Abstract
Objectives
This study aimed to determine possible associations between transcriptional and translational levels of clusterin (CLU) in the systemic and local joint environments with the severity of knee osteoarthritis (OA) and to investigate CLU mRNA expression in knee OA fibroblast-like synoviocytes (FLSs) stimulated with tumor necrosis factor-α.
Design
Circulating and synovial fluid CLU levels in 259 knee OA patients were quantified using an enzyme-linked immunosorbent assay. Relative CLU mRNA expression in 50 knee OA synovial tissues and 4 knee OA FLSs was determined using real-time polymerase chain reaction.
Results
Plasma CLU levels of knee OA patients were significantly higher than paired synovial fluid samples. Compared with early-stage knee OA patients, those with advanced-stage OA had considerably increased plasma and synovial fluid CLU levels. There were significant positive associations of plasma and synovial fluid CLU levels with radiographic severity of knee OA. Plasma CLU levels were directly correlated with its synovial fluid levels and high-sensitivity C-reactive protein levels in the patients. Receiver-operating characteristic curve analysis unveiled the potential utility of plasma CLU as a novel biomarker for knee OA severity (AUC = 0.80), with a sensitivity of 71.4% and a specificity of 73.3%. Marked upregulation of CLU mRNA expression was observed in both the inflamed synovial tissues and FLSs of knee OA.
Conclusion
Increased CLU mRNA and protein levels in the systemic and local joint environments of knee OA might reflect knee OA severity, especially systemic and synovial inflammation.
Introduction
Knee osteoarthritis (OA) is a chronic degenerative joint disease and a major cause of severe pain that leads to disability and eventual total knee replacement (TKR). Mainly afflicting the elderly worldwide,1,2 it is becoming a serious clinical concern in an ageing population. Although the specific order of pathologic alterations occurring in knee OA remains a matter of debate, synovial inflammation (synovitis) is regarded as an important pathological feature driving the progression of cartilage degeneration in knee OA. 3 Mechanistically, pro-inflammatory cytokines, including interleukin-1β (IL-1β) and tumor necrosis factor-α (TNF-α) secreted by synoviocytes, reportedly provoke the activation and production of chemokines and matrix metalloproteinases, which have been implicated in cartilage destruction.4,5 As a consequence, treating key aspects of synovial inflammation might be useful for counteracting the development and progression of knee OA. For this reason, it is imperative to better understand what causes the progressive and developmental synovitis, which could lead to not only developing more effective disease modifying treatments but also identifying potential biomarkers for the early phases of the degenerative processes in knee OA. Interestingly, being overweight and obese are increasingly appreciated as critical factors for knee OA, which are characterized by a low-grade systemic inflammatory state,6,7 thereby establishing their possible involvement in knee OA synovitis. Regarding this, accumulative data derived from preclinical and clinical studies attested the effect of obesity on synovitis mediated through enhancing the release of inflammatory molecules from adipocytes residing in fat depots throughout the body, particularly the synovium of obese arthritic mice and patients.8-10 Based on these previous findings, the possible ability of molecules that participate in metabolic alterations-mediated inflammation have great potential to become diagnostic biomarkers for the progressive synovitis of knee OA.
Clusterin (CLU, also known as apolipropotien J), acting as a regulatory molecule in energy homeostasis and inflammation, is becoming increasingly recognized as a possible mediator for obesity-associated synovitis in knee OA. As to its primary action as an ATP-independent charperon protein, CLU has been functionally implicated in a wide range of physiological processes including lipid transportation, cellular development, differentiation, and senescence. Given its overexpression in various types of tissues including the articular cartilage and the synovium,11-15 there are numerous studies demonstrating elevated circulating levels of CLU in several pathological conditions.16-19 In regard to knee OA pathology, a recent study uncovered that reduced circulating CLU levels were negatively associated with pain severity in patients with erosive hand OA. 20 From this, CLU has been hypothesized to be a predictive biomarker for knee OA progression.
To the best of our knowledge, no studies are currently available on the relevance of CLU levels in the systemic and local joint environments to knee OA severity. Consequently, the objective of this study was to determine the possible association of circulating and synovial fluid CLU levels with outcome parameters of knee OA and to examine its mRNA expression in noninflamed and inflamed synovial tissues harvested from knee OA patients. Whether aberrant mRNA expression of CLU is mediated through a pro-inflammatory cytokine like TNF-α in knee OA fibroblast-like synoviocytes (FLSs) was further investigated.
Materials and Methods
The experimental protocols conducted in conformity with the guidelines of the declaration of Helsinki were approved by the Ethical Committee on Human Research of the Faculty of Medicine, Chulalongkorn University (IRB No. 533/54) and the Faculty of Dentistry/Faculty of Pharmacy, Mahidol University (IRB No. 2019/074.2511). Written informed consent was provided by all participants prior to their enrolment in the study.
Study Population
A total of 259 primary knee OA patients who were diagnosed with knee OA according to the criteria of the American College of Rheumatology were recruited in this cross-sectional study. Participants having underlying disease such as diabetes, advanced liver or renal diseases, histories of medication interfering with bone metabolism (e.g., corticosteroids or bisphosphonates), other forms of arthritis, cancer, or other chronic inflammatory diseases were excluded from this study.
Knee radiography was taken when each knee OA participant was standing on both legs with fully extended knees and the X-ray beam centered at the concentration of the joint. Assessment of radiographic severity was performed using the Kellgren-Lawrence (KL) system, 21 being a common method used to classify the severity of knee OA, 14 and all preoperative radiographs were assessed in a blinded manner to the patients’ clinical and laboratory data. The presence of radiographic knee OA was identified as KL grade ≥2.
Clinical Assessments of Outcomes
Body Composition
Height, weight, and waist circumference (WC) were determined using standard measurement techniques. Body mass index (BMI) was calculated using body weight in kilograms (kg) divided by height in meters squared (m2). Body composition analyses including percentage of total fat mass (%) and fat mass were performed by bioelectrical impedance analysis (BIA; BC-418 Segmental Body Composition Analyzer; Tanita Corporation, Tokyo, Japan).
Knee Pain and Physical Disability
Severity of pain, stiffness, and physical disability in knee OA patients were evaluated by a visual analogue scale (VAS) and the Western Ontario and MacMaster University (WOMAC) index. A 10-cm VAS was used to assess pain related to joint movement. A score of 0 represents no pain, and 10 indicates maximal pain. In addition, WOMAC was utilized to estimate knee function and disability levels. A WOMAC score represented the sum of 3 subscales obtained from 24 questions including 5 questions on pain, 2 on stiffness, and 17 on physical function. Higher WOMAC scores indicate worse pain, more stiffness, and increased functional limitations.
Quantifications of hs-CRP and CLU Levels
Whole blood samples were collected from all participants, centrifuged, and stored immediately at −80°C for subsequent analysis. High-sensitivity C-reactive protein (hs-CRP) was analyzed by an immunoturbidimetric method using a Cobas 6000 automated analyzer (Roche Diagnostics). Synovial fluid was harvested from the most severe knee during diagnostic or therapeutic arthroscopy or total knee replacement, and then centrifuged to exclude cells and joint debris before being kept at −80°C until measurement of CLU concentrations. Plasma and synovial fluid CLU levels were quantified using a commercially available sandwich ELISA kit (R&D Systems, Minneapolis, MN), according to the manufacturer’s instruction.
Determination of CLU mRNA Expression
Synovial biopsies of 50 knee OA patients were harvested surgically at the time of an arthroplasty. Of these, 35 were defined as inflamed synovium, and 15 were identified as noninflamed synovium, based on histological findings assessed by a pathologist, who was blinded to clinical status and diagnosis of the patients, using the following relevant morphological alterations: hyperplasia of synovial lining cell layer, activation of synovial stroma, and infiltration of inflammatory cells. Total RNA was extracted from the synovial tissue using an RNeasy Mini kit (Qiagen, Hilden, Germany), with cDNA reverse transcribed using a TagMan Universal PCR Master Mix (Applied Biosystems, Inc., Foster City, CA), according to manufacturer’s instructions. Relative CLU mRNA expression was determined using quantitative real-time polymerase chain reaction (PCR) with the QPCR Green Master Mix HRox (biotechrabbit GmbH, Hennigsdorf, Germany) on a StepOnePlus Real-Time PCR System (Applied Biosystems, Inc., Foster City, CA). The primers used for CLU and glyceraldehyde 3-phosphate dehydrogenase (GADPH) amplifications are demonstrated in Supplementary Table 1. Relative CLU mRNA expressions, normalized to GADPH as an internal control, were determined using the 2−ΔΔCt method.
Cell Isolation and Culture
FLSs were isolated from the synovial tissues of 4 knee OA patients who underwent TKR using enzymatic digestion, according to standard protocols. In brief, the synovium was sectioned into small pieces using a Bard Parker blade under sterile conditions. The tissue fragments were combined with a sterile collagenase solution (Sigma, St. Louis, MO). The mixture was agitated at 37°C, 5% CO2, and 95% humidity for 6 hours. After collagenase digestion, cells were recovered through centrifugation to yield a cellular pellet that was resuspended in Dulbecco’s modified Eagle medium (DMEM; Hyclone Laboratories Inc., South Logan, UT) supplemented with 100 IU/mL penicillin/streptomycin (Hyclone Laboratories Inc.) and 10% fetal bovine serum (Hyclone Laboratories Inc.). The cell solution was transferred to a 75 cm2 tissue culture flask (Nunc, Roskilde, Denmark) containing 10 mL of supplemented DMEM. The flasks were incubated at 37°C in a humidified atmosphere of 5% CO2, with sterile medium changes performed every 3 days until the cells reached 95% confluence. Cells at the first passage (P1) were used for subsequent experiments. In experimental groups, FLSs were treated with and without TNF-α for 1, 3, and 7 days at 37°C and 5% CO2 in a humidified environment. The concentration of 10 ng/mL TNF-α (Biolegend, San Diego, CA) was selected, according to previously published studies.22-24
Statistical Analysis
Statistical analyses were accomplished with the Statistical Package for Social Sciences, version 22.0 (SPSS Inc., Chicago, IL). Comparisons among each group were executed by Mann-Whitney U tests (for 2 groups) or Kruskal-Wallis H tests (for >2 groups). Correlations were analyzed by Spearman’s rho correlation, and multivariate logistic regression models were conducted to determine the roles of confounding factors. Receiver operating characteristic (ROC) curve and the area under the ROC curve (AUC) were calculated to assess the feasibility of using plasma CLU values as a possible biomarker for knee OA severity. All data are shown as mean ± standard deviation (SD). Statistical significance for differences and correlations was set at P < 0.05.
Results
Baseline Clinical Characteristics
The demographic and clinical characteristics of 259 knee OA patients are summarized in Table 1 . The mean age of knee OA patients was 68.77 ± 8.13 years, and 86.9% of study participants were female patients. In terms of gender, female patients with knee OA showed significantly higher values of body composition markers including BMI and percentage of fat mass than male patients with knee OA (P = 0.02, P < 0.001, respectively). On the other hand, there were no significant differences in age, fat mass, scores of knee pain and physical disability, and hs-CRP levels between female and male patients with knee OA.
Baseline Characteristics of Knee OA Patients According to Gender a .
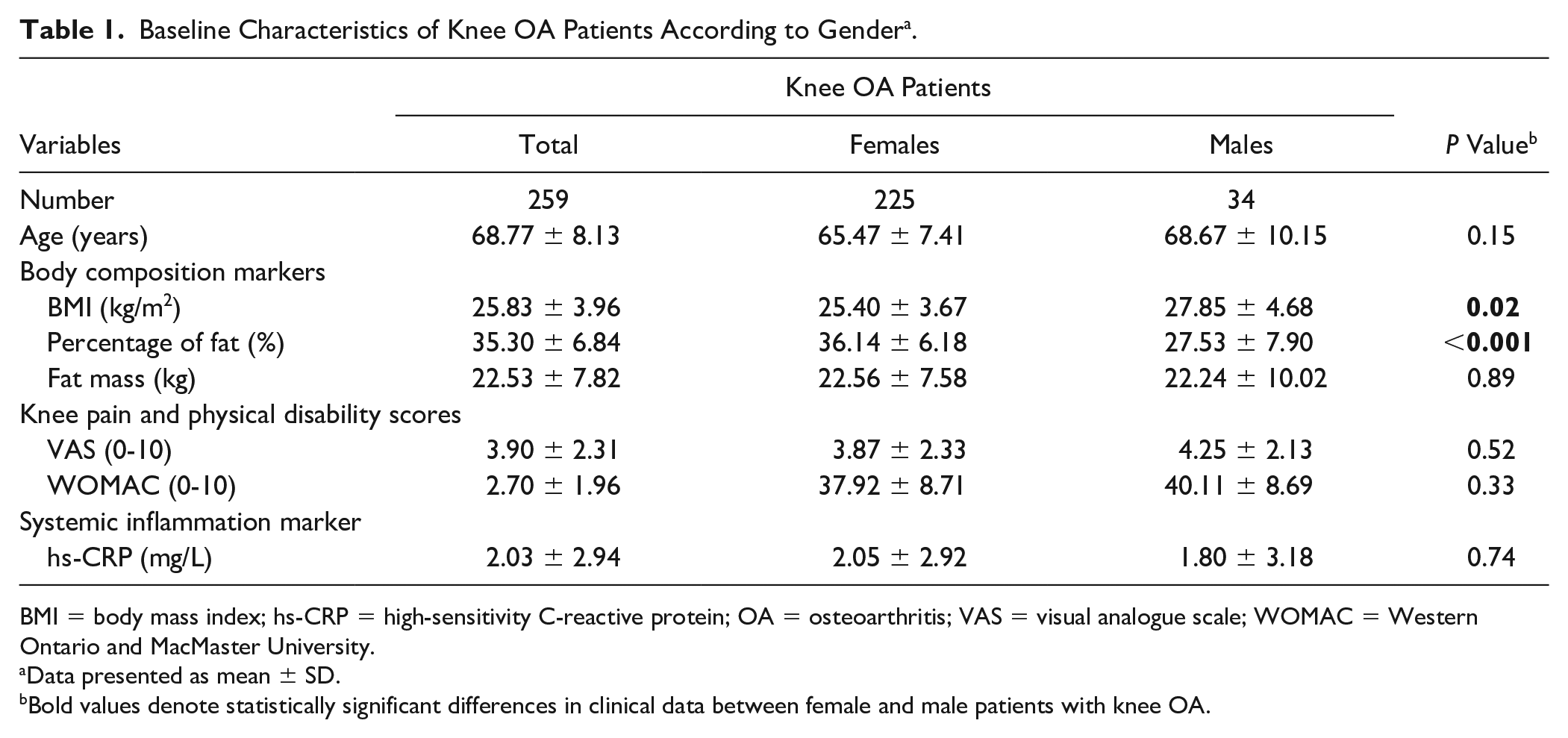
BMI = body mass index; hs-CRP = high-sensitivity C-reactive protein; OA = osteoarthritis; VAS = visual analogue scale; WOMAC = Western Ontario and MacMaster University.
Data presented as mean ± SD.
Bold values denote statistically significant differences in clinical data between female and male patients with knee OA.
Plasma and Synovial Fluid CLU Levels in Knee OA Among KL Subgroups
As shown in Fig. 1A , plasma CLU levels in knee OA patients were significantly higher than that in corresponding synovial fluid samples (P < 0.001). Given a high prevalence of women in knee OA patients, we subsequently compared circulating and synovial fluid CLU levels between female and male patients with knee OA and also found no significant differences in systemic and synovial fluid levels of CLU between groups ( Fig. 1B ). In stratified analyses by disease severity, of the 259 knee OA patients recruited in the study, 100 knee OA patients providing available data on OA grading were classified into 3 subgroups with regard to the Kellgren and Lawrence (KL) grading system (KL grade 2: 30; KL grade 3: 30; KL grade 4: 40). There was no significant difference in mean age among knee OA subgroups (KL grade 2: mean age = 70.73 ± 8.86, KL grade 3: mean age = 67.80 ± 9.06, KL grade 4: mean age = 70.05 ± 9.33). As depicted in Fig. 1C , plasma CLU levels were significantly increased in knee OA patients with KL grade 4, compared with those with KL grades 3 and 2 (P = 0.001, P < 0.001, respectively). In conjunction with this result, a marked elevation in synovial fluid CLU levels was observed in knee OA patients with KL grade 4 when compared with KL grades 3 and 2 OA patients (P = 0.002, P < 0.001, respectively; Fig. 1D ).
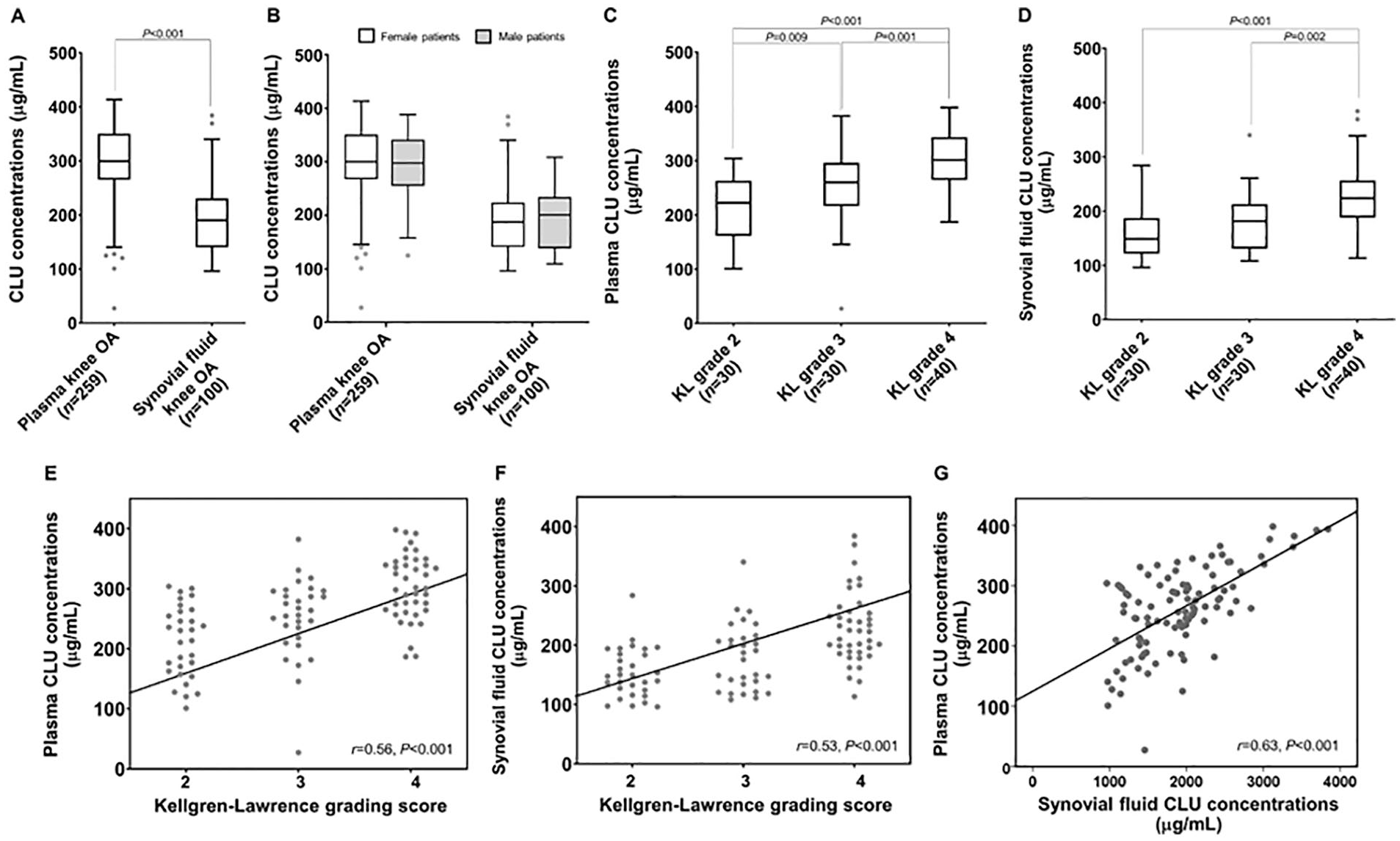
Plasma and synovial fluid CLU levels in study subjects among different groups. (
Associations of plasma and synovial fluid CLU levels with the radiographic severity of knee OA were determined subsequently. Spearman’s rho correlation analysis unveiled that plasma and synovial fluid CLU levels were positively correlated with KL grade (r = 0.56, P < 0.001; r = 0.53, P < 0.001; respectively; Fig. 1E and F ). Interestingly, there was a positive correlation between plasma and synovial fluid CLU levels in knee OA patients (r = 0.63, P < 0.001; Fig. 1G ).
Relationship between Plasma CLU Levels and Clinical Parameters of Knee OA
In addition to its association with the radiographic severity, we further investigated whether plasma CLU levels were associated with clinical variables of knee OA patients. As demonstrated in Table 2 , plasma CLU levels of knee OA patients were found to be negatively correlated with age (r = −0.28, P < 0.001), but positively associated with body composition parameters consisting of BMI values (r = 0.17, P = 0.016), percentage of fat (r = 0.33, P < 0.001), as well as fat mass (r = 0.35, P < 0.001), in addition to hs-CRP as a systemic inflammatory marker (r = 0.26, P < 0.001).
Spearman’s Rho Correlation and Multivariate Linear Regression Analyses of Plasma CLU Estimates in Knee OA Patients.
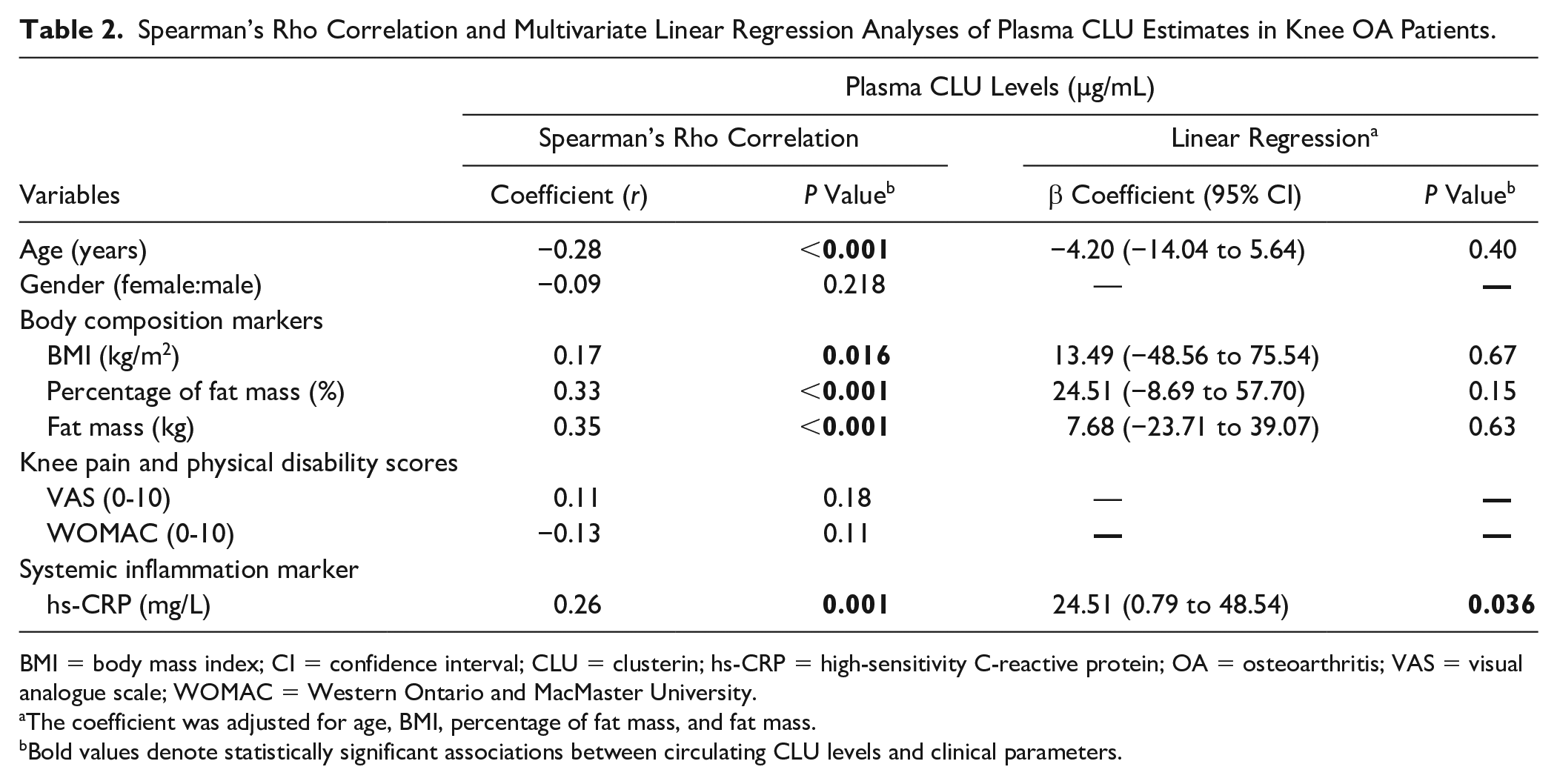
BMI = body mass index; CI = confidence interval; CLU = clusterin; hs-CRP = high-sensitivity C-reactive protein; OA = osteoarthritis; VAS = visual analogue scale; WOMAC = Western Ontario and MacMaster University.
The coefficient was adjusted for age, BMI, percentage of fat mass, and fat mass.
Bold values denote statistically significant associations between circulating CLU levels and clinical parameters.
To ascertain the independent associations between plasma CLU levels and clinical variables in knee OA, multivariate linear regression was performed. After adjusting for confounding variables including age, BMI, percentage of fat, and fat mass, an increment in plasma CLU levels was independently associated with increased levels of hs-CRP in knee OA patients (β-coefficient = 24.51; 95% confidence interval [CI]: 0.79 to 48.54; P = 0.036; Table 2 ).
Plasma and Synovial Fluid CLU as Potential Biomarkers for Knee OA Severity
We successively identified whether plasma and synovial fluid CLU levels could be employed as potential biomarkers for differentiating knee OA patients with advanced-stage (KL grade 3 or 4) from those with early-stage (KL grade 2). Based on the ROC curve, the optimal cutoff value of plasma CLU was projected to be 255.25 µg/mL, with a sensitivity of 71.4%, a specificity of 73.3%, and an AUC of 0.80 (95% CI: 0.70-0.89; P < 0.001; Fig. 2A ). Additional analysis revealed that the optimum cutoff value of synovial fluid CLU was identified at 175.00 µg/mL, which exhibited lower values of sensitivity (72.9%), specificity (70.0%), and AUC (0.77; 95% CI: 0.68-0.87; P < 0.001) than plasma CLU ( Fig. 2B ).
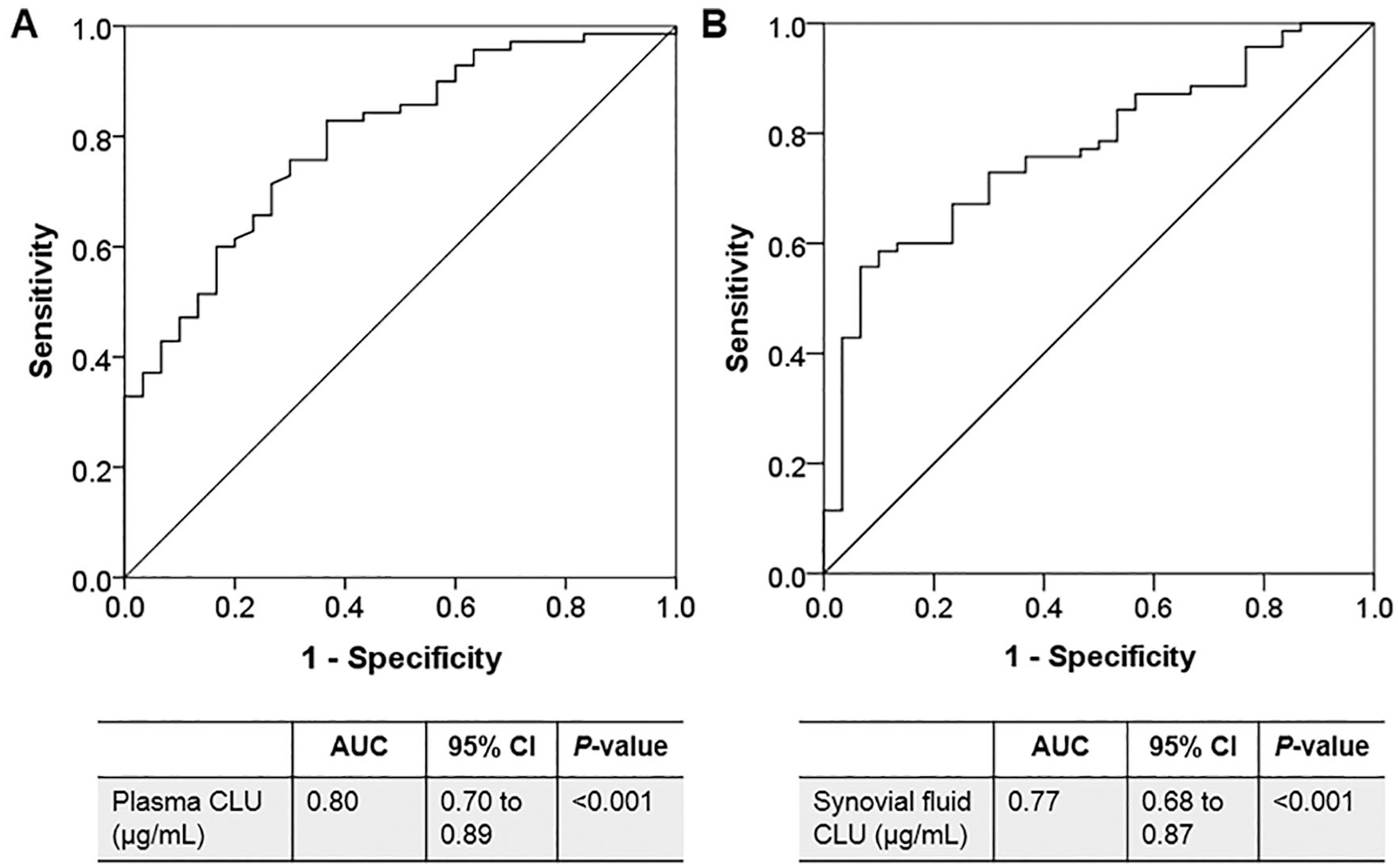
ROC of plasma and synovial fluid CLU as diagnostic biomarkers for knee OA severity. (
CLU mRNA Expression in Knee OA Synovium
Given increases in translational production of CLU in the systemic and local joint environments of knee OA, we examined CLU mRNA expression in the synovial biopsies of knee OA patients using quantitative real-time PCR. On the basis of the histopathological features for synovitis, of the 50 knee OA synovial tissues collected, 35 (70.0%) were identified as synovitis, and 15 (30.0%) were defined as no synovitis. Relative mRNA expression of CLU was observed to be remarkably up-regulated in knee OA synovitis, compared with those without synovitis (P < 0.001; Fig. 3A ).
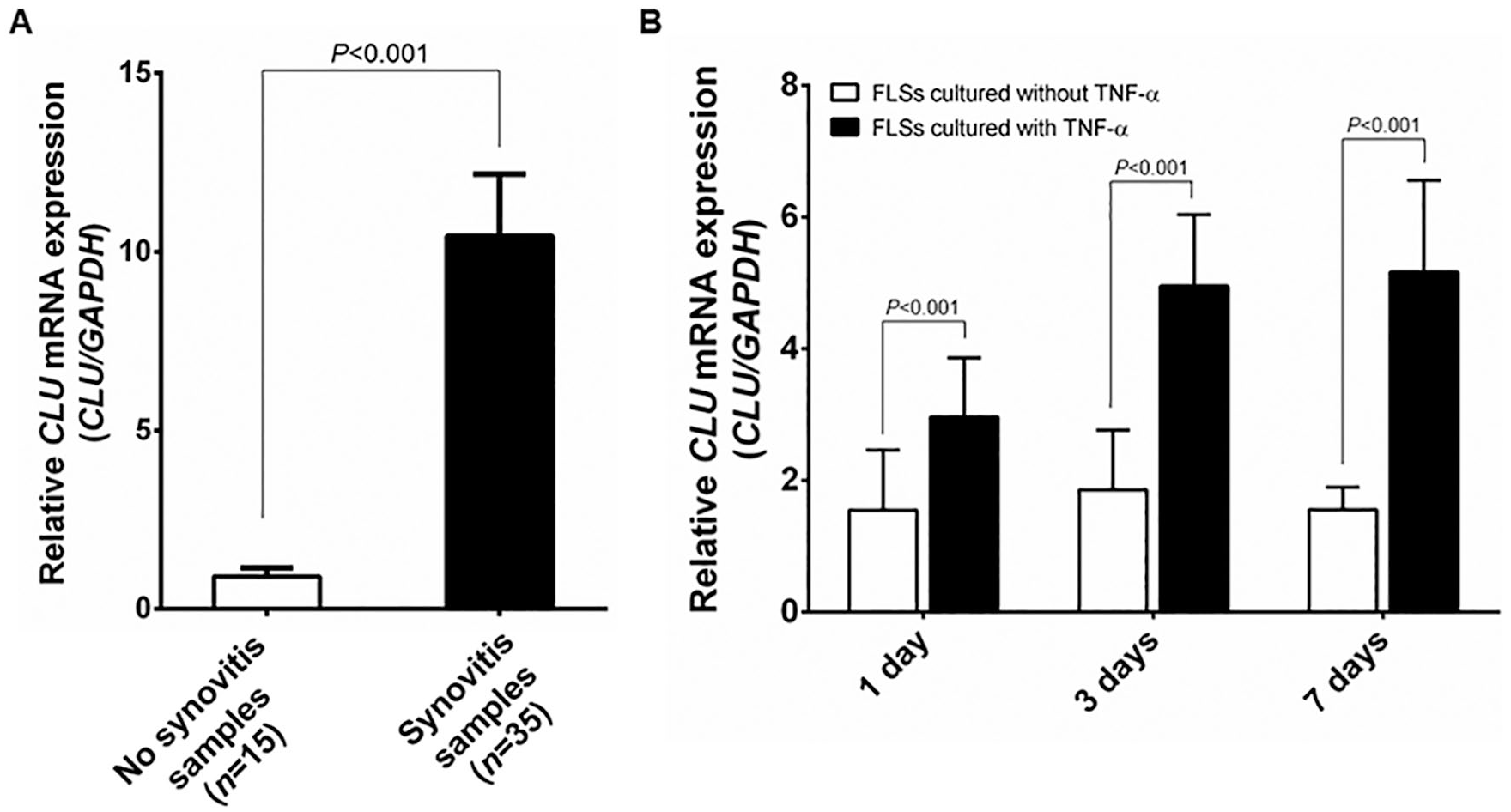
Relative CLU mRNA expression in synovial tissues and FLSs of knee OA. (
CLU mRNA Expression in Knee OA FLSs
Owing to TNF-α being a key molecule responsible for the inflammatory response in the synovium by stimulating the production of inflammatory mediators, we determined whether upregulation of CLU mRNA expression is modulated by TNF-α in FLSs isolated from knee OA synovium (n = 4, mean age 76.5 ± 7.2 years, 2 women and 2 men). Quantitative real-time PCR analysis showed that relative CLU mRNA expression was considerably increased in a time-dependent manner on days 1, 3, and 7 in knee OA FLSs cultured with TNF-α, compared with untreated knee OA FLSs (P < 0.001, P < 0.001, P < 0.001, respectively; Fig. 3B ).
Discussion
Our findings highlighted the clinical usefulness of CLU as a potential biomarker for knee OA severity—especially systemic and synovial inflammation. Synovial inflammation associated with chondrocyte apoptosis has been recognized as the pathological features involved in cartilage degeneration driving the development and progression of knee OA, thereby representing an important target for therapeutic intervention. Although there are numerous approaches conventionally employed in the detection and characterization of synovitis such as histologic or imaging assessments, these current available tools are imperfect and inadequate for early detection of the progressive synovitis. For this reason, there is an increasing awareness on the importance in identifying early phases of synovial inflammation-induced degenerative processes in knee OA. Since advances in soluble biomarkers may well yield the tools for appropriate stratification, the present study has been focused on investigating the expression and production of CLU in the systemic and local joint environments of knee OA patients.
As regards its regulatory roles in apoptotic and inflammatory processes, CLU has been proposed as a possible mediator for synovitis associated with cartilage degeneration. In regard to its anti-apoptotic action, CLU reportedly suppresses the activation of p53 signaling and stabilizes Ku-Bax complexes.16,25 In addition to this, it has been reported that CLU can alter the production of inflammatory cytokines including IL-6, IL-8, and nuclear factor kappa-B.19,26,27 In support of the significant involvement of CLU in arthritic diseases, Ritter et al. 28 drew attention to identifying biomarkers for OA progression and demonstrated that serum and synovial fluid CLU levels in OA patients were significantly associated with joint space narrowing. Furthermore, a recent study by Kropáčková et al. 20 showed that reduced circulating CLU levels were associated with pain severity in patients with erosive hand OA. These previous findings have been supported by our main results, which denoted significant increases in plasma and synovial fluid CLU levels of advanced-stage knee OA patients compared with those with early-stage knee OA and positive associations of CLU levels with radiographic grading and hs-CRP levels in knee OA patients. In line with our finding regarding its correlation with systemic inflammation, a number of clinical studies unveiled a close correlation between CLU levels and hs-CRP levels in multifactorial diseases like Alzheimer’s disease.29,30 These findings shed light on the usefulness of CLU as an important mediator for the inflammatory process in knee OA. Aside from increases in the systemic and local levels of CLU in late-stage knee OA patients, our subsequent result uncovered upregulation of CLU mRNA expression in the inflamed synovial tissues of knee OA patients compared to noninflamed synovial tissues. Consistent with our finding, prior studies described overexpression of CLU mRNA in OA cartilage and synovium.11-13,31 In parallel with increased mRNA expression of CLU in knee OA synovitis, an additional finding derived from our in vitro study showed upregulation of CLU mRNA expression in knee OA FLSs simulated with a pro-inflammatory cytokine like TNF-α, suggesting its marked relevance to inflammation in the synovium. Supporting this, an experimental study demonstrated an elevation in circulating CLU levels in hamsters in response to an acute phase of inflammation induced by TNF-α. 32 All our findings led us to consider the hypotheses that elevated levels of circulating and synovial fluid CLU might reflect knee OA severity, especially synovial inflammation, and that CLU levels may have diagnostic value as a biochemical marker of knee OA progression. To address these assumptions, we further performed ROC curve analysis revealing the clinical utility of plasma CLU as a noninvasive biomarker for discriminating knee OA patients with early-stage OA from those with advanced-stage OA. Based on this finding, CLU produced by knee OA FLSs and released into the circulation and joint fluid of knee OA could be an additional molecule involved in the pathogenesis of knee OA and cloud serve as a potential biomarker for knee OA severity. It is important to note that targeting the synovium to counteract CLU overproduction and to subsequently halt the excess accumulation of matrix-degrading mediators would be of great clinical relevance. Despite unknown mechanisms underlying the increased CLU levels in the circulation and joint fluid of advanced-stage knee OA patients, the possible reason for the significant result may come from a defensive mechanism by the body to fight against an imbalance between anabolic and catabolic processes that allow the cartilage extracellular matrix to maintain its structural integrity in knee OA. This phenomenon may help explain why increases in plasma and synovial fluid levels were found in patients with advanced-stage OA. Alternatively, apart from the joint tissues, extra-articular tissues reportedly produce and secrete CLU into the circulation,14,15 thereby significantly increasing plasma CLU levels in knee OA compared with paired synovial fluid samples of the patients.
In spite of the remarkable findings demonstrated herein, some inherent caveats of this study were unavoidable and require to be taken into consideration. One limitation of this study is the fact that this is a cross-sectional study, in which we cannot define cause-and-effect relationships. It should be emphasized that multicenter prospective cohorts are necessary to verify this observation. Unfortunately, due to ethical considerations, we were unable to harvest synovial fluid and synovial tissue samples from healthy controls, meaning the direct comparisons of mRNA and protein levels of CLU in the local joint environments between healthy controls and knee OA patients were unachievable.
Collectively, this is the first study to demonstrate significantly increased levels of CLU in the systemic and local joint environments of knee OA patients. Moreover, circulating and synovial fluid CLU levels were found to be directly associated with knee OA severity. Interestingly, ROC curve analysis uncovered the potential utility of plasma CLU as a biomarker for knee OA progression. Alongside an increase in its protein levels in the systemic and joint fluid of knee OA patients, overexpression of CLU mRNA was observed in knee OA synovitis. Additionally, an in vitro study revealed a time-dependent manner of upregulated CLU mRNA expression in knee OA FLSs stimulated with TNF-α. All findings suggest that alterations in mRNA and protein levels of CLU in the systemic and local joint environments might reflect severe complications in knee OA patients, particularly the progressive synovitis, and that circulating CLU could emerge as a noninvasive biomarker for knee OA progression. Further well-designed longitudinal studies are warranted to confirm the role of CLU as a diagnostic biomarker for knee OA severity, which would yield a specific biomarker for predicting the development and progression of knee OA. In parallel with this, future experimental studies are needed to elucidate the molecular mechanism underlying the exact role of CLU in the pathogenesis of knee OA, particularly synovitis, which could open a unique opportunity for a novel therapeutic target aimed at counteracting the progression of synovitis and eventual cartilage degeneration in knee OA.
Footnotes
Acknowledgments and Funding
The authors would like to gratefully acknowledge the Osteoarthritis and Musculoskeleton Research Unit and the Research Core Facility of the Department of Biochemistry and Chulalongkorn Medical Research Center (ChulaMRC) for kindly providing facilities. We are grateful to Dr. Artit Jinawath for his kind assessment for synovial inflammation. We also thank Dr. Thomas Mabey, University of Southampton, UK, and Dr. Jack Marshall, University of Liverpool, UK, for reviewing and proofreading the manuscript. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ratchadaphiseksomphot Fund, Faculty of Medicine, Chulalongkorn University (No. RA61/104), and the Research Chair Grant from the National Science and Technology Development Agency, Thailand. Ms. Tachatra Ungsudechachai has been partially supported by the MUPY Graduate Scholarship.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The experimental protocols conducted in conformity with the guidelines of the declaration of Helsinki were approved by the Ethical Committee on Human Research of the Faculty of Medicine, Chulalongkorn University (IRB No. 533/54), and the Faculty of Dentistry/Faculty of Pharmacy, Mahidol University (IRB No. 2019/074.2511).
Informed Consent
Written informed consent was provided by all participants prior to their enrolment in the study.
