Abstract
Objective
Meniscus tissue is composed of highly aligned type I collagen embedded with cartilaginous matrix. This histological feature endows mechanical properties, such as tensile strength along the direction of the collagen alignment and endurance to compressive load induced by weight bearing. The main objective of this study was to compare the fibrocartilage construction capability of different cell sources in the presence of mechanical stimuli.
Design
Synovial multipotent stem cells (SvMSCs) and meniscal chondrocytes (MCs) from immature and mature rabbits were maintained under similar conditions for comparative evaluation of growth characteristics and senescence tendency. The differentiation potential of cell sources, including fibrocartilage generation, were comparatively evaluated. To determine the capability of fibrocartilage generation, cultured cell sheets were rolled up to produce cable-form tissue and subjected to chondrogenic induction in the presence or absence of static tension.
Results
Although SvMSCs showed superior cell growth characteristics during
Conclusions
While SvMSCs produced fibrous tissue during default
Introduction
Meniscus is a crescent-shaped cartilage tissues located between the femur and tibia in the lateral and medial side of the patellar joint. The meniscus exhibits differential histological microstructures in the inner and outer sides of the crescent shape associated with tissue functionality. The outer side of meniscus displays typical fibrocartilage morphology with highly aligned type I collagen throughout the tissue and blood vessels penetrating the outermost layer. The inner side of the meniscus partly includes hyaline cartilage characterized by a glycosaminoglycan (GAG)-rich region.1-3 Highly aligned collagen-rich matrix can bear high tensile loads and resist shape deformation caused by loads laterally delivered from the weightbearing inner part of meniscus.2-4 The GAG-rich region in the inner meniscus performs a similar function as hyaline cartilage and shares ~50% of the weight load with articular cartilage.
4
Meniscus tissues collected from large animals include tie fiber, an additional hierarchical structure that potentiates tensile modulus of tissue by radially traversing the tissue from the outer to inner side.
3
Fibrocartilage and hyaline cartilage share common histological features, such as low cellularity and an avascular nature. Therefore, following damage, regeneration rarely occurs.1-3,5 Meniscal injuries, such as meniscal tear, are treated via meniscectomy and require orthopedic surgery in ~50% cases.
3
Accumulating meniscus tissue damage can result in compromised function due to structural deformation, in turn, causing higher weight load to articular cartilage in the joint and, eventually, increased risk of early osteoarthritis development.2-4,6,7 In view of the importance of meniscus in prevention of early-onset osteoarthritis, several approaches have been developed to repair damaged meniscus tissue. Despite advances in tissue engineering approaches to formulate the unique microstructure of meniscus, the mechanical properties of
Synovial MSCs (SvMSCs) are postnatal multipotent stem cells (MSCs) that can potentially differentiate into chondrocytes, adipocytes, and myocytes. SvMSCs present a promising cell source for meniscus reconstruction, considering the source, synovium, is a type I collagen-rich fibrous tissue, and display significantly superior chondrogenic differentiation potential compared with bone marro–derived stem cells (BMSCs).11-16
In contrast, meniscal chondrocytes (MCs) are not MSC-like cells with lack of multilineage differentiation potential. Although SvMSCs and MCs show relatively low chondrogenic differentiation potential relative to articular cartilage chondrocytes, high type I collagen expression in these cell types supports their potential utility as a promising cell source for meniscus reconstruction.11,17 Several previous reports have shown higher chondrogenic potential of meniscal fibrochondrocytes (MCs) than SvMSCs.11,18,19
In earlier studies, MCs were cultured in polyglycolic acid (PGA)–hyaluronan 20 or GelMa scaffold 21 for reconstruction of tissue-engineered meniscus. Transplantation of PGA mesh scaffold seeded with rabbit MCs into rabbit joints promoted regeneration of the tissue with proteoglycan and collagen expression. 22
As for the available donor tissue for MC cell isolation, meniscus tissue fragment could be obtained during meniscectomy, involving removal of the meniscus tear to relieve pain and prevent further damage. In case of extensive meniscus damage, restoration of its function is essential to prevent early onset of degenerative joint disease. 23 The tissues obtained from meniscectomy may effectively serve as autologous donors for cell banking in preparation for meniscus reconstruction.
In this study, 2 promising cell sources, SvMSCs and MCs, were subjected to comparative evaluation in terms of fibrocartilage matrix production capability in response to tensile stimulus as well as basic cell characteristics, including
Materials and Methods
Materials
All chemicals were purchased from Sigma-Aldrich (St. Louis, MO, USA) unless otherwise indicated.
Primary Culture of BMSCs
Primary cell cultures were generated from rabbit tissue after review and approval of the study (Approval Number: MCTTIACUC ASP 17-002/Approval Date: September 6, 2017) Institutional Animal Care and Use Committee (IACUC) of Biosolution Inc. Research Institute (Seoul, South Korea). BMSCs were isolated from femur and tibia of New Zealand white rabbits as described previously.24,25 For comparison of age-related characteristics, 4 rabbits in each age group (1 and 8 months old and weighing between 2 and 4.8 to 5 kg, classified as immature and mature, respectively), were subjected to cell yield examinations (
Primary Culture of SvMSCs and MCs
Synovial and meniscus tissues were collected from the patellofemoral joints of rabbits from each age group (immature and mature,
Cell Senescence Assay
Senescence-associated β-galactosidase (SA-β-gal) expressed by each cell type during passage progression between P4 and P8 was assessed using a specific β-Gal Assay Kit (Millipore, Billerica, MA, USA) according to the manufacturer’s instructions. Briefly, cells at each passage were fixed at 95% confluence and treated with the SA-β-gal substrate via overnight incubation in a 37°C incubator in a sealed state. After development of blue color confirmed under phase-contrast microscopy, the reagent was removed and rinsed twice with PBS. The number of β-gal-positive cells was compared between SvMSCs and MCs originating from a matching mature rabbit to reduce the errors caused by differential basal levels in each individual rabbit.
In Vitro Osteogenic Differentiation Potential
SvMSCs and MCs originating from the same mature rabbit were seeded into 6-well plates at a cell density of 2 × 105 cells/well at passage 3 and incubated overnight in standard growth medium. After overnight culture, the medium was switched to osteogenic-induction medium composed of α-MEM supplemented with 20% FBS, 10−8 M dexamethasone, 0.2 mM ascorbic acid, and 10 mM β-glycerophosphate. The medium was changed every 2 days for 4 weeks of
In Vitro Adipogenic Differentiation Potential
For evaluation of adipogenic differentiation potential, SvMSCs and MCs originating from the same mature rabbit were seeded into 6-well culture plates at a density of 2 × 105 cells/well at passage 3 and incubated overnight in standard growth medium. After overnight culture, the medium was changed to adipogenic induction medium composed of high-glucose Dulbecco’s modified Eagle medium (DMEM; Lonza, Basel, Switzerland) supplemented with 10% FBS, 1% penicillin/streptomycin, 10 µg/mL insulin, 10−7 M dexamethasone, 0.2 mM indomethacin, and 0.5 mM 3-isobutyl-1-methylxanthine. Following 2 days of culture in adipogenic induction medium, cells were incubated in adipogenic nutrition medium composed of DMEM supplemented with 10% FBS, 1% penicillin/streptomycin, 10 µg/mL insulin, and 10−7 M dexamethasone. The 2 adipogenic media were alternately replaced every 2 days. After 4 weeks of
In Vitro Chondrogenesis Assay for Hyaline Cartilage Construction
SvMSCs and MCs originating from the same mature rabbit (passage 3) were counted and added to a 15 mL conical tube (2.5 × 105 cells per tube). After seeding, cells were centrifuged at 1,500 rpm for 5 minutes at 4°C and incubated overnight in 1 mL growth medium, carefully avoiding turbulence. After overnight culture, the medium was changed to chondrogenic induction medium composed of high-glucose DMEM (Life Technologies) with 1% penicillin/streptomycin and supplemented with 10−7 M dexamethasone, 0.35 mM proline, 1× ITS-3, 0.3 mM ascorbic acid and 10 ng/mL TGF-β3 (PeproTech, Seoul, South Korea). After 4 weeks of
In Vitro Chondrogenesis Assay under Tensile Stimulus Application
To evaluate production of cartilaginous matrix under tensile stimulus application, sheet-cultured cells were rolled up to form a cable shape and static tension applied during chondrogenesis using a specific static tension chamber. Briefly, rabbit SvMSCs and MCs originating from the same mature individual (passage 3) were seeded at a density of 1 × 107 cells/dish on type I collagen-coated 125 × 125 mm square-shaped dishes (SPL Life Science, Pocheon-si, Gyeonggi-do, South Korea) in growth medium. After reaching confluence, cells were maintained via daily medium changes without passage for 10 to 14 days. Confluent cells were carefully lifted at the edge of the square dish by scraping slowly to form a cable shape. Cell cables were mounted on an acrylic static tension chamber. The initial length of cell cables was 30 mm and 10% static tension was applied. The chondrogenic induction medium was changed twice a week during culture. After 4 weeks of
DNA Quantitative Analysis
The DNA quantification assay was performed using a specific kit (Takara Bio Inc., Kusatsu, Shiga, Japan) for quantification of live cells per tissue following the manufacturer’s protocol. Briefly, tissues were incubated overnight at 37°C with proteinase K and RNase A. Dissolved tissues were reacted with Hoechst 33258 dye and DNA fluorescence measured on a Tecan ELISA reader (Spark Multimode Microplate Reader, Tecan Trading AG, Switzerland) with excitation at 346 nm and emission at 460 nm.
Quantitative Analysis of Cartilaginous Matrix Components
Quantification of Collagenous Matrix
Quantification of collagen generated during
Quantification of Glycosaminoglycans
Quantification of GAGs in tissues subjected to
Statistical Analysis
Numerical data plotted on graphs were presented as mean ± standard deviation. Differences between 2 groups were compared using a 2-tailed paired
Results
Comparison of Colony-Forming Unit–Fibroblasts Proportions in Primary SvMSCs and MCs
The frequencies of colony-forming unit–fibroblasts (CFU-Fs) in primary cultured SvMSCs and MCs were compared with those in BMSCs at passages 0 and 1. At passage 0, CFU-F numbers of BMSCs, SvMSCs, and MCs (1 × 103 cells in each group) were 0, 125.6, and 212.8 in immature animals and 1, 74.3, and 79 in mature animals, respectively. At passage 1, CFU-F numbers in BMSCs, SvMSCs, and MCs were 39, 132.2, and 77.8 in immature animals and 70.6, 178.7, and 123.8 per 1 × 103 cells in mature animals, respectively. SvMSCs and MCs from mature animals showed a higher proportion of CFU-Fs, compared with cells isolated from immature animals at passage 1 (Supplementary Figure S2). The colonies generated by SvMSCs and MCs isolated from mature animals were significantly bigger (2.41- and 13.94-fold difference, respectively) than those generated by cells from immature animals (Supplementary Figure S3). Although there was individual difference in terms of SvMSCs’ colony size, the tendency of SvMSCs’ producing relatively bigger colonies than MCs was consistently repeated in each animal (Supplementary Table S1). The colony size differences could be interpreted as the state of cellular activity, since the colony size is determined by cell population doubling number. 26 Our results indicate that SvMSCs and MCs from mature individuals are more active compared with the corresponding cell populations from immature individuals.
Comparison of In Vitro Cell Expansion Rate between SvMSCs and MCs in Immature and Mature Animals
SvMSCs and MCs were subcultured at 95% confluence and the cell number at each passage calculated to compare cumulative cell growth between the two sources. As the gold standard, BMSCs from matching animals were compared starting at passage 1, since BMSCs at passage 0 cannot be distinguished from mononuclear cells of hematopoietic lineages. SvMSCs displayed a higher
Comparison of Cell Senescence Tendencies of SvMSCs and MCs
Cell senescence characteristics according to passage progression were confirmed based on expression of SA-β-gal. Levels of SA-β-gal in SvMSCs and MCs showed a gradual increase until passages 6 and 7, respectively. At passage 7, SvMSCs showed a marked increase in SA-β-gal that continued to increase at passage 8, clearly indicative of senescence. Although SA-β-gal expression of MCs was significantly increased at passage 7, the number of SA-β-gal(+) MCs remained lower than that of SA-β-gal(+) SvMSCs at all passage time points ( Fig. 1 ), suggesting enhanced cell activity and stable functionality of MCs compared to SvMSCs.
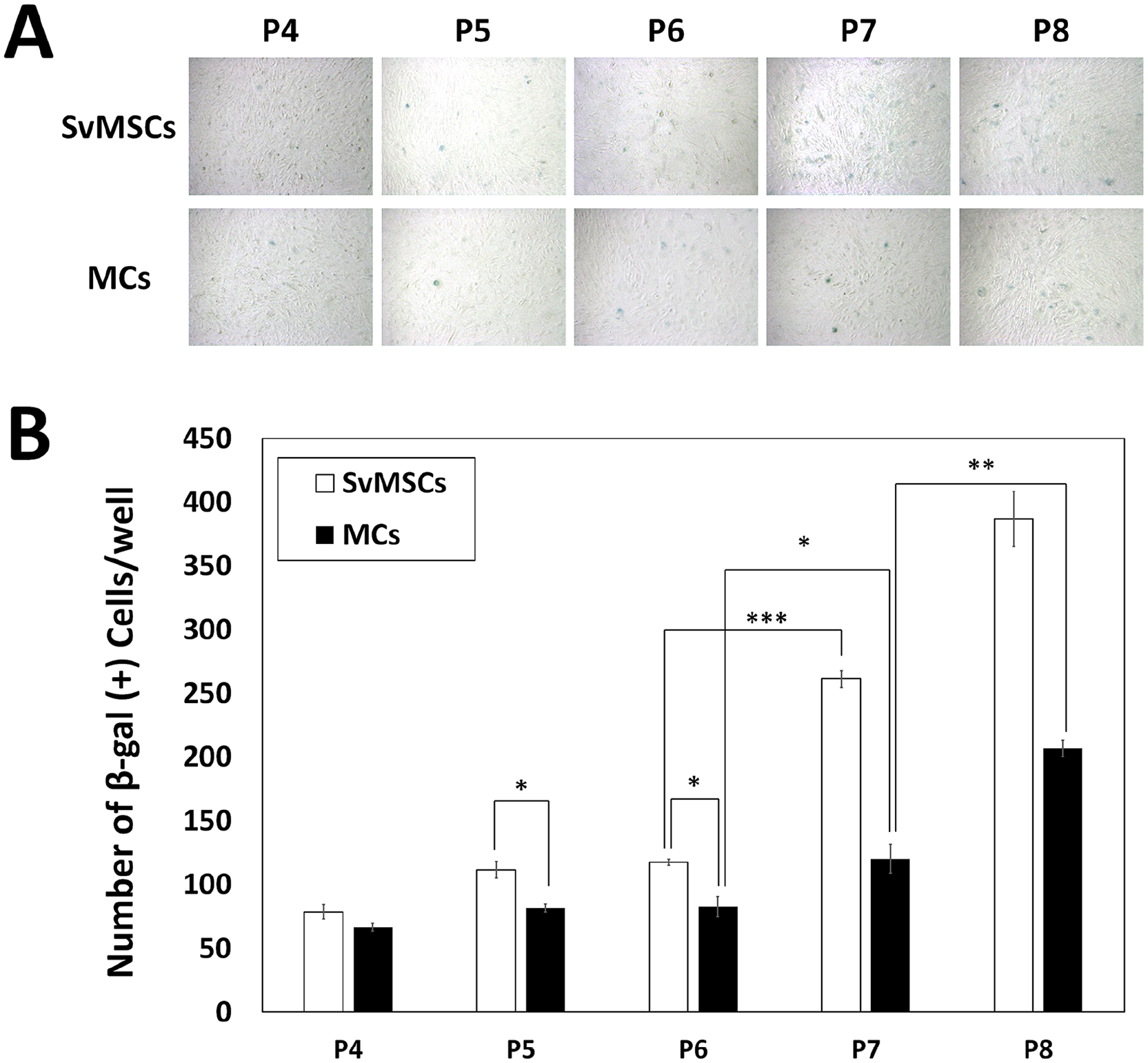
Cell senescence assay via senescence-associated β-galactosidase (SA-β-gal) staining over the course of passage progression in synovial mesenchymal stem cells (SvMSCs) and meniscal chondrocytes (MCs). The number of SA-β-gal(+) cells per well was compared at each passage (
Comparative Evaluation on In Vitro Differentiation Potential of SvMSCs and MCs
SvMSCs are classified as MSCs and show similarity to BMSCs in terms of
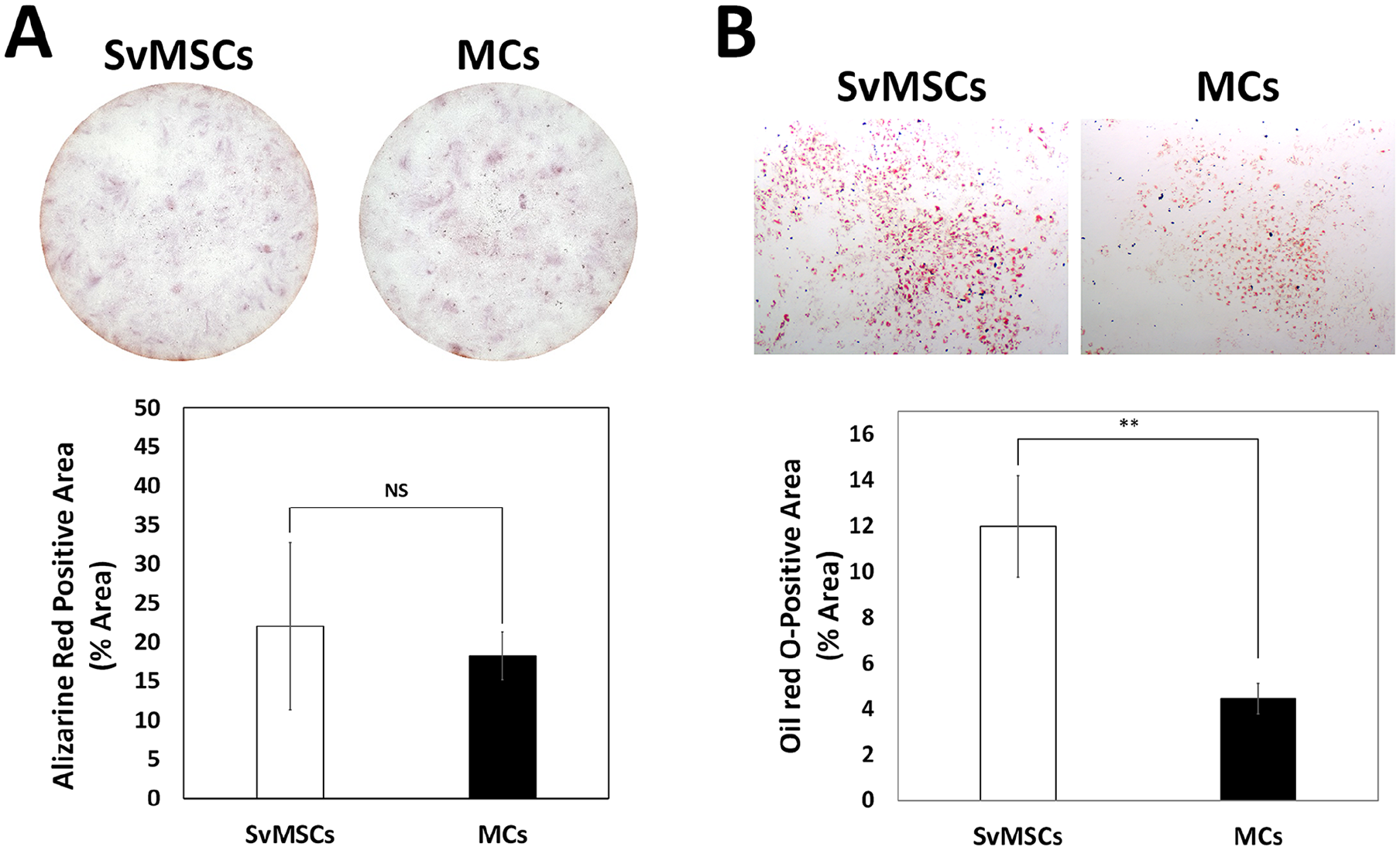
Comparison of
Comparative Evaluation of Default Chondrogenic Potential in the Absence of Mechanical Stimulus
SvMSCs is reported to show superior chondrogenic differentiation potential compared with other MSCs from various tissue sources.6,13-15 Here, the chondrogenic differentiation potential of SvMSCs and MCs was comparatively evaluated using the pellet culture method. While the pellet size in both cell types increased during the differentiation period, MCs produced bigger pellets relative to SvMSCs, which were 1.28-, 1.31-, and 1.30-fold bigger at 1, 2, and 3 weeks of chondrogenic differentiation, respectively ( Fig. 3A and B ). Histological staining showed that MCs started to produce GAG-rich cartilaginous matrix at 2 weeks of chondrogenic induction while SvMSCs failed to form a GAG-rich matrix at all time points ( Fig. 3C and D ). Based on trichrome staining, matrix produced by both cell types were devoid of fibrous matrix up to 2 weeks of chondrogenic differentiation. At 3 weeks of chondrogenic induction, SvMSCs started to show trichrome-positive fibrous tissue formation while MCs lacked the trichrome-positive area ( Fig. 3E ). At 4 weeks of chondrogenic induction, SvMSC pellets obviously formed fibrous tissue while MC pellets showed greater staining intensity of safranin O–positive GAG-rich matrix. Our findings indicate that MCs maintain superior differentiation potential toward hyaline cartilage while SvMSCs resulted in fibrous tissue formation over 4 weeks of chondrogenic induction.
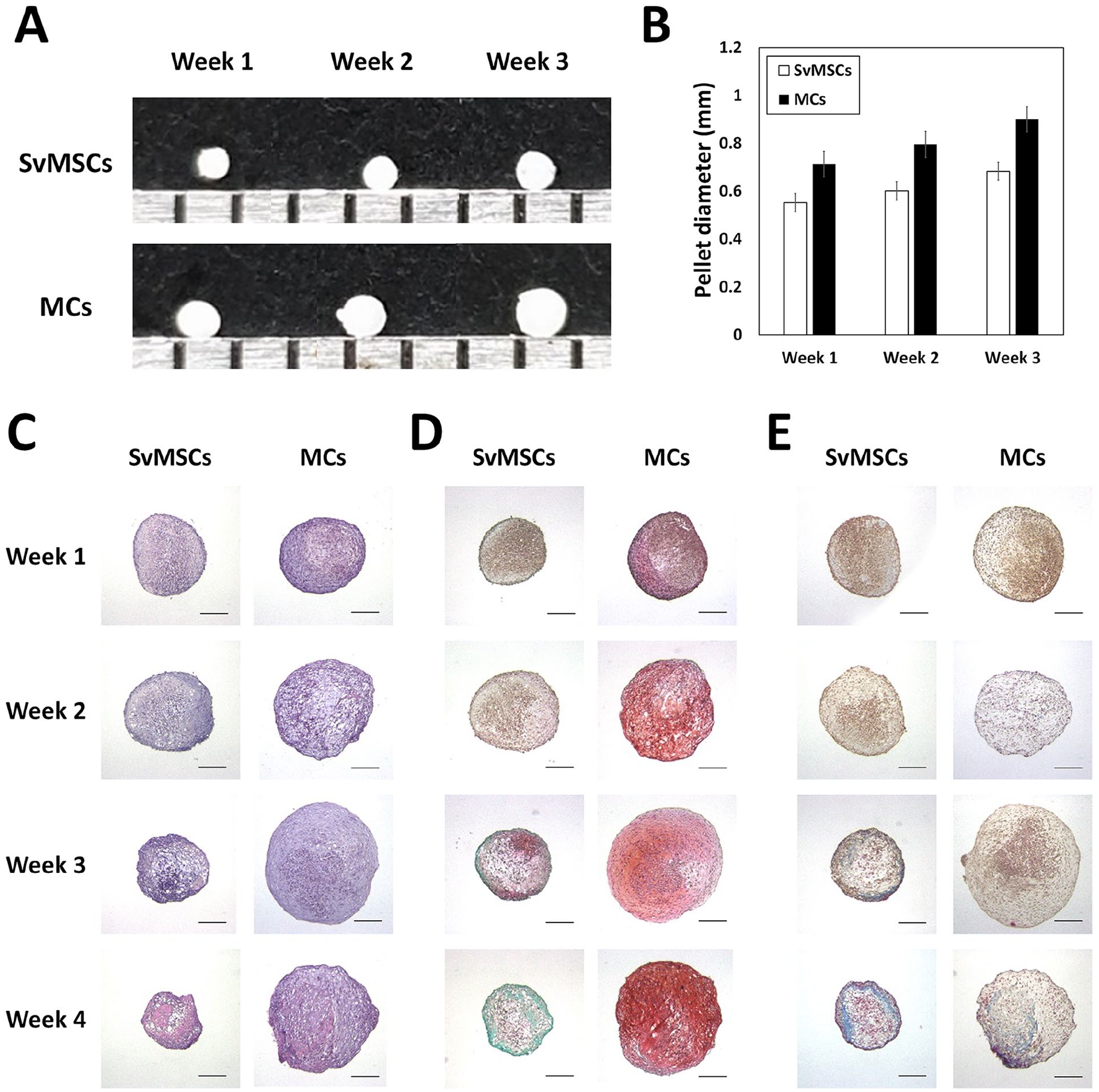
Comparison of
Comparative Evaluation of Fibrocartilage Generation Potential with Application of Tensile Stimulation
The microstructure of fibrocartilage involves uniaxial tissue alignment, which can be effectively induced via application of mechanical stimulus. However, mechanotransduction induced by tensile stimulus is reported to inhibit chondrogenic induction.27,28
To compare the cartilaginous matrix production capability of the different cell sources during tissue alignment induction by mechanical stimulation, sheet-cultured SvMSCs and MCs in growth media were rolled up to generate cable-form tissue (Supplementary Figure S6) and subjected to chondrogenic induction with tensile mechanical stimulation by 10% extension of the original length using a static tension chamber (Supplementary Figure S3). The thickness of tissue formed by MCs increased during the chondrogenic culture period while the tissue formed by SvMSCs showed no significant differences in thickness (
Fig. 4A
and
B
). In contrast to pellets of cultures subjected to chondrogenic induction without tensile stimulus (
Fig. 3
), cable-form tissues generated with SvMSCs and MCs subjected to 10% tensile stimulus did not show high-intensity safranin O(+) GAG-rich cartilaginous matrix formation (
Fig. 4C
). On the other hand, the blue color in the trichrome-stained section was indicative of fibrous matrix (
Fig. 4D
). Although SvMSCs formed trichrome(+) fibrous tissue via default chondrogenesis (
Fig. 3E
), intense staining was not observed in tissues subjected to chondrogenesis with tensile stimulation (
Fig. 4D
). Notably, MCs generated more intensive trichrome(+) fibrous tissue under tensile stimulation, compared with SvMSCs (
Fig. 4D
). For numerical comparison, equally divided tissue fragments were subjected to quantitative analysis for DNA, collagen, and GAG. Cell numbers predicted via DNA quantification showed no significant changes during chondrogenic induction (
Fig. 5A
). Quantification of collagen content revealed a significant increase in collagen production by tissues formed by MCs during
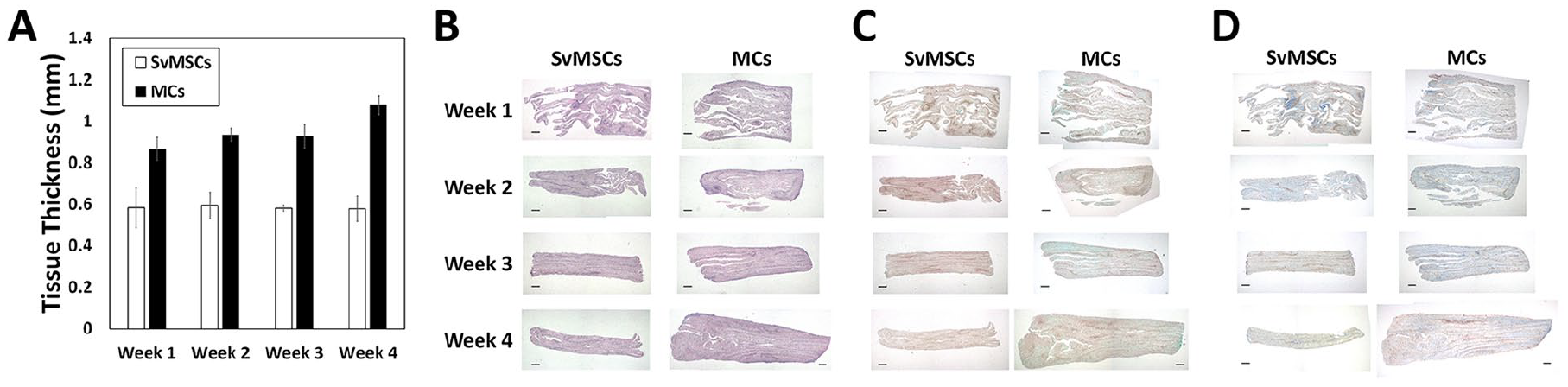
Comparison of the chondrogenic capabilities of synovial mesenchymal stem cells (SvMSCs) and meniscal chondrocytes (MCs) on application of tensile stimulation. Changes in tissue thickness were measured at time points each week (
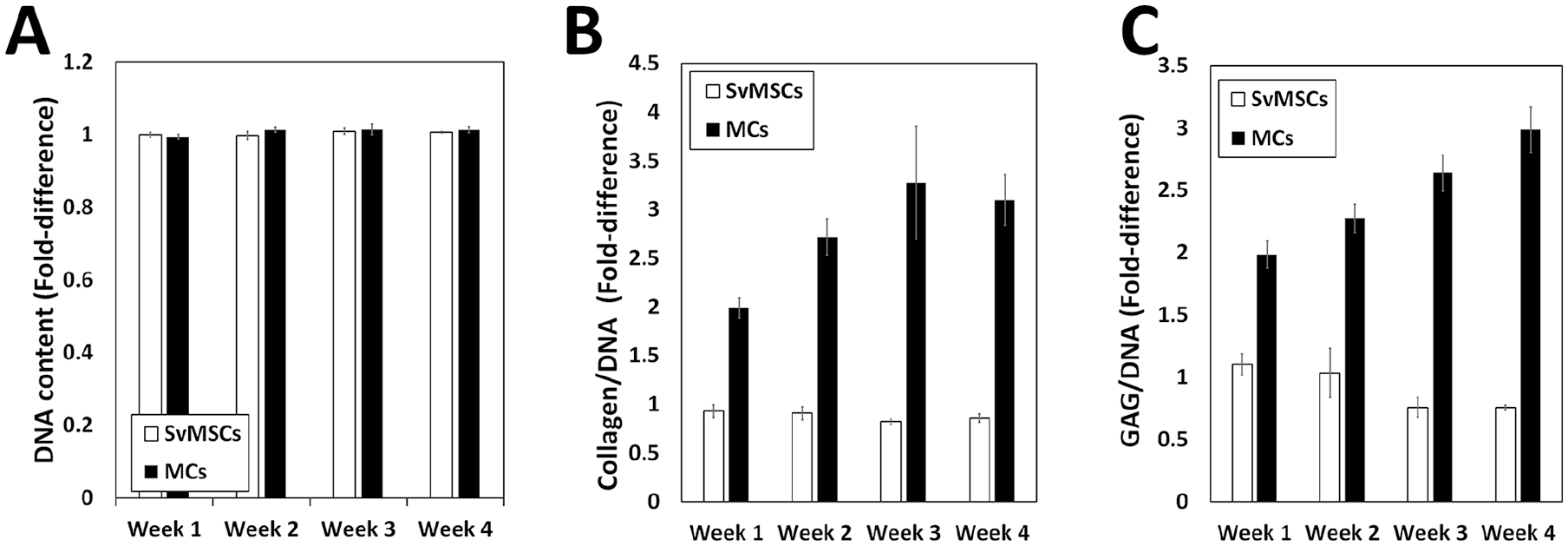
Analysis of changes in tissue components at time-points at each week in tissues subjected to fibrocartilage differentiation with tensile stimulation. Tissue fragments were divided into groups and subjected to Hoechst-based DNA quantification (
Discussion
Because of the technical challenges in reconstructing tissue-engineered meniscus, replacement with an allogeneic meniscus from cadaver donors is considered the best clinical practice for patients with severely damaged meniscus with no signs of osteoarthritis. 4 Several trials have been performed to reconstruct meniscus tissue with various cell sources and tissues, among which the most potent tissue construction strategies include mechanical training. 29 Application of tensile stimulus can promote fibrous tissue formation by inducing tissue alignment and type I collagen production. However, tensile stimulus activates mechanotransduction and induces differential responses according to cell type, such as an inhibitory effect on chondrogenesis in chondrocytes27,31 and stimulation of osteogenesis in MSCs.28,30 Therefore, cell responses to mechanical stimulation should be fully considered before utilization for meniscus reconstruction.
SvMSCs have been shown to possess MSC-like characteristics.11-16 In the current investigation study, both SvMSCs and MCs did not display alizarin red S staining after
These results indicate that evaluation of source cell functionality to achieve maximal mechanical load-bearing capability is essential for optimization of functional tissue engineering. 34 For efficient construction of tissues with mechanical functions, determination of the tissue formation characteristics of source cells in response to mechanical stimuli is critical, not only for the tissue construction process but also to replicate their response to the continuous mechanical stress imposed by the local environment following transplantation.
To elucidate the characteristics of
In general, cells isolated from younger animals grow faster and show higher activity compared with those isolated from older animals. In our study, cells isolated from immature animals produced more colonies at primary culture (P0) in the CFU-F assay. However, once established, cells isolated from mature animals produced greater numbers and sizes of colonies than cells isolated from immature animals at passage 1 (Supplementary Figures S2 and S3). These characteristics may account for the higher number of cells from mature animals in terms of accumulative cell growth. Cell functionality can be enhanced if mechanical stress exerts beneficial effects by stimulating activities, such as survival, proliferation, and matrix production. In fact, mechanical stress experienced during the postnatal development process activates chondrocytes to acquire functionality resulting in tissue maturation and property changes. 36
Although no direct comparison of the responses of SvMSCs and MCs to mechanical stimuli has been reported in the literature, comparison of chondrogenic potentials of the 2 cell types revealed consistency with previous reports. Pellets of human meniscal fibrochondrocyte cultures showed better cartilaginous matrix formation based on GAG production than synovium-derived MSCs. 18 Human synovium-derived MSCs are additionally phenotypically unstable with low expression of type II collagen and Sox-9.11,19
In the current study, matrix production characteristics in response to static tension were examined in a simple acrylic chamber designed for small-scale tissue cultivation. To generate functional tissue-engineered meniscus, culture conditions need to be optimized. First, the mechanical dose should allow sufficient cartilaginous matrix formation while maintaining tissue alignment to limit the chondroinhibitory effect of mechanotransduction.27,28,37 Second, differential application of combined mechanical stimuli may be required to materialize the tissue alignment and replicate the unique tissue characteristics of meniscus. 29 Third, scaling up cultivation of tissue-engineered cartilage devoid of the necrotic inner core caused by undernutrition remains a considerable challenge. Therefore, several additional strategies require consideration to generate transplantable functional meniscus tissue using the tissue engineering approach.
To overcome the challenges in meniscus reconstruction, utilization of the optimal cell source is critical to reproduce target tissue properties. In our experiments, SvMSCs failed to show tissue growth during
Supplemental Material
R2_-_Supplementary_information – Supplemental material for Comparative Evaluation of Synovial Multipotent Stem Cells and Meniscal Chondrocytes for Capability of Fibrocartilage Reconstruction
Supplemental material, R2_-_Supplementary_information for Comparative Evaluation of Synovial Multipotent Stem Cells and Meniscal Chondrocytes for Capability of Fibrocartilage Reconstruction by Jisoo Lee, Seoyoung Jang, JunPyo Kwon, Tong In Oh and EunAh Lee in CARTILAGE
Footnotes
Supplemental Material
Author Contributions
JL: Contribution to experiment, analysis, drafting of the article. SJ: Contribution to experiment, analysis, drafting of the article. JPK: Contribution to experiment. TIO: Contribution to concept and design, interpretation of the data, drafting of the article, final approval of the article. EL: Contribution to concept and design, experiment, analysis, interpretation of the data, drafting of the article, final approval of the article.
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Basic Science Research Program through the National Foundation of Korea (NRF) funded by the Ministry of Education (2018R1D1A1A02047934), Basic Science Research Program through the National Foundation of Korea (NRF) funded by the Ministry of Science and ICT (2018R1D1A1B07047508), and a grant of the Ministry of Health and Welfare of Korea (HI18C2435).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Institutional Animal Care and Use Committee (IACUC) of Biosolution Inc. Research Institute (Seoul, South Korea) (Approval Number: MCTTIACUC ASP 17-002/Approval Date: September 6, 2017).
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
