Abstract
Objective
To establish a method for investigating the permeability of calcified cartilage zone (CCZ) and to observe solute transport between articular cartilage (AC) and subchondral bone (SB) through intact CCZ in vivo.
Design
We developed a novel fixing device combined with un-decalcified fluorescence observation method to address the permeability of CCZ in live mice. Twenty-four Balb/c female mice aged 1 to 8 months were used to observe the development of CCZ. Eighty-four Balb/c female mice (aged 1 or 6 months) with mature or immature CCZ of distal femur were used to investigate the permeability of intact CCZ in vivo. Diffusivity of rhodamine B (476 Da) and tetramethyl-rhodamine isothicyanate-dextran (TRITC-Dextran, 20 kDa) was tested from AC to SB in 0 minutes, 1 minute, 15 minutes, 30 minutes, 1 hour, and 2 hours. None diffused knee joints (0 minutes) served as blank control, while in vitro immersion of distal femurs in rhodamine B or TRITC-Dextran for 72 hours served as positive control.
Results
CCZ was well developed in 6-month mice. Both tracers penetrated immature CCZ down to SB in less than 1 hour in live mice, while the diffusion of both tracers decreased rapidly at tidemark in all testing time points.
Conclusion
Current study provided direct evidence of blocking effect of CCZ in solute transportation during short diffusion period in live animal, indicating the important role of CCZ in joint development and microenvironment maintenance.
Introduction
Osteochondral tissue composed of articular cartilage (AC), calcified cartilage zone (CCZ), and subchondral bone (SB) is a key integrated unit in modulating skeletal function and maintaining joint health. 1 Although these tissues are regarded as a unit, the components vary. The AC mainly consists of water (>70%) and 2 major organic components, type II collagen and aggrecan;2-4 the SB is mainly constructed by type I, type X collagen, and hydroxyapatite; 5 the CCZ contains components from both AC and SB and serves as mechanical supporter in between.6,7 It has been suggested that CCZ helps transform shear stress into compressive and tensile stress during weight-bearing and joint motion. 8 Moreover, CCZ is also able to attenuate load forces on cartilage according to its unique undulating interface and biocomposite formed by the calcified cartilage and underlying bone. 9 Despite the well-established mechanical function of CCZ, few studies have shed light on its permeability. It is commonly accepted that cartilage is avascular, aneural, and hypoxia, 10 while bone is rich in blood supply and aerobic. 11 The CCZ may act as a barrier that interrupts the transfer of interstitial fluid between AC and SB, 12 leading to the stabilization of the physiological microenvironment of hyaline cartilage. However, the exact permeability of CCZ and its regulatory role in AC and SB cross-talk is unclear.
Understanding the permeability of CCZ is essential to reveal the pathogenesis of joint diseases such as osteoarthritis (OA). During the development of OA, changes in SB such as sclerosis, formation of cysts, alteration in trabecula integrity, and development of osteophytes13,14 are normally observed. Turnover and remodeling of SB may release growth and inflammatory factors, affecting the overlying cartilage. 15 Due to the potential cross-talk pathway, 16 degenerated cartilage may in turn accelerate the pathogenic remodeling of SB, resulting in the initiation and progression of OA. 17 To date, studies on the permeability of the CCZ are scarce and obscure. Although pillaries, canals which may contain blood vessels, were observed in CCZ,16,18-20 the direct evidence of solute transport between AC and SB through CCZ is lacking. In early studies, it was thought that no substance including hydrogen gas, 21 water, and sodium fluorescein (376 Da) 22 could communicate at the osteochondral interface in vivo. However, these conclusions have been challenged recently. Studies using fluorescence loss induced by photobleaching (FLIP) showed that diffusivity of sodium fluorescein was quantified to be 0.07 ± 0.03 µm2/s between SB and calcified cartilage in murine, suggesting that the CCZ was permeable to small molecules. 23 This observation was also confirmed by Arkill and Winlove, 24 who demonstrated that CCZ was permeable to small solutes such as fluorescein and rhodamine in horse joint. It is noteworthy that, in order to analyze and observe the precise dynamic of fluorescein dye in the CCZ, sample dissection and pretreatments were often required in these in vitro studies, which might result in inevitable changes of the native character in the tissue and impair the accurateness of the observation. Therefore, new observation methods that can measure the permeability of intact CCZ in live condition are needed.
In this study, to address the permeability of CCZ in vivo, custom designed fixing device and un-decalcified fluorescence observation method were employed. Furthermore, in order to investigate the role of CCZ in regulating the nutrition and cytokine diffusion between AC and SB, mature mice (6 month) with well-developed CCZ and young mice (1 month) with developing CCZ were selected as experimental animals. We hypothesized that intact mature CCZ would serve as a barrier to block the AC-SB cross-talk and play a central role in modulating cartilage microenvironment during joint development and pathological progression.
Materials and Methods
Animal
All animal procedures were in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals and were approved by the Animal Ethics Committee of Third Military Medical University (Army Medical University). Twenty-four Balb/c female mice (aged 1-8 months) were used to observe the development of CCZ. Eighty-four Balb/c female mice (aged 1 or 6 months) were used to investigate the permeability of CCZ. All mice were purchased from the Experimental Animal Center of Third Military Medical University.
Histologic Analysis of CCZ Development
To observe the development of CCZ, 24 Balb/c mice aged 1 to 8 months were selected, 3 mice in each month. After all mice were sacrificed using carbon dioxide euthanasia, knee joints were dissected and fixed in 4% paraformaldehyde for 3 days, followed by decalcification in 10% ethylenediaminetetraacetic acid (EDTA) for 2 weeks. Samples were subsequently embedded in paraffin and sectioned at 5 µm in sagittal plane. In order to investigate the morphology of CCZ, sections were stained with hematoxylin-eosin (H&E) and safranin O-fast green. Zen software (version 2012, Carl Zeiss) was utilized to measure the thickness of AC (the distance between cartilage surface and tidemark) and CCZ (the distance between tidemark and cement line).
Live Animal Permeability Experiment
In order to investigate the role of CCZ in regulating substance diffusion between AC and SB, 84 mice aged 1 month or 6 months were separated into the immature-CCZ group and mature-CCZ group. To mimic the diffusion of amino acid, glucose, or inflammatory cytokine between AC and SB, rhodamine B (467 Da, 0.1%, Solarbio, Beijing, China) and tetramethyl-rhodamine isothicyanate-dextran (TRITC-Dextran, 20 kDa, 0.1%, Ruixi, Xi’an, China) were tested. Animals were then divided into 4 subgroups: mature-CCZ + rhodamine B, mature-CCZ + TRITC-Dextran, immature-CCZ + rhodamine B, immature-CCZ + TRITC-Dextran. To investigate the relationship between substance permeability and diffusion time, time points of 1 minute, 15 minutes, 30 minutes, 1 hour, and 2 hours were selected. None diffused knee joints (0 minute) served as blank control, while distal femurs immersed in rhodamine B or TRITC-Dextran for 72 hours served as positive control.
To fix the exposed knee joint of live mice and provide support for the container of fluorescence dye, a fixing device equipped with 2 universal manipulators connected to the “L” stainless steel plate was custom designed. During diffusion experiment, animal was anesthetized with pentobarbital sodium (1%) at a dosage of 50 mg/kg (intraperitoneal [IP] injection); the right knee joint capsule was opened, and the femoral condyle was detached from the tibial plateau. The soft tissue was removed while the integrity of the cartilage surface and CCZ was maintained. The exposed femoral condyle was fixed through the pore (diameter of 3 mm) at the center of a petri dish on the device mentioned above, and the gap between femoral condyle and petri dish pore was sealed with adhesive (UHU, Germany) to avoid any leakage. Rhodamine B or TRITC-Dextran solution was then dropped into the petri dish until the AC was merged (Suppl. Fig. S1). Samples at different time points were collected and kept at −80°C for further analysis. All experiments were performed under anesthesia, until mice were sacrificed to harvest femur samples by anesthetic overdose, and all efforts were made to minimize animals suffering during and after operation.
To avoid redistributing the fluorescent dyes in osteochondral samples during traditional histological analysis, samples were first freeze-dried at −55°C for 12 hours, followed by embedding with MC-Plastic I embedding kit (Sunbio, Nanjing, China) at 4°C overnight. Samples were then cut at medial femoral condyle in sagittal plane, grinded with sandpapers until being smooth enough for confocal microscope observation (Zeiss LSM 780, Standort Göttingen, Germany). The planes were first examined 5 to 20 µm below the cutting surface using 488/520 nm excitation/emission to record the distribution of fluorescent dyes, then stained with 4′,6-diamidino-2-phenylindole (DAPI, 5 µg/mL, Beyotime, Shanghai, China) and sodium fluorescein (0.1%, Aladdin) for 30 minutes to visualize the tidemark and cement line. The samples were then scanned with confocal microscope again using 360/460 nm excitation/emission for DAPI and 495/519 nm for sodium fluorescein, respectively. Images were emerged with the previously acquired images in Photoshop (version 2017.0.0, Adobe). The experimental process is shown in Figure 1 .
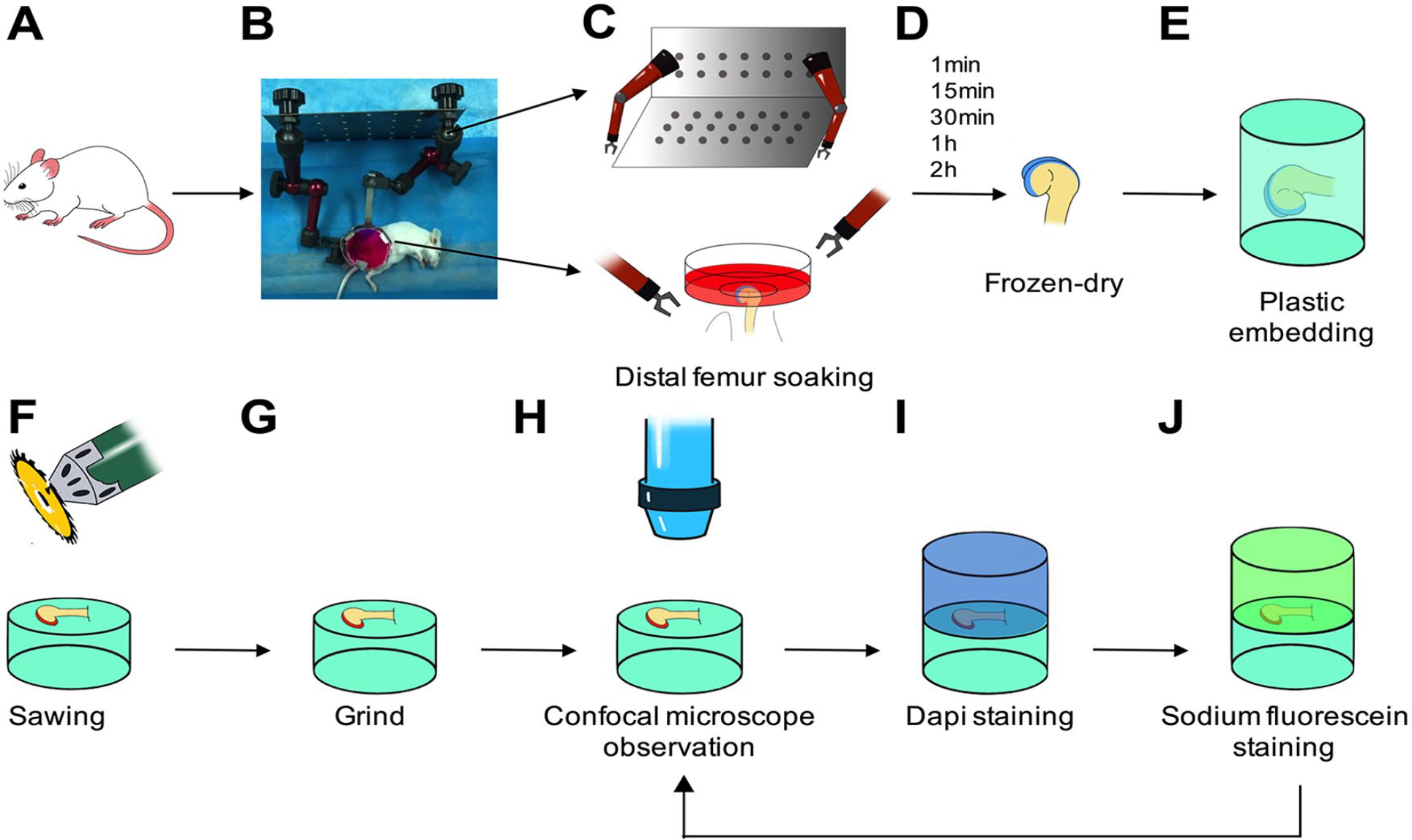
The procedure of the experiments. The right knee joint capsule of mouse was opened and the femoral condyle was detached from the tibial plateau. The exposed femoral condyle was fixed through the petri dish on the custom designed device. Rhodamine B or tetramethyl-rhodamine isothicyanate-dextran (TRITC-Dextran) solution was then dropped into the petri dish until the AC was submerged (
Image Analysis
Fluorescence intensity measurement from cartilage surface to deep layer was done by Zen software. In detail, three 10 × 140 µm rectangular areas covered from the top of AC to SB were randomly chosen in each sample. The mean fluorescence intensity of the area with 10 µm width and 0.41 µm length was measured continuously from top to bottom in the rectangle. To calculate the relative fluorescence intensity (RFI) value of rhodamine B or TRITC-Dextran, fluorescence intensity at each measurement was normalized by the differences between maximum fluorescence intensity in the positive control group and minimum fluorescence intensity in the blank control. A linear graph was drawn to display the trend of distance-fluorescence value based on the RFI value ( Fig. 2 ). In each section, the mean fluorescence intensity of AC was represented by the mean RFI of the top 20 µm from the cartilage surface to the middle zone. To compare the fluorescence intensity of the immature-CCZ group and mature-CCZ group at the same depth, the RFI at the distance of every 10 µm between 0 and 100 µm depths from AC surface was selected at the 2-hour time point of each subgroup.
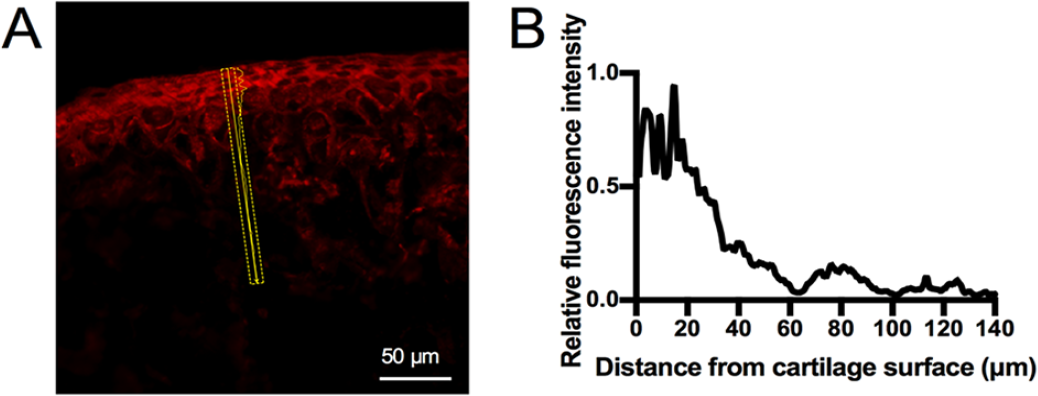
Image analysis of live animal permeability experiment. Representative image from 2-hour diffusion of rhodamine B in immature-CCZ group (1-month old mouse). A 10 × 140 µm rectangular area covering articular cartilage (AC) and subchondral bone (SB) and perpendicular to AC surface was randomly chosen (
Statistical Analysis
Data analysis and graphing was performed with GraphPad (version 7, GraphPad Software). One-way ANOVA analysis was used to exam the differences in the thickness of total cartilage between 1-month and 6-month mice. The differences of cartilage layer RFI between 72 hours and other time points were analyzed by 2-way ANOVA test, followed by Bonferroni post hoc correction. In addition, multiple t tests were used to compare RFI at the same depth of the 2-hour time point in the immature-CCZ group and mature-CCZ group. A P value less than 0.05 was considered to indicate statistically significant difference.
Results
Development of CCZ in Mice
The histological observation on the development of CCZ from 1-month to 8-month mice was performed ( Suppl. Fig. S2 ), and the representative images of 1-month and 6-month mice are shown in Figure 3 . The CCZ of the knee joint in 1-month mouse was immature. Although hypertrophic chondrocyte was noticed in the deep zone cartilage, tidemark, and cement line were not present ( Fig. 3A ). At the age of 6 months, the joint was mature; the typical structures of AC, CCZ, and SB along with the tidemark and cement line were clearly observed ( Fig. 3B ). The thickness of total cartilage (AC and CCZ) in the femur of 1-month mouse was 93.5 ± 24.8 µm, and that of 6-month mouse was 70.9 ± 14.5 µm (P > 0.05, n = 12; Fig. 3C ). In the joint of the 6-month mouse, the thickness of AC was 27.6 ± 6.2 µm, 58.1 ± 12.8 µm in femur and tibia; the thickness of CCZ was 43.3 ± 12.8 µm, 58.0 ± 19.7 µm in femur and tibia (n = 12; Fig. 3D ). Based on the histological observation, 1-month and 6-month mice were selected for immature-CCZ group and mature-CCZ group, respectively.
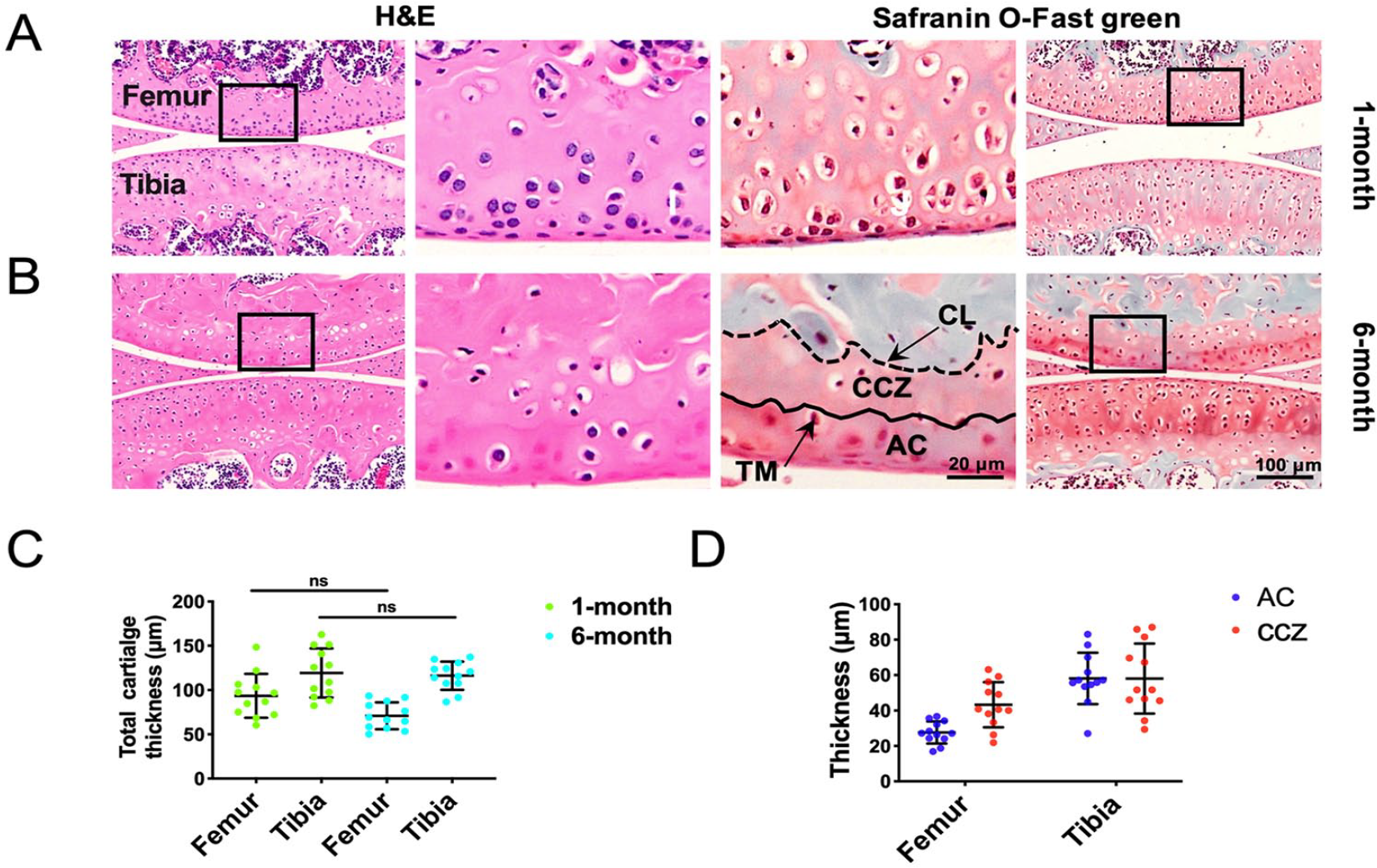
Histologic analysis of CCZ development. Representative images of hematoxylin-eosin (H&E) and safranin O-fast green staining in 1-month (
Permeability of CCZ in Live Mice
Using the custom designed animal fixing device and un-decalcified fluorescence observation method, the distribution of fluorescence tracer in the samples at different time points was observed under confocal microscope. As shown in Figure 4 , SB and AC were visible with the fluorescein sodium and DAPI staining. After 1 hour of diffusion, rhodamine B was present in full-thickness of AC, and in SB tissue of immature-CCZ group ( Fig. 4D ). While in mature-CCZ group, rhodamine B was present in full-thickness of AC, and absent in SB plate and underlying trabecula bone tissue ( Fig. 4E and F ), suggesting blockage of rhodamine B diffusion toward SB by mature tidemark ( Fig. 4H ).
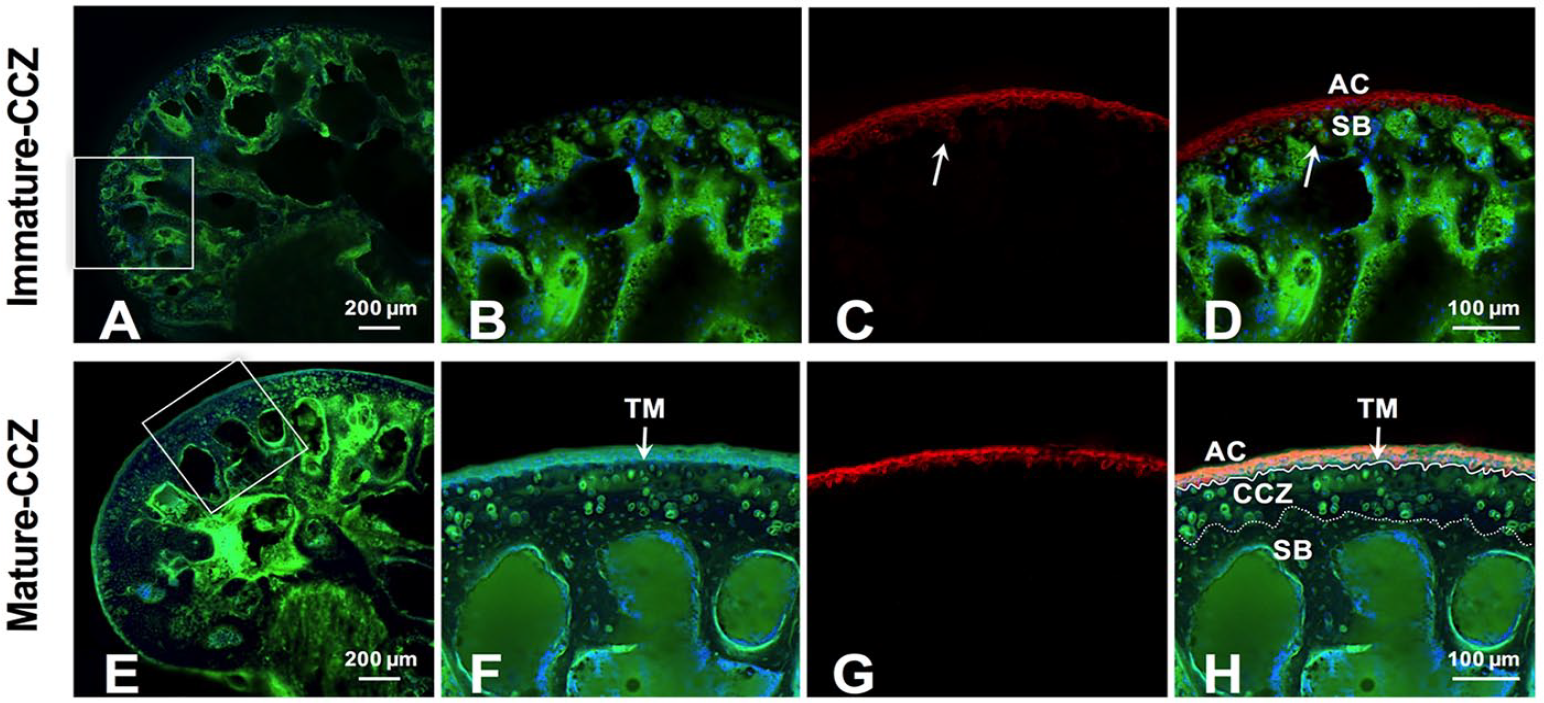
Representative images of the fluorescence tracers’ distribution in distal femur at the time point of 2 hours. AC and SB were visualized in distal femur of 1-month and 6-month mouse joints with sodium fluorescein (green) and DAPI (blue) staining (
Rhodamine B Transport in Distal Femur
The transport of rhodamine B in distal femur of live mice from AC surface to SB at different time points in the 2 groups was analyzed. As shown in Figure 5A and B , the RFI of AC gradually increased as the diffusion prolonged in both groups. By investigating the time required by rhodamine B to reach the saturated concentration in AC layer, the mean RFI of cartilage within 20 µm from the surface was measured. Compared with the positive control group (72 hours), no significant difference of RFI in AC was found after 1 hour diffusion period for the mature-CCZ group ( Fig. 5F ), and after 30 minutes for immature-CCZ group ( Fig. 5E ), suggesting that minimum time frame was required by rhodamine B to reach the saturated concentration in AC of both groups. In the immature-CCZ group, the RFI of osteochondral unit gradually decreased from the AC surface to SB at all time points ( Fig. 5C ). In contrast, a drastic decrease of RFI was noticed at the distance of 20 µm to the AC surface in the mature-CCZ group ( Fig. 5D ). The 2-hour diffusion quantitative results indicated that there was a significant difference of rhodamine B intensity between the mature-CCZ group and immature-CCZ group below 20 µm from the AC surface ( Fig. 5G , P < 0.05), suggesting absence of rhodamine B below tidemark in the mature-CCZ group.
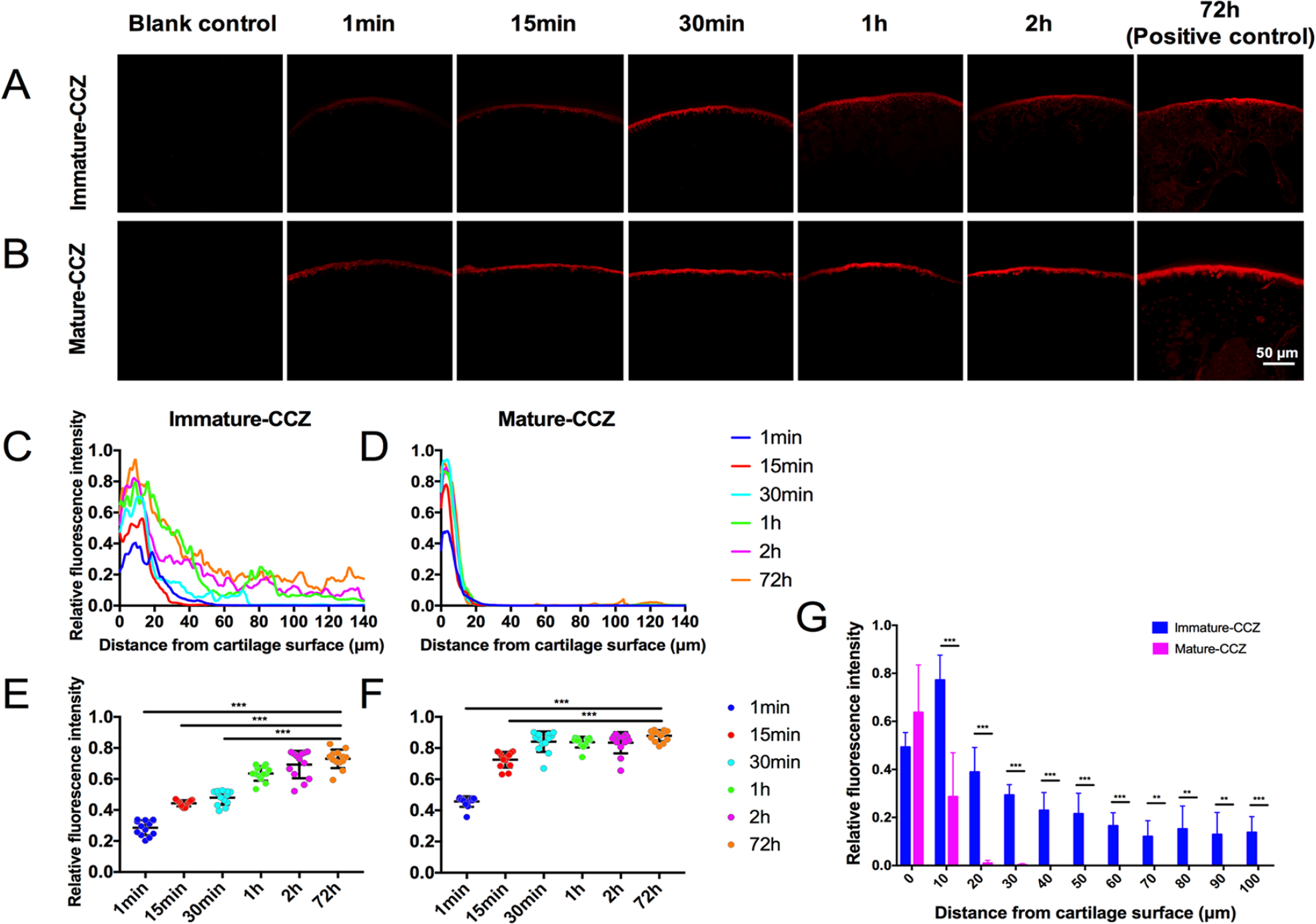
The transport of rhodamine B in distal femur of live mice. The distribution of rhodamine B (red) at different time points in immature-CCZ group (
TRITC-Dextran Transport in Distal Femur
The transport of TRITC-Dextran in the distal femur of live mice is shown in Figure 6A and B . The RFI of AC gradually increased as the diffusion prolonged in both groups. Similarly, the mean RFI of cartilage within 20 µm from the surface was measured to investigate the time required by TRITC-Dextran to reach the saturated concentration in AC layer. Compared with the positive control group (72 hours), no significant difference of RFI in AC was observed after 30-minute diffusion period for the mature-CCZ group ( Fig. 6F ), and after 1 hour for the immature-CCZ group ( Fig. 6E ). The RFI of 0 to 1 hour time points decreased sharply at the distance of 20 µm from the cartilage surface in both groups ( Fig. 6C and D ). The 2-hour diffusion quantitative results indicated that there were no statistical differences in the RFI of TRITC-Dextran at 0 and 10 µm below the AC surface between groups, but significant differences in the RFI of TRITC-Dextran at 20 µm below the AC surface between groups ( Fig. 6G ). While the RFI value of TRITC-Dextran was undetectable below 30 µm from AC surface in the mature-CCZ group, quantitative data indicated low concentration of TRITC-Dextran at the same region of immature-CCZ joints, suggesting penetration of TRITC-Dextran through immature-CCZ tissue.
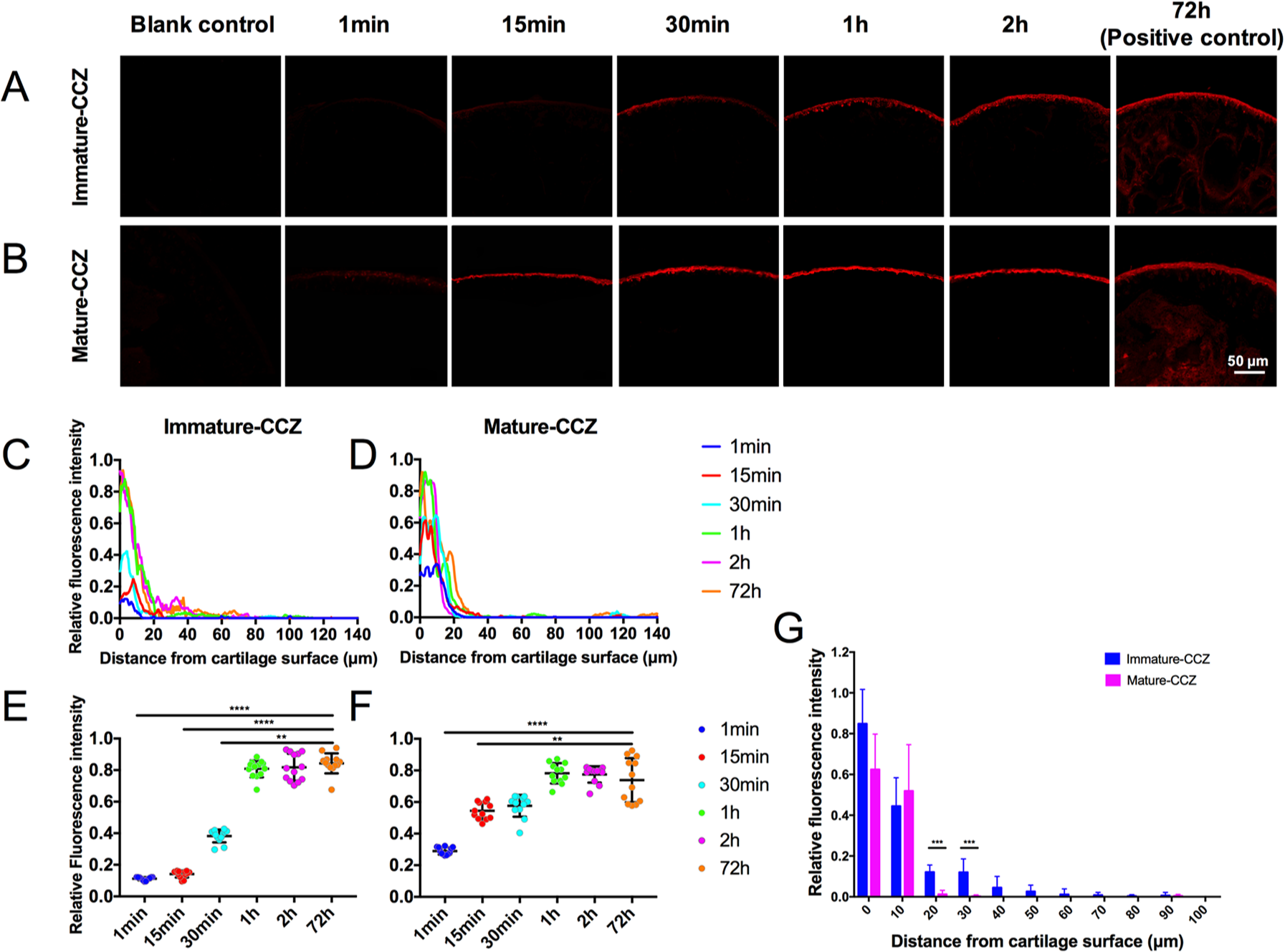
The transport of TRITC-Dextran in distal femur of live mice. The distribution of TRITC-Dextran at different time points in immature-CCZ (
Discussion
As far as we know, this study was the first attempt to explore the permeability of CCZ in live mice by utilizing a live mouse fixing device and an un-decalcified fluorescence observation method. According to our data, the intact mature CCZ blocked the solute of molecular weight of 479 Da (rhodamine B) and 20 kDa (TRITC-Dextran) transport from AC to SB within 2 hours in vivo. The diffusion of both tracers dramatically decreased near the tidemark rather than gradually attenuated within the CCZ tissue. These data suggested that tidemark blocked further diffusion due to its dense organic component as calcification front14,25 and unique deposition of inorganic constituents such as trace element. 26 However, some previous studies claimed that tidemark and CCZ was permeable to low-molecular-weight substance.23,24,27 The reason for this contradiction may lay on the involvement of procedures. In order to perform further analyses, cryo-sectioning and drilling, which may possibly alter the natural property of CCZ or destroy the integrity of tidemark, are often required before diffusion process. In contrast, this study preserved the natural status and integrity of CCZ and recreated the diffusion process in live mice.
In the immature joints, which contained developing tidemark and CCZ, the rhodamine B rapidly diffused to SB tissue in a short time period (1 hour), indicating small nutrition molecules such as amino acids and glucose could easily exchange between AC and SB during joint development. These results were consistent with a previous study in which the nutrition supply of AC in immature animal was mainly from underlying bone tissue, 10 while the nutrition exchange in the AC of mature joint was conducted between AC and synovial fluid. 11 Interestingly, although the diffusion of TRITC-Dextran (20 kDa) rapidly decreased at the developing CCZ interface, evidence of diffusion of TRITC-Dextran toward SB tissue was still observed in the immature joint ( Fig. 6A and G ), indicating gradual translation of calcified cartilage from semipermeable to impermeable to large molecules such as paracrine-acting signaling molecules or hormones during joint development.28,29 As a key mediator, parathyroid hormone-related protein (PTHrP) confined in the AC surface postnatally30,31 has been proven to promote chondrocyte proliferation and suppress the maturation of pre-hypertrophic chondrocytes, thereby preventing further mineralization,28,32-35 while Indian hedgehog (Ihh) secreted by hypertrophic and pre-hypertrophic chondrocytes in the growth plate 33 promotes hypertrophy in adjacent chondrocytes.36-38 Spatiotemporal expression patterns and inverse concentration gradient of these two counteracting factors, combined with hormones stimulating articular chondrocyte hypertrophy and mineralization from SB such as triiodothyronine (T3), 39 might subsequently define the position of calcification front (tidemark) and result in fully calcified underlying CCZ structure.
It is well accepted that increased vascular communication channels, fissures, and microcracks through the CCZ in OA could facilitate molecular transport, suggesting enhanced crosstalk of molecules between AC and SB. 40 Bone turnover seems to play a pivotal role in the pathogenesis of OA. 13 Thinning and increased porosity of the SB were significantly related to cartilage damage in the early stage of OA. 14 The mediators releasing form SB remodeling may transmit through broken osteochondral barrier and activate the cascade of cartilage degeneration. In this study, TRITC-Dextran (20 kDa) was utilized to imitate pro-inflammatory cytokines and chemokines, such as interleukin 1 (IL-1, 17 kDa) 15 and tumor necrosis factor-α (TNF-α, 17 kDa) 41 due to similar molecular weights. Our data indicated that compared with mature CCZ, diffusion of TRITC-Dextran from AC to SB was significantly higher at 20 µm below the AC surface in immature CCZ, suggesting that cytokines may not diffuse freely in the barrier when physiologic structure CCZ is present. Nevertheless, immature CCZ does not represent many features such as cracks, composition alternation of CCZ in OA tissue. Role of CCZ in regulating inflammatory loop between AC and SB in OA pathogenesis still needs further investigation.
There are also some limitations in our study. First, we aimed to imitate the CCZ permeability in live condition, but diffusion period longer than 2 hours was difficult to perform due to the anesthetic effect. Blocking effect of mature CCZ during long period of diffusion was thus undetermined. Second, as the limb was fixed during the experimental process, the effect of joint motion on the diffusion of solutes was not tested. Since the mechanical force during joint movement is an important factor for cartilage nutrition exchange,42,43 whether the joint motion promotes the permeability of CCZ needs further investigation. Third, due to the influence of growth plate on the distribution of fluorescence tracer (data not shown), indicators were only diffused from AC to SB in this study. To distinguish the roles of tidemark and calcified cartilage in regulating solute transportation, a novel method performing diffusion experiment from SB to AC, while preserving the integrity of the SB structure, is needed.
In conclusion, utilizing a live mouse fixing device and an un-decalcified fluorescence observation method, this study investigated the permeability of CCZ to small and large solutes in live mice. Our results indicated a rapid decrease of permeability for both fluorescence tracers at the front of mature CCZ (tidemark). Although the detailed mechanism was unaddressed, our results have provided direct evidence of blocking effect of CCZ in solute transportation during short diffusion period in live animal, indicating the important role of CCZ during joint development and microenvironment maintenance in mature joint.
Supplemental Material
supplementary_figS1 – Supplemental material for Observation of Solute Transport between Articular Cartilage and Subchondral Bone in Live Mice
Supplemental material, supplementary_figS1 for Observation of Solute Transport between Articular Cartilage and Subchondral Bone in Live Mice by Yang Huang, Cheng Chen, Fuyou Wang, Guangxin Chen, Shidi Cheng, Zhexiong Tang, Zheng Li, Xiaoyuan Gong and Liu Yang in CARTILAGE
Supplemental Material
Supplementary_figS2 – Supplemental material for Observation of Solute Transport between Articular Cartilage and Subchondral Bone in Live Mice
Supplemental material, Supplementary_figS2 for Observation of Solute Transport between Articular Cartilage and Subchondral Bone in Live Mice by Yang Huang, Cheng Chen, Fuyou Wang, Guangxin Chen, Shidi Cheng, Zhexiong Tang, Zheng Li, Xiaoyuan Gong and Liu Yang in CARTILAGE
Supplemental Material
Supplementary_figure_legends – Supplemental material for Observation of Solute Transport between Articular Cartilage and Subchondral Bone in Live Mice
Supplemental material, Supplementary_figure_legends for Observation of Solute Transport between Articular Cartilage and Subchondral Bone in Live Mice by Yang Huang, Cheng Chen, Fuyou Wang, Guangxin Chen, Shidi Cheng, Zhexiong Tang, Zheng Li, Xiaoyuan Gong and Liu Yang in CARTILAGE
Footnotes
Correction (August 2024):
Figure 6B-1h has been replaced since its original publication.
Supplemental Material
Author Contributions
Yang Huang, Xiaoyuan Gong, and Yang Liu contributed to the conception and design of the study. Yang Huang, Guangxin Chen, Shidi Cheng, Zhexiong Tang, Fuyou Wang, and Zheng Li participated in acquisition of the data. Yang Huang, Xiaoyuan Gong, Yang Liu, and Cheng Chen participated in analysis of the data. All authors contributed to interpreting the data, drafting or revising the manuscript, and have approved the submitted version of the manuscript.
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by grants from the National Natural Science Foundation of China (31130021, 8157090269, 31800784), Chongqing Social Welfare Innovation Project (cstc2016shmszx130021), and the Foundation of Southwest Hospital (SWH2016JSTSYB-54). The authors thank Xin Chen from the Center for Joint Surgery, Southwest Hospital, Third Military Medical University, for the language support for this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All animal procedures were approved by the Animal Ethics Committee of Third Military Medical University (Army Medical University).
Animal Welfare
All animal procedures were in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
