Abstract
Introduction
Hip osteoarthritis is a prevalent condition responsible for important pain and disability. Most available guidelines for nonsurgical management of hip osteoarthritis recommend a combination of nonpharmacological and pharmacological treatment modalities. Intraarticular corticosteroid injections have been used for decades, although evidence is quite scarce, and many controversies remain.
Methods
This article reviews the available literature from Medline and Embase and discusses the evidence for intraarticular corticosteroid injections in hip osteoarthritis, where only 5 randomized controlled trials were found in the literature. These are analyzed in this article, which also aims to explain the main characteristics and features of glucocorticoids, along with their contraindications and potential adverse effects.
Results
Available randomized controlled trials show that intraarticular corticosteroid injections provide pain relief and functional improvement in hip osteoarthritis. This efficacy has not been shown with intraarticular hyaluronic acid injections.
Conclusion
This review shows that intraarticular corticosteroid injections are efficacious in hip osteoarthritis and that this benefit can last up to 12 weeks.
Introduction
Hip osteoarthritis (OA) is one of the most frequent osteoarticular conditions affecting the elderly. 1 Radiographic evidence of hip OA is found in nearly 5% of the population aged over 65 years, 2 while the estimated lifetime risk of symptomatic hip OA is approximately 25%. 3 It is responsible for important pain and disability. Nowadays, the prevalence of total hip arthroplasty (THA) is increasing. 4 However, efficient nonsurgical therapeutic options remain rather limited and often controversial.
Conservative management of OA relies on a combination of nonpharmacological and pharmacological modalities.5-7 Among those, intraarticular (IA) corticosteroid (CS) injections have been used for many decades, with the first report of IA CS use dating back to 1940; the practice was then established by Hollander in 1951. 8 Nowadays, they have become commonly recommended in the management of large joint osteoarthritis,6,7,9 including the hip. However, there are rather few studies addressing IA CS injections in hip OA while the subject has been extensively studied in the knee. Thus, it appears important to review the real efficacy and tolerability of IA CS injections in hip OA.
Methods
Medline and Embase were searched from inception to February 2019. We selected only human trials. Keywords included corticosteroid, hip, osteoarthritis, efficacy and tolerability, which gave 781 results. After reading the titles, 727 articles were excluded due to duplicate results and data irrelevant to the subject. Of the 54 remaining articles, 6 were eliminated due to lack of abstract and 15 for nonrelated data upon reading the abstracts ( Fig. 1 ). Reference lists of the 33 selected articles were also hand searched. Among these remaining 33 articles, 5 were randomized controlled trials (RCT) studying 346 patients with symptomatic hip osteoarthritis, of whom 134 had received an IA CS injection. Eventually, these 5 trials were included in this review.
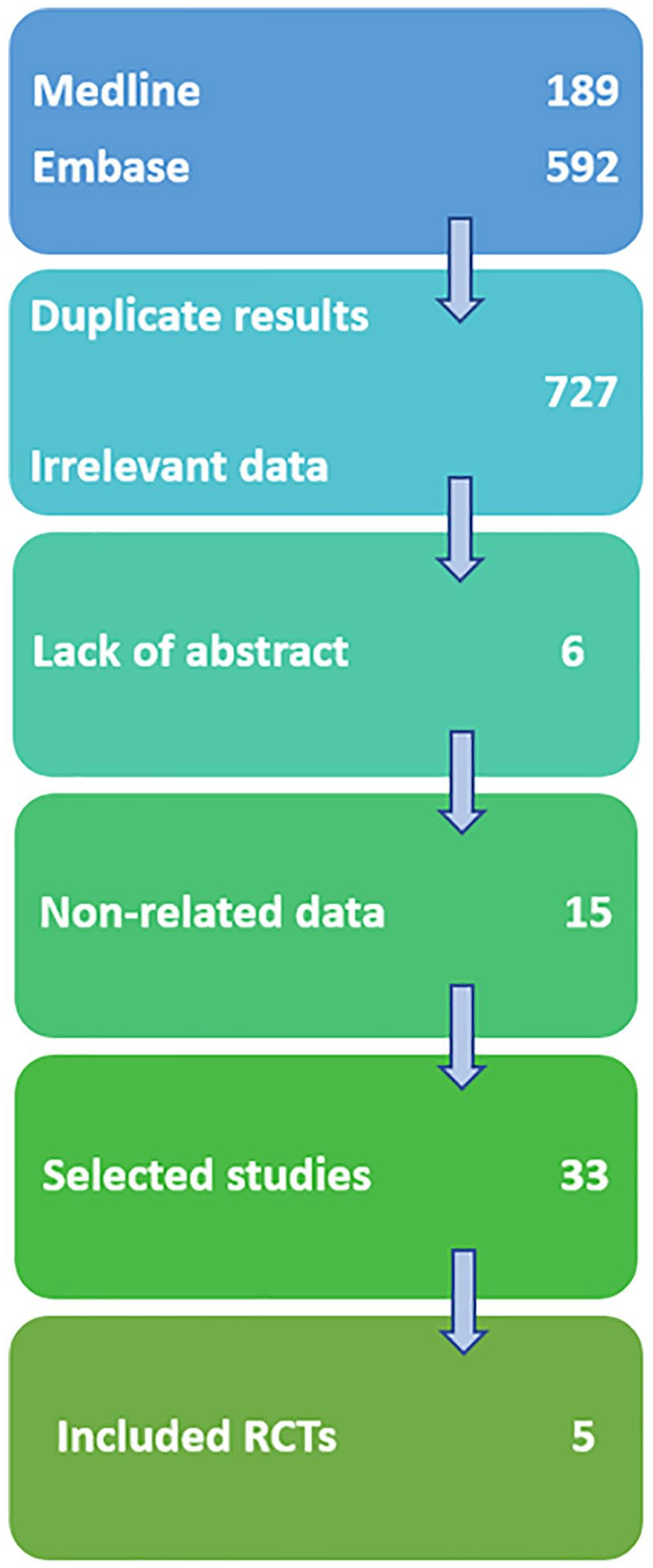
Flowchart (RCTs = randomized controlled trials).
Mechanism of Action of Corticosteroids
Corticosteroids have a complex mechanism of action, with immunosuppressive and anti-inflammatory effects. When injected intraarticularly, they have both local and possible systemic effects. By binding to nuclear steroid receptors, they interrupt the inflammatory and immune cascades at many levels and modulate several pro-inflammatory cytokines involved in cartilage damage and degradation. 10 They also interfere with the production of inflammatory mediators such as prostaglandins and leukotrienes. Ultimately, they lead to downregulation of immune function. 11
In OA, synovitis plays a key role in pain and disease progression. 12 While in systemic diseases, such as rheumatoid arthritis, it is logical to target the synovitis through a systemic approach, in a single joint affected by the osteoarthritic process it seems more interesting to favor the local administration of anti-inflammatory drugs like CS.
Corticosteroid Preparations
Approved CS for IA use can be particulate or nonparticulate. Particulate preparations are non–water soluble because they contain CS ester and require hydrolysis 13 by specific enzymes (esterases) to release their active compound and exert their action. Consequently, they theoretically last longer in joints,11,14 while water-soluble nonparticulate molecules do not need hydrolysis; they tend to have a quicker onset of action but are rapidly eliminated from the joint.
Most frequently used CS include methylprednisolone, betamethasone acetate, and triamcinolone (acetonide and hexacetonide). Methylprednisolone is a methyl derivative of prednisolone while betamethasone and triamcinolone are fluorinated; they are all particulate compounds 11 ; they last longer in the joint than nonparticulate soluble preparations (dexamethasone, prednisolone acetate), which are rapidly cleared from the synovial fluid.
Recently, a new CS has been approved for IA use: triamcinolone acetonide extended release, with microsphere technology prolonging its persistence in the joint, hence increasing its duration of action. 15 This molecule has been studied in the knee in double-blind RCTs and has been found to induce a prolonged response, with both pain and functional limitation decreasing up to 12 weeks after the injection.15,16 Detectable levels of the drug in the synovial fluid were still present up to 12 weeks after one single IA injection. 15 Moreover, there were no serious adverse effects with this extended release formulation, which seems very promising, even though it has not been studied yet in other joints.
Contraindications and Precautions for Use
The most important contraindication to IA CS is the presence of infection, and it is the same for any joint, including the hip. Infection, whether it is systemic or local, including overlying skin infection, 17 is an absolute contraindication to IA CS use. Similarly, injecting through a skin lesion should be avoided. 18 An IA fracture also contraindicates CS injection. 17
Coagulopathies and anticoagulant use should be considered with caution; while they can be overlooked for some superficial joints, for deep ones, especially the hip, they should definitely be taken into account. For example, injections in knees and shoulders in patients taking warfarin have been shown to be safe with international normalized ratio (INR) values up to 5.5. 19 However, the hip is a much deeper joint, and it is hardly accessible for compression. Coxo-femoral joint injection is considered to carry a moderate risk of bleeding.20,21 Consequently, experts recommend withholding anticoagulants before hip IA CS injections in the hip, and bridging heparin therapy should be discussed depending on every patient’s case. 20 However, aspirin treatment can be safely maintained, while other antiaggregant should be withheld and replaced by aspirin, if possible, to allow the IA hip injection. 21
Other absolute contraindications for IA CS injections are known allergy to the used product or to one of its excipients, while relative contraindications injection also include poorly controlled hypertension and hyperglycemia. 22
Adverse Effects
IA CS injections can have local and systemic adverse effects. Local side effects are more common and include postinjection flare, septic arthritis, skin depigmentation and fat atrophy, and tendon rupture if the injection was not performed strictly intraarticularly. 11 Infection is the most feared and most severe complication of IA CS injections. Yet, with proper sterile technique, it seldom occurs, with incidence rates as low as 1 in 50 000 injections in some articles.14,23 On the other hand, postinjection flare, though benign, is the most commonly encountered reaction to IA CS use, occurring in up to 26% of patients.18,24,25 It develops hours after the procedure and can be quite painful and inflammatory, mimicking an infection, but usually resolves spontaneously within 3 days.11,14
Skin hypopigmentation and local fat atrophy can occur with less soluble agents, particularly with triamcinolone hexacetonide, and is usually due to inaccurate needle placement and extraarticular steroid injection.11,14 They usually develop 1 to 4 months after the procedure and are generally transient, although they might take several months to regress. 24
There are a few case reports of rapid destructive (RD) OA of the hip after IA CS. 26 However, there is no proof that this rare and poorly understood condition was associated with the procedure, and not a natural evolution of the patient’s disease, with no relation to the IA CS injection. A retrospective cohort trial published in 2018 27 found that RD OA of the hip occurred in 21% of patients receiving IA CS in the hip, but it only looked at procedures performed in the radiology department and only included 109 individual hips (129 injections). Nonetheless, the authors raised concern for the possibility of this complication occurring after an IA CS injection, and suggest informing patients of this risk, among others, before performing the injection. 27
Similarly, osteonecrosis of the femoral head has also been sporadically reported following IA CS injection.22,28-30 Interestingly, those cases occurred more often after injections in other joints. But, as for RD hip OA, the causality with IA CS is not clearly elucidated, and osteonecrosis is generally considered to be linked to a systemic CS administration or a more severe underlying disease, rather to a complication of the injection itself. However, there is no solid data to support this.
Another concern with IA CS injection in the hip is the possible increased risk of infection in patients undergoing subsequent THA. This has been especially discussed with multiple injections. 31 However, in hip OA, strong evidence is lacking, and it is generally believed that IA CS injections, overall, do not lead to increased rates of infection in case of joint arthroplasty. 32 Still, authors generally used to recommend avoiding IA CS within at least 2 months before hip replacement surgery, 22 and more recent data recommend waiting for 3 months after an IA injection of steroids before performing this surgery.33,34
Possible systemic side effects of IA CS include facial flushing, which has been reported in up to 15% of patients.11,35 It usually occurs 2 to 30 hours after steroid injection but does not last more than 36 hours. It has also been reported more frequently with triamcinolone preparations, even though it may be associated with any corticosteroid compound. 35
Transient hyperglycemia has been reported in diabetics following IA CS injections.14,36 It usually subsides around 5 days after the injection. 11 Diabetic patients should be informed about the expected rise in their blood glucose level.
Injection in the Hip
The hip is a deep joint. Consequently, anatomic landmarks are less reliable than in other more superficial joints such as the knee. Furthermore, the hip joint is adjacent to important neurovascular structures, and non–image-guided injection might carry a risk of injury to the femoral artery, femoral nerve, and lateral femoral cutaneous nerve. 22 For this reason, fluoroscopic or ultrasonographic (US) guidance are useful adjuncts in IA hip injection and are especially interesting when using nonsoluble steroid preparations that require a strict IA placement. Both methods allow a proper visualization of the needle during injection. The advantage of US guidance is the absence of ionizing radiations and the visualization of soft tissue. Furthermore, no contrast is required for US-guided injections.37,38
Corticosteroid Injection in the Hip: International Guidelines
IA CS injections are now part of many recommendations for pharmacological management of hip OA. While EULAR recommendations published in 2005 stated that they “may” be considered in flares of hip OA, in case of failure of nonsteroidal anti-inflammatory drugs (NSAIDs) and analgesics, 9 newer guidelines issued by the American College of Rheumatology in 2012 6 recommend their use the same way they recommend using analgesics, including opioids and oral NSAIDs. Indeed, according to these recommendations, patients with hip OA “should” use either paracetamol, oral NSAIDs, tramadol, or IA CS. Furthermore, guidelines published by the American Academy of Orthopedic Surgeons in 2017 state that strong evidence supports their use in symptomatic hip OA for short-term pain relief and functional improvement. 39 The newest international guidelines have been issued in 2019 by OARSI (Osteoarthritis Research Society International). While IA CS are recommended for knee OA, such recommendation does not exist for the hip where nonpharmacological treatments are generally preferred over pharmacological interventions—including IA steroid injections. Indeed, IA injection—which requires strict aseptic conditions—is more difficult and may be less accessible in the hip. Moreover, it might have a poorer safety profile and has not reached a sufficient efficacy compared to IA CS injection for knee OA. 40
Corticosteroid Injection in the Hip: The Evidence
IA CS injections have been used widely in hip OA despite the paucity of available data, compared with knee OA. In fact, only few trials have been conducted in symptomatic hip OA, and some evidence has emerged from a few randomized trials during the last 2 decades, but its quality remains relatively poor. 41
In 1988, Flanagan et al. reported that the benefit of IA CS injections in hip OA was too transient. It did provide pain relief, especially night pain, but this effect rapidly declined, and at 3 months, 50% of the patients who received IA steroids and local anesthetics (LA) reported being worse. 42 However, this trial carried a major bias, for patients were all awaiting total hip replacement and were informed that those getting worse after IA injection would be given priority for this surgery. Nonetheless, although the authors concluded that IA CS injections in the hip were useless, and even harmful, since most patients reported a worsening of their symptoms after the injection, this trial did show that, among patients with hip OA receiving IA CS, 75% had pain relief at 1 month and 33% maintained this good response at 3 months. 42
In 2004, a double-blind RCT conducted by Kullenberg et al. compared 80 mg of triamcinolone acetonide to LA injected intraarticularly, under fluoroscopic guidance, for symptomatic hip OA. 43 Three and 12 weeks after the injection, there was a significant decrease in pain at rest and weight-bearing for patients who received IA CS, with the largest differences seen at 3 weeks. 43 Indeed, pain at rest decreased from 5.3 on a visual analogue scale (VAS) at baseline to 1.3 at 3 weeks in the IA CS group, whereas it only reached 5.0 with LA. Similarly, at 3 weeks, pain at activity was also reduced from 6.9 to 2.5 in the CS group while it did not decrease in the LA group (from 6.9 to 7.0). Moreover, at 3 weeks, hip range of motion improved in all directions, as well as functional ability, and analgesics consumption decreased with steroids compared to LA, and all these results were statistically significant. Importantly, there were no adverse effects reported in patients treated with IA CS injections. 43
Then, in 2006, Qvistgaard et al. conducted a prospective, double-blind RCT comparing an injection of 1 mL of methylprednisolone (40 mg), under US guidance, to the same volume of saline water. 44 The primary outcome was pain on walking, and it showed a statistically significant decrease with IA steroids, 14 and 28 days post-injection, whereas there was no such improvement with saline. Indeed, with IA CS injections, pain on walking decreased from 45 on a VAS at baseline to 33 at 14 days, and 30 at 28 days, while it actually increased (46) at 14 days with saline water, then was barely diminished (43) at 28 days. 44 All these results were statistically significant. However, there was no significant difference after 3 months, which lead to the conclusion that IA CS injections in hip OA provided only short-term pain relief, and this transient improvement failed to be maintained beyond 1 month. 44
On the other hand, in 2007, Lambert et al. showed sustained efficacy for IA CS injections in hip OA. In a double-blind RCT comparing fluoroscopically guided injections of IA steroids and LA to LA alone, they not only showed a significant difference between groups, favoring steroids, but this discrepancy was maintained at 3 months post-injection. 45 Notably, the compound used in this trial was triamcinolone hexacetonide, which is highly insoluble and known to last longer in the joint. The primary outcome in this study was based on the Western Ontario and McMaster Universities OA Index (WOMAC) and was defined as a 20% improvement in pain (WOMAC20) at 2 months post-injection. 45 Significant differences in WOMAC pain scores were found between the 2 groups at month 2: in the IA CS group, mean WOMAC at baseline was 310.1 and it decreased to 157.4, while in the placebo group, it was 314.3 at baseline and only reached 306.5 two months later, with a P value <0.0001. 45 WOMAC20 was also significantly greater in the steroid arm: at 2 months, 67.7% of patients were responders, compared to only 23.8% in the placebo group. Significant differences were also observed in WOMAC50 (a 50% decrease in WOMAC pain score), WOMAC stiffness and physical functions scores, and quality of life scores (Short Form-36). 45 Most important, all those benefits lasted up to 3 months, and there were no significant differences in adverse events and complications between groups. 45
Compared to the efficacy of IA CS injections in knee OA, which seems quite short-lived, their effect in the hip is more prolonged. Indeed, in a more recent Cochrane review, it was shown to last 6 weeks in the knee (“small to moderate” effect) and there was no effect at 13 weeks. 46
In 2010, Atchia et al. showed similar results. They compared US-guided IA hip injections of 120 mg of methylprednisolone acetate (MPA) to those of normal saline and to standard care (no injections) in patients with hip OA. 47 Of note, they only evaluated the response at 8 weeks. The results were strongly in favor of steroid IA injections, for both pain relief, using a Numerical Rating Scale (NRS), and WOMAC scores, and these achievements were maintained at 8 weeks. 47 Effect sizes of the efficacy of IA CS were large at 1 week (1.5 for NRS pain, 1.9 for WOMAC pain and 1.3 for WOMAC function) and decreased by 8 weeks (0.5, 0.6, and 0.4 respectively), though the results were still in favor of IA steroid injections. 47
Prognostic Factors of Response
Importantly, in their study, Atchia et al. evaluated the possible effect of preexisting synovitis, which was possible to visualize with US, on the response to injections, and showed that the presence of synovitis was a significant predictor of response to IA CS injections at 4 and 8 weeks. 47 Indeed, in the CS group, composed of 19 patients, 14 were responders at week 1, 11 at week 4, and 7 at week 8. Among those, 10 out of 14, 10 out of 11, and 7 out of 7 had a synovitis at baseline. Interestingly, no patient without synovitis had a sustained response over 8 weeks. 47 Before this study, there was no solid data pertaining to the possible factors influencing response to steroid IA injection.
Radiographic severity of OA could also be related to the degree of benefit provided by IA CS injections in the hip. Back in 1997, an open study by Plant et al. suggested that there might be no benefit for IA CS in “atrophic” hips. 48 The concept of atrophic hip pattern in OA was first described by Solomon in 1976 49 and is equivalent to grades 0 and 1 of the Kellgren-Lawrence (KL) scale (0 = Normal radiograph and 1 = Doubtful narrowing of the joint space and possible osteophyte lipping).50,51 Looking at this possible relation between radiographic damages and pain relief after IA CS injections in hip OA was the aim of a retrospective study conducted by Deshmukh et al. in 2011. 51 It showed that, while age and gender were not significant predictors of response, radiographic severity was linked to the efficacy of IA steroids. Indeed, the authors showed that the patients with severe hip OA—that is, grade 3 and 4 hip OA based on the KL scale—benefited the most from IA CS injections, and were much more likely to experience pain relief compared to patients with mild OA—that is, grade 0 and 1 of the KL scale. 51
Of note, other studies had suggested that patients with less severe imaging findings could have a better response, which might seem logical since patients with less damage in their joint and less stiffness or disability might experience more improvement once inflammation decreases after local administration of CS. However, those results were not statistically significant, 52 while the data produced by Deshmukh et al., though emanating from a retrospective study, reached statistical significance. 51
Obesity is also considered to be a predictor of nonsufficient or insufficient response, although there is no solid evidence for that. Patients with lower body mass index (BMI) were found to have greater chances of improving after IA CS injections in the hip, but these results were not significant. 52 Yet it may seem like common sense that higher BMI could reduce the extent of pain relief, especially at weight-bearing, but to date, there is no data demonstrating this. Consequently, due to the lack of clear evidence, it is believed that patients’ characteristics, such as BMI, but also age or gender, do not correlate with pain relief after an IA CS injection in the hip. 53
Dose and Volume of Injectate
In 2007, Robinson et al. published the results from a pragmatic clinical study that evaluated 2 different dosages of CS. They used 40 mg and 80 mg doses of methylprednisolone, which were injected in the hips of patients with hip OA, under fluoroscopic guidance. Although it was not randomized nor placebo-controlled, it managed to show some superiority for the higher dose, which provided a more sustained benefit. Indeed, both doses induced significant improvement in WOMAC pain and stiffness scores at 6 weeks, but only the 80 mg dose allowed to maintain this response at 12 weeks. 52 Further studies and RCT are required to provide evidence regarding this finding.
Volume of injectate could possibly play a role in the potential response to IA hip injections, for increasing the volume of fluid injected (often by adding a LA) could improve the distribution of the corticosteroid in the synovium. 53 That was the rationale behind an RCT conducted in 2012 by Young et al. who gave patients with hip OA IA injections of either 1 mL of triamcinolone acetonide (40 mg) and 2 mL of LA, or that same combination with an additional volume of 6 mL of saline water. Procedures were fluoroscopically guided. Both groups showed a significant improvement, with WOMAC scores decreasing for pain, stiffness, and activities of daily living, but the study failed to show a statistically significant difference between 3 and 9 mL volumes. Eventually, despite a similar efficacy of both injection volumes, the authors stated that using a higher one was not harmful, and hence recommended using volumes between 3 and 9 mL in the hip. 53
Repeated Injections and Injection Rate
There is no evidence-based consensus on how often a joint should be injected and how many times it can be treated with IA steroids.24,54 Traditionally, the most frequently accepted limit is that of 3 to 4 injections per joint in 1 year, especially in weight-bearing joints.18,54 Some authors recommend 1 injection every 1 to 3 months, but all data suggest not giving more than 4 yearly injections per joint. 24
Comparison with Hyaluronic Acid IA Injections
Hyaluronic acid (HA) is one of the main components of cartilage matrix and synovial fluid. Aging decreases HA concentration by 33% to 50%14,55 and, along with OA, reduces the quality of HA in the synovial fluid. 56 Hence, it is thought that viscosupplementation (VS) might improve cartilage biochemistry in patients with OA 54 and might even have chondroprotective effects, even though these have only been shown in vitro and have not been proven in human trials.14,55,57
However, the use of HA in the hip remains quite controversial since studies in this field have shown conflicting results. Additionally, no evidence exists to recommend one type of VS over another, although it is thought that high-molecular-weight HA might have a better clinical efficacy. 58
Evidence from RCT comparing IA CS injections to VS in the hip is quite scarce. The trial conducted by Qvistgaard et al. included HA among compared modalities. 44 They randomized patients into 3 arms of treatment, one treated with IA CS (1 injection of methylprednisolone, 1 mL, followed by 2 sham injections), one with saline water (3 injections), and the third with HA (Hyalgan; 3 injections of 2 mL each). While this trial showed significant improvement with steroids as opposed to saline water, VS failed to show any statistically significant efficacy on pain on walking, compared to saline treatment. 44
On the other hand, in 2010, Spitzer et al. compared IA MPA and HA in patients with mild to moderate hip OA (KL grades 2 and 3). Patients were given 2 HA injections or 1 MPA injection followed by one simulated injection. 59 This study showed similar WOMAC responses in patients with milder disease (KL grade 2) but better response with VS in more advanced OA (KL grade 3). Importantly, both treatments were equally safe, and no difference was noted in adverse events between the 2 groups. 59
The trial conducted by Atchia et al. also studied HA. It actually divided included patients into 4 treatment arms: no injection, IA saline water, VS (Durolane, 3 mL, single injection), and IA injection of MPA. The results were strongly in favor of CS, which provided pain relief and function improvement, while the administration of HA failed to produce any demonstrable benefit. 47
This seemed to corroborate the results of a study conducted by Richette et al. in 2009, who showed that one single injection of HA (Adant), under fluoroscopic guidance, was no more efficient than a saline injection. 60
Of note, due to the use of fluoroscopic guidance in Richette’s trial, contrast was also injected, which might have diluted the concentration of injected HA. Additionally, these studies might have been too short (12 weeks or 3 months, or less), for some authors consider that the potential effect of VS is much slower than that of CS, but it might last longer, while the efficacy of IA CS tends to wean off more rapidly. 61 Further long-term, high-quality studies are required in this field.
Discussion
IA CS injections in hip OA are part of most international recommendations.6,7,9,39,40 However, our review shows that the number of RCTs is limited, and the quality of the evidence remains relatively poor. 41 In fact, the latest available guidelines, issued by the OARSI in 2019, emit no recommendation when it comes to IA CS in the hip, and remain globally against pharmacologic interventions in hip OA. 40 Among the 5 available RCTs, the earliest one 42 produced negative results at 3 months post-injection of CS (despite good initial results) yet these results were biased. On the other hand, the 4 remaining studies43-45,47 showed positive results favoring IA CS, as compared to placebo and HA,44,47 though relatively short-lived (≤3months) 46 ( Table 1 ).
Summary of the Main Results in the 5 Randomized Controlled Trials (RCTs) Selected and Included in Our Review.
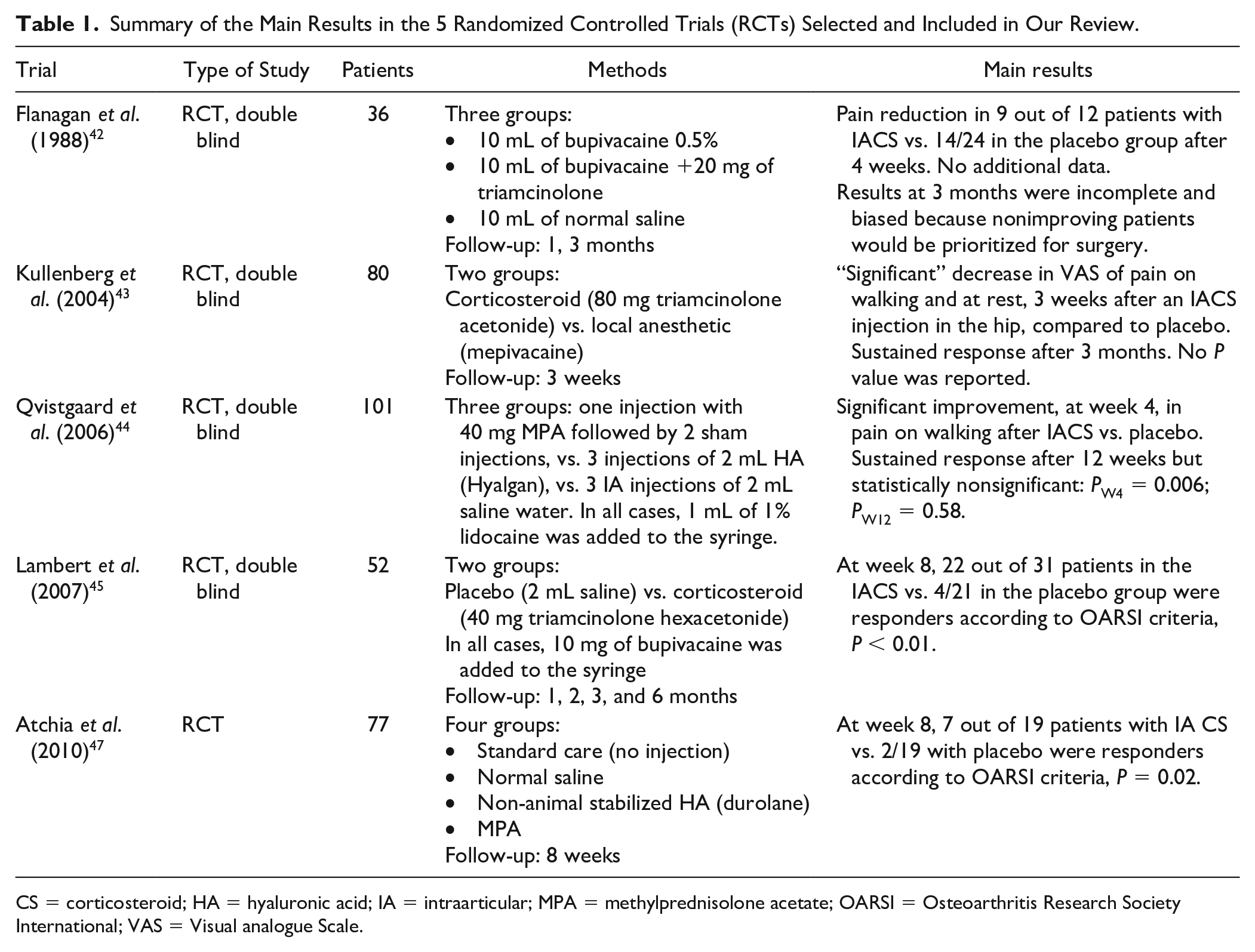
CS = corticosteroid; HA = hyaluronic acid; IA = intraarticular; MPA = methylprednisolone acetate; OARSI = Osteoarthritis Research Society International; VAS = Visual analogue Scale.
Importantly, IA hip injections, unlike those performed in the knee, where anatomical landmarks are more reliable, require some expertise and ideally need imaging guidance, by fluoroscopy or US. 37
Even in the absence of contraindications for IA steroid injections, poorly controlled hypertension or hyperglycemia, and coagulopathies11,14 must be acknowledged.
There are very few studies addressing injected dose and volume. It seems that 80 mg of methylprednisolone gives better results than 40 mg. 52 No solid data exist for doses of other products. The recommended volume is 3 to 9 mL, higher volumes failing to show superiority compared to 3 mL. 53
The presence of synovitis is an important predictor of response while an atrophic radiographic pattern seems related to a lack of improvement after IA CS injections in the hip.46,50 Additionally, there is no sufficient data concerning the impact of obesity on the efficacy of IA CS in hip OA. 52 There are no studies allowing to determine what is the ideal profile of patients who would benefit from these injections, and in whom IA CS would be likely to achieve a good response.
It is common sense to recommend IA CS injections in hip OA in patients in whom surgery is not yet indicated or when there is a contraindication to surgery. However, the optimal interval between repeated injections and the maximum allowed number of such injections have not yet been elucidated. Similarly, there seems to be a lack of consensus regarding the feasibility of these procedures when a THA is already indicated; the required time between an IA CS injection and a THA is not yet clearly known, although most authors recommend waiting at least 3 months.
It is also common sense to consider local interventions like IA CS in patients with symptomatic hip OA who have multiple comorbidities contraindicating other pharmacologic interventions, notably the use of NSAIDs or even analgesics such as acetaminophen or tramadol.
Of note, LA are often injected along with CS. Nevertheless, high concentrations of LA have been shown to be cytotoxic for chondrocytes, especially with older molecules such as bupivacaine. 11 One might then consider avoiding their usage in IA CS injections to prevent this potential chondrotoxicity.
Globally, IA CS injections in the hip have shown better results than IA injections of HA, but this superiority was only studied over 12 weeks.44,47 The potential effect of VS may, in fact, be slower than that of CS, but it might last longer, while the efficacy of IA CS tends to wean off more rapidly. 61
Future Agenda
Further studies are required to determine the ideal dose of CS, but also the recommended rate and the maximal number of allowed injections. Trials should also be conducted to elucidate the innocuity of these injections before a THA, the good and bad prognostic factors and the association between some patients’ characteristics and the expected response to IA CS in hip OA. Only one study considered the presence of synovitis and studied its relation to efficacy of IA injections. 47 This should be further studied, especially with the growing availability of musculoskeletal US, which should allow a better and more precise evaluation of synovitis in the studied hip joints.
There is also a lack of high-quality studies in mild hip OA, and this issue should be addressed in the future.
Similarly, there is no solid data regarding the potential effect of repeated IA CS on the hip cartilage, and further RCTs are needed to study the possibility of a disease-modifying effect of these injections. A potential toxicity of CS on the cartilage has been described in knee OA, in a recent study, 62 though this trial used IA CS given every 3 months, over 2 years. It is not known if this deleterious effect was due to the steroid itself or to the repeated injections, and there is no such data in hip OA.
Conclusion
IA CS injections in hip OA provide significant benefit by inducing pain relief and increased range of motion, improving functional ability, and decreasing analgesics consumption. 43 They are recommended in symptomatic hip OA by most international guidelines and have shown better responses than HA. However, the number of trials addressing this highly prevalent disease remain quite limited and further studies are needed to better elucidate all the issues related to IA CS injections in hip OA.
Footnotes
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
