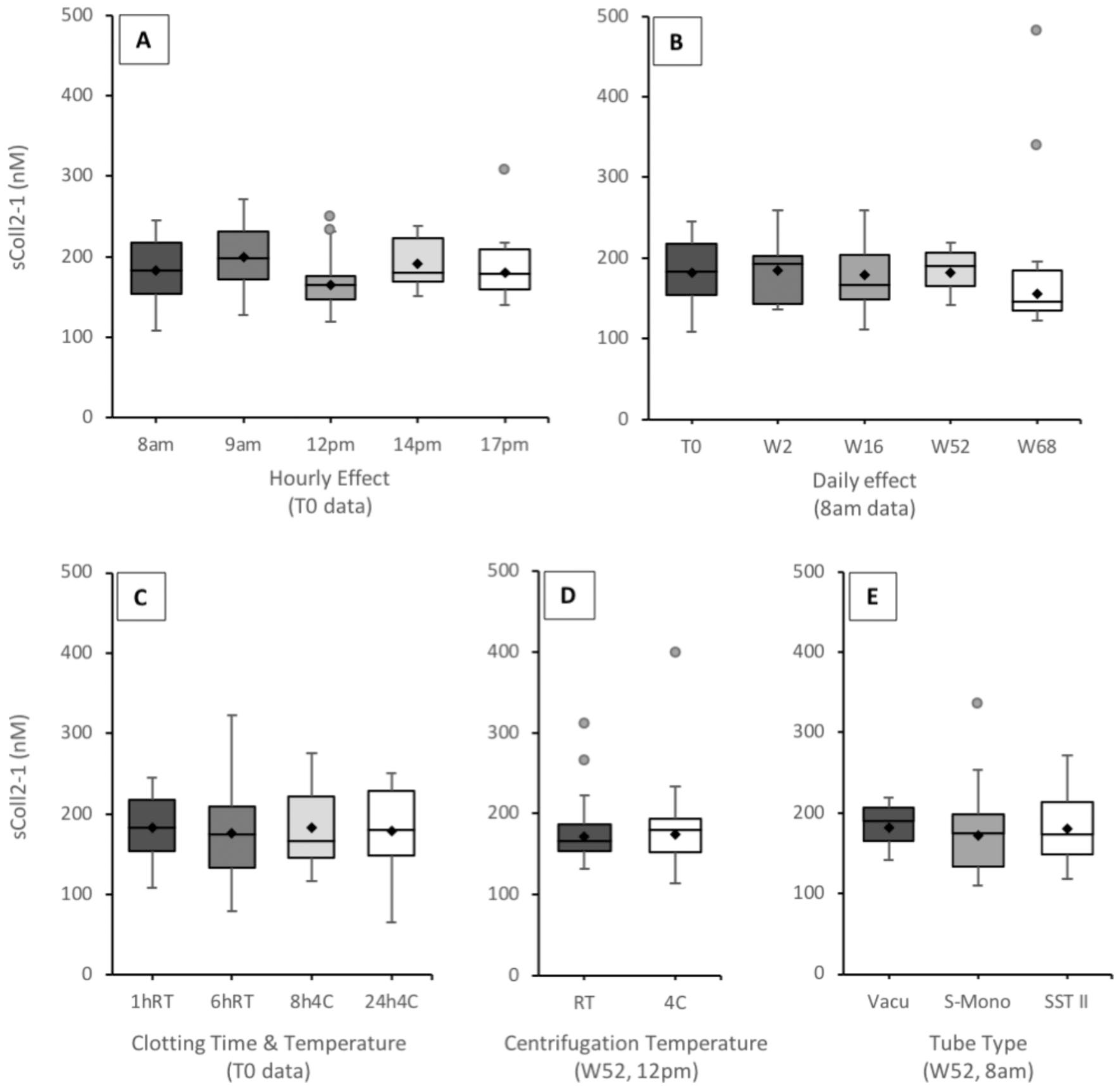
Coll2-1 serum concentrations (nM) at different times of the day (
Influence of Sampling
Influence of sampling protocol was assessed on blood samples collected from healthy subjects. Clotting time and temperature (1 hour at RT, 6 hours at RT, 8 hours at 4°C and 24 hours at 4°C) did not significantly influence Coll2-1 levels (baseline 8 am, n = 14, P = 0.98, η2 = 0.004 with 90% CI = [0, 0]; W2 8 am, n = 15, P = 0.98, η2 = 0.003 with 90% CI = [0, 0]; W16 8 am, n = 14, P = 0.22, η2 = 0.081 with 90% CI = [0, 0.175]; W52 8 am, n = 12, P = 0.86, η2 = 0.017 with 90% CI = [0, 0.047]) ( Fig. 2C ). Similar conclusion was drawn when values from all days were pooled (T0, W2, W16, W52 8 am, n = 55, P = 0.98, η2 = 0.029 with 90% CI = [0, 0]). Similarly, temperature of blood centrifugation (RT or 4°C), evaluated at W52 and 12 pm only, did not affect Coll2-1 levels (n = 13, P = 0.91, η2 = 0.001 with 90% CI = [0, 0.042]) ( Fig. 2D ). Finally, the 3 types of clotting tubes (Vacutainer, S-Mono, SST II advanced with gel separator) used at W52, did not significantly affect the level of Coll2-1 (8 am, n = 13, P = 0.98, η2 = 0.001 with 90% CI = [0, 0]; 12 pm, n = 13, P = 0.76, η2 = 0.015 with 90% CI = [0, 0.084], under standard sampling procedure, that is, clotting during 1 hour at RT and centrifugation at RT) ( Fig. 2E ). Similar observation was performed when all tested conditions were combined at W52 (pooled data from 8 am, 12 pm and all clotting/ centrifugation conditions, n = 72, P = 0.87, η2 = 0.051 with 90% CI = [0, 0.018]). Altogether, these data indicated that neither tube type, clotting time and temperature, nor centrifugation temperature significantly influenced on Coll2-1 values.
Osteoarthritis Condition and Within-Subjects Differences
In the absence of any significant association with all sampling conditions, all data of Coll2-1 from healthy subjects were pooled to evaluate intersubjects differences. A general linear model evaluating all collected data was applied on Coll2-1 values (n[data] = 631, n[days] = 5, n[hour of collection] = 5, n[blood processing] = 6, n[tubes] = 3, n[subjects] = 15). This model confirmed the absence of significant association with all tested parameters (P[days] = 0.58, P[hour of collection] = 0.85, P[blood processing] = 0.93, P[tubes] = 0.38) while significant differences were shown between subjects with mean values ranging from 160 to 270 nM (P[subjects] < 0.001) ( Fig. 3 ).
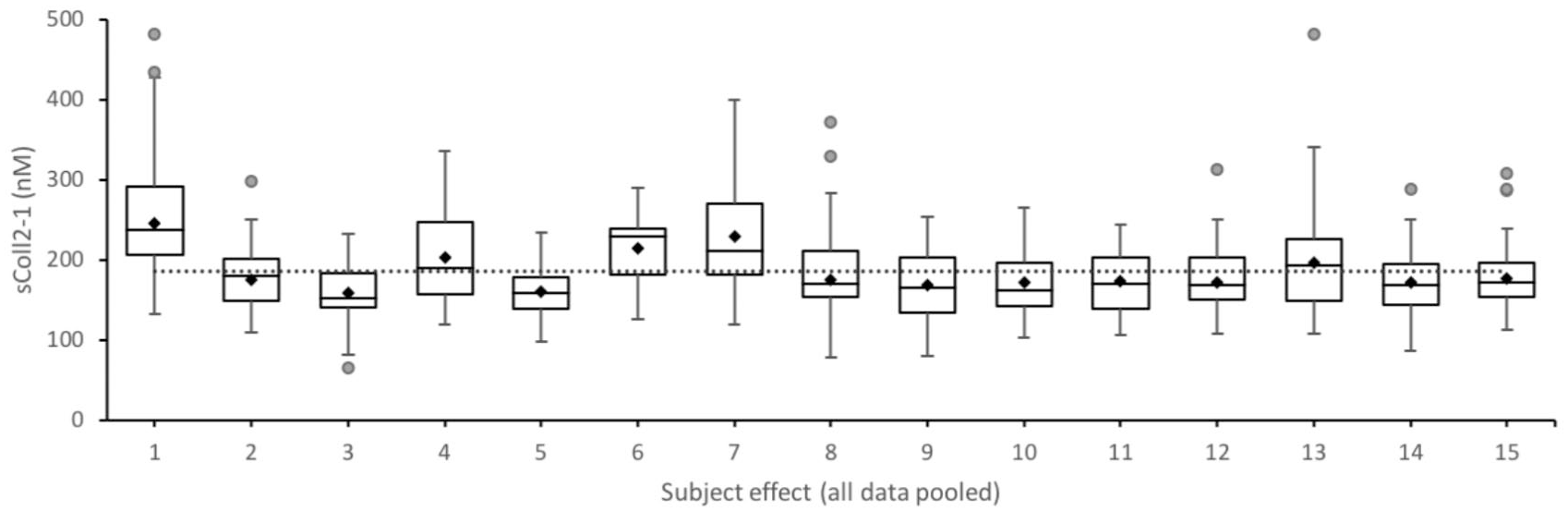
Coll2-1 serum concentration (nM) across all healthy subjects (significant subject association, P < 0.001). Dotted line represents the mean for all subjects. Boxplots are built with minimum, lower quartile (Q1), median, upper quartile (Q3) and maximum values. ♦indicates the mean and • are outliers.
At baseline, serum Coll2-1 levels were significantly higher in OA than in healthy subjects (OA: 481 ± 136 nM vs Healthy 192 ± 32 nM, P < 0.001) ( Fig. 4 ). However, BMI and age of the 2 populations are significantly different. Therefore, multiple regression analysis was performed to make adjustments for these 2 parameters. As a result, both age and OA status were significant predictors of Coll2-1 concentration, with P = 0.02 for age and P < 0.0000 for OA status. On the contrary, BMI was not a significant predictor (P = 0.86). Regression parameters are R2 = 0.59, F(3, 124) = 459.34, P < 0.0000. Residue normality was verified, demonstrating that regression was not biased. Finally, pooled data of healthy and OA subjects showed no significant association with sex (female: 449 ± 164 nM vs male: 455 ± 157 nM, P > 0.1).
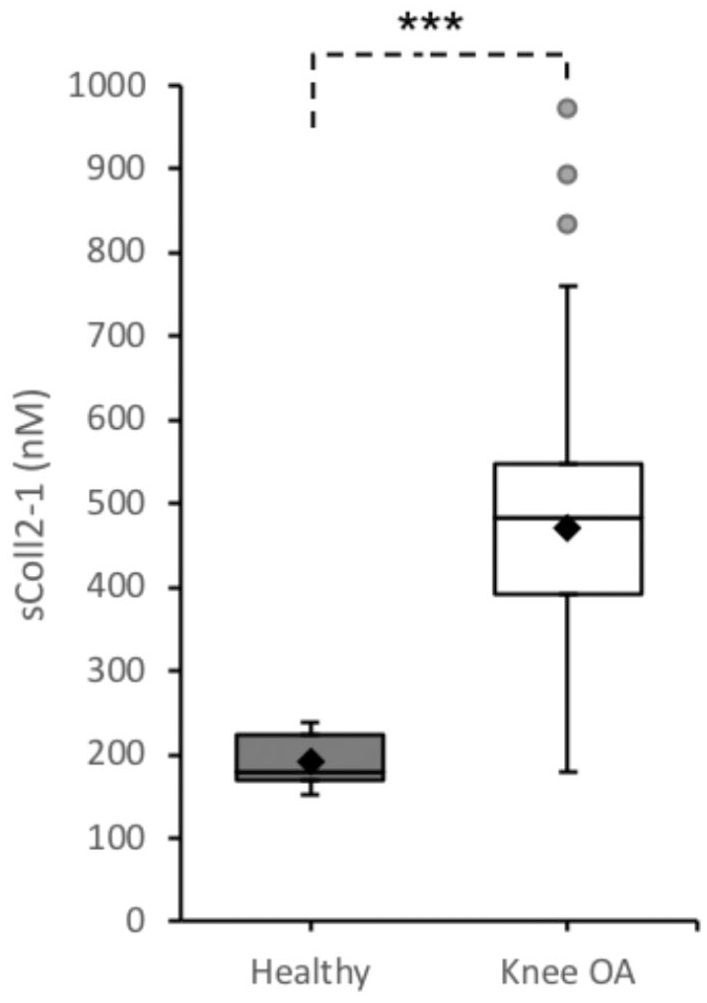
Coll2-1 serum concentration (nM) in healthy and knee osteoarthritis (OA) populations. Significative difference between the 2 groups (***P < 0.001). Boxplots are built with minimum, lower quartile (Q1), median, upper quartile (Q3) and maximum values. ♦indicates the mean and • are outliers.
Discussion
According to Food and Drug Administration recommendations, analytical, preanalytical, and clinical factors are part of stability evaluation of an assay (guidance on bioanalytical method and validation, 2018). This study aimed to evaluate preanalytical factors such as the influence of sampling collection and handling on levels of serum Coll2-1, a peptide located in the triple-helical part of type II collagen, the most specific protein of the articular cartilage. Regarding analytical and clinical factors, Coll2-1 is already a well-documented biomarker of cartilage degradation in OA.6,8-13,15 Clearly, this study demonstrated that Coll2-1 serum levels were not influenced by the sampling conditions, the timing of blood sampling, the seasons and the daily life activity. In contrast to urinary CTX-II, 16 CTX-I, 17 and PIIANP, 18 serum Coll2-1 has no diurnal variation, which is a great advantage in the management of biomarkers sampling in clinical trials. Clearly, these data suggest that Coll2-1 is less susceptible to the effects of timing and sampling conditions and daily physical activity than others and therefore offers an advantage in the context of clinical trials incorporating biologic parameters. The opposite effects of preanalytical factors seen between serum collagen and urinary collagen markers are most compatible with stimulation of glomerular filtration rate by food intake. 19 It was also found that uCTX-II is highly correlated to urinary bone resorption markers. 17 Bone turnover is highly sensitive to physical activity, posture, and nycthemeral variation explaining the diurnal variation of uCTX-II. This finding contributes to increase the robustness of the data obtained with Coll2-1, as variation of the biomarker is not linked to the study environment, but to the effect of disease or treatment on cartilage degradation. Summary of the recommendations for the use of Coll2-1 in clinical trials is provided in Table 3 .
Recommendations for sColl2-1 Preanalytical Factors in Clinical Trials.
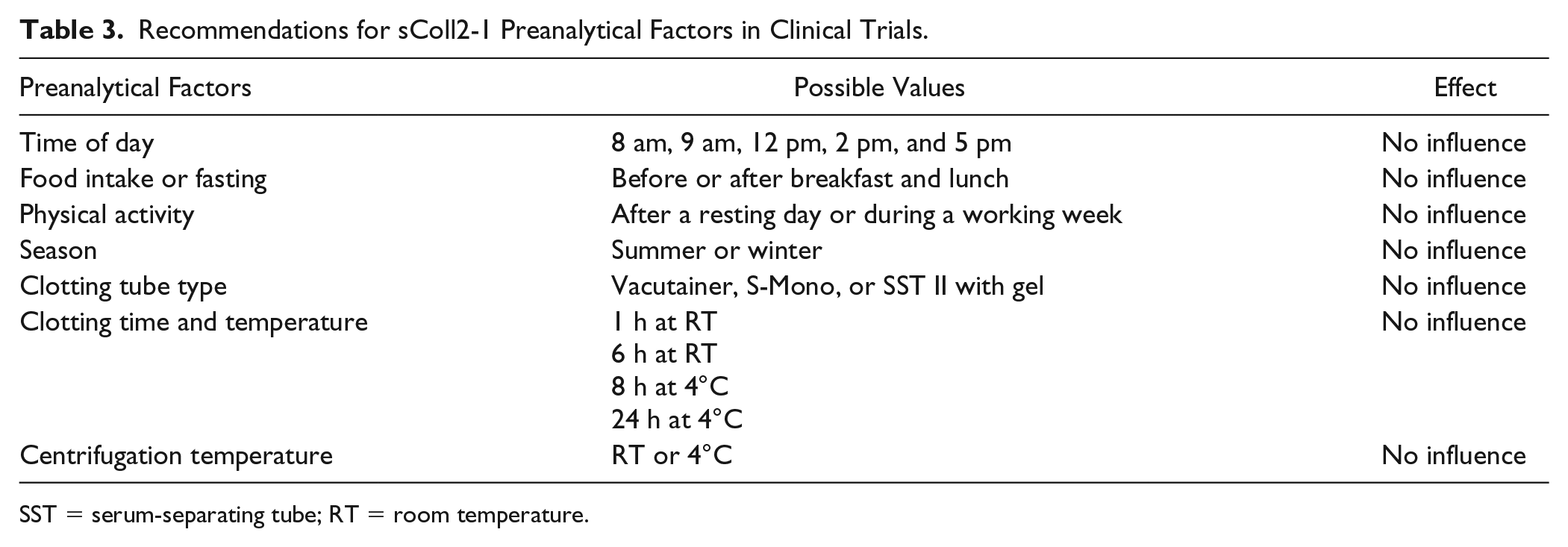
SST = serum-separating tube; RT = room temperature.
Another important observation was the absence of serum Coll2-1 variation with BMI. This result also contrasts with those of other commonly used biomarkers. However, Coll2-1 is significantly associated to age, as many other OA biomarkers. Indeed, serum COMP, HA, MMP3, PIIANP, CTX-I and urinary CTX-II among others, were shown to be associated with age and/or BMI.20-25 In contrast to serum CTX-I 17 and COMP, 21 serum Coll2-1 is not associated to sex. Finally, we found that Coll2-1 serum concentration is significantly increased in OA, confirming that serum Coll2-1 variation is associated to OA disease. Clearly, our results indicate that, even after age and BMI adjustments, Coll2-1 levels are higher in OA patients indicating that Coll2-1 increase in serum of OA patients is well related to the disease.
There were several limitations of this study. First, sample size of healthy volunteers is small that nevertheless confirms the robustness of serum Coll2-1 value because of the high precision and reproducibility of the immunoassay. The time interval between the various body fluid samplings may not have captured the maximal magnitude of the diurnal variation. More time points had to be performed to cover daily variation. Finally, healthy subjects were relatively young as compared with OA subjects.
In conclusion, Coll2-1 assay is sufficiently robust for measuring Coll2-1 in serum of subjects in clinical trials. Coll2-1 levels are affected neither by subject specific conditions such as fasting, circadian rhythm, seasonality, nor by sampling process factors such as type of clotting tube, clotting patterns and centrifugation temperature. Of course, further studies to investigate correlation of Coll2-1 with clinical and/or imaging features are necessary to qualify this biomarker as drug development tool but these findings already support the use of Coll2-1 in clinical trials investigating the effects of OA treatments.
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: PRODIGE study (NCT02070224) was performed in the framework of a convention between the Walloon region and ARTIALIS SA (Convention No. 6905).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: YH is the founder and the President of ARTIALIS SA, Spin-off Company of the University of Liege. He has also received fees from Bepharbel, GSK, KiOmed Pharma SA (formerly Synolyne Pharma SA), Nestle, Flexion therapeutics, IBSA, BioIberica, Laboratoires Expanscience, Royal canin, MagPharm, LABRHA, Pfizer, Thuasne, and Tilman SA. ACH, BC, EC, SP, LG, AL and SV are employees of ARTIALIS S.A.
Ethical Approval
The protocol has been approved by the Central Ethics Committee (EC) from the University Hospital of Liege (B707201317939)
Informed Consent
Written informed consent from all individuals in accordance with the International Conference on Harmonisation Good Laboratory Practice (ICH GLP)
